As signal-transmitting and effector molecules, proteins play a leading role in any biological process, including follicular development and competent oocyte formation. During follicular development, proteins are associated with signal transmission and the induction of molecule production to express behaviors related to fertilization and early embryo development processes (Ishak et al. 2022). However, protein expression and reproductive parameters vary dramatically between species, even though they are phylogenetically close, such as cattle and buffalo, both classified as bovids. Reproductively, buffaloes have been reported to have a later puberty, shorter estrous cycles, and a longer gestational period than cows (Gimenes et al. 2015). Some proteins have been characterized and associated with these aspects (Valckx et al. 2015). However, very few studies have focused on studying them within the physiological and comparative contexts between species, allowing the proposal of theories about their role in reproductive parameters.
In this sense, there are several proteins in follicular fluid (FF) that are related to aspects such as nuclear and cytoplasmic maturation of the oocyte, parameters that are specific to each species and may vary according to the time of the estrous cycle (Filipiak et al. 2016). Evaluating these variations is the basis for searching for potential oocyte and follicle quality markers. The ovary has two functions: 1) an endocrine function associated with the production of steroidal hormones and proteins and 2) another related to the production of gametes. The second is carried out by two mechanisms: oogenesis and folliculogenesis (Valckx et al. 2015). Folliculogenesis begins during fetal life, becoming active around day 140 of gestation in cattle. The follicle grows from 25-30 µm to reaching about 15 mm at ovulation in a continuous growth process. However, not all of them grow. Most of them suffer from atresia during their reproductive life. The follicle is composed of the oocyte and the granulosa cells surrounding it; during development, these multiply and become the cells that produce the FF, which will be accumulated in a structure called antrum (Fair 2003). Follicular deviation has been defined as the moment during the growth of a follicle cohort when one acquires more LH receptors. This follicle continues its development towards ovulation; meanwhile, all others within the cohort that have not been chosen regress. Studies in buffalo have shown that follicular deviation occurs when follicles are around 7 mm. It is assumed that this size may define two moments during development and, thus, the physiology of the follicle (Gimenes et al. 2011).
The FF is an exudate of blood plasma modified by the metabolic activity of granulosa cells and possesses different proteins, glycoproteins, glycosaminoglycans, and steroids (Gordon 2003). The composition is variable and depends on the physiological state of the follicle and the time of the estrous cycle of an individual. Its composition has been reported as being affected by environmental conditions and the age of the animal (Iwata 2017). In the FF, there are proteins such as albumin, polypeptides, and lysosomal enzymes, in addition to ions, ascorbic acid, and steroids, including estradiol and progesterone. Furthermore, there are also gonadotropins, such as LH, FSH, alpha and dimeric inhibins (between 34 kDa and >160 kDa), prolactin, high-density proteins, glycosaminoglycans, and growth factors (Neira-Rivera et al. 2020). They all compose a complex mechanism that influences follicular dynamics and contributes to oocyte maturation and growth. Schweigert et al. (2006) found in human beings some differences between FF proteins and serum proteins associated with the reproductive process, mainly because some are synthesized by the interaction between follicle components, including theca cells, granulosa, and the oocyte itself. Moreover, the authors found peptides between 6.9 and 13.8 kDA in higher concentrations in FF than in serum. Albumin and immunoglobulins, predominantly IgG, have been identified as the most abundant proteins in FF from humans, pigs, canines (Fahiminiya et al. 2010), and horses (Fahiminiya et al. 2011).
In this context, the knowledge derived from the study of FF proteins can contribute to the development of embryo production systems in species and, in this case, to the development of buffalo production systems. This study aimed to evaluate if there are differences in FF protein profiles in cow and buffalo follicles before and after the follicular deviation, as a contribution to the knowledge of the species and to the improvement of in vitro embryo production programs, especially in buffaloes.
