Introduction
Collagenous gastritis is a rare type of gastritis, with fewer than 300 cases reported in the literature. It is characterized by the subepithelial deposition of a thick collagen band and a variable presence of inflammatory infiltrate in the lamina propria1,2. Due to its rare presentation profile, epidemiological data are scarce, and most information found in the literature corresponds to case reports. Some publications mention the association of collagenous gastritis with other pathologies, such as collagenous colitis, lymphocytic colitis, celiac disease, and other autoimmune diseases. However, the pathophysiology, prognosis, and treatment have not been precisely clarified.
Case Report
The case involves a 15-year-old female patient, the daughter of non-consanguineous parents; the father had a history of splenomegaly of uncertain etiology, with no other significant medical history. She was under follow-up by pediatric hematology for a case of iron deficiency anemia (microcytic hypochromic) two years prior to the gastroenterology consultation. Clinically, she reported symptoms of pica (excessive consumption of ice). She received iron salt supplementation on several occasions with improvement in erythrocyte indices, but the anemia recurred when supplementation was discontinued.
She had a history of irregular menstrual cycles, so a gynecological etiology for the anemia was suspected; this was ruled out due to lack of improvement with hormonal management and normal studies. Hematology investigated and ruled out hemolysis, primary and secondary hemostasis disorders, as well as platelet dysfunction (Figure 1). For a period, the patient remained asymptomatic and under hematology control, but the symptoms of pica, asthenia, and lethargy reappeared, and moderate anemia with very low ferritin was again found. A fecal occult blood test was requested, management with iron salts was restarted, and the patient was referred for evaluation by pediatric gastroenterology.
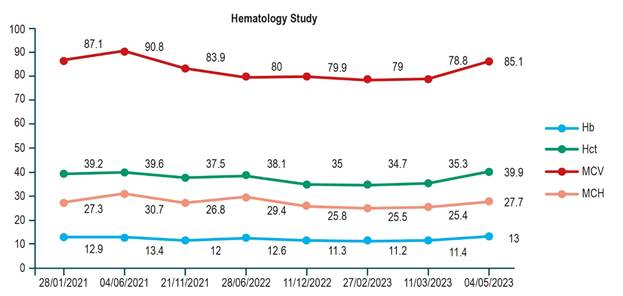
Image property of the authors.
Figure 1 Hemoglobin curve. Hb: hemoglobin; MCH: mean corpuscular hemoglobin; Hct: hematocrit; MCV: mean corpuscular volume.
During the gastroenterology consultation, the patient reported long-standing abdominal pain associated with the anemia. On physical examination, the only relevant finding was an anal fissure. Anthropometric measurements were normal for her age (weight: 49.1 kg; height: 157.5 cm; height for age [H/A]: P24, -0.7 standard deviation [SD]; body mass index [BMI]: P39, -0.29 SD). During the consultation, she provided other paraclinical results: normal thyroid function and normal abdominal ultrasound. Further workup was considered to rule out inflammatory bowel disease, taking into account the persistence of anemia, systemic symptoms (asthenia and lethargy), and the finding of an anal fissure (Table 1).
Table 1 Laboratory Results
| Laboratory | Test Value | Reference Range |
|---|---|---|
| Hemoglobin (g/dL) | 11.4 | 12.3-15.3 |
| MCV (fL) | 78.8 | 80-100 |
| Iron (μg/mL) | 35.68 | 30-109 |
| IgA (g/L) | 1.81 | 0.4-2.93 |
| Gliadin Antibodies IgG (U/mL) | 71.3 | Positive >12 |
| Gliadin Antibodies IgA (U) | 28.7 | Positive >20 |
| Transglutaminase Antibodies IgA (U/mL) | 2 | Positive >10 |
| Fecal Calprotectin (μg/g) | 798.4 | Elevated >200 |
| Endomysial Antibodies IgA | Negative | >1/5 |
| Endomysial Antibodies IgG | Negative | >1/5 |
IgA: immunoglobulin A; IgG: immunoglobulin G; MCV: mean corpuscular volume. Table prepared by the authors.
An upper gastrointestinal endoscopy (UGIE) (Figure 2) and a total ileocolonoscopy were performed. The significant finding was a cobblestone-appearing mucosa in the gastric body with severely thickened folds and a spared antrum with usual appearance. The other segments (esophagus, duodenum, ileum, and colon) also appeared normal. Due to the significant elevation of calprotectin (798 μg/g), an enterography MRI was requested, which was normal.
