Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
Agronomía Colombiana
Print version ISSN 0120-9965
Agron. colomb. vol.32 no.2 Bogotá May/Aug. 2014
https://doi.org/10.15446/agron.colomb.v32n2.43265
http://dx.doi.org/10.15446/agron.colomb.v32n2.43265
1 Nataima Research Center, Corporación Colombiana de Investigación Agropecuaria (Corpoica). Espinal (Colombia). omontenegro@corpoica.org.co
2 Department of Agronomy, Faculty of Agricultural Sciences, Universidad Nacional de Colombia. Bogota (Colombia).
Received for publication: 2 May, 2014. Accepted for publication: 30 July, 2014.
ABSTRACT
This study was conducted to assess fruit and seed yield, oil content and oil composition of Jatropha curcas fertilized with different doses of nitrogen and potassium in Espinal (Tolima, Colombia). The yields ranged from 4,570 to 8,800 kg ha-1 of fruits and from 2,430 to 4,746 kg ha-1 of seeds. These yields showed that the fertilizer dose of 150 kg ha-1 N + 120 kg ha-1 K increased fruit production by 92% and seed production by 95%, which represents an increase of about 100% in oil production, which increased from 947 to 1,900 kg ha-1. The total oil content in the seeds ranged from 38.7 to 40.1% (w/w) with a high content of the unsaturated fatty acids oleic (> 47%) and linoleic acid (> 29%). The highest content of oleic acid in the seed oil was from the unfertilized control plants and plants with an application of 100 kg ha-1 of N and 60 kg ha-1 of K, with an average of 48%. The lowest content of oleic acid was registered when a low dose of nitrogen and a high level of potassium were applied at a ratio of 1:2.4 and doses of 50 kg ha-1 N + 120 kg ha-1 K, respectively. Low contents of the saturated fatty acids palmitic (13.4%) and stearic (7.26%) were obtained, making this oil suitable for biodiesel production. The nitrogen was a more important nutrient for the production and quality of oil in J. curcas than potassium under the studied conditions of soil and climate.
Key words: vegetable oils, fatty acids, biodiesel, Euphorbiaceae, fertilization.
RESUMEN
Este estudio se realizó para evaluar la respuesta a la fertilización con N y K en la producción de frutos, semillas, contenido y composición de aceite de Jatropha curcas, en El Espinal (Tolima, Colombia). Las producciones oscilaron entre 4.570 y 8.800 kg ha-1 de frutos y las semillas entre 2.430 y 4.746 kg ha-1. Estos rendimientos mostraron que la fertilización con dosis de 150 kg ha-1 de N + 120 kg ha-1 de K, incrementó la producción de frutos en 92% y la de semillas en 95%, lo cual representa un aumento del 100% en la producción de aceite, desde 947 a 1,900 kg ha-1. Las semillas presentaron un contenido de aceite entre el 38,7 al 40,1% (w/w), con altos contenidos de ácidos grasos insaturados oléico (>47%) y linoléico (>29%). Los mayores contenidos de ácido graso oleico se obtuvieron en los aceites de las semillas provenientes de plantas testigo sin fertilización y en plantas con la aplicación de 100 kg ha-1 de N y 60 kg ha-1 de K, con promedio del 48%. El menor nivel del ácido oleico se presentó cuando se aplicó un nivel bajo de nitrógeno y alto de potasio, en relación 1:2,4, con dosis de 50 kg ha-1 de N + 120 kg ha-1 de K. Los contenidos de ácidos grasos saturados palmítico y esteárico fueron bajos, con 13,4 y 7,26%, respectivamente, lo que indica que el aceite obtenido resulta adecuado para la producción de biodiesel. El nitrógeno resultó más importante en la producción y calidad de aceite que el potasio, en las condiciones de clima y suelos estudiados.
Palabras clave: aceites vegetales, ácidos grasos, biodiesel, Euphorbiaceae, fertilización.
