Introduction
Venous thromboembolism (VTE) is an entity that interlinks deep vein thrombosis (DVT) and pulmonary thromboembolism (PTE). Although DVTs usually occur in the lower extremities, they can also occur in the splenic bed, upper extremities and brain 1. Venous thromboembolism is common, affecting 10 million people per year (1-2 cases per 1,000 people) worldwide, and is the third main cause of cardiovascular disease, after ischemic heart disease and cerebrovascular disease 2. The global incidence of VTE is growing along with life expectancy; in fact, after age 45, the risk of VTE is 8% 3. The growth in associated comorbidities (like obesity, heart failure, cancer, etc.), together with a greater use of imaging tests, increases VTE detection 4.
The likelihood of experiencing VTE does not vary by sex, although it can be twice as high in men as in women, when causes related to pregnancy and estrogen therapy are excluded 5. The causes have been defined in many ways. Recently, healthcare professionals have been urged to identify provoking and/or triggering etiological factors within the previous three months, like immobility, trauma, surgery, cancer, etc. On the other hand, unprovoked embolic events occur without these incidents 1. Anywhere from one third to one half of VTE episodes have no identifiable triggering factor and, therefore, remain as unprovoked events 6. Alteration of any of the three factors described in Virchow's triad since 1856 (endothelial damage, blood stasis and hypercoagulability) helps identify the etiopathogenesis 7.
In 1957, R. May and J. Thurner described an anatomical condition that included the three abnormalities described by Virchow 8. Cockett and Thomas depicted it clinically in 1965. May-Thurner syndrome (MTS) is described as a compression of the left common iliac vein (LCIV) by the right common iliac artery (RCIA) against the fifth lumbar vertebra which can cause venous flow obstruction and pain, edema and DVT in the left lower extremity 9. Anecdot-ally, it can involve the right iliac vein or even be bilateral 10. It is more prevalent in women (5:1), generally those of reproductive age 11. The incidence and prevalence of MTS are unknown, and it is commonly underdiagnosed, although it accounts for 2-5% of DVT cases 9. There are no standardized diagnostic criteria. Diagnostic imaging plays an essential role in identifying this condition in patients with suggestive signs and symptoms 12. Below, we describe an unusual case of an elderly woman with an imaging report compatible with this condition.
Case presentation
A 71-year-old woman presented to the emergency room with a complaint of sudden-onset pain and hard edema in her left leg, with functional limitation. A review of systems indicated that she had experienced moderate pleuritic right chest pain three days prior, with no dyspnea, palpitations, orthopnea, fever and/or weight loss. Her medical history was remarkable for type 2 diabetes mellitus, treated with insulin glargine, insulin glulisine and dapagliflozin, along with chronic venous insufficiency, with no monitoring or treatment, as well as bilateral blindness due to glaucoma. She had no significant surgical, allergic or exposure history. She had last been hospitalized six years before for a urinary tract infection that caused diabetic ketoacidosis. On her obstetrical-gynecological history she denied miscarriages or stillbirths (G3P3).
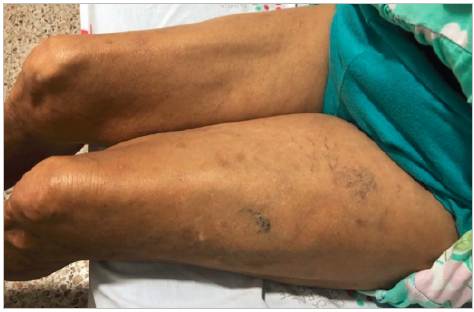
Her vital signs were normal on admission. On physical exam, she had no palpable breast nodules. Her lower extremities were asymmetrical due to edema in the left lower extremity, mainly in the thigh, with telangiectasias (Figure 1). The Homans and Pratt signs were positive in this extremity, with preserved popliteal and pedal pulses.