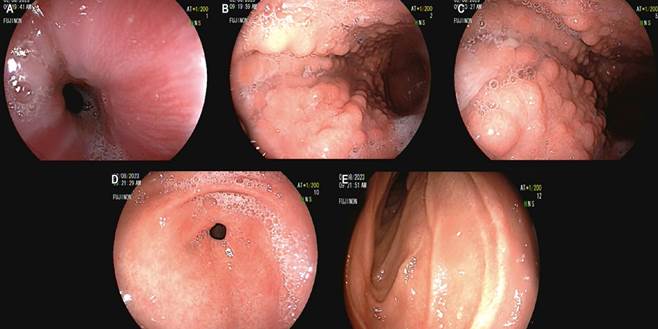
Image property of the authors.
Figure 2 Esophagogastroduodenoscopy. A. Normal mucosa. B and C. Body with thickened folds and cobblestone-appearing mucosa. D. Normal antrum. E. Normal duodenum.
The initial report of the gastric mucosa biopsies (Figure 3) described moderate, diffuse, chronic, non-active, non-atrophic gastritis with an absence of Helicobacter pylori. Given the severe mucosal alteration observed during endoscopy and the unusual finding of exclusive involvement of the body without findings in the antrum, a second histological review was requested. Special stains were performed and reported an increased chronic inflammatory infiltrate in the lamina propria of the body and patchy thickening of the subepithelial collagen with entrapped lymphocytes, findings compatible with collagenous gastritis, a diagnosis that explained the patient’s clinical picture. The remaining biopsies from the different segments, including the antrum and intestine, were normal.
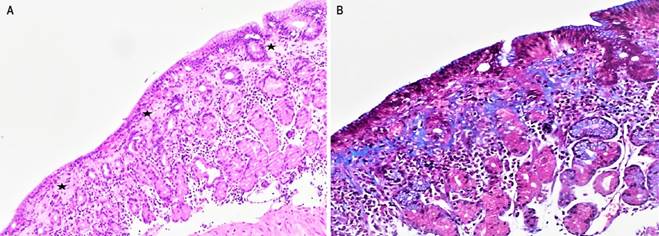
Image property of the authors.
Figure 3 Histological review with stains. A. Hematoxylin and eosin (20X). Body mucosa with an irregular subepithelial collagen band (stars) approximately 80 to 180 microns thick, surrounding some capillary vessels and accompanied by chronic inflammation, with occasional intraepithelial lymphocytes (less than 25 per 100 epithelial cells). B. Masson’s trichrome (20X). Highlights the collagen band in blue, extending irregularly to the middle third of the lamina propria.
The patient was also evaluated by pediatric rheumatology, where further workup for autoimmune gastritis and Immunoglobulin G4 (IgG4)-related disease as a cause of the collagenous gastritis was considered; these results were normal. Treatment was initiated with a high-dose proton pump inhibitor (PPI). Due to the elevated fecal calprotectin and positive anti-gliadin IgA antibodies, a trial of wheat restriction was considered for a possible associated gluten sensitivity. Following this, there was an improvement in abdominal pain and resolution of the anemia.
Discussion
Collagenous gastritis is a rare disease. A study conducted in Sweden reported an incidence of collagenous gastritis in pediatrics of 0.25/100,000 inhabitants and a prevalence of 2.1%2. There are no other epidemiological data1,2. In most pediatric cases, mucosal inflammation is restricted to the stomach, unlike in adults, where the intestine or colon is frequently also affected.
Clinically, in the pediatric population, most cases present with severe iron deficiency anemia or recurrent abdominal pain; however, a study from Italy, which compiled 71 pediatric cases of collagenous gastritis from the literature, mentioned other symptoms such as dyspepsia, nausea, dysphagia, diarrhea, constipation, and weight loss. In adults, the predominant symptoms are diarrhea and malabsorption, considering the association with collagenous colitis2-4.
