Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
Revista colombiana de Gastroenterología
Print version ISSN 0120-9957
Rev Col Gastroenterol vol.28 no.1 Bogotá Jan./Mar. 2013
Hepatotoxicity due to green tea consumption (Camellia Sinensis): A review
Eliana Palacio Sánchez (1), Marcel Enrique Ribero Vargas (1), Juan Carlos Restrepo Gutiérrez MD. (2)
(1) Student at the Medicine Faculty of the Universidad de Antioquia in Medellín, Colombia. Member of the Gastro-hepatology Group at the Universidad de Antioquia in Medellín, Colombia.
(2) Internist and Hepatologist in the Hepatology and Liver Transplant Unit of the Hospital Pablo Tobón Uribe in Medellín, Colombia. Tenured Professor in the Medicine Faculty of the Universidad de Antioquia in Medellín, Colombia. Mail: jcrestrepo@une.net.co; jcrestrepo@hptu.org.co
Received: 27-06-12 Accepted: 18-12-12
Abstract
As consumption of green tea has increased in recent years, so too have reports of its adverse effects. Hepatotoxicity is apparently caused by enzymatic interaction that leads to cellular damage and interference with biological response systems and metabolic reactions. This review article introduces the morphological characteristics and biochemical components of the green tea plant, camellia sinensis. Analysis of clinical trials, in-vitro trials and pharmacodynamic and pharmacokinetic studies then shed light on some of the mechanisms by which green tea causes hepatic damage. Examples are the chemical interactions with enzymes such as UDPGT, alcohol dehydrogenase and cytochrome P450 and interactions with the mitochondrial enzyme and immune systems. These forms of cellular lesions are correlated with case reports in the scientific literature which clarify the spectrum of hepatic damage associated with the consumption of green tea. This analysis finds that even though the mechanisms by which green tea causes hepatic toxicity are still a mystery, certain catechins of camellia sinensis and interactions at the cellular and mitochondrial levels may be responsible for this toxicity. On this basis, social and political preventive measures regarding intake of this natural product at levels can be justified.
Keywords
Hepatotoxicity, green tea, camellia sinensis, catechins.
INTRODUCTION
Green tea is obtained from the Camellia sinensis plant which is a bush or small perennial tree. Its main root is 4 to 5 cm long green leaves and it has yellowish-white flowers are cut for harvesting when the plant is no more than two meters tall (Figure 1).
The exact date when Camellia sinensis was first cultivated is unknown. According to Chinese tradition, approximately 4,000 years ago Emperor Shen Nung was warming up a pot of water under a tree, and some of its leaves fell into the water. He drank the entire pot and felt invigorated. On the spot, he decided to promote its cultivation throughout the empire.
Tea was first processed in the United States in 1650 although the Camellia sinensis genus entered the country in 1744 in the gardens of Georgia. The first attempts at cultivation were without success, so further attempts were needed for successful cultivation (1). Camellia sinensis is currently cultivated in tropical and subtropical climates in many parts of the world (2).
There are three main types of tea. Fresh leaves are picked from the plant, then left to dry under the sun or hot air, and then ground into small pieces. When the processing of the leaves stops without undergoing oxidation and fermentation, green tea is obtained. If the leaves are exposed to the sun or air for longer periods of time after maceration, oxidation occurs as a result of exposure to the environment and black tea is obtained. If the process of oxidation is shortened, then Oolong Tea may be extracted (1).
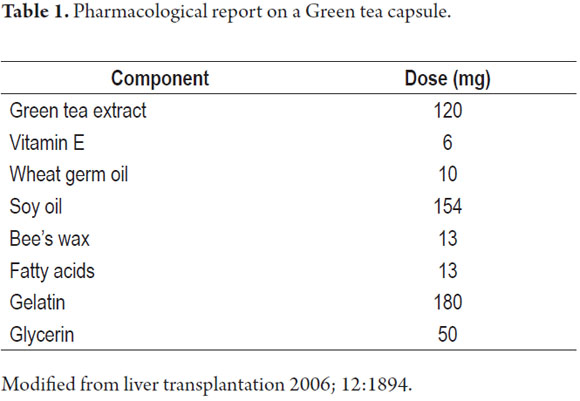
Many healing benefits have been attributed to green tea. Among these are improvement of asthenia, diarrhea, bronchitis, asthma, hyperlipidemia, cellulitis, and abscesses as well as weight reduction. Also, another group of healthy people defends its use as a tool that helps improve health and prevents disease. Nevertheless, the necessary scientific evidence to support these claims has not yet been gathered. Most studies of green tea's benefits are in vitro or animal studies without sufficient clinical trials to support the use of green tea in humans. The possible benefits have led to health improvements which have increased green tea's unsupervised availability, accessibility and intake. Its global consumption is surpassed only by water (3). At present there are also liquid and capsule presentations which unlike conventional infusions contain high concentrations of possibly toxic substances and other excipients. At the Queen Elizabeth II Health Sciences Center in Canada an acute liver failure case was reported from green tea consumption. Upon pharmacological analysis the components listed in Table 1 below found.
