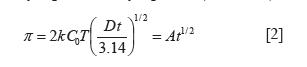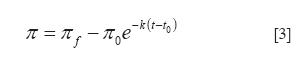Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
Revista Colombiana de Química
Print version ISSN 0120-2804On-line version ISSN 2357-3791
Rev.Colomb.Quim. vol.40 no.3 Bogotá Sept./Dec. 2011
INFLUENCE OF 1-BUTANOL, 1,2-BUTANEDIOL AND 1,2,3,4-BUTANETETROL ON THE ADSORPTION OF b-LACTOGLOBULIN AT THE AIR-WATER INTERFACE
INFLUENCIA DE 1-BUTANOL, 1,2-BUTANODIOL Y 1,2,3,4-BUTANOTETROL EN LA ADSORCIÓN DE b-LACTOGLOBULINA EN LA INTERFASE AIRE-AGUA
INFLUÊNCIA DE 1-BUTANOL, 1,2-BUTANODIOL E 1,2,3,4-BUTANOTETROL NA ADSORÇÃO DE b-LACTOGLOBULINA NA INTERFACE AR-ÁGUA
Carmen M. Romero1,2, Alberto R. Albis3, Néstor E. Mendieta1
1 Universidad Nacional de Colombia, sede Bogotá, Facultad de Ciencias, Departamento de Química, Grupo de Termodinámica Clásica, Laboratorio de Investigaciones Básicas, Calle 44 # 45-67 Bloque B9, Bogotá, Código Postal 111321 - Colombia.
2 Universidad Nacional de Colombia, sede Bogotá, Facultad de Ciencias, Departamento de Química, Grupo de Termodinámica Clásica, Laboratorio de Investigaciones Básicas, Calle 44 # 45-67 Bloque B9, Bogotá, Código Postal 111321 - Colombia. cmromeroi@unal.edu.co
3 Universidad del Atlántico. Facultad de Ingeniería, Departamento de Ingeniería Química, Barranquilla, Colombia.
Recibido: 20/11/11: Aceptado: 30/12/11
ABSTRACT
In this work, a systematic study on the effect of 1-butanol, 1,2-butanediol and 1,2,3,4-butanetetrol (erythritol) on the surface tension of β-lactoglobulin in aqueous solution at pH 6.5 and 298.15 K is presented.
The experimental data were used to calculate the surface pressure and were adjusted to different protein adsorption models at the liquid-air interface to explain the behavior of β-lactoglobulin in aqueous solution. The results show that the alcohols produce a significant effect on the adsorption behavior of the protein at the interface that is related to the number of hydroxyl groups.
Key words: β-lactoglobulin, 1-butanol; 1,2-butanediol; 1,2,3,4-butanetetrol, surface tension, adsorption.
RESUMEN
En este trabajo se presenta un estudio sistemático del efecto de 1-butanol, 1,2-butanodiol y 1,2,3,4-butanotetrol (eritritol) sobre la tensión superficial de la β-lactoglobulina en solución acuosa a 298,15 K.
Los datos experimentales fueron usados para calcular la presión superficial y se ajustaron a distintos modelos de adsorción en la interfase líquido-aire para explicar el comportamiento de la β-lactoglobulina en solución acuosa. Los resultados muestran que los alcoholes tienen un efecto significativo en el proceso de adsorción de la proteína en la interfase, relacionado con el número de grupos hidroxilo del alcohol.
Palabras clave: β-lactoglobulina, 1-butanol; 1,2-butanodiol; 1,2,3,4-butanotetrol; tensión superficial, adsorción.
RESUMO
Neste trabalho, um estudo sistemático sobre o efeito do 1-butanol, 1,2-butanodiol e 1,2,3,4-butanotetrol (eritritol) sobre a tensão superficial da β-lactoglobulina em soluçãoaquosa em pH 6,5 e 298,15 K é apresentado. Os dados experimentais foram utilizados para calcular a pressão de superfície e foram ajustados para diferentes modelos de adsorção de proteínas na interface líquido-ar para explicar o comportamento de β-lactoglobulina em soluçãoaquosa. Os resultados mostram que os alcoóisproduzem um efeito significativo sobre o comportamento de adsorção da proteína na interface que está relacionado com o número de grupos hidroxila.