Introduction
Global energy consumption depends on fossil fuel sources, especially from the petrochemical industry, coal, and natural gas, in addition to the sources provided by hydroand nuclear power plants. In recent decades, emissions of greenhouse gases have caused great concern due to their impact on the environment, thus, increasing the search for renewable energy sources. The production of biodiesel from vegetable oils is one of the possible options for reducing environmental pollution and decreasing dependency on fossil fuels (Verrastro and Ladislaw, 2007; Achten et al., 2008). The use of plants as a source of energy to replace fuel is an important economical alternative; however, it is highly controversial because it can affect the food security of a country due to the substitution of food crops in some areas (Harinder and Becker, 2009). In order to avoid competition with food crops, non-food crops used as sources for biofuels must be adapted to marginal and non-productive lands.
The oils extracted from seeds or fruits of non-edible crops serve as raw materials for biodiesel production; among the non-edible oilseed species used for biodiesel production are crops such as Jatropha curcas, Pongamia pinnata, Madhuca indica, Ricinus communis and Cynara cardunculus (Sengo et al., 2010; Bankovic-Ilic et al., 2012). In recent decades, the cultivation of J. curcas. has generated great interest since it can serve as raw material for the production of oils for biodiesel (Brittaine and Lutaladio, 2010). J. curcas (Euphorbiaceae) is a perennial shrub that reaches a height of up to 5 m and grows well in tropical and subtropical climates in areas of low precipitation and poor soils, and has a productive period that may exceed 30 years. This plant is native to Central America and was extended to Asia and Africa by Portuguese traders in the 16th century through the islands of Cape Verde and Guinea-Bissau as a medicinal plant (Brittaine and Lutaladio, 2010); currently, it has widespread exploitation in many tropical and subtropical regions of the world.
J. curcas produces flowers in inflorescences; the fruits are ovoid to ellipsoidal in shape, yellow when ripe and usually contain three black seeds, about 2 cm long and 1 cm wide. The black seed coat protects the endosperm which contains between 27 and 43% non-edible oil. The first commercial uses of J. curcas oil were reported in Lisbon as the artisanal production of soap and fuel for oil lamps, having been brought from Cape Verde industries (Kumar and Sharma, 2008). Currently, J. curcas is used as a commercial crop for the production of biodiesel or as a cover crop for soil protection and soil recovery. J. curcas oil can be easily converted into biodiesel through a process of trans-esterification, generating a biofuel that meets US and European Union standards of quality (Azam et al., 2005; Achten et al., 2008).
J. curcas has been reported as a drought-resistant species, well adapted to arid and semi-arid climates, capable of maintaining and restoring soil fertility in regions with degraded soils and, therefore, is a promising crop for preventing desertification and improving socio-economic development in poor areas of rural economy (Foidl et al., 1996; Francis et al., 2005; Zahawi, 2005). The production of J. curcas seeds can range from 0.4 to 12 t ha-1 per year (Achten et al., 2008). Although the plant survives under soil and climate conditions with severe limitations and is well adapted to marginal areas, its productivity is greatly increased if planted in well-drained soils of medium fertility, under more favorable climatic conditions and suitable agronomic management (Achten et al., 2010). Also, the control of pests and diseases is not currently considered as a limitation to achieve acceptable yields (Openshaw, 2000).
J. curcas has low competition with traditional crops for production areas. As many species of the Euphorbiaceae family, this plant contains phytotoxins, mainly terpenoids and alkaloids (Sauerwein et al., 1993; Makkar and Becker, 2009). All products, including oil, seed cake, leaves, and stems are toxic for humans and animals. Although seed cake from J. curcas contains 50-62% proteins, it is not suitable for animal consumption due to the presence of glycoprotein curcin or jatrophin, a toxin that inhibits protein synthesis (Pal et al., 2012). J. curcas contains two types of curcin (curcin and curcin-L) which were detected in all plant parts including leaves, seed cotyledons, stems, and roots (Pal et al., 2012). However, several studies have shown that the main responsible agents for the toxicity of J. curcas are phorbol esters that are tetracyclic diterpenoids found in the seeds at high concentrations, products that are potentially harmful to health due to their ability of tumor promoting activity (Goel et al., 2007; Abhilash et al., 2011). Also, the seeds and seed cake contain toxic and antinutritional compounds, including flavonoids vitexin, isovitexin, and 12-deoxyl-16-hydroxyphorbol (Aregheore et al., 2003). To detoxify seed cake for usage as a livestock feed, several biological, thermal, and chemical treatments have been proposed (Aregheore et al., 2003; Phasukarratchai et al., 2012; Phengnuam and Suntornsuk, 2013). However, seed detoxification is not a simple process and is unlikely to be economically viable on a small scale.
