Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
Revista colombiana de Gastroenterología
Print version ISSN 0120-9957
Rev Col Gastroenterol vol.31 no.4 Bogotá Oct./Dec. 2016
Cholestasis in Pediatrics
Mónica D'Amato G. MD (1), Patricia Ruiz N. MD (2), Karen Aguirre R. MD. (2), Susana Gómez Rojas MD.(3)
(1) General Practitioner, Pediatrics Resident at the Universidad Pontificia Bolivariana in Medellín Colombia
(2) Pediatric Gastro-hepatologist at Pablo Tobón Uribe Hospital in Medellín, Colombia
(3) Pediatrics Resident at the Universidad Militar Nueva Granada in Bogotá, Colombia
Received:Â Â Â 14-01-16Â Accepted:Â Â Â 01-11-16
Abstract
Cholestasis always indicates a pathological process that can result in chronic liver dysfunction, the necessity of liver transplantation and even death. (1) Cholestasis is a process in which there is a decrease in biliary flow, histological evidence of deposition of bile pigments in hepatocytes and bile ducts, and an increase in the serum concentrations of products excreted in bile. Cholestasis can occur at any age. It is caused by alteration of the formation of bile by the hepatocytes or by obstruction of the flow in the intrahepatic or extrahepatic biliary tracts. Neonatal cholestasis occurs in the first (3) months of life with elevated serum levels of direct bilirubin, cholesterol and bile acids. (2) In our environment, the most frequent cause is idiopathic neonatal hepatitis, followed by infectious causes. Obstructive causes have the worst prognoses. Among them, the most common is biliary atresia in which progressive obliteration of the extra hepatic biliary tract, parenchymal damage and intrahepatic biliary tract cause cirrhosis and death before the patient reaches three years of age. (1-3) The prognosis improves with surgical management if it is performed within the first 45 to 60 days of life. (3) The most common presentation is a healthy full-term newborn who develops jaundice, hypocolony, firm hepatomegaly and splenomegaly between the 2nd and 6th weeks of life. The principal serum marker that supports diagnosis is elevation of gamma glutamyltransferase while ultrasound of the liver following fasting has the greatest sensitivity and specificity. The only method for confirming the diagnosis is exploratory laparotomy with direct vision of the bile duct by intraoperative cholangiography. (4)
In addition to correcting the cause and treating cholestasis, nutritional management with an adequate diet with high levels of medium chain triglycerides and fat-soluble vitamins is important for management of all patients with cholestasis. (5)
Keywords
Cholestasis, jaundice, biliary atresia, hepatitis.
Introduction
From 60% to 80% of newborns develop jaundice and one out of every 2,500 to 5,000 live births develop cholestasis. This is a process through which biliary flow decreases, there is histological evidence of deposition of bile pigments in hepatocytes and bile ducts, and serum concentrations of products excreted in bile increases. Cholestasis is caused either by alteration in the formation of bile by the hepatocytes or obstruction of the bile flow in the intrahepatic or extrahepatic biliary ducts. This which always indicates some pathological process (2) that can have sequels such as chronic hepatic dysfunction and decreased quality of life and which can result in a need for liver transplantation or even death. (1, 2, 3, 5).
In light of this, it is of vital importance that pediatric patient care staff understand neonatal cholestasis in order make early diagnoses, timely evaluations by pediatric hepatology and choose the appropriate treatment.
This review focuses on neonatal cholestasis limited to the clinical and/or biochemical onset of cholestasis in the first 3 months of life.
