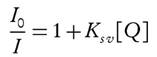Introduction
The 1-hydroxypyrene-3,6,8-trisulfonic acid (HPTS) is a fluorescent dye which contains three sulfate groups (Fig. 1) and has been used as an apoplastic marker (Wang et al. 1994, Wright et al. 1996).It is well known that its physiological pH HPTS would be highly charged thus preventing the entry to the cell and decreasing its range of applications. The pH detection at the intracellular level has become a challenge for the scientific community because it is a significant variable in understanding the action mechanism of different types of drugs within a specific c ell. The study of the interactions between intracellular dyes and nanostructures is of crucial interest to understand the nature of the interaction and thus develop new types of optical biosensors. Recently, nanotechnology has provided different nanostructures to functionalize many biomolecules to facilitate the entry into the cell (Baties et al. 2013).
Carbon nanotubes (CNTs) are unique tubular structures with diameters in the range of nanometers and large diameter/length ratio which have attracted the attention of researches due to the numerous applications in the biomedical field (Liu et al. 2009, Wang et al. 2009; Xing et al. 2010). One of the advantages possessed by the CNTs is the possibility of effectively of crossing biological membranes. In an elegant study, it was shown that the ability of different types of CNTs to penetrate the cell and how this ability can be exploited for the use in a variety of biological systems and devices as biosensors (Xing et al. 2011). Chemically functionalized CNTs has been used to evaluate cellular internalization in the presence of different cellular uptake inhibitors (Kostarelos et al. 2007). The ability of CNTs have allowed advancements in the gene therapy research area.
In an interesting work, (Zhao et al). developed a pH sensor system using single-walled carbon nanotubes (SWCNTs) functionalized with HPTS and coated with a layer of zinc phthalocyanine (Zhao et al. 2009). CNTs are promising candidates to facilitate the entry of HPTS to the cells and to be used as intracellular probes. Therefore, understanding the mechanism of interactions between SWCNTs and HPTS is of prime importance. CNTs have been used in the determination of fluorescence quenching mechanism of different dyes due to its ability as a quencher molecule. In a similar work, the effect of structural defect of SWCNTs and multiwall carbon nanotubes (MWCNTs) on their quenching fluorescence properties was studied (Singh et al. 2012). Ahmad et al. studied the static quenching mechanism of Rhodamine B by SWCNTs (Ahmad et al. 2009). Chiu et al. studied the quenching effect of different dyes by SWCNTs (Chiu et al. 2011).
To the best of our knowledge, there are no results regarding the quenching effect of SWCNTs on the fluorescence properties of HPTS. In this study, we have prepared and characterized a conjugate between SWCNT and HPTS dye to investigate the quenching effect of the SWCNTs using a fluorescence method based on Stern-Volmer plots.
Materials and methods
Materials
SWCNTs with diameters ranging from 0.8 nm to 2 nm and lengths between 500 nm and 2000 nm were purchased from Unydim, California. Folic acid and 1-hydroxypyrene-3,6,8-trisulfonic acid (HPTS) were obtained from Sigma-Aldrich Corp. All chemicals used in this work were of analytical grade.
Synthesis
Solubilization of single-walled carbon nanotubes
5 mg of SWCNTs were sonicated for 20 min in 5 mL of a 5 mM aqueous solution of folic acid (FA). FA solution was prepared as described by Castillo and coworkers (Castillo et al. 2013). FA molecules were dissolved in water with an addition of 50 /µL of NaOH (1 M) due to the poor solubility of FA. The FA powder (0.029 g) was mixed with H2O (25 mL) until a final pH of 10 was reached and magnetically stirred until a yellow-to-clear transition was observed. The solution was centrifuged at 2800 rpm for 20 min; the supernatant was collected and carefully separated from the solid. After aggregation, a large bundle of nanotubes was found. The conjugate was washed several times with MilliQ water and centrifuged to eliminate excess free FA. Finally, the SWCNT solution was stored at 4 °C. To estimate the concentration of SWCNT/FA suspension, a similar methodology used by Regev (Attal et al. 2006) was employed.
Synthesis of the single-walled carbon nanotube-HPTS conjugate
4 mg of HPTS was dissolved in 10 mL distilled water to a final concentration of 1 mM. Then aliquots of 10 [uL of HPTS were added to different concentrations of SWCNTs/FA suspensions (10µg/mL, 20µg/mL, 40µg/mL, 100 µg/mL). The solutions were magnetically stirred for 30 min and mixing before fluorescent measurements.