The initial laboratory tests reported a complete blood count, electrolytes and kidney function tests within normal limits, except for random central blood sugar (319 mg/dL) and elevated C-reactive protein (CRP) at 7.19 mg/dL (reference range 0-0.5). An electrocardiogram showed no tachycardia, chamber overload or signs of injury, ischemia or necrosis. Troponin T was negative (0.009 ng/mL, reference 0-0.0014).
Venous Doppler of the lower extremities confirmed the presence of occlusive thrombotic material in the common femoral vein. She was hospitalized with a diagnosis of unprovoked DVT, and treatment was started with enoxaparin adjusted to 1 mg/kg (50 IU subcutaneously every 12 hours). Her insulin treatment was adjusted, and complementary studies were ordered.
The patient underwent computed tomography (CT) of the chest and abdomen, with contrast. The chest CT showed asymmetric enhancement of the right basal parenchyma and a hypoperfused medial area indicating ipsilateral atelectasis, conclusive for acute PTE in the right posterior basal segmental artery, with an apparent ipsilateral posterior basal infarction (Figure 2).
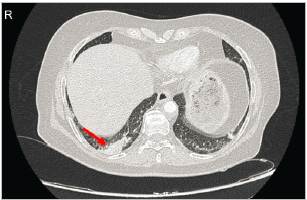
Figure 2 Axial section of the chest CT with contrast showing an area of right basal hypoperfusion (red arrow), which is conclusive for acute PTE of the right posterior basal segmental artery with apparent right posterior basal infarction.
The abdominal CT showed an enlarged left common iliac vein extending toward the ipsilateral iliofemoral vein. The apparent reduction of the common iliac vein's lumen below the RCIA was notable, indicating a mural thrombus extending toward the confluence of the iliac veins at the inferior vena cava, ending in thrombosis of the iliac vein and left iliofemoral vein, with suspected May-Thurner syndrome (MTS) (Figures 3a and 3b).
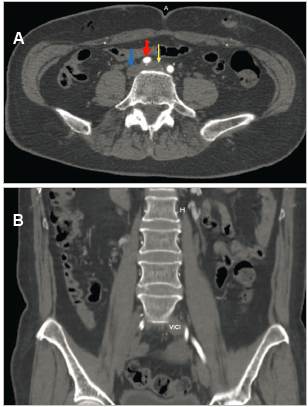
Figure 3 A (Top): left common iliac vein compression (yellow arrow) by the right common iliac artery (red arrow), causing venous dilation (16 mm), with mural thrombi inside. Inferior vena cava (blue arrow). B (Bottom): coronal abdominal-pelvic section showing left common iliac vein (LCIV) thrombosis with an increased diameter compared to the contralateral vein.
Tumor marker screening (alpha-fetoprotein [AFP], carcinoembryonic antigen [CEA], CA 125, and CA 199) was within normal limits. Autoimmunity tests were negative (antinuclear antibodies [ANAs], anti-Scl-70, anti-centromere antibodies, IgM/IgG anti-beta2 glycoprotein and rheumatoid factor), and complement C3 and C4 levels were normal. The lupus anticoagulant screening test was prolonged according to the reference range (48.8, with a control of 34.9), with normal coagulation times. A urinalysis ruled out proteinuria.
A transthoracic echocardiogram showed mild concentric hypertrophy of the left ventricle (LV) with preserved systolic function and type I diastolic dysfunction (left ventricular ejection fraction [LVEF] of 62%), with preserved right ventricular systolic function and no indirect signs of pulmonary hypertension.
Endovascular interventionism reported complete occlusion and multiple apparently chronic common femoral and left primitive iliac collaterals, compatible with chronic occlusion secondary to vascular compression, compatible with MTS. Thrombectomy/embolectomy was rejected in favor of medium caliber vessel repair through recanalization using a stiff hydrophilic guidewire and multipurpose catheter, with angioplasty using a 12 x 100 mm venous stent, achieving adequate patency of the left common iliac vein.