Abbreviations
- ALP: alkaline phosphatase
- ALT: alanine transferase
- AST: aspartate transferase
- CCK: cholecystokinin
- CF: cystic fibrosis
- CMV: cytomegalovirus
- CPK: creatinine phosphokinase
- DB: direct bilirubin
- EBV: Epstein Barr virus
- ERCP: endoscopic retrograde cholangiopancreatography
- FTN: full-term newborn
- GGT: gamma glutamyltransferase
- GIT: gastrointestinal tract
- HAV: Hepatitis A virus
- HBV: hepatitis B virus
- HCV: hepatitis C virus
- HIV: human immunodeficiency virus
- Ig: immunoglobulin
- LDH: lactate dehydrogenase
- PCR: polymerase chain reaction
- PFIC: Progressive familial intrahepatic cholestasis
- PTNB: preterm newborn
- TB: total bilirubin
- MCT: medium chain triglycerides
- PT: prothrombin time
- TPN: total parenteral nutrition
- TORCH: toxoplasmosis, rubella, cytomegalovirus, herpes virus
- PTT: partial thromboplastin time
- UTI: urinary tract infection
Definitions
When a newborn has jaundice, it must be determined whether it meets the criteria for pathological jaundice and cholestasis. (3, 5)
- Jaundice starts within the first 24 hours of life.
- It is associated with choluria, light colored stools, acholia which are symptoms of cholestasis.
- TB increases by more than 5 mg/dl/day
- TB is greater than 13 mg/dl for full term infants and greater than 15 mg/dl for preterm babies
- Jaundice lasts more than 14 days in FTNs and more than 21 days preterm infants.
- DB is greater than 2 mg/dl or 20% of TB.
Cholestasis is defined as an alteration in biliary flow that can lead to hepatic dysfunction characterized by the following factors. (3, 6)
- Elevation of DB in blood over 2 mg/dl or more than 20% of total bilirubin.
- Elevated serum cholesterol and bile acids, but these are rarely measured in Colombia
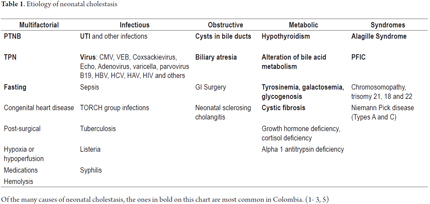
Etiology
Although neonatal cholestasis has numerous causes (Table 1), (9) the most common diagnosis is idiopathic neonatal hepatitis (6) which is a diagnosis that does not identify any specific cause. Usually it presents late together with acholia and is most common among PTNBs with antecedents of intrauterine growth retardation and abnormalities in prenatal ultrasound. The prognosis is good and generally resolves within the first year. (8)
The next most frequent causes are infections, but many authors assign infectious causes to idiopathic neonatal hepatitis in which the causative organism cannot be identified. (4)
Obstructive causes have the worst prognoses and are susceptible to change with timely surgery.
Physiopathology
Hepatobiliary system immaturity
During acquisition of biliary system competence, hepatobiliary system immaturity may develop. It also increases the likelihood of jaundice as a manifestation of a hepatic or systemic disease. (7, 10)
Infections
- Infections involve direct cholestatic action of bacterial toxins (in gram negatives, mainly E. coli), release of cytokines such as IL1 and TNF alpha which decrease transport and which are fibrogenic and can directly affect the liver. (7,8)
- Neonatal hepatitis due to congenital infections from microorganisms of the TORCH group, parvovirus B19, tuberculosis, and/or listeria. (5)
- Postnatal hepatitis due to CMV, herpes 1, 2 and 6, Coxsackievirus, Echovirus, Adenovirus, varicella (5). In infants, classical hepatitis viruses are not a cause of cholestasis except when there is liver failure due to HBV (usually after 45 days of life). (7)
Total parenteral nutrition
Prolonged use of total parenteral nutrition (TPN) for more than 15 days predisposes patients to development of cholestasis which occurs in approximately 56.7% of these patients. (11) TPN leads to cholestasis for the following reasons:
- Interruption of enterohepatic circulation during fasting which leads to decreased production and secretion of bile acids.
- Decreased contraction of the gallbladder.
- Decreased production of intestinal hormones such as CCK which is also affected by loss of hormone-producing cell mass through intestinal resection.