Measurements and tool techniques
Scanning electron microscopy images To characterize the functionalization of SWCNT with FA, Scanning Electron Microscopy (SEM) images were recorded using a Quanta FEG SEM microscope (Oregon, USA) with a maximal spatial resolution of 1.0 nm.
Atomic force microscopy images
All atomic force microscopy (AFM) images were recorded using an XE-150 Premier cross-functional AFM (Park Systems, Korea) with a PPPNVHR AFM probe (Nanosensors, Switzerland).
Ultraviolet-visible (UV-Vis) absorption spectrum
UV-Vis absorption spectrum was collected at room temperature in a quartz cell with a 1.0 cm path length with an Agilent-HP 8453 spectrophotometer.
Raman scattering spectroscopy
Raman measurements were performed using a Thermo Scientific DXR Raman microscope. An optical microscope was coupled to a single grating spectrometer with 5 cm-1 FWHM spectral resolution and ±2 wavenumber accuracy. A frequency-stabilized single diode laser was operated at 532 nm. Raman spectra were recorded using a 10x long working distance objective, 0.1 mW laser power and 15 s signal accumulation times. The power density was kept low (~12 kW/cm2) of the sample to minimize photo induced and thermal effects.
Fluorescence methods
Fluorescence measurements were made on standard luminescence spectrophotometer Perkin Elmer LS-55 using quartz cuvettes (10 mm path length). The fluorescence emission spectra were recorded in the range of 400-600 nm following an excitation wavelength at 458 nm. Both excitation and emission bandwidths were adjusted at 2.5 nm. The HPTS concentration was fixed at 10 µM, and the SWCNT/FA concentrations were varied from 10 to 100 µg/mL. To evaluate the effect of the temperature on the HTPS-SWCNT interaction, fluorescence emission was recorded at three different temperatures (5 °C, 25 °C and 45 °C).
Results and Discussion
The conjugate was prepared by sonication of a solution of FA (3.5 mM) with 5 mg of SWCNT. Fig. 2a and Fig. 2b show the dispersion SWCNT and FA before and after the ultrasonication step. It can be appreciated the debundled of SWCNT by the action of FA solutions. To obtain a clearer image of the debundling of SWCNT by AF, Fig. 2c shows an AFM image of several nanotubes covered by FA. On the other hand, the Fig. 2c is showed a picture of the solutions of SWCNT-FA, free HPTS, and HPTS conjugated to the nanotubes. Fig. 2d shows UV-vis absorbance spectra to SWCNT/FA of the free HPTS, SWCNT/FA, and HPTS conjugated. The HPTS conjugated to the nanotubes showed an absorption peak at 240 nm which corresponds to the Plasmon absorption of tubular graphite (Attal et al. 2006). An electronic transition around 375 nm absorbance band was observed, and probably this corresponds to the characteristic peaks of FA (See UV-vis spectrum of FA, supporting information SI). Additionally, at 402 nm a band belonging to HPTS is observed (Fig. 2b, white line).
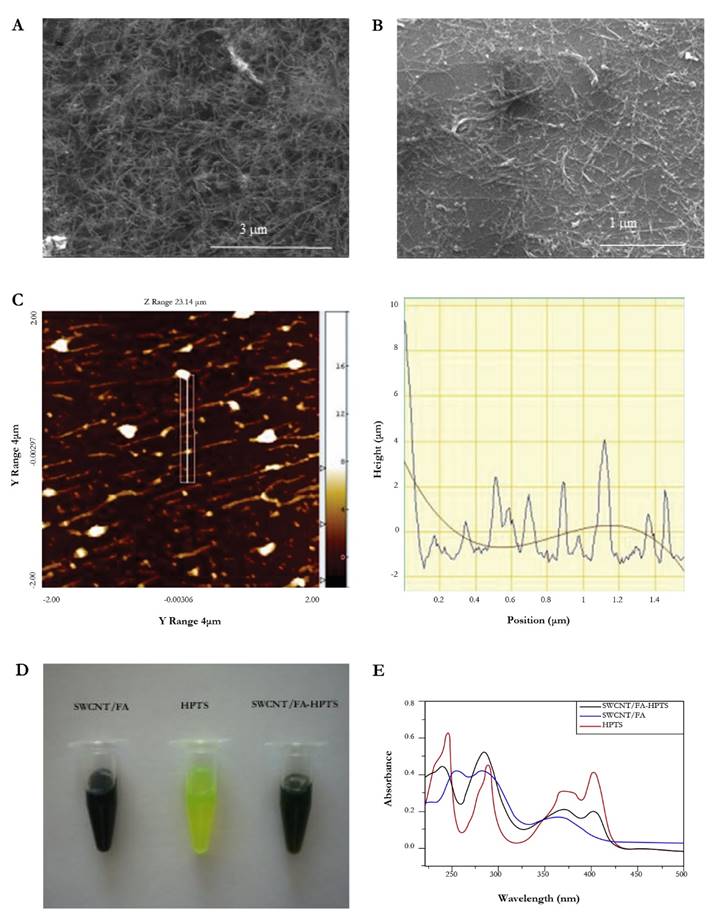
Figure 2 SEM image of the conjugate SWCNT before (a) and after (b) the modification with FA; (c) topographic image on the silicon substrate of SWCNT covered by FA using non-contact mode image; (d) images of solutions of SWCNT/FA, HPTS and SWCNT/FA-HPTS and (e) UV-vis absorbance spectra of the SWCNT/FA-HPTS (black line), SWCNT/FA (blue line) and free HPTS (red line) (ultra-purified water was used as a reference solution).