These components have certain influences on health that represent great risks due to their persistent use which in some cases can be fatal. In the last few years cases of liver toxicity associated with green tea consumption have been reported in the main medical literature data bases. For this reason, we have reviewed major scientific medical knowledge bases regarding mechanisms of possible liver damage resulting from green tea and spectrums of liver toxicity according to the latest case reports on liver damage, and report about the safety of green tea consumption.
GREEN TEA COMPONENTS
The variety of components in green tea is very large. Its components include gallocatechin gallate (GCG), gallocatechin (GC), catechin gallate (CG), catechin (C), and flavonoids such as kaempferol, quercetin and myricetin. Other important components include theanine, derived from amino acid, the xanthine alkaloid caffeine, theophylline, theobromine, saponins, and tannins. Green tea has more than 300 other substances (4, 5). However, the main active components to which both beneficial and adverse health effects have been attributed are the four polyphenolic catechins: epigallocatechin gallate (EGCG), epicatechin gallate (ECG), epigallocatechin (EGC), epicatechin (EC) (6). A very important benefit that is attributed to catechins, especially to epigallocatechin gallate (EGCG), is the property of decreasing or maintaining body weight due to its capacity to induce and stimulate thermogenesis and the oxidation of fats (7). Nevertheless, a metaanalysis published in the International Journal of Obesity concluded that the effect of green tea on the reduction or maintenance of weight is very slight (8).
GREEN TEA ABSORPTION AND ELIMINATION
Multiple clinical trials have been performed with volunteer patients that have made it possible to clarify absorption of green tea catechins to some extent.
In one of these, Yang et al. administered different doses of catechins to 18 volunteers. Doses of 282 mg of EGCG, 564 mg of EGC and 846 mg CE, the three main green tea catechins, were administered. The maximum levels of catechins detected in plasma were approximately 1.5h and 2.5h indicating good oral absorption. Twenty four hours later these substances were undetectable. The maximum plasma concentration of EGCG measured was 321 ng/mL, maximum plasma concentration of EGC was 550 ng/mL, and maximum plasma concentration of CE was 190 ng/mL. The catechin with the longest lifespan was EGCG which lasted five hours. This is the catechin found to be most involved in the processes of liver damage, as cited below. EGC and CE were detected in urine, which indicates renal excretion. There are no considerations about the counterproductive effect on patients with liver failure regarding kidney excretion since the study was performed on healthy people (9). It is important to consider that in pharmacokinetic studies patients did not present adverse effects. Nevertheless, small sample sizes ranging from 18 to 60 patients provide limited numbers for concluding that the use of green tea is safe (10, 11, 12, 13).
