Introduction
Sulfur (S) is considered the fourth major plant nutrient along with nitrogen, phosphorus and potassium. Sulfur is important for rapeseed production, and S deficiencies frequently constrain rapeseed yield (Jan et al., 2008). Rapeseed requires about 1.5 kg of S to produce 100 kg ha-1 of seed (Kumar et al., 2002). Therefore, a 3000 kg ha4 crop would require approximately 45 kg S ha-1. To obtain optimum yields of high-quality rapeseed seed, S needs to be an important part of balanced fertilization along with other nutrients (Jackson, 2000; Malhi and Gill, 2002; Kandil and Gad, 2012; Sharifi, 2012). Sulfur is essential for the synthesis of amino acids including cystine and methionine (a component of vitamin A), and it activates special enzyme systems in plants (Balint and Rengel, 2009).
Regarding protein formation during rapeseed growth and development, S can also increase seed yield and improve oil content (Zhao et al., 1993; Jan et al., 2002; Sattar et al., 2011). Sulfur is also involved in the synthesis of chlorophyll and is also required in plants of the family Cruciferae for the synthesis of volatile oils (Marschner, 2012). Castellano and Dick (1991) find that photosynthesis-related proteins such as the Rubisco protein and chlorophyll and N and S content in leaves significantly increase with sulfur levels up to 50 kg S ha-1 compared to 0 kg S ha-1. Ahmad et al. (2000) report that sulfur application significantly increased acetyl-CoA concentration, acetyl-CoA carboxylase activity, and soluble protein and starch content in developing seeds. Plants take sulfur primarily in the form of S04 -2 by a specific transport protein (Thompson et al., 1986). Shallow soils with low organic matter content are likely to provide little sulphate (Holmes, 1980). Total chlorophyll content and peroxidase activity increases with higher sulfur levels (Khanpara et al., 1993). Abiotic stresses contribute to the formation of reactive oxygen species (ROS), superoxide radical (O2 -), hydrogen peroxide (H2O2) and hydroxyl radical (OH-), with the last one as the most cytotoxic. These ROS cause perturbation of basic metabolic pathways and damage membranes and organic molecules, mainly proteins, DNA, and pigments (Fridovich, 1986; Imlay and Linn, 1988) as well as sulfur containing amino acids in proteins (Hernandez et al., 2000). Plants use different strategies to solve this problem. For instance, one strategy is that plants increase the activity of antioxidant enzymes. The toxic superoxide radical is rapidly dismutated by superoxide dismutase (SOD) to H2O2, a product that is relatively stable and can be detoxified by catalase (CAT) and guaiacol peroxidases (Grant and Loake, 2000). Increased SOD activity is known to confer oxidative stress tolerance (Bowler et al., 1992). The balance between ROS production and activities of antioxidative enzymes determines whether oxidative signaling and/or damage will occur (Moller et al., 2007). The activities of these an-tioxidant enzymes are reported to increase under various environmental stresses (Hernandez et al, 1995; Hernandez et al., 2000). There are many reports in the literature that underline the intimate relationship between enhanced or constitutive antioxidant enzyme activities and increased resistance to environmental stresses in Brassica and other plant species (Sreenivasulu et al, 2000; Rameeh et al., 2004; Khanna-Chopra and Selote, 2007). Sulfur application will improve seed and oil quality and is also a key factor for oil formation. In the present study, four sulfur levels: 0, 12, 24 and 36 kg ha-1 along with 115 kg N ha-1 were applied to evaluate economic yield and also enzyme activity of rapeseed under rainfed conditions.