- Toxic components such as phytosterols used in TPN favor cholestasis, and lipid components used in TPN favor the release of proinflammatory B4 leukotrienes. (11, 12)
Extrahepatic Biliary Atresia
Extrahepatic biliary atresia is a cause of cholestasis in 35% to 41% of patients. Fibrosis and progressive obliteration of the extra hepatic biliary tract occur and lead to damage of the parenchyma and intrahepatic biliary tract. This ultimately leads to cirrhosis and death before three years of age. The prognosis improves with surgical management, provided that it is performed within the first 45 to 60 days of life. (3, 5, 7)
The prenatal form is believed to be due to genetic (but not hereditary) alterations, The perinatal form involves extrahepatic and intrahepatic inflammation and subsequent fibrosis and has sometimes been associated with other alterations in various organs (syndromic) such as situs inversus, heart disease, polysplenia and preduodenal portal veins. (3, 13)
There are three types of atresia defined according to the alteration in the hepatobiliary system. (13)
I. Choledochal atresia
IIa. Atresia of the common hepatic duct
IIb. Cystic and bile duct atresia
III. Atresia of the hepatic duct onwards towards the intrahepatic biliary tract. It is present in 90% of patients and is not correctable.
Usually, healthy full-term newborns begin to have jaundice and light colored stools between the second and sixth weeks of life. Subsequently, they develop firm hepatomegaly and splenomegaly. Between the second and third months of life there is general deterioration of infants health with portal hypertension which progresses to cirrhosis and hepatic failure. (13)
Biochemical signs of atresia include: (14)
- Elevation of GGT is the main marker.
- Elevation of TB and DB, AF, transaminases and the ALT/AST ratio.
Diagnostic Aids (3, 5, 13-14)
- Liver ultrasound after the patient fasts for four to six hours. Signs of atresia include collapsed vesicles and the triangular cord sign (hepatic hyperechoic) with a sensitivity of 85% and specificity of 100%. It is a dependent operator. (13, 15)
- Hepatic scintigraphy with prior administration of phenobarbital (5 mg/kg/day for 3 to 5 days) when there is no evidence of intestinal excretion at 24 hours. High sensitivity (83% to 100%) but low specificity (33% to 80%). (15)
- Duodenal aspirate is a diagnostic method for obstructive causes with a sensitivity similar to scintigraphy. The concentration of bilirubin in duodenal fluid obtained by catheter is analyzed. It is considered to be positive for obstruction when this concentration is lower than the concentration in serum. It is not widely used because of the time it takes and because it has not demonstrated superiority to scintigraphy. (15)
- Liver biopsies can identify cholestasis, ductal proliferation and portal fibrosis. Sensitivity of 79% to 98% depending on the training of the pathologist. (13-15)
- Exploratory laparotomy and intraoperative cholangiography can confirm only with direct view of the bile duct. (13-15)
Treatment consists of surgical rerouting of biliary flow directly to the intestine to allow drainage. This is called the Kasai technique and consists of portoenterostomy between the permeable intrahepatic biliary tract and a jejunal Roux which should be performed to decrease the possibility of reflux of the intestinal contents into the bile duct. (3, 7, 13).
Of the patients who undergo this surgery, 30% will have bile ducts partially restored, but another 30% will not have any bile duct restoration. Both groups require liver transplantation in the following months. More than 2/3 of the remaining 40% for whom total biliary flow is restored will also require liver transplants. Nevertheless, the Kasai technique can be used as a bridge that allows better nutritional and clinical conditions before transplantation. (16, 17) The best results have been reported when Kasai surgery is performed before eight weeks of life. (7)
The most common complication is acute bacterial cholangitis. (16) For this reason, antibiotics are indicated postoperatively and during the first year of life. Cholestasis may also develop and require treatment. Despite the fact that Kasai surgery treats cholestasis, (16) if the patient has already developed liver cirrhosis and presents portal hypertension and its complications, liver transplantation will probably be required. (16) However, the success of the Kasai technique should be measured by the number of patients who do not need a transplant and also by those who receive better muscular nutrition and are able to wait until they are older before undergoing transplantation. (16)
Alagille Syndrome