MATERIALS AND METHODS
Animals and sampling
For this study, 25 ovaries from Bos indicus cows and 20 from Bubalus bubalis buffaloes were collected from a slaughterhouse in Medellín, Antioquia Department, Colombia. The follicles of each ovary were measured with a graduated ruler and classified according to their size as large or small, i.e., larger, or smaller than 7 mm. After measurement, the FF was aspirated with an 18-gauge needle attached to a 10 mL syringe. In large follicles, the volume obtained was sufficient for analysis, while in small ones, it was necessary to make a pool of aspirated follicles (3 to 5). The FF obtained was centrifuged at 13,000 rpm for 30 min at 4 °C, and phenylmethylsulfonyl fluoride (PMSF) dissolved in dimethyl sulfoxide (DMSO) was added as a protease inhibitor. FF samples were stored at -20 °C for subsequent analysis.
Protein quantification
The total protein concentration in FF was determined through the Bradford method (Bradford 1976) in 96-well Elisa microplates. The measurement was performed at 620 nm in a Biotec Cytation Elx 800 spectrophotometer. Then, 200 µL of FF samples were used to remove albumin and immunoglobulins, and the ProteoPrep Blue Albumin and IgG Depletion Kit from Sigma (Catalog No PROTBA-1KT) was used, according to the manufacturer’s instructions.
Electrophoresis in denaturing gels
For denaturing electrophoresis in sodium dodecyl sulfate-polyacrylamide gels (SDS-PAGE), 7 cm mini gels with an acrylamide/bis-acrylamide concentration of 15% were used according to the Laemmli method (Laemmli 1970). The weight marker Precision Plus Protein™ Dual Xtra from BioRad (Bio-Rad), with a range between 2 to 250 kDa, was used. Electrophoresis was performed in a Mini Protean II chamber (Bio-Rad) with a constant voltage of 140 V for 75 min to determine the molecular weight of the protein bands. The gels were developed by staining with 0.025% Coomassie Brilliant Blue for 2 h, 10% acetic acid, and 30% methanol destaining solution. The gels were digitized in an Image Gel Doc™ documenter (Bio-Rad). They were analyzed with the Image Lab software (Bio-Rad) to determine the molecular weights by comparison with the weight marker and the relative amounts of the bands evidenced through the measurement of the optical density in pixels obtained from the area and depth of each band.
RESULTS AND DISCUSSION
Follicle fluid was collected from the ovaries of 25 bovines and 20 buffaloes. Between 10 and 500 µL/ovary of FF were obtained. It was classified by the size of the follicle from which they were obtained. Finally, FF from 13 buffalo samples (five from large and eight from small follicles) and 16 cow samples (eight from large and eight from small follicles) were analyzed. The initial protein concentration and the one after removing albumins and immunoglobulins (depletion) were determined. Total protein concentration in FF from small and large follicles was 53.82±21.79 mg µL-1 for bovine follicles and 57.2±19.74 mg µL-1 in buffalo follicles. There were no significant differences in total protein concentration between follicles of the same size or species or follicles of different sizes for both species (P>0.05) (Figure 1A). In all samples, after the depletion of the most abundant proteins (albumin and immunoglobulins), the total protein concentration decreased by about 70% (Figure 1B). This was done to find protein bands with less concentration that were not detected when most proteins were not eliminated. However, there was no difference (P>0.05) in the number of detected bands among the samples to which the removal procedure was performed.
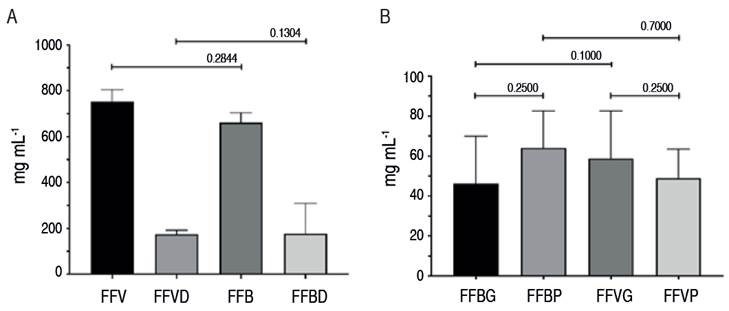
Figure 1 A. Total protein from cow (FFV) and buffalo (FFB) follicle fluid before (original) and after depletion (FFVD, FFBD, respectively). B. Total protein concentration in cows and buffaloes from big (>7 mm; FFVG, FFBG, respectively) and small follicles (<7 mm; FFVP, FFBP, respectively).