Raman scattering spectroscopy (RSS) is a technique mainly used to observe vibrational, rotational and other low-frequency modes in a system (Rindzevicius et al. 2017). In this study, we employed RSS to evaluate the effect SEM image of the conjugate SWCNT before (a) and after (b) the modification with FA; (c) topographic image on the silicon substrate of SWCNT covered by FA using non-contact mode image; (d) images of solutions of SWCNT/FA, HPTS and SWCNT/FA-HPTS and (e) UV-vis absorbance spectra of the SWCNT/FA-HPTS (black line), SWCNT/FA (blue line) and free HPTS (red line) (ultra-purified water was used as a reference solution). of the HPTS on the structure of CNT and thus show its functionalization. Fig. 3 shows the presence of two Raman peaks located at 1537 cm-1 (D band) and 1591 cm-1 (G band).
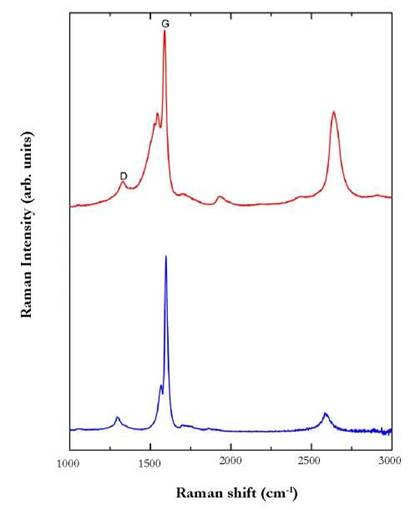
Figure 3 Raman spectra of SWCNT-FA (blue line) and SWCNT-FA in the presence of HPTS (laser 532 nm 0.1 mW).
These vibrations modes have been used to evaluate the presence of surface defects on the walls of CNTs. In a recent report, Koh et al. 2011 analyzed the presence of (i) the disorder-induced D band, and (ii) the G band to investigate dispersion states and covalent functionalization of SWCNTs and to map COOH groups on oxidized SWCNTs. Hence, inelastic scattering spectra can provide a more detailed evidence of the vibrational Raman modes induced by the coating of HPTS around CNTs.
The interaction of SWCNT-FA with HPTS was evaluated by measuring the intensity ratio between the D and G-bands. The ID/IG intensity ratios for the SWCNTs-FA and the HPTS functionalized nanotubes were found to be 0.098 and 0.21, respectively. These relatively small variations of the Id/Ig ratios can be interpreted as an indication of a noncovalent interaction character between HPTS and carbon nanotubes functionalized with FA that inhibits the formation of significant structural defects.
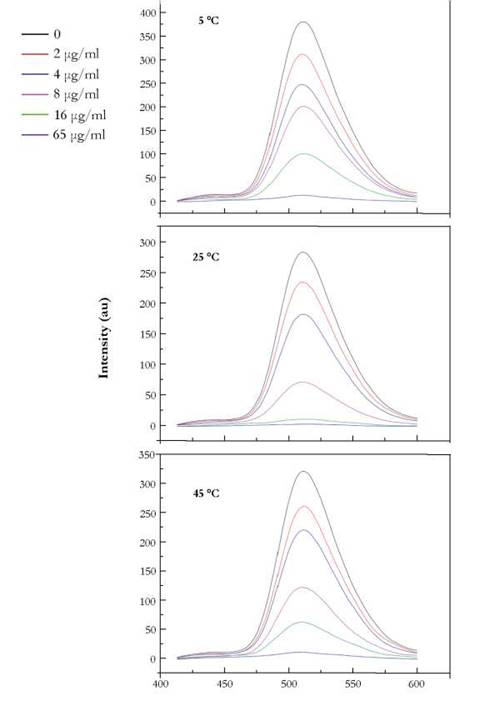
The fluorescence spectrum of HPTS in the presence of different concentration of SWCNT at different temperatures is presented in Fig. 4. At a fixed concentration of HPTS, the addition of SWCNT produced a decrease in the fluorescence intensity of HPTS at 510 nm, SWCNTs have been considered as an efficient quencher (Zhiu et al. 2010) of a variety of dyes decreasing their fluorescence by a variety of electron transfer mechanisms (Ahmad et al. 2009).