Alagille syndrome includes a scarcity of bile ducts due to a mutation in the JAG1 gene (located on chromosome 20) which can either be de novo or an autosomal dominant inheritance. De novo mutations are very common. It is associated with cardiac alterations (most commonly peripheral stenosis of the pulmonary artery), butterfly vertebrae, posterior embryotoxon (an eye disorder), and characteristic facial deformations including hypertelorism, bulging foreheads and prominent chins (triangular face). (3, 7)
The definitive diagnosis is made by identifying the mutation through sequencing the gene. It can be found in the laboratory through identification of hyperbilirubinemia, elevation of serum bile acids, hypercholesterolemia, elevation of GGT and transaminases (moderate). (9)
There is no specific treatment. Cholestasis should be treated and coagulation should be managed with Vitamin K supplements. Without treatment patients develop malnutrition, rickets and short stature. (9) They are at increased risk for early development of hepatocellular carcinoma, and 50% of children will require transplantation. (9)
Progressive familial intrahepatic cholestasis (PFIC)
Progressive familial intrahepatic cholestasis (PFIC) accounts for 10% to 15% of cholestasis cases in children. (18) These are de novo or inherited autosomal recessive mutations in the genes encoding the canalicular membrane transport systems of hepatocytes that alter formation of bile and result in poor secretion of bile salts. (3, 7)
There are three types of PFIC mutations on chromosomes 18q21-22, 7q21. Types 1 and 2 are characterized by jaundice in the neonatal period, intense pruritus that is not related to the degree of jaundice, firm hepatomegaly, splenomegaly and nutritional deterioration. Type 3 can develop in older children. (18) GGT is decreased in Types 1 and 2, but elevated in Type 3. (18)
A diagnosis is made by detection of the genetic mutation, and cholestasis must be treated. (18) Patients with Type 1 PFIC who develop cirrhosis can benefit from liver transplantation. Pruritus can alter the quality of life. (18)
PFIC stimulates the production of lithiasis and formation of thicker bile which obstructs the bile ducts. (1-5)
- Medications: furosemide or ceftriaxone
- Hemolysis: isoimmunization or other cause
- Surgery
Hormone Alterations
Thyroid, growth hormone and cortisol deficiencies cause hepatocellular malfunctions. (1-5)
Alpha 1 Antitrypsin Deficiency
Alpha 1 antitrypsin deficiency occurs as the result of a mutation on chromosome 14 that leads to alteration in production and accumulation of α1-antitrypsin by hepatocytes. A history of low birth weight is common, the deficiency manifests in the first months of life with low levels of alpha 1 antitrypsin in the blood (less than 100 mg/dl). It is confirmed by genetic study. (7)
Inborn Errors of Bile Acid Metabolism
Absence of synthesis of primary biliary acid with intense early onset pruritus indicates inborn errors of bile acid metabolism. (8)
- Deficiency of 3-Ã-hydroxy-Î-5C27-steroid dehydrogenase/isomerase.
- Deficiency of Î-4-3-oxosteroid 5-Ã-reductase.
- Deficiency of dihydroxyalkanoic 24, 25 cleavage enzyme.
Cystic Fibrosis
Cystic fibrosis is an autosomal recessive disease characterized by alteration in the movement of sodium and chlorine transmembrane into exocrine and endocrine glands due to a mutation of the CFTR (CF transmembrane conductance regulatory protein) gene that alters the normal functioning of various organs including the lungs and pancreas. It also affects the intrahepatic biliary tract and produces focal biliary cirrhosis that causes neonatal cholestasis in 2% of patients. The presence of cholestasis in a CF patient is a phenotype with a poor prognosis. (19, 21, 20)
Metabolic Diseases (12)
- Tyrosinemia appears as cholestasis and prolongation of clotting time that are not corrected after initial administration of vitamin K. It is due to deficiencies of succinyl and acetone with elevation of serum tyrosine and phenylalanine. (24)
- Galactosemia appears as malnutrition with hypoglycemia with reducing sugars in urine with lactose intake. It is diagnosed by measuring levels of galactose 1-P-uridyltransferase in red blood cells (without prior transfusion of red blood cells). (24)
- Neonatal hemochromatosis is indicated by hepatomegaly, cholestasis, saturation elevation of transferrin and ferritin and is confirmed by hepatic biopsy. (24)
- Wolman's disease is indicated by diarrhea, dyslipidemia, adrenal calcifications and cholestasis and is diagnosed by measuring acid lipase in a skin biopsy. (24)
Clinical Manifestations (24)
- Jaundice: Yellow coloration of the skin
- Choluria: Yellow hyperpigmentation, orange-brown, urine
- Hypocholia: Light colored stools
- Acholia: Total stool discoloration (white stools)
- Hepatomegaly
- Splenomegaly
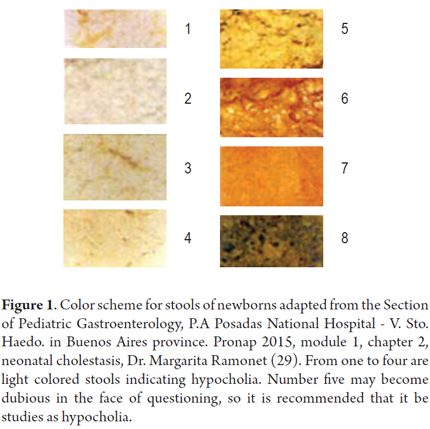
The figure below shows the schematic relation of different colors of stools. Numbers one through four are light colored stools. Number 5 looks yellow but may cause doubt on the questioning, so it is recommended that it be studied as a hypocholia (Figure 1).