In Colombia, studies on this crop are still scarce and preliminary; however, there has been an expansion of cultivation of J. curcas in Colombia, attaining a high presence in the areas of Guajira, Llanos Orientales and some areas in the warm valleys of the upper Magdalena (Campuzano, 2008). This increase in the cultivated area has generated a demand for information on the fertilizer needs of this plant for macro- and micronutrients; however, this information is currently absent in Colombia.
The fertilizer recommendations of a crop depend on the nutritional demands of the plants in the phases of vegetative and reproductive development, the efficient use of the applied fertilizers, and the nutrients supplied by the soil (ICA, 1992). Therefore, the aim of this study was to determine the response in the production of fruits and seeds, seed oil content and oil quality of the J. curcas M-3 genotype fertilized with different doses of nitrogen and potassium in the soil and climate conditions of Espinal, Tolima, Colombia.
Materials and methods
Experimental site conditions
The study was established at the Nataima Research Center of Corpoica (Espinal, department of Tolima, Colombia) located at 4°11'14" N and 74°57'22" W, with an altitude of 371 m, average temperature of 28°C, relative air humidity of 70%, and annual precipitation of 1,270 mm; according to Holdridge (1967), this area is classified as tropical dry forest (bs-T).
During the establishment of the experiment in the field in May, June, and July of 2012, an average temperature of 27.8°C and relative air humidity above 75% were registered; the transplanting in the field was carried out in May 2012 taking into consideration the start of the rainy season. Comparing the average rainfall for the region in this period with the averages of the last 30 years, the rainfall was slightly lower in the season of crop establishment, accentuating the dry period between the months of June and September 2012 (Tab. 1). However, this factor might not be considered limiting for normal crop development because the experimental area had supplemental irrigation and the application of two surface irrigations was necessary, with each one equivalent to 35 mm of precipitation. During the first half of 2013, rainfall higher than the historical average occurred in March, April and May; during this period, the plants had an increased production of new vegetative and reproductive structures. For the second half of 2013, rainfall below historical averages was recorded during the months of September and October (Tab. 1).
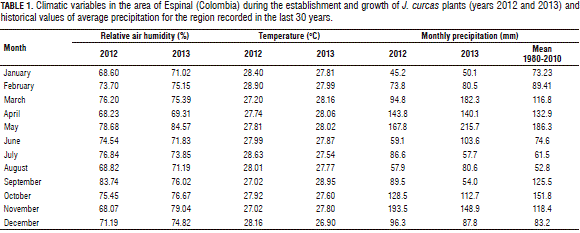
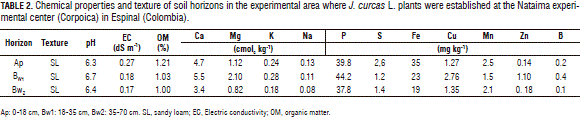
The soil, where the plants were established, had an arable depth that exceeded 1 m, sandy loam texture, with good external drainage. The content of total soil nitrogen was low, with nitric and ammonia contents that corresponded to 2.26 and 2.03 mg kg-1, respectively. The main properties of the soil are presented in Tab. 2.
Plant growth and fertilization treatments
The study started in February 2012 with plant establishment in the nursery. The seeds of the elite M-3 genotype used for the experiment were obtained through the breeding program of J. curcas developed by the Corpoica in 2006. The seedlings grew in 32 x 12 cm black plastic bags with a capacity of 4 kg of substrate under shade cloth, ensuring 60% shade. One seed was planted per bag with a total of 1,500 planted bags. The substrate was prepared with sandy loam soil, burned rice husks, and organic compost mixed at a 2:1:1 ratio (w/w), respectively, and had a 5.5 pH, 4.1% organic matter, 67.7 mg kg-1 of available phosphorus, and 0.55 cmol kg-1 of available potassium. The irrigation was applied 2 times per week. Eight days after germination, the cloth shade was removed and the seedlings were given free sun exposure. Weed control was done manually and phytosanitary controls for pests and/or diseases were not required.
