Services on Demand
Journal
Article
Indicators
 Cited by SciELO
Cited by SciELO  Access statistics
Access statistics
Related links
 Cited by Google
Cited by Google  Similars in
SciELO
Similars in
SciELO  Similars in Google
Similars in Google
Share
DYNA
Print version ISSN 0012-7353
Abstract
PAEZ, MANUEL; LANS, EDINELDO and CANTERO, PLINIO. DENSITIES AND VOLUMETRIC PROPERTIES OF THE MIXTURE N,N-DIMETHYLFORMAMIDE (DMF) + 1-BUTANOL AT SEVERAL TEMPERATURES. Dyna rev.fac.nac.minas [online]. 2013, vol.80, n.177, pp.132-141. ISSN 0012-7353.
Densities of binary mixture of N, N-dimethylformamide (DMF) + 1-Butanol were determined using a vibrating-tube densimeter DMA 5000, over the entire range of molar fractions in the range temperatures from 283.15 K to 313.15 K and 1.011 bar. Excess molar volumes (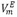 ), partial molar volumes (
), partial molar volumes (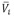 ), partial molar volumes at infinite dilution (
), partial molar volumes at infinite dilution (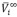 ), excess partial molar volumes (
), excess partial molar volumes (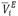 ) of the solute and solvent were calculated. The excess molar volumes were correlated using the Redlich-Kister equation. Additionally, the virial coefficients (bv) were calculated according to the McMillan-Mayer theory. The values obtained for these parameters were discussed in terms of the interactions present in solution.
) of the solute and solvent were calculated. The excess molar volumes were correlated using the Redlich-Kister equation. Additionally, the virial coefficients (bv) were calculated according to the McMillan-Mayer theory. The values obtained for these parameters were discussed in terms of the interactions present in solution.
Keywords : Density; Excess molar volumes; Interactions.