Laboratory
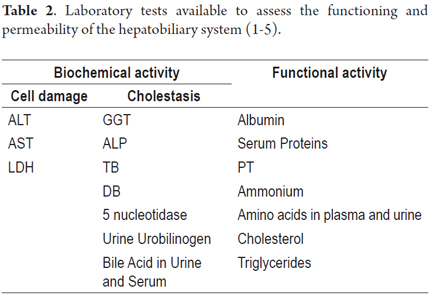
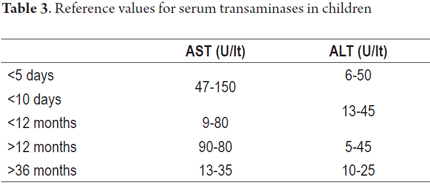
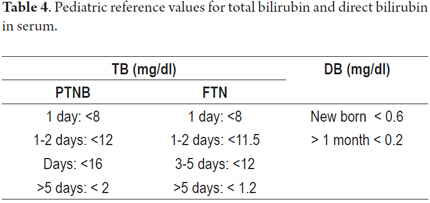
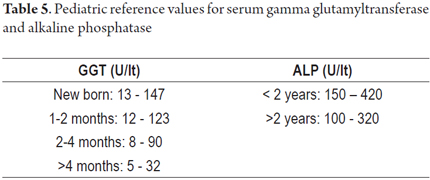
The tests shown in Tables 2, 3, 4, 5 and 6 are available for assessment of the functioning and permeability of the hepatobiliary system. Each of them has reference values which vary according to the patient's age. (22, 23)
What Tests Should Be Used?
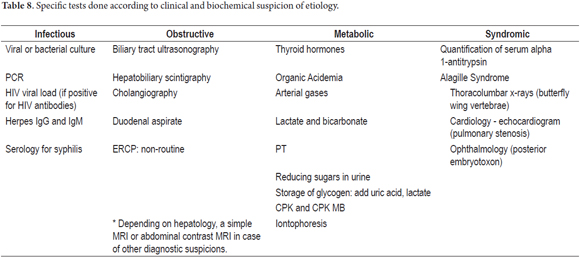
Although the recommended scheme for diagnosis is given in Tables 7 and 8, it should be noted that all patients should be evaluated for pediatric hepatology. (3, 5, 13).
Levels of fat-soluble vitamins A, D and E should be checked after 2 months of life which is when decreased serum levels can begin to be measured since these levels may have previously been supplemented by maternal inputs. (22, 23)
Treatment of Cholestatic Syndromes
Is the management of patient support, apart from managing its cause when it can be treated.