INTERACTIONS OF GREEN TEA CATECHINS WITH THE URIDINE DIPHOSPHATE - GLUCURONOSYLTRANSFERASE ENZYME
As already mentioned, the main components of green tea are EGCG, ECG, EGC and CE catechins. However, only EGCG and EGC stand out because they alone can interact with the UDPGT enzyme. To understand this interaction it is necessary to understand the possible mechanisms that the hepatocyte has to metabolize these substances. The biotransformation of EGCG and EGC has yet to be completely studied, though authors such as Li et al., in 2001, have described that EGCG and EGC undergo a process of methylation, glucuronidation and sulfation, although which of the three processes predominates has not yet been defined. More recently, Hong Lu et al. together with the American Society for Pharmacology and Experimental Therapeutics (ASPERT) have proposed that glucuronidation predominates in metabolizing the main catechins of green tea. This conclusion was based on the finding that EGC-Omonoglucuronides were most commonly found in searches for metabolites in urine, plasma and bile. This indicates that glucuronidation is the most frequently used means by which hepatocytes transform EGC and EGCG catechins. Apparently UDPGT 1A with the UDPGT 1A1, 1A3, 1A8 and 1A9 subfamily are the most active in this process of glucuronidation. These glucuronidated metabolites are cleaved by the β-glucuronidase which means that glucuronidated metabolites of these catechins are accumulated in the hepatic tissue as a result of chronic intake of green tea since β-glucuronidase is overcome and inhibited by the green tea's catechins. The accumulated metabolites apparently have the property of capturing free radicals, a process which may lead to liver toxicity, damage to the hepatocyte's mitochondria and difficulty in metabolizing other substances such as arachidonic acid. This in turn is a possible precursor of inflammatory products through the cyclooxygenase and lipoxygenase pathways (14).
INTERACTIONS WITH THE CYTOCHROME P450 SYSTEM
The interactions of the main catechins in green tea in enzymatic systems such as that of cytochrome P450 have also been evaluated in scientific literature. Animal studies have shown that the components of green tea can inhibit cytochrome the P450. Despite this, preclinical studies comparing green tea consumers and non-consumers which have evaluated the concentrations of metabolites from medications processed by the cytochrome P450 in plasma and urine have found that people who consume green tea do not have any significant inhibition even though the CYP3A4 member seems slightly affected (15). However, these studies have had limited sample sizes and limited number of medications evaluated for this enzyme. It is therefore necessary to perform more studies to come to any conclusions about the role of green tea catechins in relation to the cytochrome P450 enzymatic system.
INTERACTIONS WITH GLYCOPROTEIN P
Glycoprotein P is a dependent ATP pump found in the small intestine, bloodâbrain barrier (BBB), hepatocytes and tumoral tissues among other places. Its function is to transport substances such as xenobiotics, cellular metabolites and antitumor medications to the exterior of cells which means that it favors resistance to multiple antitumor drugs by removing them from the cancerous cell.
This is why in-vitro trials have been performed in order to understand the interaction of green tea catechins with this transporter. These experiments have included medications such as tamoxifen, doxorubicin, verapamil and diltiazem. The results have shown that the main catechins of green tea may inhibit functioning of glycoprotein P which would decrease the resistance level of tumor tissue to antineoplastic agents (16, 17, 18). This must be viewed with caution since there is no consensus on dosages, the time these catechins should be used, adverse effects and the specific tumors that might be treated to avoid resistance from these cancerous cells to antitumor agents. Consensus has not yet developed because there are still no long term clinical trials in humans with the epidemiological and ethical conditions necessary to provide sufficient scientific support.
In addition to the functions already mentioned, glycoprotein P also eliminates various medications and compounds such as bilirubin conjugated by hepatocytes (19, 20). When catechins inhibit this action of glycoproteins action it may result in toxic effects on hepatocytes since it could lead to intrahepatic cholestasis and systemic toxicity (21).
INTERACTIONS WITH HEPATOCYTE MITOCHONDRIA
In vitro and in vivo trials have found possible interactions between hepatocyte mitochondria and the green tea catechins. Giuseppe Galati et al. of the University of Toronto used an in vivo cellular model to demonstrate hepatotoxicity resulting from the components of camellia sinensis. The study concluded that the main mechanism of liver toxicity is formation of oxygen-free radicals which leads to mitochondrial dysfunction and cellular toxicity. It is interesting that EGCG, the most abundant green tea phenol, was the catechin which produced the largest number of free radicals and caused the greatest mitochondrial damage (22, 23).
INTERACTIONS WITH THE ALCOHOL DEHYDROGENASE ENZYME
In vitro trials have demonstrated that the triterpenoid saponins components of the camellia sinensis plant, isolated primarily from the roots of the plant through chromatography experiments (24), have the capacity to inhibit the alcohol dehydrogenase enzyme. In these experiments ethanol is added to a solution with a phosphate buffer, then a solution of the ADH enzyme is introduced and serial measurements are taken every 5 minutes of ethanol absorption which reflects the activity of the alcohol dehydrogenase enzyme. Results showed that in the solution with saponins the alcohol dehydrogenase enzyme was up to 50% less active than in a control solution control without saponins (25, 26). As it is well known, in-vitro studies have no great clinical validity, but certain green tea substances can be proposed to inhibit alcohol dehydrogenase. For people who consume alcohol and green tea this would increase the average life of ethanol levels in the blood, creating higher toxicity levels in the liver while increasing other systemic effects of ethanol.