Palavras-chave: β-lactoglobulina, 1-butanol; 1,2-butanodiol; 1,2,3,4-butanotetrol; tensão superficial; adsorção.
INTRODUCTION
Protein stability is a consequence of a delicate balance between intermolecular interactions of the protein with solvent molecules, which are dominant at high dilution and intramolecular interactions between the functional groups of the protein. While most researchers agree that the hydrophobic effect plays a key role in stabilizing proteins, there is not a definite explanation concerning whether and to what extent a given type of interaction determines the native conformation of a protein (1-3).
Several proposals have been presented to explain the effect of cosolvents on protein stability. Between them, some authors propose that the stabilizing effect of osmolytes such as polyols and sugars is a consequence of the increase in the surface tension of the solvent (1-3), based on experimental results that suggest that, with few exceptions, additives that increase the surface tension of water also stabilize proteins (4). However other studies do not show a linear dependence of denaturation temperatures with the increase in the solvent surface tension (5,6) and the correlation between thermal stabilization and the change in surface tension of the solvent remains to be an unresolved problem that requires the knowledge of the adsorption characteristics of proteins and is worth of systematic research using well characterized model macromolecules.
β-Lactoglobulin has been considered a model globular protein (7-13). It is one of the most investigated proteins and its structural and dynamic properties have been extensively studied. β-Lactoglobulin is one of the main whey proteins and has several applications in food and pharmaceutical industries. In its native state is a globular protein (7-10) with a molecular weight of 18362 g mol-1, 162 amino acid residues, two disulfide bonds, and an isoelectric point around 5.2. X Ray diffraction and NMR studies show that it is a predominantly β-sheet protein; the secondary structure of this protein consists of 9 strands of beta structure, an α -helix segment and three helicoidal turns (11-14). It has been reported that between pH 5.2 and 7.5 b-lactoglobulin exists in a dimeric form while under pH 3.0 and above pH 8.0 the protein exists as a monomer (15). However, other results clearly show that near neutral pH the monomeric form of b-lactoglobulin is dominant (16).
Several studies on the influence of alcohols and polyols on b-lactoglobulin thermal stability have been developed. The results show that polyols improve the conformational stability of proteins while alcohols induce protein denaturation and the effect decreases as the number of OH groups increases (16-18).
Proteins are surface active substances that lower surface tension of water and tend to adsorb at the water-air interface. Adsorption of β-lactoglobulin in aqueous solution has been studied in buffers (17,19,20) and in the presence of different cosolvents such as denaturanting agents (20), polysaccharides (21), and polyols (22). The effect of sugars has been explained as a consequence in the increase in the surface tension of water considering it the major factor in the stabilization of proteins. In the case of polyols, some authors suggest that surface tension of water does not has a major effect in protein stabilization and other effects such as preferential hydration and solvophobic effects are considered to be the responsible for protein stabilization (23-24) while other studies suggest that the surface tension of water has a fundamental role (25).
Protein adsorption is a complex dynamic process that is affected by protein structure, intermolecular forces between the adsorbed molecules and the solvent, solute-solute and solvent-solvent interactions and the presence of other substances (25-29). Dynamic surface tension is very sensitive to adsorption. As the protein is adsorbed at the liquid-air interface, the surface tension decreases until the equilibrium value is attained. Thus the process can be followed from time evolution of surface pressure Π defined as the difference between the surface tension of the protein solution γ and the surface tension of the solvent γ0:
Adsorption involves different processes before equilibrium surface tension is attained. Two empirical models have been proposed to describe protein adsorption. One of the models involves two kinetic surface tension regimes and the other three regimes, being the first step in both of them the diffusion of the protein from the bulk toward the interface. Additionally, in some cases an induction period is observed in which the surface tension remains nearly equal to that of the pure solvent.
The diffusion controlled step is usually represented by equation (21, 30-33):
where k is the Boltzmann constant, C0 is the protein concentration in the bulk, D is the diffusion coefficient of the protein in the solvent, T is the absolute temperature, A is a constant and t is the drop lifetime at which the surface pressure p is measured.