The analysis of the electrophoretic profiles shows that the undepleted FF samples from cows showed a total of 19 protein bands in the small follicles (maximum number of bands), while the buffalo FF samples had a maximum of 11 bands. Large FF from undepleted cows presented a total of 11 protein bands (maximum number of bands), while buffalo FF samples had a maximum of 13 bands. The detected bands had a molecular weight range between 5 and 250 kDa (Figure 2).

Figure 2 Electrophoretic profile of follicle fluid proteins without depletion in A. cow ovaries and B. Buffalo ovaries. Line 1 (left): Molecular weight marker (kDa); line 2 (center): Large follicles (>7 mm); line 3 (right): Small follicles (<7 mm).
All the band proteins were grouped according to their molecular weight before depletion. Buffaloes showed more bands than cows (24 vs. 31). However, there were more bands in the cow samples (32) than in those of buffaloes (22) after the depletion procedure was performed. Moreover, in all the evaluated groups, the highest amount of protein in FF corresponded to the group of less than 40 kDa, specifically in the 13/21 (cows) and 15/25 (buffaloes) groups without depletion, and in the 23/32 (cows) and 13/22 (buffaloes) groups that were depleted (Table 1 and Figure 3).

Figure 3 Comparison of uni-dimensional electrophoretic profiles of follicle fluid proteins with and without depletion. A. Cows: Line 1 Molecular Marker (MW) (kDa); lines 2 and 3>7 mm; line 4 <7 mm undepleted; lines 5 and 6>7 mm; line 7<7 mm depleted. B. Buffaloes: Line 1 MW (kDa); line 2>7 mm depleted; line 3 undepleted.
Table 2 shows the number of proteins grouped by molecular weight within the categories analyzed. The qualitative results (relative % proportion of number of bands) did not coincide with the quantitative results (proportion of each band within the total). Proteins smaller than 20 kDa were not so abundant. In contrast, those between 20 and 6.0 kDa were more abundant.
Table 2 Effect of the species and depletion procedure on the protein proportion in follicle fluid of cows (V) and buffaloes (B).

Concerning protein concentration in FF of buffaloes, the results obtained in this study (57.2±19.74 µg µL-1) are lower than those reported by Behera et al. (2016) in buffaloes in India (61.5 µg µL-1) without alterations in their estrous cycle. In Colombia, Neira-Rivera et al. (2020) reported that in cows, the average total protein concentration of FF (<3 mm) was 60.6±16.6 µg dl-1, specifically 50.6±21.0 µg dl-1 in follicles with a size between 3 to 6 mm, and 53.4±16.3 µg dl-1 in follicles of more than 6 mm. These values are similar to those obtained in this study.
In contrast, Shabankareh et al. (2013) reported a decrease in total protein concentration as follicle size increases in cows, suggesting that the cause was a dilution of the proteins due to the increase in follicle fluid volume. Other authors have reported that in buffaloes, there are no significant differences in the amount of follicle protein between cyclic (0.49±1.07 µg µL-1) and acyclic animals (0.6±0.28 µg µL-1).
After depletion, 72.3% of the total proteins present in the FF were immunoglobulins and albumin, with no statistical differences between species or the follicular sizes evaluated. Fahiminiya et al. (2010) reported that some previously undetected proteins were observed after albumin depletion. Therefore, they were not analyzed, and the authors suggested a possible bias in the analysis. Furthermore, these are associated with specific events of follicular development and their physiological role, i.e., oxidative stress and as scavengers of reactive oxygen species. Figure 1 shows that in the depleted samples, there is a decrease in the number of bands between 3 and 75 kDa, which are albumin and immunoglobulins.