INTERACTIONS WITH THE IMMUNE SYSTEM
There are only a few trials that evaluate the interactions of Green tea catechins with the components of the immune system. Last year an essay was published with evidence that EGCG production of oxygen-free radicals leads to activation of calcium channels which in turn leads to release of the ion into the extracellular space. Mast cells then pro-
duce greater quantities of IL-13 and TNF α with all of the repercussions that his entails for the immune system. These include regulation of Tp lymphocytes, stimulation of growth, differentiation and regulation of the activities of B lymphocytes, production of other inflammatory cytokines, and mediation of the TNFα inflammatory response (27).
SPECTRUM OF LIVER TOXICITY
The mechanisms explained in this review can lead to a miscellany of liver damage associated with consumption of green tea. The spectrum can go from an alteration of liver enzymes to acute liver failure requiring liver transplantation (28, 29). Histological examination of liver explants has shown that some areas of parenchyma are conserved while others have centrilobular or panlobular necrosis. In some cases an eosinophil reaction is observed while mixed inflammatory infiltrates are observed in other cases (30, 31, 32, 33, 34).
Not all cases in which liver damage is associated with green tea consumption have acute or linear presentations. In some cases insidious manifestations with intermittent elevation of liver enzymes have been observed. These cases reflect inflammations associated with consumption of camellia sinensis. When the patient suspends intake, the normal values of biological markers reappear. In some cases, after multiple expositions to this type of inflammation the liver is unable to recover its functions and enters into fulminant liver failure (35, 36). In some cases alterations of the biochemical profile are presented at the expense of the gamma-glutamyl transferase enzyme and alkaline phosphatase. These modifications occurred in some patients over 5 consecutive year indicating that the spectrum of liver damage is wide, but still uncertain. It is important to highlight that in the presentation of these cases other causes that may generate these types of liver alterations have been rigorously discarded. In some publications methods such as the RUCAM have been used. This enables the evaluation of the association between the consumption of a medication and the appearance of alterations and liver deterioration and thus can show causal relations between green tea consumption and patients' pathologies in cases reporting fulminant liver failure (37, 38).
DISCUSSION
The consumption of green tea has increased in the last 5 years and with it an association between this consumption and liver damage has emerged. Multiple cases without fulminant liver damage have been reported, and then in 2001 a scenario in which a patient suffered acute liver damage was presented. This problem led France and Spain to prohibit commercial distribution of camellia sinensis based products in 2003 (39). Added to this, entities such as the United States Pharmacopeia (USP) systematically reviewed the safety of green tea consumption in 2008. This review showed that cases of green tea toxicity had been reported in many countries and that the rate of adverse effects for pharmaceutical specialties such as Exolise ® was 1 in every 100,000 consumers. It suggested public health measures to prevent these figures from rising and affecting public health care systems (40).
The clinical evidence is not restricted to products such Exolise ® but applies to diverse pharmaceutical specialties currently being marketed which have had cases of toxicity attributed to them (41). As a consequence alarms are going off in the medical and health care professions around the world as this attention is turned toward preventing this health problem from evolving and advancing and to conducting the research needed to clarify the spectrum of damage and take measures for maximum prevention (42).
In Colombia, INVIMA (Instituto Nacional de Vigilancia de Medicamentos y Alimentos National Medication and Food Surveillance Institute) is responsible for monitoring and authorizing medications, including over the counter and ânaturalâ products, and the marketing of these products. This national control agency accepts food preparations that contain small amounts of green tea which are not harmful to health. However, many applications for distribution of ânaturalâ medications and products containing green tea have received negative responses from INVIMA. This review found only one ânaturalâ product which has been approved by INVIMA. This product, called Thever, advertises that it reduces cardiovascular risk and its label warns that it should not be used in patients with liver or kidney disease due to the issues discussed in this article. Despite this, there are many preparations in the gray and black markets in Colombia without INVIMA approval. Since these products do not report concentrations or net composition, consumers buying these products with the idea of obtaining beneficial effects are unaware of the components of the products (43-45). The authors of this review believe that the INVIMA should deny registration to this variety of formulas containing green tea. Unfortunately, there is no national policy to remove such unapproved products from the market.