After some time, protein concentration at the interface increases and the rate of the process is controlled by the adsorption and rearrangement of adsorbed molecules. At this stage, the exposure of some part of the protein to the air can produce reorientation and conformational changes in the protein. This process can be represented by one or two steps that are usually represented by semi empirical first-order equations (30, 34).
where πf , π0 and π are the surface pressures at the final adsorption time of each step, at the initial time t0 and at any time t of drop formation, and k is the first-order kinetic constant.
For small globular proteins at low concentration, adsorption at the air-water interface usually follows a two steps model: Diffusion and rearrangement. However, the adsorption rate depends also on the nature of the solvent (pH, cosolvents) and factors such as temperature and pressure, so the adsorption model has to be determined from experimental behavior of the protein.
In the present work, the influence of the cosolvents 1-butanol, 1,2-butanediol and 1,2,3,4-butanetetrol (erythritol) on the dynamic surface tension of β-lactoglobulin in aqueous solution at 298.15 K and pH 6.5 is considered. Time evolution of surface pressure is used to analyze the effect of the number of hydroxyl groups of the alcohols on the adsorption dynamics of the protein.
MATERIALS AND METHODS
The materials used in this work were the following: β-lactoglobulin 90 % (Sigma),1-butanol ≥ 99.5 % (Merck), 1,2-butanediol 98 % (Aldrich), and 1,2,3,4-butanetetrol (erythritol) ≥99 % (Sigma). Water content of the alcohols and liquid polyols was determined by the Karl Fischer's method, and they were degassed before used. Water was doubly distilled, treated according to literature (34), and degassed before used to obtain water with conductivity lower than 2 mS.cm-1.
Solutions were prepared by weight using a Mettler balance AT 201 with sensitivity of 10−5 g in the lower range. Polyol solutions were prepared in molar fractions xOH of 0.005; 0.010; 0.015, and 0.020 but for butanol the highest molar fraction used was 0.015 due to its low solubility. Solutions of b-lactoglobulin (BLG) with mass fractions wBLG of 9.86 .10-5 and 6.39.10-5 were prepared dissolving the protein in the aqueous solutions. The final pH in all cases was 6.5.
Surface tension g measurements were determined using a LAUDA TVT2 drop volume tensiometer based on the principle of the pending drop volume, with temperature control better than 0.01 K and uncertainty of + 0.1 mN'm−1 in surface tension. The volume of the syringe used for the measurements was 2.5 mL and the tip used has an external diameter of 1.395 mm. Surface tension of a solution of β-lactoglobulin in water of mass fraction 9.86 .10-5 and the aqueous polyol solutions were measured using the dynamic method with a time of drop formation of 60 s and reported values are the average of 18 to 20 measurements. The surface tension of b-lactoglobulin with mass fractions of 9.86.10-5 and 6.39.10-5 in the aqueous solvents was measured using the quasi-static method (35-37). The surface tension value corresponds to the average of at least three independent measurements.
Density of solutions was measured using an Anton Paar vibrating tube densimeter DSA 5000 calibrated using dry air and distilled water at 298.15K + 0.01 K. The uncertainty in density measurements is + 5.10-6 g.cm-3.
RESULTS AND DISCUSSION
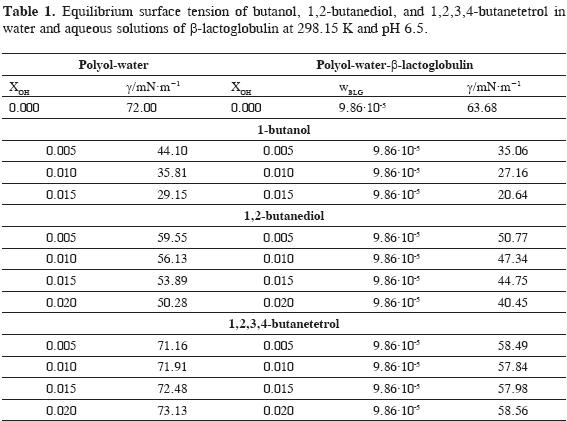
Experimental data obtained in this work for equilibrium surface tension g at 298.15 K of aqueous solutions of 1-butanol, 1,2-butanediol and 1,2,3,4-butanetretol as a function of mole fraction xOH, and the values for aqueous solutions of polyols in the presence of b-lactoglobulin at 9.84.10-5 mass fraction wBLG are presented in Table 1.