Collagenous gastritis is histologically characterized by the presence of a subepithelial collagen band >10 μm associated with a chronic inflammatory infiltrate in the lamina propria. The inflammatory infiltrate can vary, with a predominance of lymphocytes (pattern similar to lymphocytic gastritis), plasma cells, and eosinophils (eosinophil-rich pattern), the latter described more frequently in pediatric presentations. However, in our case, the infiltrate was mixed among lymphocytes, plasma cells, and eosinophils, without a predominance of any cell line, and the intraepithelial lymphocytosis was discrete. An atrophic gastritis pattern, more common in adults, has also been described.
Collagenous gastritis with a predominance of eosinophilic infiltrate is part of the differential diagnosis of eosinophilic gastritis; therefore, eosinophilia should prompt a search for a subepithelial collagen band when present5. Several available studies describe the persistence of the subepithelial collagen band during follow-up, which has been found up to 10 years after the initial biopsy. To detect this collagen band, some immunohistochemical studies (tenascin, type IV collagen) have been described as potentially useful in the histological diagnosis of collagenous gastritis6 and may have superior sensitivity to hematoxylin-eosin and trichrome for its detection; however, they are not routinely available in pathology laboratories, and studies proving their superiority over histochemistry are lacking.
The pathophysiology of this condition is still unknown; however, the most accepted theory considers the subepithelial collagen deposition to be a repair process in response to chronic inflammation caused by intraluminal toxic or infectious agents3. Other potential causes of this inflammation to consider are allergic reactions, medications, and an increase in IgG4-type plasma cells7. Another well-regarded hypothesis is the presence of primary vascular anomalies that increase vascular permeability, resulting in protein extravasation and collagen deposition, in addition to a fibrotic reaction secondary to a primary inflammatory process in susceptible individuals4. Although the etiology of collagenous gastritis is unclear, some studies demonstrate that the collagen isolated in the subepithelial bands corresponds mainly to types III and VI collagen8. Furthermore, it is considered that the collagen band present in both the gastric and colonic mucosa is a consequence of the same pathogenic factor in susceptible patients, and the host response would be greater at the gastric level in both the pediatric population and the adult population with colonic predominance9.
Regarding endoscopic findings, mucosal edema and a nodular appearance in the body, antrum, and gastric fundus are most frequently found; hypertrophic mucosa with erythema in the gastric body; and nodular changes in the bulb, which may raise suspicion for other diseases. Other described endoscopic findings are polypoid lesions and hyperplastic, coarse folds2,3.
Case reports in the literature document the association of collagenous gastritis with celiac disease, as well as other autoimmune disorders such as collagenous enteritis and colitis, Sjögren’s syndrome, systemic lupus erythematosus, rheumatoid arthritis, autoimmune thyroiditis, type 1 diabetes mellitus, and common variable immunodeficiency. These entities are more associated with the adult phenotype of collagenous gastritis than the pediatric one, although the reason for this association is unclear. In patients diagnosed with collagenous gastritis, studies should be considered to exclude these pathologies10-12.
A specific therapy has not yet been established, although most pediatric patients are treated with iron supplementation and high-dose PPIs. In refractory cases, elimination diets, systemic and topical steroids, and local anti-inflammatories (mesalamine) have been used, with clinical improvement in most cases13.
In our patient’s case, she presented with chronic anemia associated with abdominal pain, asthenia, and lethargy. These are symptoms that can be confused with various gastrointestinal pathologies, but the persistence of chronic anemia without a clear etiology led to endoscopic studies. These revealed the severe alteration with hypertrophic and nodular-appearing mucosa exclusively in the body, as reported in the literature, and at the histopathological level, the presence of patchy subepithelial collagen with entrapped lymphocytes, which diagnosed collagenous gastritis. Since it is mentioned that this pathology may be related to celiac disease, pertinent studies were performed, which found positive anti-gliadin IgG antibodies. While these are not diagnostic of celiac disease, they suggest possible gluten sensitivity. Therefore, a therapeutic trial of gluten restriction was considered, in addition to management with a high-dose PPI, which produced an improvement in the patient’s symptoms.
Conclusion
A specific treatment or long-term monitoring recommendations for this disease are not yet established in the literature, considering the possibility of association with autoimmune pathology or malignant potential in the context of chronic inflammation. Although collagenous gastritis is a rare disease, it should be considered in the differential diagnoses to be evaluated in children with gastrointestinal symptoms and persistent iron deficiency anemia. Similarly, at the histological level, special stains can be useful to highlight the characteristic thickened subepithelial collagen band associated with this entity.











 text in
text in 