Materials and methods
A field experiment was carried out in a farm in Aben-dankash located in Sari, Iran (53°7' E, 36°32" N, 60 m a.s.l.) during the 2006-2007 cropping seasons. The soil was classified as a deep loam soil (Typic Xerofluents, USDA classification), which maintained an average of 280 g clay kg-1, 560 g silt kg-1, 160 g sand kg-1, and 22.4 g organic matter kg-1 with a pH of 7.3. Soil samples were found to have 45 kg ha-1 of mineral nitrogen (N) in the upper 30-cm profile. The experiment received 50 kg P ha-1 and 75 kg K ha-1. The average temperature was around 15.5°C and the average rainfall was 36.7 mm. Seeds of the rapeseed cultivar Hy-ola401 were planted on October 18, 2006. Seeds were sown with a uniform seed rate of 5 kg ha-1 in all plots with the help of a hand hoe in straight rows. The experiment was set in a randomized complete block design with four replicates. The treatments under study included different amounts of ammonium sulfate (containing 21% nitrogen and 24% sulfur) and urea fertilizer (containing 46% nitrogen): S0: 250 kg ha-1 urea; S1: 227 kg ha-1 urea+50 kg ha-1 ammonium sulfate; S2: 204 kg ha-1 urea + 100 kg ha-1 ammonium sulfate; and S3: 182 kg ha-1 urea + 150 kg ha-1 ammonium sulfate. S0, S1, S2 and S3 included 0, 12, 24 and 36 kg ha-1 S, respectively, and all treatments maintained 115 kg N ha-1. All cultural practices were uniformly applied to all plots. All plant protection measures were adopted to keep the crop free from insect pests. Seed yield (adjusted to kg ha-1) was recorded based on three middle rows in each plot. For enzyme assays, frozen leaves were ground to fine powder with liquid nitrogen and extracted with ice-cold 0.1 M Tris-HCl buffer (pH 7.5) containing 5% (w/v) sucrose and 0.1% 2- mercaptoethanol (3:1 buffer volume/FW). The homogenate was centrifuged at 10,000 g for 20 min, at 4°C, and the supernatant was used for enzyme activity. Regarding the enzyme assay, superoxide dismutase activity which has the ability to inhibit the phothochemical reduction of nitro-blue tetrazolium (NBT) (Beauchamp and Fridovich, 1971), was determined according to the method by Dhindsa et al. (1980). For the SOD assay, the reaction mixture contained 50 mM K-phosphate buffer (pH 7.8), 13 mM methionine, 75 µM NBT, 0.1 µM EDTA, 4µM riboflavin and the required amount of enzyme extract. The reaction was initiated by adding riboflavin and placing the tubes under two 15 W fluorescent lamps for 15 min. A complete reaction mixture without enzyme, which gave the maximal color, served as control. Peroxidase activity was assayed adopting the method by Polle et al. (1994). According to this method, Peroxidase activity was determined at 436 nm by its ability to convert guaiacol to tetraguaiacol (E = 26.6 mM-1 cm-1) The reaction mixture contained 100 mM K-phosphate buffer (pH 7.0), 20.1 mM guaiacol, 10 mM H2O2, and the enzyme extract. The increase in absorbance was recorded by the addition of H2O2 at 436 nm for 5 min. CAT activity was determined by monitoring the disappearance of H2O2 at 240 nm (E = 40 mM-1 cm-1) according to the method by Aebi (1984). The reaction mixture contained 50 mM K-phosphate buffer (pH 7.0), 33 mM H2O2 and the enzyme extract. To estimate sugar and starch contents in leaves, chopped leaves were fixed in boiling 80% ethanol. Sugar contents were estimated colorimetrically by the method described by Nelson (1994) at 500 nm. Data recorded were analyzed statistically, using analysis of variance (ANOVA) with appropriate techniques for randomized complete block design (Steel and Torrie, 1980). For the analysis, an Excel worksheet was programmed. All the analyses were performed using the software SAS version 9 (SAS Institute Inc., 2004).
Results and discussion
Significant mean square, which indicates significant different effects of sulfur levels, were found for the traits including leaf nitrate reductase, root nitrate reductase, leaf and root sugars, root peroxidase, leaf catalase, and seed yield (Tab. 1). Leaf peroxidase activity was not affected by sulfur levels. Sulfur application along with seed yield that improves oil quality is a critical factor for oil formation. Sulfur shortage adversely decreases yield as well as protein and enzyme synthesis (Scherer, 2001).
TABLE 1 Randomized complete block (RCBD) analysis of variance for the studied traits.
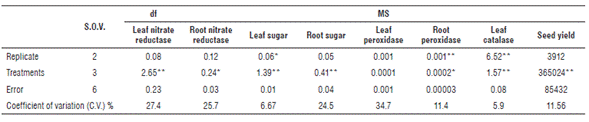
S.O.V.: source of variance, df: degree of freedom, MS: mean squares. *, ** Significant at P<0.05 and 0.01, respectively.
Means comparisons of leaf nitrate reductase as influenced by different levels of sulfur is presented in Table 2 and Figure 1. Sulfur application induced a significant increase in activity of leaf nitrate reductase. Leaf nitrate reductase activity varied from 0.82 to 2.90 mM No2- g-1 in S0 (control) and S3 (36 kg S ha-1), respectively. Mean values of leaf nitrate reductase activity were classified into two statistical groups for the application of four S levels. This trait related to S0 and S1 determined the same group, and for S2 and S3 it was also classified in the same statistical group. Ahmad et al. (2000) report that sulfur application significantly increases acetyl-CoA concentration acetyl-CoA carboxylase activity, soluble protein and starch content in developing seeds. A significant positive correlation was determined between leaf nitrate reductase activity and sugar content in leaf and root of rapeseed (Tab. 3). Therefore, any variation for this enzyme will have considerable effect on leaf and root sugar contents. Root nitrate reductase activity ranged from 0.33 to 1 mM No2 - g-1 in S0 and S3, respectively. A significant positive correlation between root nitrate reductase activity and sugar content in leaf and root of rapeseed was observed (Tab. 3).
TABLE 2 Mean comparison of yield components, seed yield and oil percentage.
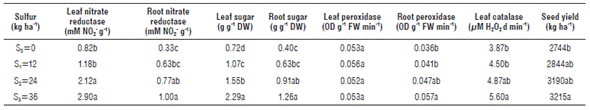
S0, S1, S2 and S3 included 0, 12, 24 and 36 Kg S ha-1, respectively, and all treatments maintained 115 Kg N ha-1.