The results obtained for surface tension of butanol and 1,2,3,4-butanetetrol in water agree well with literature data (36, 38, 39) but the value reported for 1,2-butanediol is lower than the results obtained in this work (39).
From the experimental data it can be observed that 1-butanol and 1,2-butanediol lead to a decrease in the surface tension of water being the larger change observed with butanol. The addition of 1,2,3,4-butanetetrol induces a very small change in surface tension and shows a complex behavior that does not follow a clear trend.
The results in Table 1 show the effect of β-lactoglobulin at 9.86.10-5 mass fraction on the equilibrium surface tension of the aqueous solutions of polyols. As expected, the protein lowers the surface tension of water indicating that protein molecules tend to adsorb at the interface due to its amphiphilic nature giving as result a positive surface excess. The decrease in the surface tension is larger in the presence of 1-butanol followed by 1,2-butanediol while 1,2,3,4-butanetetrol produces a small change in surface tension and shows a complex behavior that does not follow a clear trend. A similar behavior has been reported for b-lactoglobulin in the presence of sorbitol (22).
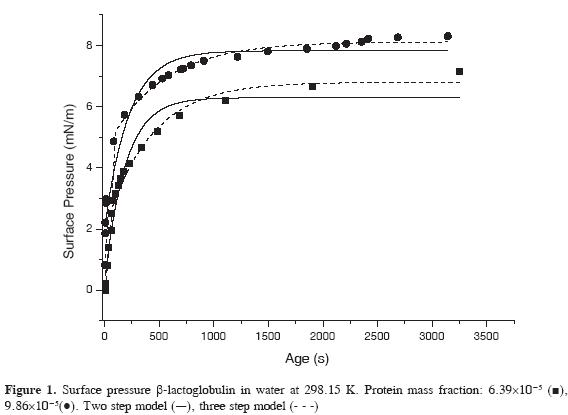
Protein adsorption was followed measuring the change of surface tension as a function of time using two different mass fractions of protein: 9.86.10-5 and 6.39.10-5, respectively. The experimental protein adsorption profiles were adjusted to the two and three steps models using the Origin® software.
The results are shown in Figure 1 for the selected protein concentrations. Surface pressure increases as protein concentration becomes larger and induction period is not observed before the adsorption process. The time dependence of the change in surface pressure depends on protein concentration and using dynamic surface measurements it has been shown that for low concentrations of β-lactoglobulin an induction period is observed while it is not present at higher protein concentrations (40, 41).
Experimental data were fitted to the two general models and they are well described by a three step model. After a rapid diffusion step the rate of β-lactoglobulin adsorption is controlled by the penetration and by rearrangement and partial unfolding of the protein at the interface. The diffusion controlled step is too rapid to be measured accurately using the quasi-static method (less than 30 s) and the other two steps follow a logarithmic behavior.
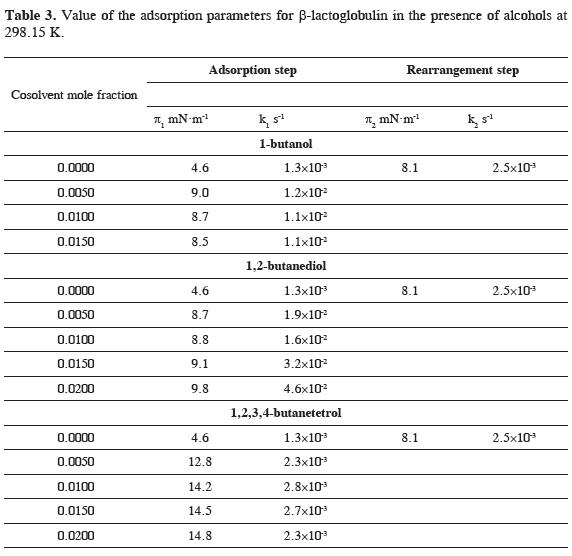
Table 2 shows the adsorption parameters of the two logarithmic steps of the model. π1 and k1 are the final pressure and the kinetic constant of the penetration process and π2 and k2 are the final pressure and the kinetic constant associated to the protein rearrangement at the liquid-air interface.