Around the world free intake of ânaturalâ products has been reported to be associated with up to 10% frequency of liver toxicity, and products that contain green tea are at the top of the list in order of frequency (46). Therefore, we believe that measures to restrict green tea consumption would benefit the entire Colombian population. Such measures should not be limited to a special group, since the profile for liver safety and the specific type of patients who might be impaired by or who might benefit from the intake of camellia sinensis are as yet unknown.
CONCLUSIONS
Green tea toxicity is an emerging problem with multiple reports proving this association. However, there is little knowledge about mechanisms causing damage and few political and social measures taken so far to monitor distribution and intake. Most studies reviewed herein examined mechanisms of toxicity which had been evaluated in-vitro or in animals. This establishes the necessity of performing more causal studies, clinical trials and pharmacokinetic trials to evaluate the safety of green tea consumption. In the meantime, safety measures must be taken by administrative and regulatory entities to prevent liver toxicity.
1. Odom D. Camellia sinensis the tea plant. The Camellia Journal 2007; 18-20. [ Links ]
2. Weisburger JH. Tea and health: a historical perspective. Cancer Lett 1997; 114(1-2): 315-317. [ Links ]
3. Schönthal AH. Adverse effects of concentrated green tea extracts. Mol Nutr Food Res. 2011; 55(6): 874-885. [ Links ]
4. Cooper R, Morre DJ, Morre DM. Medicinal benefits of green tea: part II. Review of anticancer properties. J Altern Complement Med 2005; 11(4): 639-652. [ Links ]
5. Vuong QV, Golding JV, Nguyen M, Roach PD. Extraction and isolation of catechins from tea. J Sep Sci 2010; 33(21): 3415-3428. [ Links ]
6. Nikaidou S, Ishizuka M, Maeda Y, Hara Y, Kazusaka A, Fujita S. Effect of components of green tea extracts, caffeine and catechins on hepatic drug metabolizing enzyme activities and mutagenic transformation of carcinogens. Jpn J Vet Res 2005; 52(4): 185-192. [ Links ]
7. Dulloo AG, Duret C, Rohrer D, Girardier L, Mensi N, Fathi M, Chantre P, Vandermander J. Efficacy of a green tea extract rich in catechin polyphenols and caffeine in increasing 24-h energy expenditure and fat oxidation in humans. Am J Clin Nutr 1999; 70(6): 1040-1045. [ Links ]
8. Hursel R, Viechtbauer W, Westerterp-Plantenga MS. The effects of green tea on weight loss and weight maintenance: a meta-analysis. Int J Obes (Lond) 2009; 33 (9): 956-961. [ Links ]
9. Yang CS, Chen L, Lee MJ, Balentine D, Kuo MC, Schantz SP. Blood and urine levels of tea catechins after ingestion of different amounts of green tea by human volunteers. Cancer Epidemiol Biomarkers Prev 1998; 7(4): 351-354. [ Links ]
10. Lee MJ, Wang ZY, Li H, Chen L, Sun Y, Gobbo S, Balentine DA, Yang CS. Analysis of plasma and urinary tea polyphenols in human subjects. Cancer Epidemiol Biomarkers Prev 1995; 4(4): 393-399. [ Links ]
11. Pietta P, Simonetti P, Gardana C, Brusamolino A, Morazzoni P, Bombardelli E. Relationship between rate and extent of catechin absorption and plasma antioxidant status. Biochem Mol Biol Int 1998; 46(5): 895-903. [ Links ]
12. Unno T, Kondo K, Itakura H, Takeo T. Analysis of (-)-epigallocatechin gallate in human serum obtained after ingesting green tea. Biosci Biotechnol Biochem 1996; 60 (12): 2066-2068. [ Links ]