Drugs that Promote Biliary Flow
Ursodeoxycholic acid is the drug of choice. It is administered in three oral doses per day totaling 10-20 mg/kg/day. By increasing hydrophilic bile acids that displace hydrophobic acids that are hepatotoxic it has a cytoprotective and stabilizing effect on the hepatocyte membrane. (8, 26)
Other drugs to be considered by hepatology are phenobarbital (3-5 mg/kg/day oral, divided into 3 doses) and resincolestiramina (0.25-0.5 g/kg/day, in 2 doses before meals). (25) These two drugs are not the first choice. Phenobarbital increases the independent biliary flow of bile salts, so that it is not clear whether its main effect is action on biliary flow or sedative effect. (25) Cholestyramine is an anion exchange resin which blocks absorption of bile salts and disrupts the whole hepatic circulation. The dose required for this effect is very high and is not well tolerated by newborns. (8)
Nutrition
Diet, vitamin supplementation and formula choices should be agreed upon with a pediatric nutritionist. (25, 26, 28).
Diet
Suggest to the mother that she first offer a formula with MCT and then the maternal breast. The diet should have the following characteristics. (27)
- Hypercaloric: 120 to 150% of the needs for the child's age.
- Normal protein (2 to 3 g/kg/day).
- Hypoproteic only in cases of suspected metabolopathy (1 to 2 g/kg/day).
- Normal fat enriched with MCT from 40% to 70% of the total dietary fat.
- Liposoluble vitamins: the ideal is to have control of serum levels, but the results should not be expected to start supplementing them (1, 6).
- Vitamin K: 5-10 mg oral; Intramuscular or intravenous 0.2 to 0.3 mg/kg/day, 2-3 times per week.
- Vitamin E: 75-100 mg/day, oral or 0.2-0.5 mg/kg/day parenteral or 50 U/kg/day.
- Vitamin D3: 1,200-5,000 IU/day, or 500 U/kg/day.
- Vitamin A: 5,000-10,000 IU/day or 1500 U/kg/day.
Depending on development of nutritional status and ideally with serum levels:
- Supplement water-soluble vitamins 1-2 times according to the recommendations for the age.
- Calcium supplement: 50 mg/kg/day. (11, 27, 28)
REFERENCES
1. Balistreri W. Neonatal cholestasis. J Pediatr. 1985;106:171–84. [ Links ]
2. Suchy FJ. Approach to the infant with cholestasis. En: Suchy FJ, Sokol RJ, Balistreri WF, editors. Liver diseases in children, 2nd ed. Philadelphia: Lippincott, Williams and Wilkins, 2001; p. 187-194. [ Links ]
3. Götze T, Blessing H, Grillhösl C, Gerner P, Hoerning A. Neonatal cholestasis – differential diagnoses, current diagnostic procedures, and treatment. Front Pediatr. 2015;3:43. [ Links ]
4. Moyer V, Freese DK, Whitington PF, Olson AD, Brewer F, Colletti RB, et al. Guideline for the evaluation of cholestatic jaundice in infants: recommendations of the north American society for pediatric gastroenterology, hepatology and nutrition. J Pediatr Gastroenterol Nutr. 2004;39(2):115–28. doi:10.1097/ 00005176-200408000-00001. [ Links ]
5. NASPGHAN. Guideline for the Evaluation of Cholestatic Jaundice in Infants: Recommendations of the North American Society for PediatricGastroenterology, Hepatology and Nutrition. J Pediatr Gastroenterol Nutr 2004;39(2):115-128. [ Links ]
6. Rodríguez Miguélez JM, Figueras Aloy J. Ictericia neonatal. En: Protocolos de Neonatología. Asociación Española de Pediatría. Sociedad Española de Neonatología. 2008; p. 371-373. [ Links ]
7. Frauca E, Remacha G, Muñoz B. Colestasis en el lactante. Tratamiento en Gastroenterología, Hepatología y Nutrición Pediátrica. SEGHNP. Madrid. Ed 3ª. Editorial Ergon. 2012, 44: 575-590. [ Links ]