The results presented reveal that the constants for the penetration and for the rearrangement steps are nearly the same for the two protein concentrations and that rate of the penetration process is much higher than the rate of rearrangement of β-lactoglobulin at the liquid-air interface.
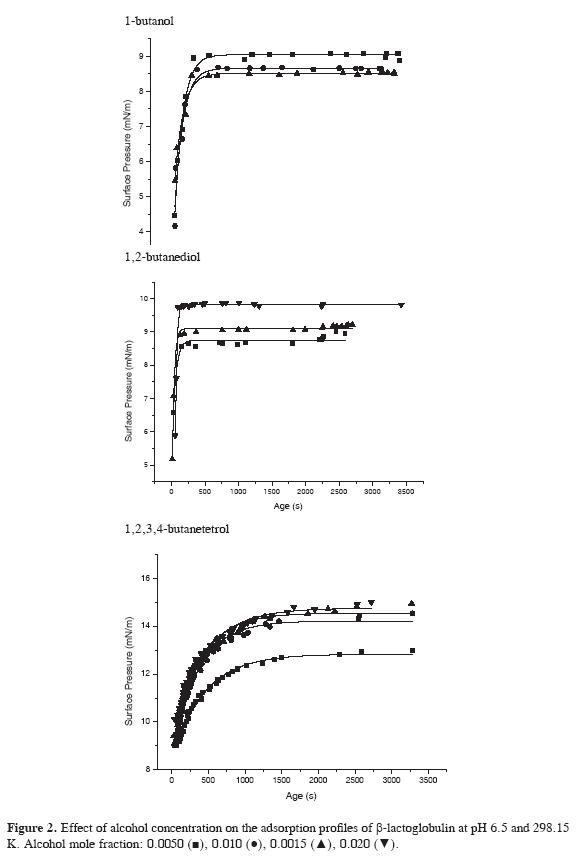
Adsorption at the air-water interface in the presence of alcohols was determined following the change of surface tension as a function of time for solutions of β-lactoglobulin of mass fraction of 9.86.10-5 in the aqueous solvents at 298.15 K and pH 6.5. The effect of alcohol concentration on the adsorption behavior of the protein is shown in Figure 2. The presence of alcohols increases surface pressure when compared with the behavior of the protein in water and the effect is larger as alcohol concentration increases. The change in surface pressure for polyols depends clearly on the number of OH groups. The largest increase is observed with 1,2,3,4-butanetetrol followed by 1,2-butanediol and the smallest change is observed in the presence of 1-butanol.
The experimental protein adsorption profile in the presence of 1-butanol, 1,2-butanediol and 1,2,3,4-butanetetrol was adjusted to the two and three steps models. In the presence of alcohols, the experimental data fitted well the two steps model, as it is shown in Figure 2 indicating that the three alcohols induce an important modification in the adsorption behavior of the protein. The diffusion controlled step is fast and occurs below the lowest experimental time measured as for the protein in water. After the diffusion stage, the observed behavior shows that adsorption of β-lactoglobulin is controlled by penetration of β-lactoglobulin at the liquid-air interface. The absence of the rearrangement regime suggests that the conformational changes in protein structure are very rapid and depend on the nature of the cosolvent. Table 3 shows the adsorption parameters of the two steps model.
The equilibrium surface pressure π1 for the penetration step increases with the addition of butanol and butanediol being the largest change observed in the presence of 1,2,3,4-butanetetrol. This behavior shows that the increase in surface pressure is largest for the more hydrophilic and non surface active cosolvent, while for the hydrophobic and surface active compounds butanol and butanediol, the change is smaller. In the case of butanol, after an initial increase, the surface pressure tends to become lower as the concentration of the cosolvent increases.
The kinetic constants follow a different behavior with cosolvent concentration. The presence of butanol induces a fast initial increase of k1 and at higher concentration its value remains constant and the kinetic constant for butanediol becomes larger as concentration increases. This behavior suggests that protein conformation changes are faster in the presence of butanol, which has a strongest denaturating effect. 1,2,3,4-butanetetrol causes a very small change of the constant indicating that the adsorption regime of the protein is not affected by the polyol because it is preferentially excluded from the liquid-air interface and does not induce changes in protein conformation.