The transplanting to soil in the field was done when the plants reached an average height of 30 cm and developed 5-6 true leaves (40-45 d after sowing) and at a distance of 2 m between the rows and 3 m between the plants.
Twelve fertilization treatments with different doses of nitrogen and potassium fertilizers were evaluated under a randomized complete block design with four replicates and 24 plants per plot and one edge row, for a total of 1,666 plants/ha, with 1,184 plants used for variable evaluation. The fertilizer treatments were combined in a factorial matrix design (32) and included control treatments for each potassium or nitrogen dose and an absolute control (Tab. 3).

The nitrogen and potassium fertilizer doses were established taking into account the results of the studies performed by Kalannavar (2009), who reported that applications of 100 kg N, 100 kg P2O5 and 150 kg K2O ha-1 per year in poor soils significantly increased seed yields of Jatropha up to 3,937 kg ha-1. Patil and Parameshwarappa (2007) reported significant increases in plant height, stem base diameter, number of branches, and seed yield up to 1,475 kg ha-1 year with the application of 80:80:80 kg ha-1 N:P2O5:K2O. Considering these references as estimates for J. curcas demands for N and potassium and using nutrient availability from the soil (Tab. 2), fertilizer doses for N between 50 and 150 kg ha-1 and for potassium between 60 and 180 kg ha-1 were established. The N and K were applied as urea and potassium chloride, respectively, in the ring matter across the root zone at a distance between 30 and 40 cm from the trunk and covered with soil. Other mineral nutrients (P, Ca, Mg, S, and micronutrients) were applied prior to the transplanting by mixing them with the soil removed from the hole with the same dose for all the treatments, which were calculated taking into account the results of soil analysis (ICA, 1992).
The total dose of fertilizers was fractioned in each treatment and the fertilization was performed every three months starting one month after seedling transplanting to the field. Weed control at the early stages (first 6 months) of the field experiment was performed manually around the plants between the rows. No pest control was performed during the course of the experiment. Mature fruits were harvested periodically on a monthly basis for 9 months starting from December 2012 to August 2013.
Variables evaluated
Fruit and seed yield, total oil contents in seeds
To evaluate the fruit and seed production, five plants of each of the 48 experimental units were assessed monthly. After the harvest, the fruits were weighed and air dried in shade, the seeds were removed, weighed and dried to 14% moisture and packed in paper bags and preserved at a temperature of 17°C. To evaluate the total oil content in the seeds, whole seeds were air dried and subjected to drying in an oven at 70°C for 72 h to constant weight, then ground with a mill for particles smaller than 2 mm. The oil content was determined according to Nielsen (2003), taking approximately 20 g of seeds placed in cellulose thimbles (filters No. 84). Then, the thimbles with samples were placed in a Soxhlet extraction device for 6 h continuous, using 250 mL of benzine as a solvent with a density of 0.640-0.655, fixed residue of 0.002%, maximum 0.02% water content and boiling point of 55-75°C. Extracted fatty acids were collected and stripped of the solvent by evaporation using rotary evaporation equipment (digital rotovapor Heidolph- 4011, Heidolph Instruments GmbH & Co, Germany) at 60°C to eliminate the excess solvent present in the samples. The oil extracted from the seeds was weighed and stored in amber glass jars at 18°C. The amount of oil in the seeds was calculated on a seed dry basis (kg of oil dry matter per 100 kg of seeds).
Oil contents in seeds through mechanical extraction
Air dried whole seeds were subjected to drying in an oven at 70°C for 72 h to constant weight. The oil content was determined in a sample of approximately 150 g placed in canvas filter bags cf-16 and extracted using a hydraulic press (PR-M-A-40 cm, C-Genpar) with a capacity of 10 t pressure and resistance to heating at 90°C, pressing time of 10 and 5 min of re-pressing. The extracted oil was collected in amber glass jars. The amount of oil in the seeds was calculated on a dry basis (kg of oil dry matter per 100 kg of seeds). These samples were stored at 17°C for subsequent analysis of the percentage and composition of fatty acids.