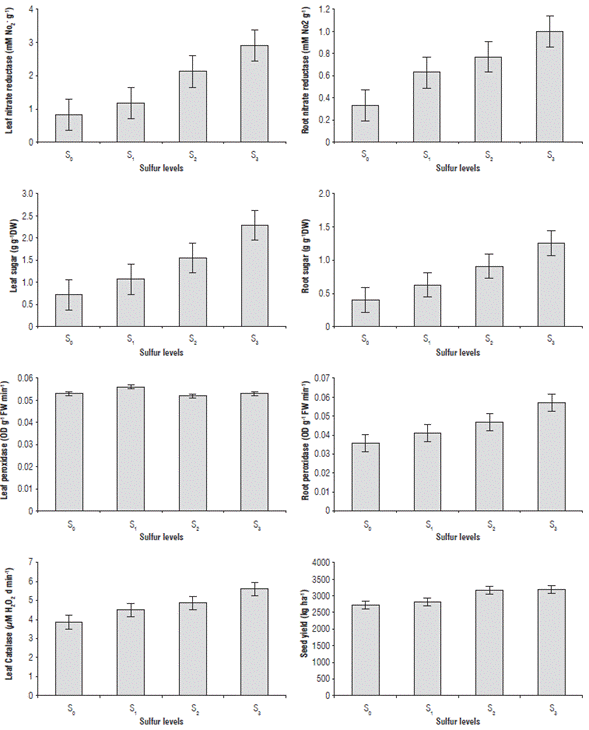
FIGURE 1 Means of plant heights, yield components, seed yields and oil contents of rapeseed var. Hyola401 under different sulfur levels (S0, S1, S2 and S3 including 0, 12, 24 and 36 kg S ha-1, respectively). Error bars indicate 95% confidence intervals.
TABLE 3 Pearson correlation coefficient estimates of biochemical traits and seed yield in different levels of sulfur used on a rapeseed variety.
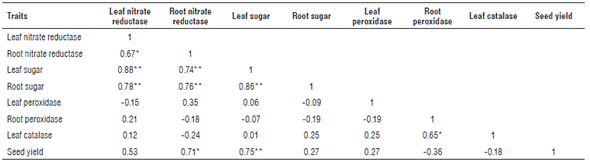
*, ** Significant at P<0.05 and 0.01, respectively.
Due to a significant positive correlation between activity of root nitrate reductase and seed yield, increasing this enzyme in the roots followed by sulfur application will have considerable effect on rapeseed seed yield. Leaf sugar was positively affected by sulfur levels, and it varied from 0.72 to 2.29 g g-1 DW for S0 and S3, respectively. A highly significant positive correlation (0.75**) was determined between leaf sugar content and seed yield; therefore, increasing sulfur levels had a direct increasing effect on_jeaf sugar content, which had an escalating effect on seed yield. Root sugar content was positively affected by sulfur levels and changed from 0.40 to 1.26 g g-1 DW for S0 and S3, respectively (Tab. 2). Sulfur levels had an increasing effect on root peroxidase activity and this enzyme ranged from 0.036 to 0.057 OD g-1 FW min-1 in roots. Leaf catalase activity was significantly affected by sulfur levels, and the mean value of this enzyme activity varied from 3.87 to 5.60 µM H2O2 dmin-1 for S0 and S3. Bashir et al. (2015) indicate that the activity of ascorbate peroxidase, glutathione reductase and catalase declined in Cd-treated and S-deficient plants, but it was upregulated in the presence of sulfur.
Sulfur application significantly increased seed yield compared to control (S0 level) and it ranged from 2744 to 3215 kg ha-1 in S0 and S3 (Tab. 3). S3 (36 kg ha-1 S) increased seed yield on 17%. Sulfur application improves seed yield quantity and oil quality and also sulfur shortage adversely decreases yield, protein and enzyme synthesis (Scherer, 2001; Rehmanuh et al., 2013).
Conclusion
All the traits except leaf peroxidase activity were significantly affected by sulfur levels. A significant positive correlation between leaf nitrate reductase and sugar content in rapeseed leaves and roots was found. Therefore, any variation of this enzyme will have considerable effect on leaf and root sugar contents. Due to a significant positive correlation between root nitrate reductase activity and seed yield, increasing this enzyme in roots by sulfur application will have a considerable effect on rapeseed seed yields. Leaf sugar was positively affected by sulfur levels and its high mean value was observed at level S3. A highly significant positive correlation determined between leaf sugar content and seed yield (0.75**) was observed. Thus, increasing sulfur levels had a direct increasing effect on leaf sugar content, which had an accelerating effect on seed yield. Sulfur application significantly increased seed yield compared to S0 level, and it ranged from 2744 to 3215 kg ha-1 in S0 and S3.