CONCLUSIONS
In the present work, a systematic study on the effect of 1-butanol, 1,2-butanediol and 1,2,3,4-butanetetrol on the surface tension of β-lactoglobulin in aqueous solution at pH 6.5 and 298.15 K is presented. The protein lowers the surface tension of water indicating that protein molecules adsorb at the interface giving as result a positive surface excess. The decrease in the equilibrium surface tension is larger in the presence of 1-butanol followed by 1,2-butanediol while 1,2,3,4-butanetetrol produces a small change.
The adsorption profile of b-lactoglobulin in water, determined from the dynamic surface pressure, is well described by a three step model. An initial and rapid diffusion step followed by the penetration regime and the rearrangement and partial unfolding step of the protein at the interface. The presence of the alcohols induces important changes in the adsorption behavior of the protein. The diffusion controlled step is fast as for the protein in water and after the diffusion stage, adsorption of β-lactoglobulin is controlled by penetration of β-lactoglobulin at the liquid-air interface. The absence of the rearrangement regime suggests that if the alcohols induce conformational modifications in protein structure, they are very rapid. The adsorption behavior of the protein suggests that protein conformation changes are faster in the presence of butanol, while 1,2,3,4-butanetetrol does not induce changes in protein native structure.
ACKNOWLEDGMENTS
This work was supported by Grant DIB-9315 from Universidad Nacional de Colombia.
REFERENCES
1. Zweifel, M.E.; Barrick, D. Relationships between the Temperature Dependence of Solvent Denaturation and the Denaturant Dependence of Protein Stability Curves. Biophys. Chem. 2002. 101-102: 221-237. [ Links ]
2. Cooper, A. Thermodynamics of Protein Folding and Stability. En: Protein: A Comprehensive Treatise. Vol. 2, Stamford, JAI Press Inc. 1999. pp. 217-270. [ Links ]
3. Scharnagl, C.; Reif, M.; Friedrich, J. Stability of Proteins: Temperature, Pressure and the Role of the Solvent. Biochim. Biophys. Acta. 2005. 1749 (2): 187-213. [ Links ]
4. Lin, T. Y.; Timasheff, S. N. On the Role of Surface Tension in the Stabilization of Globular Proteins.Protein Sci. 1996. 5 (2): 372-381. [ Links ]
5. Romero, C. M.; Albis, A. Influence of Polyols and Glucose on the Surface Tension of Bovine α-Lactalbumin in Aqueous Solution. J Solution Chem. 2010. 39 (12): 1865-1876. [ Links ]
6. Auton, M.; Chris, A.; Ferreon, M.; Bolen, D. W. Metrics that Differentiate the Origins of Osmolyte Effects on Protein Stability: A Test of the Surface Tension Proposal. J. Mol. Biol. 2006. 361 (5): 983-992. [ Links ]
7. Forge, V., Hoshino, M; Kuwata, K.; Arai, M.; Kuwajima, K.; Batt, C. A.; Goto, Y. Is Folding of β-Lactoglobulin Non-hierarchic? Intermediate with Native-like b -Sheet and Non-native a-Helix. J. Mol. Biol. 2000. 296 (4): 1039-1051. [ Links ]
8. D'Alfonso, L.; Collini, M.; Baldini, G. Does β ;-Lactoglobulin Denaturation Occur via an Intermediate State? Biochem. 2002. 41 (1): 326-333. [ Links ]
9. Busti, P.; Gatti, C. A.; Delorenzi, N., Thermal Unfolding of Bovine b -Lactoglobulin Studied by UV Spectroscopy and Fluorescence Quenching. Food Res. Int. 2005. 38 (5): 453-550. [ Links ]