The patient was admitted to the intensive care unit for monitoring and was transferred to the general floor after 24 hours due to her satisfactory progress, with double anti-platelet therapy (a 100 mg acetylsalicylic acid tablet and a 75 mg clopidogrel tablet every 24 hours by mouth) and anticoagulation treatment (60 mg of enoxaparin every 12 hours subcutaneously) for seven days. She was subsequently discharged on oral antiplatelet therapy (clopidogrel) and direct anticoagulation (rivaroxaban) for an indefinite period, contingent on ambulatory follow up with vascular surgery, cardiology and internal medicine
Discussion
May-Thurner syndrome predominantly affects women in their twenties to forties, with few cases outside this range 13. The disease may be asymptomatic or characteristically present with lower extremity pain and edema (usually on the left), claudication and progression toward chronic venous insufficiency, manifesting in skin color changes and varicose veins 14 (Figure 1). In the case we have described, the patient had a history of chronic venous insufficiency that had not been studied or treated. Despite not reporting constitutional syndrome information, tomography was ordered, initially directed toward a neoplastic cause, finding low-risk PTE and a structural/anatomical abnormality of the iliac vein, which was highly suggestive of MTS.
May-Thurner syndrome results from LCIV compression by the RCIA immediately after the origin of the abdominal aorta and before the iliofemoral vein junction. The constant pressure exerted by the artery compresses the vein against bony structures, typically lower lumbar vertebrae, giving rise to venous "spurs" or stenosis. Although left-sided involvement is the most frequent, as in our case, contralateral venous involvement is also possible, less frequently 15.
This condition is mainly diagnosed through imaging. Initially, Doppler ultrasound is used to visualize the vascular defect in the common iliac vein. However, due to the anatomical depth and being operator-dependent, its sensitivity is only 47% 16. In our case, the diagnosis was made through abdominal and pelvic vessel tomography. The findings can be confirmed with intravascular ultrasound (IVUS), recently considered as the possible gold standard. This helps detect thrombi in the vessel lumen and characterize them, helping determine the need for thrombolytic treatment. In addition, it favors proper stent selection by measuring lumen diameters and identifying the confluence of the iliac vein 12.
Anticoagulation is essential for treatment; delays in administering it are associated with a higher risk of PTE, which occurred in our case. Anticoagulation should be continued for 6-12 months after endovascular treatment, to prevent a new thromboembolic event 16. No anticoagulant is preferable to another. Heparins are just as effective as coumarin anticoagulants or direct oral anticoagulants (DOACs), and therefore may be used 17. In practice, the choice of anticoagulation will be guided by the individualized treatment, considering aspects like the clinical setting, glomerular filtration rate (GFR), availability, and each patient's risk of bleeding 18.
A combination of endovascular treatment (using thrombectomy, thrombolysis and/or angioplasty with stent placement) and anticoagulation is generally preferred, instead of depending solely on the pharmacological management of symptomatic patients (12), with VTE rates of less than 10% reported for combination therapy versus 80-90% with medical treatment alone 19. In our report, thrombectomy/ embolectomy was not performed due to chronic endovascular changes, opting instead for repair and recanalization through angioplasty and stenting in the collaterals. Once endovascular treatment has been performed, the patients should receive antiplatelet therapy (preferably with clopidogrel) for six weeks to three months, and low-dose aspirin indefinitely, with periodic follow-up using ultrasound or venous tomography to confirm stent patency 14.
Conclusion
This article highlights the importance of considering MTS as an underdiagnosed cause of DVTs, even in unusual age groups, considering that there is often an undetected history of venous insufficiency. The search for an etiology, taking the DVT risk conditions (Virchow's triad) into account, can be key. Prompt identification of the condition, supported by diagnostic imaging, is crucial for proper treatment, which requires a combination of medical (anticoagulant) and endovascular treatment, with a lower rate of post-thrombotic syndrome. There is no consensus on anticoagulant and anti-platelet treatment, and therefore it should be individualized, keeping the local guidelines or recommendations in mind.











 text in
text in