13. Li C, Meng X, Winnik B, Lee MJ, Lu H, Sheng S, Buckley B, Yang CS. Analysis of urinary metabolites of tea catechins by liquid chromatography/electrospray ionization mass spectrometry. Chem Res Toxicol 2001; 14(6): 702-707. [ Links ]
14. Lu H, Meng X, Li C, Sang S, Patten C, Sheng S, Hong J, Bai N, Winnik B, Ho CT, Yang CS. Glucuronides of tea catechins: enzymology of biosynthesis and biological activities. The American Society for Pharmacology and Experimental Therapeutics. Drug Metab Dispos 2003; 31(4): 452-461. [ Links ]
15. Muto S, Fujita K, Yamazaki Y, Kamataki T. Inhibition by green tea catechins of metabolic activation of procarcinogens by human cytochrome P450. Mutat Res 2001; 479(1-2): 197-206. [ Links ]
16. Leslie EM, Deeley RG, Cole SP. Multidrug resistance proteins: role of P-glycoprotein, MRP1, MRP2, and BCRP (ABCG2) in tissue defense. Toxicol Appl Pharmacol 2005; 204(3): 216-237. [ Links ]
17. Jodoin J, Demeule M, Beliveau R. Inhibition of the multidrug resistance P-glycoprotein activity by green tea polyphenols. Biochim Biophys Acta 2002; 1542(1-3): 149-159. [ Links ]
18. Zhou S, Lim LY, Chowbay B. Herbal modulation of P-glycoprotein. Drug Metab Rev 2004; 36(1): 57-104. [ Links ]
19. Kageyama M, Fukushima K, Togawa T, Fujimoto K, Taki M, Nishimura A, Ito Y, Sugioka N, Shibata N, Takada K. Relationship between excretion clearance of rhodamine 123 and P-glycoprotein (Pgp) expression induced by representative Pgp inducers. Biol Pharm Bull 2006; 29(4): 779-784. [ Links ]
20. Tanigawara Y. Role of P-glycoprotein in drug disposition. Ther Drug Monit 2000; 22(1): 137-140. [ Links ]
21. Lin LC, Wang MN, Tsai TH. Food-drug interaction of (-)-epigallocatechin-3-gallate on the pharmacokinetics of irinotecan and the metabolite SN-38. Chem Biol Interact 2008; 174(3): 177-182. [ Links ]
22. Nakagawa Y, Moldéus P, Moore GA. Relationship between mitochondrial dysfunction and toxicity of propyl gallate in isolated rat hepatoctyes. Toxicology 1996; 114(2): 135- 145. [ Links ]
23. Galati G, Lin A, Sultan AM, O'Brien PJ. Cellular and in vivo hepatotoxicity caused by green tea phenolic acids and catechins. Free Radic Biol Med 2006; 40(4): 570-580. [ Links ]
24. Kumar NS, Rajapaksha M. Separation of catechin constituents from five tea cultivars using high-speed counter-current chromatography. J Chromatogr A 2005; 1083(1-2): 223-228. [ Links ]
25. Manir MM, Kim JK, Lee BG, Moon SS. Tea catechins and flavonoids from the leaves of Camellia sinensis inhibit yeast alcohol dehydrogenase. Bioorg Med Chem 2012; 20 (7): 2376-2381. [ Links ]
26. Varughese T, Manir MM, Rahaman M, Kim JK, Lee BG, Moon SS. Tea triterpenoidal saponins from the roots of camellia sinensis has inhibitory effects against alcohol dehydrogenase. Planta Med 2011; 77(18): 2029-2036. [ Links ]
27. Inoue T, Suzuki Y, Ra C. Epigallocatechin-3-gallate induces cytokine production in mast cells by stimulating an extracellular superoxide-mediated calcium influx. Biochem Pharmacol 2011; 82(12): 1930-1939. [ Links ]
28. Gloro R, Hourmand-Ollivier I, Mosquet B, Mosquet L, Rousselot P, Salamé E, Piquet MA, Dao T. Fulminant hepatitis during self-medication with hydroalcoholic extract of green tea. Eur J of Gastroenterol Hepatol 2005; 17(10): 1135-1137. [ Links ]
29. Adeline NS. Toxicity of Green Tea Extracts Used for Weight Loss. Eur J Emerg Med 2009; 18(2): 43-48. [ Links ]