10. Jung, Y.M.; Czarnik-Matusewicz, B.; Ozaki, Y. Two-Dimensional Infrared, Two-Dimensional Raman, and Two-Dimensional Infrared and Raman Heterospectral Correlation Studies of Secondary Structure of beta-Lactoglobulin in Buffer Solutions. J. Phys. Chem. B. 2000. 104 (32): 7812-7817. [ Links ]
11. Qin, B. Y.; Bewley, M. C.; Creamer, L. K.; Baker, H. M.; Baker, E. N.; Jameson, G. B. Structural Basis of the Tanford Transition of Bovine, β-lactoglobulin. Biochem., 1998. 37 (40): 14014-14023. [ Links ]
12. Iametti, S.; Scaglioni, L.; Mazzini, S.; Vecchio, G.; Bonomi, F. Structural Features and Reversible Association of Different Quaternary Structures of beta-Lactoglobulin. J. Agric. Food Chem. 1998. 46 (6): 2159-2166. [ Links ]
13. Manderson, G.A.; Creamer, L.K.; Hardman, M.J. Effect of Heat Treatment on the Circular Dichroism Spectra of Bovine beta-Lactoglobulin A, B, and C. J. Agric. Food Chem. 1999. 47 (11): 4557-4567. [ Links ]
14. Apenten, R.K.O.; Khokhar, S.; Galani, D. Stability Parameters for β-Lactoglobulin. Thermal Dissociation and Unfolding in Phosphate Buffer at pH 7. Food Hydrocolloids. 2002. 16 (2): 95-103. [ Links ]
15. Lozano, J.M.; Giraldo, G.I.; Romero, C.M. An Improved Method for Isolation of β-Lactoglobulin. Int. Dairy J. 2008. 18 (1): 55-63. [ Links ]
16. Romero, C.M.; Lozano, J.M.; Giraldo, G.I.; Sancho, J. Thermal Stability of β-Lactoglobilin in Presence of Aqueous Solutions of Alcohols and Polyols. Int. J.Biol. Macromol. 2007. 40 (5): 423-428. [ Links ]
17. Suttiprasit, F.; Krisdahasima, V.; McGuire, J. The Surface Activity of α-Lactalbumin, β-Lactoglobulin, and Bovine Serum Albumin: I. Surface Tension Measurements with Single-component and Mixed Solutions. J. Colloid Interface Sci. 1992. 154 (2): 316-326. [ Links ]
18. Herskovits, T.T.; Gadegbeku, B.; Jaillet, H. On the Structural Stability and Solvent Denaturation of Proteins: I. Denaturation by the Alcohols and Glycols. J. Biol. Chem. 1970. 245 (10): 2588-2598. [ Links ]
19. Waniska, R.; Kinsella, J.E. Surface Properties of beta-Lactoglobulin: Adsorption and Rearrangement during Film Formation. J. Agric. Food Chem, 1985. 33 (6): 1143-1148. [ Links ]
20. Bramanti, E.; Allegrini, C.; Onor, M.; Raspi, G.; Skogerboe, K.J.; Synovec, R.E. Flow Injection Analysis with Diode Array Absorbance Detection and Dynamic Surface Tension Detection for Studying Denaturation and Surface Activity of Globular Proteins. Anal. Biochem. 2006. 351 (1): 100-113. [ Links ]
21. Baeza, R.; Carrera-Sanchez, C.; Pilosof, A.M.R.; Rodríguez-Patino, J.M. Interactions of Polysaccharides with β-Lactoglobulin Adsorbed Films at the Air-Water Interface. Food Hydrocolloids. 2005. 19 (2): 239-248. [ Links ]
22. Chanasattru, W.; Decker, E.A.; McClements, D.J. Impact of Cosolvents (Polyols) on Globular Protein Functionality: Ultrasonic Velocity, Density, Surface Tension and Solubility Study. Food Hydrocolloids. 2008. 22 (8): 1475-1484. [ Links ]
23. Gerlsma, S.Y. The Effects of Polyhydric and Monohydric Alcohols on the Heat Induced Reversible Denaturation of Chymotrypsinogen A. Eur. J. Biochem. 1970. 14 (1): 150-153. [ Links ]
24. Timasheff, S.N. The Control of Protein Stability and Association by Weak Interactions with Water: How Do Solvents Affect These Processes? Annu. Rev. Biophys. Biomol. Struct. 1993. 22: 67-97. [ Links ]