8. Cera D, Vera J, Vargas M, Rodríguez F, Troncoso G. Ictericia colestásica del lactante y del lactante menor. En: Vera J, Suárez M, Briceño G. Guías de gastroenterología y nutrición pediátrica basadas en la evidencia. Bogotá. Editorial Distribuna; 2006;11: 265-294. [ Links ]
9. Mieli-Vergani G, Howard ER, Mowat AP. Liver disease in infancy: A 20-year perspective. Gut. 1991; 32(Suppl):123-8. [ Links ]
10. Hondal E. Colestasis del recién nacido y del lactante. Rev Cubana de Pediatr. 2010;82(3): 49-61. [ Links ]
11. Cavicchi M, Beau P, Crenn P, Degott C, Messing B. Prevalence of liver disease and contributing factors in patients receiving home parenteral nutrition for permanent intestinal failure. Ann Intern Med 2000;132:525-32. [ Links ]
12. Kaufman S. Prevention of parenteral nutrition – associated liver disease in children. Pediatr transplantation 2002;6(1):37-42. [ Links ]
13. Chardot C. Biliary atresia. Orphanet J Rare Dis. 2006;1(28):1-9. [ Links ]
14. Kim M, Park Y, Han S, Yoon C, Hwang E, Chung K. Biliary atresia in neonates and infants: triangular area of high signal intensity in the porta hepatis at T2-weighted MR cholangiography with US and histopathologic correlation. Radiology 2000;215 (2): 395-401. [ Links ]
15. Donat Aliaga E, Polo Miquel B, Ribes-Koninckx C. Atresia de vías biliares. An Pediatr. 2003;58(2):168-73. [ Links ]
16. Lien TH, Chang MH, Wu JF, Chen HL, Lee HC, Chen AC, et al. Effects of the Infant Stool Color Card Screening Program on 5-Year Outcome of Biliary Atresia in Taiwan. Hepatology. 2011;53(1):202-208. [ Links ]
17. Petersen C. Pathogenesis and treatment opportunities for biliary atresia. Clin Liver Dis 2006;10:73-88. [ Links ]
18. Ortíz-Rivera CJ. Colestasis intrahepatica familiar progresiva. Gastrohnup. 2015;17(2):117-125. [ Links ]
19. Guía de práctica clínica para la prevención, diagnóstico, tratamiento y rehabilitación de fibrosis quística. Guía No. 38. Bogotá, Colombia: Ministerio de Salud de Colombia; 2014. [ Links ]
20. Bush A, Alton E, Davies J, Griesenbach U, Jaffe A. Cystic Fibrosis in the 21st Century. Prog Respir Res. Basel, Karger 2006;34:2–10. [ Links ]
21. Rowland M, Bourke B. Liver disease in cystic fibrosis. Curr Opin Pulm Med 2011;17(6):461–466. [ Links ]
22. Batres A, Maller E. Laboratory assessment of liver function and injury in childre. At Suchy F, Sokol R, Balistreri W. Liver Diseases in children. Ed Lippincott Williams and Wilkins; 2001;7: 155-169 [ Links ]
23. Arcara K. Blood Chemistries and Body fluids. At The Harriet Lane handbook. Ed Elsevier. Edition 19th; 2011; 27: 639-647. [ Links ]
24. Leonard JV, Morris AA. Diagnosis and early management of inborn errors of metabolism presenting around the time of birth. Acta Paediatr. 2006;95(1):6. [ Links ]
25. Romero M, Godínez T, Yescas B, Fernández C, Echániz A, Reyna R. Efectividad del ácido ursodesoxicólico versus fenobarbital para el tratamiento de la colestasis en prematuros. Ensayo clínico cruzado. Bol Med Hosp Infant Mex. 2010; 67(5):422-429. [ Links ]
26. Smart K, Alex G, Hardikar W. Feeding the Child With Liver Disease: A Practical Guide: Chronic Liver Disease. J Gastroenterol Hepatol 2011;26(5):810-815. [ Links ]
27. Nightingale S, et al. Optimización del manejo nutricional de los niños con enfermedad hepática crónica. Pediatr Clin N Am 2009;56: 1161-1183. [ Links ]
28. Lee Ng V, Balistreri W. Treatment options for chronic cholestasis in infancy and childhood. Curr treat Options Gastroenterol 2005;8: 419-430. [ Links ]
29. Ramonet M. Colestasis neonatal. En: Programa Nacional de Actualización Pediátrica 2015. Buenos Aires: Sociedad Argentina de Pediatría; 2015. p. 43-72. [ Links ]











 text in
text in