Fatty acid profile in seeds
The samples obtained from physical extraction were used and the analysis was performed by the official method of the European Commission (1991) for determining the fatty acid methyl esters (FAM E) using gas chromatography: Perkin Elmer Auto system 9000 (GC) equipped with FID and a capillary column of fused silica SPTM -2380 (60 m x 0.25 mm x 0.2 m thick film). The detector and the injector temperatures were 260 and 250°C, respectively. The carrier gas was hydrogen. The column was programmed to a temperature of 165°C for 45 min, followed by a rate of 7.5°C per min to 230°C, an isotherm for 15 min, a second rate of 10°C per min up to 250°C, and a final isotherm of 20 min. The identification of the different fatty acids was performed by comparison of retention times of FAM E standards (GLC-10-1891 FAM E mix 1AM P, Sigma-Aldrich) analyzed under the same conditions. The results were expressed as the percentage of each peak area relative to total area. The samples were analyzed for each of the 12 treatments with three replicates for each fertilization treatment.
Statistical analysis
Analysis of variance (ANOVA) was performed using the SAS v. 6. The means comparisons were obtained using a Tukey test at a probability level P≤0.01.
Results and discussion
Fruit and seed yields
Significant differences in fruit and seed production of the J. curcas plants with different fertilization treatments were observed (Tab. 4), with the highest fruit production, 8,805.8 kg ha-1, and seed yield of 4,764 kg ha-1 obtained with the application of nitrogen 150 kg ha-1 + 120 kg ha-1 of K. The lowest yield was observed with 120 kg ha-1 of K and no nitrogen application (nitrogen control) and corresponded to 4,571 kg ha-1 of fruit and 2,430 kg ha-1 of seed yields. In nitrogen control, the fruit production was only 51% of the fruit yield obtained in 150N;120K, whereas the fruit yields in nitrogen control were similar to the yields in the absolute control without fertilization (absolute control). The low yields in nitrogen control can be explained by a deficiency of nitrogen and indicate a high demand of J. curcas for nitrogen for seed/fruit production. Nitrogen deficiency could also impose a low demand of plants for other nutrients, such as P, K, S, Ca and Mg, since the organs limited in nitrogen may not provide high sink strength (Mengel and Kirkby, 1987; Akbarian et al., 2010). Furthermore, in nitrogen control (no nitrogen application), a calcium and/or magnesium deficiency in the plants might have been induced by the application of potassium. Although the calcium and magnesium contents in J. curcas plants were not measured in this experiment, the absorption rate of these minerals can be greatly restricted by competition with other cations, such as potassium (Marschner, 1997).
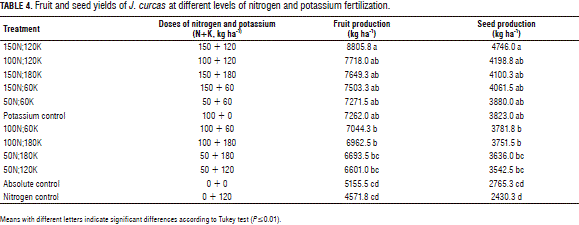
The low yields in absolute control (Tab. 4) were related to the low nitrogen contents in the soil determined at the time of establishment of the experiment, indicating a marked deficiency of this element and a misbalance in the relations between the macronutrients available in the soil. The J. curcas plants in the absolute control were smaller as compared to the plants in the other treatments and developed visual symptoms of nitrogen deficiency in the leaves. The absence of nitrogen in soil can severely reduce plant height, stem diameter, and leaf formation in J. curcas, resulting in visible symptoms of nitrogen deficiency (Marrone, 2009); according to Furoc-Paelmo et al. (2012), plant height in J. curcas is positively correlated with the contents of nitrogen in soil.
The trend that could be observed in Tab. 4 indicates that high yields occurred in treatments with higher doses of N. The treatment 150N;120K, with the highest fruit yield, was obtained with a N:K ratio in the fertilizer equal to 1:0.8, suggesting that, in the vegetative stage and at the early stages of reproductive development, J. curcas absorbed a higher amount of N than K. This is consistent with the assertions of Malavolta (1987) that trees/shrubs can absorb a large amount of nitrogen at the stage of vegetative growth because leaves and branches act as sinks for carbohydrates that are further re-translocated to fruits in the production stage.