25. Kaushik, J.; Bhat, R. Thermal Stability of Proteins in Aqueous Polyol Solutions: Role of the Surface Tension of Water in the Stabilizing Effect of Polyols. J. Phys. Chem. B. 1998. 102 (36): 7058-7066. [ Links ]
26. Sadana, A. Protein Adsorption and Inactivation on Surfaces. Influence of Heterogeneities. Chem. Rev. 1992. 92 (8): 1799-1818. [ Links ]
27. Sengupta, T.; Razumovsky, L.; Damodaran, S. Energetics of Protein-Interface Interactions and Its Effect on Protein Adsorption. Langmuir 1999. 15 (20): 6991-7001. [ Links ]
28. Yampolskaya, G.; Platikanov, D. Proteins at Fluid Interfaces: Adsorption Layers and Thin Liquid Films. Adv. Coll. Interface Sci. 2006. 128-130: 159-183. [ Links ]
29. Madgassi, S. Surface Activity of Proteins: Chemical and Physicochemical Modifications. New York, Marcel Dekker Inc. 1996. [ Links ]
30. Guzey, D.; McClements, D.J.; Weiss, J. Adsorption Kinetics of BSA at Air-Sugar Solution Interfaces as Affected by Sugar Type and Concentration. Food Res. Int. 2003. 36 (7): 649-660. [ Links ]
31. Rao, C.S.; Damodaran, S. Is Surface Pressure a Measure of Interfacial Water Activity? Evidence from Protein Adsorption Behavior at Interfaces. Langmuir. 2000. 16 (24): 9468-9477. [ Links ]
32. Alvarez, J.M.; Pizones, V.; Carrera, C.; Rodríguez, J.M. The Role of Static and Dynamic Characteristics of Diglycerol Esters and β-Lactoglobulin Mixed Films Foaming. 1. Dynamic Phenomena at the Air-Water Interface. Food Hydrocolloyds. 2008. 22 (6): 1105-1116. [ Links ]
33. Ward, A.F.H.; Torday, L. Time-dependence of Boundary Tensions of Solutions I. The Role on Diffusion in Time-effects. J. Chem. Phys. 1946. 14 (7): 453-461. [ Links ]
34. Weisseberger, A.; Rossiter, B.W. Physical Methods of Chemistry. New York, Wiley-Interscience. 1972. Vol. 4. [ Links ]
35. Medrano, A.; Abirached, C.; Panizzolo, L.; Moyna, P.; Añón, M.C. The Effect of Glycation on Foam and Structural Properties of β-Lactoglobulin. Food Chem. 2009. 113 (1): 127-133. [ Links ]
36. Razafindralambo, H.; Blecker, C.; Delhaye, S.; Paquot, M. Application of the Quasi-static Mode of the Drop Volume Technique to the Determination of Fundamental Surfactant Properties. J. Colloids Interface Sci. 1995. 174 (2): 373-377. [ Links ]
37. Dunning, H.N.; Washburn, E.R. Diffusion Coefficients and Some Related Properties of the Butyl Alcohols in Aqueous Solutions. J. Phys. Chem. 1952. 56 (2) 235-237. [ Links ]
38. Habrdov., K.; Hovorka, S.; Bartovsk., L. Concentration Dependence of Surface Tension for Very Dilute Aqueous Solutions of Organic Nonelectrolytes. J. Chem. Eng. Data. 2004. 49 (4): 1003-1007. [ Links ]
39. Hawrylak, B.; Andrecyk, S.; Gabriel, C.; Gracie, K.; Palepu, R. Viscosity, Surface Tension, and Refractive Index Measurements of Mixtures of Isomeric Butanediols with Water. J.Solution Chem. 1998. 27 (9): 827-841. [ Links ]
40. Krägel, J.; Wüstneck, R.; Clark, D.; Wilde, P.; Miller, R. Dynamic Surface Tension and Surface Shear Rheology Studies of Mixed β-Lactoglobulin/Tween 20 Systems. Colloids and Surfaces A: Physicochemical and Engineering Aspects. 1995. 98 (1-2): 127-135. [ Links ]
41. Wüstneck, R.; Krägel, J.; Miller, R.; Fainerman, V.B.; Wilde, P.J.; Sarker, D.K.; Clark, D.C. Dynamic Surface Tension and Adsorption Properties of β-Casein and β-Lactoglobulin. Food Hydrocolloids. 1996. 10 (4): 395-405. [ Links ]