Introduction
Juvenile idiopathic arthritis (JIA) is the most frequent chronic rheumatic disease in children. The disease is characterized by chronic inflammation of the joints and develops more frequently in children under 16 years of age. Many aspects of the etiology of the disease are still unknown,1,2 with a variable clinical evolution ranging from total remission to significant disability. According to some series of patients followed for 10 or more years, one-third of the patients achieve remission, 60% present with some type of sequelae, and about 30% develop severe functional limitations. 2,3
Pain is critical to make the diagnosis and is considered a marker for improvement of inflammation and good prognosis for future rehabilitation of the child. Therefore, pain modulation is essential for the management of inflammation, for preventing deformities and for ade quate growth and development. 4,5
Chronic pain experienced from such an early age leads to difficulty in modulating pain during a crisis, and medical practitioners are forced to use various pain control strategies and are faced with the challenge of administering a safe and effective approach to this group of patients. 5
Ketamine has been used as a pain-relieving strategy, particularly in adults, when the anti-inflammatory effect becomes relevant in some cases. There are some reports about the use of Ketamine in children, but not specifically for JIA-associated pain; this may be then an option for the management of refractory cases.
Description of a clinical case
Patient information and clinical record
A 7-year-old male patient, attending elementary school, 22kg, mestizo, with a history of enthesitis-related JIA, diagnosed 1 and a half years ago and managed with acetaminophen and NSAIDs, with poor ambulatory pain control using oral medication for over 1 year. Consequently, pediatric rheumatology added methotrexate 1 month before being admitted to the hospital. There was no evidence in the medical record about prior hospitalizations, but there was evidence of multiple visits due to poor pain control with significant enthesitis and gate limitations that forced the patient to quit school for the last 2 months.
Clinical findings and diagnostic evaluation
The patient was admitted with clinical chronic pain of >1 year of evolution that intensified 1 month ago. The patient experienced morning pain in the distal lower limbs, associated with stiff hands and feet that improved through the day; the pain increased progressively in intensity to up to 8/10 in the verbal rating scale for pain (VRS) with no improvement with ambulatory therapy with ibuprofen at a dose of 30mg/kg/d and methotrexate.
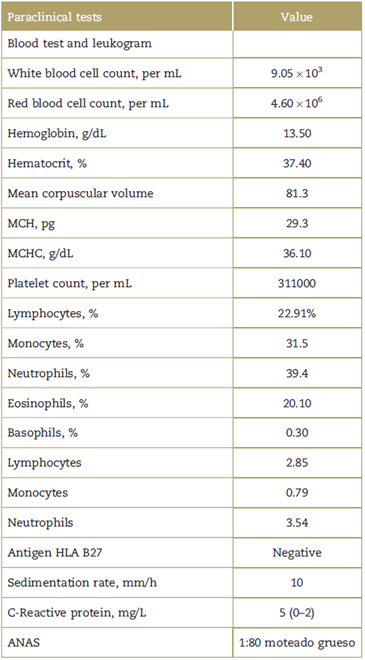
The physical examination evidenced bilateral tarsal pain with edema and enthesitis, pain at palpation on the head of the metatarsals and clinical signs of bilateral sacroiliitis, difficult walking, and stiffness that limited the patient for active and passive movements. The para-clinical tests performed at admission are listed on Table 1.
Therapeutic intervention
Because of the evidence of inflammation, pediatric rheumatology started hospital management with prednisolone (0.1mg/kg) and sulfasalazine (20mg/kg) associated with naproxene (125 mg q12h) and dipyrone (30 mg/ kg q8h), with no improvement and with gastric intolerance to naproxene which was then switched over to tramadol (1mg/kgq8h) plus acetaminophen (250 mg q6h), but there was no significant analgesic improvement after 6 days of treatment. Consequently, an evaluation was requested to the Institution's Pain Relief Clinic. In view of the fact that the first and second line analgesia medications had already been tried at the time of the evaluation, intermittent infusion (8 pm to 8 am) of Ketamine (0.1mg/k/h) was initiated in order not to limit the patient's mobility during the day; otherwise, the patient had to be connected to the infusion pump. The expectation was improved functionality upon good pain control, in combination with the pre-established doses of tramadol with rescue doses (not used), together with acetaminophen and dipiyone, as a strategy to modulate central sensitization and probably the inflammation secondary to acute-chronic somatic pain.
Follow-up and results
The following day after initiating the infusion, significant improvement was noted (over 50%); the child had no pain during the night and according to the mother night pain used to be always present. The child was able to move around during the day, which as already mentioned was difficult before. After 3 days with this therapeutic regimen, significant pain improvement was noted (VRS 3/10) as well as improved functional ability with no ketamine-associated adverse events reported (ie, nightmares or halluci nations). The child was then discharged from hospital.
The mother expressed her satisfaction with the pain control achieved in her son.
Discussion
The use of ketamine at analgesic doses in children, as part of analgesia management, has proven to be safe and effective in other settings, such as cancer and non-cancer (non-inflammatory) patients, reducing the pain intensity with a lower score in the visual analogue scale and free from any significant side events, with doses between 0.05 and0.5mg/k/h.6-8
Ketamine, inadditiontoitsN-Methyl-D-aspartate(NMDA) receptor antagonistic effect, also acts at different levels of the inflammatory cascade, interacting with the cells, the production of cytokines and the regulation of inflammatory mediators that provide ketamine with its anti-inflammatory effect. Some reports claim that at a dose of 0.25 mg/kg there is a decrease in the Interleukin (IL) 6 levels and at a dose of 0.5 mg/kg there is reduced Tumor Necrosis Factor (TNF)-alpha and increased nitric oxide (NO) production. 9-11
The nature of the structures where pain originates seems to make a difference in the ability of Ketamine to relief the pain12; however, mechanisms ranging from local inflammation of the terminal nerve fibers and then progressing to the release of neuropeptides and prosta-glandins, glutamate activation with its NMDA receptor, and finally activation of the glial cells that participate in central sensitization phenomena and chronicity of pain, increasingly show a smaller gap from inflammation to nociception and neuropathic pain. 13-16
However, with so many mechanisms associated with the generation of central sensitization phenomena in chronic pain of inflammatory origin, 15,17 it is impossible to attribute improvement just to 1 single drug, and hence multimodal management continues to be the mainstay of treatment.
The use of an analgesic Ketamine infusion in this case was based on failure to the initial analgesic therapy, the safety reported in other pediatric scenarios and its anti-inflammatory effect described in other publications; using analgesic doses (0.1mg/kg/h), together with a multimodal strategy, it was possible to control severe pain and the limitations of a child with decompensated underlying inflammatory disease.
Notwithstanding the fact that Ketamine has been used for over 30 years, there are no studies or reports providing enough evidence to advocate the routine use of ketamine in the management of chronic pain due to JIA.
Conclusions
This case report provides valuable information regarding the use of Ketamine as a central sensitization and inflammation modulator that could be extrapolated to a similar population of rheumatologic patients and design new research trials to generate the missing evidence.
This case report has been endorsed by the Health Research Ethics Committee of the Institution.
Ethical disclosures
Protection of human and animal subjects. The authors declare that the procedures followed were in accordance with the regulations of the relevant clinical research ethics commit-tee and with those of the Code of Ethics of the World Medical Association (Declaration of Helsinki).
Confidentiality of data. The authors declare that they have followed the protocols of their work center on the publication of patient data.
Right to privacy and informed consent. The authors declare that no patient data appear in this article.











 text in
text in