It is worth mentioning that the results in our study were obtained with J. curcas plants that did not reach their maximum production stage. At the same time, the ratio between the elements extracted by the plants depends on different factors and varied with the plant age. In woody plants, trunk and branch biomass increases with age, so potassium extraction increases and the ratio between the extracted N and K by the tree could be changed towards the high accumulation of potassium (Taiz and Zeiger, 2006). Additionally, J. curcas is a deciduous species, and the demands of N and K in these plants could depend, among other factors, on defoliation rates and rates of new leaf formation, contents of N and K in abscised leaves, and patterns of N and K recycling from old leaves (Millard and Grelet, 2010).
Total oil content in seeds
No statistically significant differences in the percentages of seed oil contents were observed among the treatments (Tab. 5). The percentages of seed oil, oscillating between 37.2 and 40.1%, were consistent with those obtained in the studies in other regions of the world. Islam et al. (2012) assessed the content of J. curcas seed oil in 21 accessions collected in Malaysia, Borneo, India, Indonesia, Cape Verde, South Africa, Philippines, Vietnam, and Thailand, where contents varied from 40.0% in materials from Malaysia to 48.4% in Vietnam. In another study on the variability of 82 accessions of J. curcas in five zones of peninsular India, the oil content ranged from 31.5 to 45.5% and the level of unsaturated fatty acids in oils ranged from 75.5 to 85% (Bhagat et al., 2011). A higher oil content of 61 to 64% in J. curcas seeds was reported by Emil et al. (2010) in materials from Malaysia, Indonesia, and Thailand.
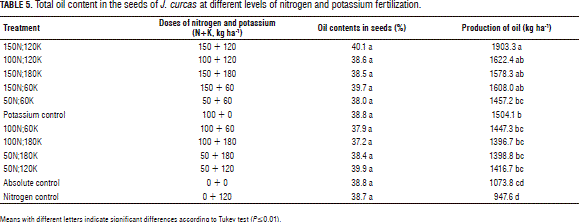
The highest oil yield, 1,903.3 kg ha-1, in this experiment was obtained in 150N;120K, corresponding to the application of 150 kg ha-1 of N + 120 kg ha-1 of K; the lowest oil yield, 947.6 kg ha-1 , was observed in nitrogen control, corresponding to the application of 120 kg ha-1 K without a nitrogen application; the last treatment had an oil yield lower than that obtained in the unfertilized control (Tab. 5). This could be explained as a result of low nitrogen absorption by the plants since they did not receive nitrogen fertilizer. Also, an application of potassium fertilizer might only have induced a deficiency of calcium and/or magnesium since these elements are antagonistic during absorption by the roots.
The accumulation of nutrients in different organs (stems, leaves, flowers, and fruits) and tissues provides an important background to estimate the nutritional needs of plants and serves as a basis to determine the amount of nutrients to be supplied to plants through fertilization (Laviola and Dias, 2008). In this experiment, the treatment that resulted in the highest oil yield was obtained with a N:K ratio in the fertilizer equal to 1.0:0.8, indicating a higher demand of J. curcas plants for nitrogen than for potassium. Nitrogen is an element that is linked to the formation of amino acids, amides, peptides and amines and its assimilation is closely related to the formation of seeds (Mengel and Kirkby, 1987). These photoassimilates are translocated to fruits and constitute part of proteins and fatty acids accumulated in seeds. According to Laviola and Dias (2008), N:K ratios in J. curcas plants are equal to 2.3:1 in leaves and 1.4:1 in fruits, which indicates that, in the production stage, the plants have an increased demand for potassium. At the same time, the accumulation of nitrogen in J. curcas seeds is superior to that of other macronutrients since the seed cake of J. curcas contains about 4.6-6.0% N, 2.6-3.0% P, 1.0-1.4% K, 1.3% Mg, and 0.7% Ca (Lieth, 1975; Chikpah and Demuyakor, 2013).