For protein banding patterns, more protein bands were identified in buffalo FF samples than in cow FF before depletion (25 vs. 21). However, after depletion, 34 protein bands were found in FF from cows vs. 22 from buffaloes. Neira-Rivera et al. (2020) found 25 bands in cows, a lower number than reported in this study.
When the bands were grouped by molecular weight, 76.4% in cows and 60% in buffaloes correspond to proteins below 40 kDa (Table 2), maintaining the same ratio after depletion with 71.8 and 59.0%, respectively. Similar results can be observed when comparisons are made with data grouped by follicle size (Table 2). Notably, the relative number of bands is higher in those with molecular weights higher than 40 kDa. Fu et al. (2016) reported that the bands identified by SDS-PAGE in FF from swamp buffaloes were between 10 and 200 kDa. They managed to identify 363 proteins, of which 153 were related to some metabolic or signaling pathway. Additionally, they found 11 proteins with differential expression between large and small follicles involved in inhibiting serine and threonine proteases, oxidation, and the complement cascade. Subsequently, the authors identified some candidate proteins as molecular markers of follicle quality. Finally, the same authors compared the proteins identified in the buffalo FF with those of human serum and found that of 349 proteins identified in the buffalo FF, 217 were shared with human serum, and 132 were exclusive to the FF.
This work found differences between the number of proteins identified in the species studied and the size of the follicle from the samples. Since follicular development has the same pattern, it can be affirmed that the species studied perform the same function with different proteins and that these are responsible for the differences in follicular development or preparation for ovulation.
Since the follicle structure acts as a barrier to the diffusion of proteins larger than 100 kDa, it is possible that those found in the FF do not necessarily reflect what is happening in the bloodstream, allowing the granulosa cells to exert their function in follicle development. This has been evidenced by reports describing that the protein concentration can be up to to twice as high in healthy follicles compared to atretic follicles (Clarke et al. 2006).
Several proteins have been identified in the FF, such as follicle-stimulating hormone (30 kD), inhibin (32 kDa), some growth factors (<30 kDa), transforming factor β (7.5 kDa), fibroblast growth factors (16-17 kDa), uterine serpins (52 kDa), albumin (62-69 kDa), immunoglobulins with their two heavy chains (50-70 kDa) and two light chains (23 kDa), transferrin (78-80 kDa), complement factors (31.8 kDa), gelsolin (80 kDa), arsenic methyl transferase (42.5 kDa), vitamin D (54.9 kDa), gelatinase B (91.5 kDa), lactotransferrin (78.05), and osteopontin (45 kDa) (Neira-Rivera et al. 2020). All these proteins perform some function in the development of the follicle, ovulation, or the atresia process. Many may be included in the protein bands found in this research, but this needs to be confirmed in future works using two-dimensional gel electrophoresis and mass spectrometry. It can be evidenced that the proteins of low molecular weight are those most associated with reproductive processes, while those of higher molecular weight correspond to the basal functions of the organism.
Regarding the effect of FF proteins on reproductive function, Ghosh et al. (2005) immunized goats with protein fractions larger than 30 kDa in buffalo FF and observed a lengthening of the cycle and a delay in the estrus onset. Another aspect associated with the functions of the proteins identified in human FF, which may have a plasma origin, is that they are related to inflammation, reaffirming the theory that it assimilates the ovulatory process into an inflammatory process (Zamah et al. 2015).
CONCLUSIONS
The diversity observed among species may reflect the significant number of possibilities that nature has to exert a function and events associated with follicle growth and their consequences on oocyte maturation. This forces researchers to be careful when extrapolating results and protocols from one species to another. Quantitative and qualitative differences exist in the proteins present in the follicular fluid evaluated from cows and buffaloes, both classified as Bovidae, and their follicle sizes (large and small). This information can contribute to the explanation of the physiological differences between both species.
