Fig. 4. shows the fluorescence intensity decreases gradually with the increase of SWCNT concentration. This probably indicates some interaction between HPTS and the nanotubes.
The fluorescence quenching is described by the Stern-Volmer equation:
where Io and I are the fluorescence intensities of the probe in the absence and presence of the quencher, respectively. Several cases have documented the Stern-Volmer plots with a linear trend. However, other studies have shown a positive deviation from this linearity (Toprak et al. 2011). There are two possible reasons for the deviation from this linearity: the first is due to the simultaneous presence of dynamic and static quenching; the second is due to the formation of a chemical complex (static quenching) or by excited-state collisions (dynamic quenching) (Adenier et al. 2002).
To identify the quenching mechanism as being dynamic or static, we collected fluorescence emission data at different temperatures. The spectra were recorded for various concentrations of SWCNT containing HPTS after incubating the mixture at various temperatures (5 °C, 25 °C and 45 °C).
Fig. 5. shows the S-V plots at different temperature. It can be seen from Figure 4 that the quenching efficiency correlates inversely with temperature, in the range between 0-8 ug/mL, strongly evidenced the presence of a static quenching mechanism. However, it is worth to mention that at a concentration of 66 ug/mL SWCNT there is a noticeable deviation from the linearity probably due the quenching is nearly completed. Also, the above concentration of 66 ug/mL, an opposite behavior, is observed, meaning probably the presence of a dynamic quenching. However, to confirm this observation further experiments with a narrow interval of concentrations between 60 ug/mL and 100 ug/mL are necessary to carry out.
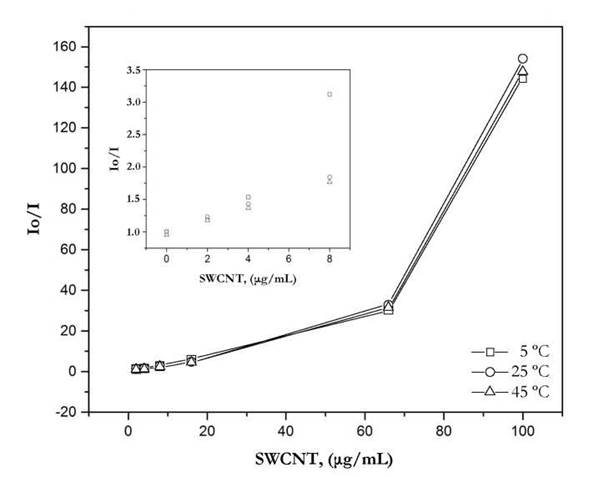
Figure 5 Stern-Volmer plots of HPTS quenching with SWCNT at different temperatures. Inset: Stern-Volmer plots at low concentrations of SWCNT.
At higher temperatures, the probability of a collision between the HPTS and carbon nanotubes will be increased leading to a decrease in the fluorescence.
The increase in temperature favors the equilibrium to shift towards the non-complexed form of the HPTS decreasing the fluorescence quenching efficiency. I n a recent work, Ahmad et al. 2 009 s tudied the mechanism between CNTs and Rhodamine B showing a static quenching mechanism between the dye and carbon nanotube.
This paper is the first study to determine the quenching mechanism between HPTS and SWCNTs opening new insights into the use of the system HPTS-SWCNT for sensor probes applications.
Conclusions
The fluorescence quenching of HPTS by single walled carbon nanotubes was analyzed by a fluorescence m ethod. We h ave s hown t hat SWCNTs quench HPTS by a static mechanism by forming a stable ground state complex which decreases the efficiency quenching at higher values of temperature. The use of SWCNT integrated to probe fluorescent as HPTS will facilitate the entry to the membrane cells opening new perspectives for creating novel pH intracellular biosensors for biomedical applications.