Fatty acid composition
The content of saturated and unsaturated fatty acids in the J. curcas seeds is presented in Tab. 6, expressed as a percentage in weight by weight ratio (w/w). The seed oil had a high proportion of unsaturated fatty acids of 78.07%, while saturated fatty acids were present in smaller amounts of 21.48% (Tab. 6). Oleic fatty acid (C 18:1), with an average content of 47.54%, and linoleic acid (C 18:2), with 29.91%, were the principal ones in the composition of the fatty acids, while saturated fatty acids had lower contents, with palmitic (C 16:0) and stearic (C 18:0) acids being the more representative ones with 13.39 and 7.26%, respectively. Therefore, J. curcas seed oil can be classified as oleic-linoleic, which agrees with the results obtained by Akbar et al. (2009) and Rodrigues et al. (2013), who also confirmed that unsaturated oleic and linoleic fatty acids predominate in J. curcas oil. Likewise, Emil et al. (2010) reported that J. curcas seed oil contains a high percentage of the unsaturated fatty acids oleic (42.4 to 48.8%) and linoleic (28.8 to 34.6%) and a low percentage of the saturated fatty acids palmitic (13.2 to 14.5%) and stearic (7.0 to 7.7%) in materials obtained from Malaysia, Indonesia, and Thailand. The accessions of J. curcas grown in Nepal contained approximately 80% unsaturated fatty acids with 46% oleic and 32% linoleic acids (Chhetri et al., 2008). This information differs from the results reported by Pedraza and Cayón (2010) who demonstrated that linoleic acid had the highest content reaching 42.7% on average and that oleic acid only reached 33.7%. The oil content in seeds and its composition are influenced by the accession or ecotype and the conditions of climate and soil where the crop is grown (Pedraza and Cayón, 2010; Rodrigues et al., 2013). Stress conditions, such as low temperatures, drought or nutrient deficiencies could increase the contents of saturated fatty acids in plant oils (Taiz and Zeiger, 2006; Upchurch, 2008).
According to European Standard EN 14214, a quality biodiesel, among other characteristics, includes a linolenic acid content in the oil lower than 12% and a cetane number higher than 51 (Ramos et al., 2009). Cetane number is employed as a parameter of diesel fuel quality related to the ignition delay time and combustion quality; a high cetane number ensures good fuel ignition properties and lowers the formation of smoke (Ramos et al., 2009). The cetane number in J. curcas seed oil is 40-45 (Kumar and Sharma, 2008) and linearly increases with an increase in the contents of unsaturated fatty acids (Ramos et al., 2009).
The high contents of unsaturated fatty acids provide a higher quality J. curcas oil for processing into biodiesel through the process of trans-esterification as it requires lower temperatures for the process, but may present higher oxidation in the short-term (Pedraza and Cayón, 2010). Likewise, J. curcas oil, as compared with palm oil, (Elaeis guineensis) has advantages because palm oil contains about 50% unsaturated fatty acids, being less effective in the trans-esterification process (Habib, 2000).
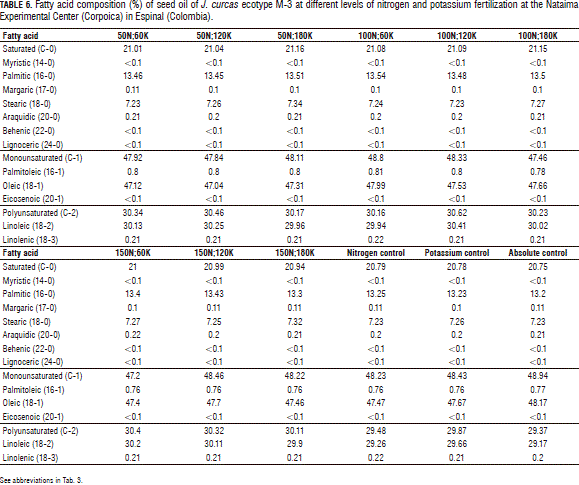
For all fatty acids measured in the J. curcas seed oil in this experiment, no statistically significant differences in the application of different levels of nitrogen and potassium fertilization were found. The higher contents of oleic fatty acid were found in the seed oil of the control plants without fertilization (absolute control) and 100 kg ha-1 of N and 60 kg ha-1 of K (100N;60K), which averaged 48.17 and 47.99% oil, respectively; these values were slightly higher than the amounts obtained with the other fertilizer treatments. The content of oleic fatty acid obtained in 50 kg ha-1 of nitrogen + 120 kg ha-1 K (50N;120K), was 47.04%, the lowest percentage in comparison with other treatments (Fig. 1A).