30. Molinari M, Watt KD, Kruszyna T, Nelson R, Walsh M, Huang WY, Nashan B, Peltekian K. Acute Liver Failure Induced by Green Tea Extracts: Case Report and Review of the Literature. Liver Transpl 2006; 12(12): 1892-1895. [ Links ]
31. Dueñas Sadornil C, Fabregas Puigtió S, Durández R. Hepatotoxicity due to Camellia sinensis. Med Clin (Barc) 2004; 122(17): 677-678. [ Links ]
32. Garcia Moran S, SaezRoyuela F, Gento E, Lopez Morante A, Arias L. Acute hepatitis associated with Camellia tea and Orthosiphon stamineus ingestion. Gastroenterol Hepatol 2004; 27(9): 559-560. [ Links ]
33. Pedros C, Cereza G, Garcia N, Laporte JR. Liver toxicity of Camellia sinensis dried etanolic extract. Med Clin (Barc) 2003; 121(15): 598-599. [ Links ]
34. Bonkovsky HL. Hepatotoxicity associated with supplements containing Chinese green tea (Camellia sinensis). Ann Intern Med 2006; 144(1): 68-71. [ Links ]
35. Annals of Internal Medicine. Porcel JM, Bielsa S, Madronero AB. Hepatotoxicity associated with green tea extracts. Electronic letter. May 2005. Accessed at 31 July 2009. Available at: www.annals.org on. [ Links ]
36. Federico A, Tiso A, Loguercio C. A case of hepatotoxicity caused by green tea. Free Radic Biol Med 2007; 43(3): 474. [ Links ]
37. Danan G, Benichou C. Causality assessment of adverse reactions to drugs-I. A novel method based on the conclusions of international consensus meetings: application to drug-induced liver injuries. J Clin Epidemiol 1993; 46(11): 1323-1330. [ Links ]
38. Mazzanti G, Menniti Ippolito F, Moro PA, Cassetti F, Raschetti R, Santuccio C, Mastrangelo S. Hepatotoxicity from green tea: a review of the literature and two unpublished cases. Eur J Clin Pharmacol 2009; 65(4): 331-341. [ Links ]
39. Ministerio de Sanidad y Consumo. Sistema Español de Farmacovigilancia. Hidalgo A, Manso G, Revuelta P, Sánchez L. Nota informativa de la Agencia Española del Medicamento: Suspensión de la comercialización de la especialidad farmaceútica Exolise®. Ref: 2003/03. Disponible en: www.aemps.gob.es [ Links ]
40. Sarma DN, Barrett ML, Chávez ML, Gardiner P, Ko R, Mahady GB, Marles RJ, Pellicore LS, Giancaspro GI, Low Dog T. Safety of green tea extracts: a systematic review by the US Pharmacopeia. Drug Saf 2008; 31(6): 469-484. [ Links ]
41. Dara L, Hewett J, Lim JK. Hydroxy cut hepatotoxicity: a case series and review of liver toxicity from herbal weight loss supplements. Worls J Gastoenterol 2008; 14(45): 6999-7004. [ Links ]
42. Javaid A, Bonkovsky HL. Hepatotoxicity due to extracts of Chinese green tea (Camellia sinensis): A growing concern. J Hepatol 2006; 45 (2): 334-336. [ Links ]
43. Ministerio de la Protección Social. INVIMA, Instituto Nacional de Vigilancia de Medicamentos y Alimentos. En línea www.invima.gov.co. Citado en Octubre de 2012. [ Links ]
44. Amariles P, González L, Giraldo NA. Prevalence of Self-Treatment with Complementary Products and Therapies for weight Loss: A Randomized Cross-Sectional Study in Overweight and Obese Patients in Colombia. Curr Ther Res Clin Exp 2006; 67 (1):66-78. [ Links ]
45. Amariles P, Angulo N, Agudelo J, Gaviria G. Hepatitis associated with aqueous green tea infusions: a case study. Farm Hosp 2009; 33(5): 289-291. [ Links ]
46. Avelar Escobar G, Méndez Navarro J, Ortiz Olvera NX, Castellanos G, Ramos R, Gallardo Cabrera VE, Vargas Alemán Jde J, Díaz de León O, Rodríguez EV, Dehesa Violante M. Hepatotoxicity associated with dietary energy supplements: use and abuse by young athletes. Ann Hepatol 2012; 11(4): 564-569. [ Links ]











 text in
text in