The plants that received fertilization corresponding to the application of 100 kg ha-1 of N + 120 kg ha-1 of K (100N;120K) had the highest average content of linoleic acid, 30.41%, slightly higher than the amounts of linoleic acid obtained with the other fertilization treatments. At the same time, the plants that received no fertilizer had the lowest percentage (29.17%) of this fatty acid (Fig. 1B).
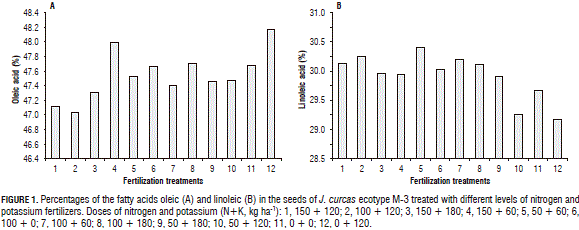
In relation to saturated fatty acids, the higher levels of palmitic acid were registered for plants receiving applications of 100 kg ha-1 of N and 60 kg ha-1 of K (100N;60K); 13.54% was slightly higher than the amounts obtained with the other fertilizer treatments. The lowest percentage (13.2%) of this fatty acid was obtained in the seed oil from the unfertilized control plants (absolute control) (Fig. 2A).
The highest content of stearic acid was found in the seed oil from the plants receiving 50N;180K and 150N;180K, with averages of 7.34 and 7.33%, respectively, which were statistically equal but slightly higher than the percentages obtained in the other fertilization treatments. The percentage of stearic fatty acid in the seed oil from the control plants without fertilization (absolute control) was 7.23%, the lowest percentage compared to the ones in the fertilization treatments (Fig. 2B).
Conclusions
The fertilization of J. curcas plants with nitrogen and potassium at a dose of 150 + 120 kg ha-1 increased fruit and seed production by more than 90%, as compared with the unfertilized control, and represents an almost two-fold increase in oil production that rose from 947 up to 1,900 kg ha-1.
Fertilization with N and K improved the yield of vegetable oil in J. curcas, increasing the total number of fruits and seeds produced per plant, but did not affect the oil content in the seeds, which presented oil contents ranging from 38.7 to 40.1% (w/w) without presenting statistically significant differences due to the effect of fertilizations. These oils were rich in unsaturated fatty acids such as oleic acid (≈ 47%) and linoleic acid (≈ 29%). A high level of oleic fatty acid was present in the oils of seeds from the unfertilized control plants and plants with applications of 100 kg ha-1 of N and 60 kg ha-1 of K, averaging about 48%. The lowest level oleic fatty acid content occurred when a low nitrogen and potassium ratio 1:2.4 was applied respective to the doses of 50 kg ha-1 of N + 120 kg ha-1 of K. The content of the saturated fatty acids palmitic and stearic were low, ranging from 13.4 to 7.26%, respectively, making these oils profitable for biodiesel production.
Acknowledgements
The authors express their gratitude to the Nataima Research Center (Corpoica) and its facilities for the development of this research. We also thank Dr. Fernando Campuzano and Dr. Eduardo Barragan and are grateful for the financial support of this project and also to research assistant Mr. Gentil Garcia for his excellent assistance in data measurements in this study.
Literature cited
Abhilash, P.C., P. Srivastava, S. Jamil, and N. Singh. 2011. Revisited Jatropha curcas as an oil plant of multiple benefits: critical research needs and prospects for the future. Environ. Sci. Pollut. Res. 18, 127-131. [ Links ]
Achten, W.M.J., L. Verchot, Y.J. Franken, E. Mathijs, V.P. Singh, R. Aerts, and B. Muys. 2008. Jatropha bio-diesel production and use. Biomass Bioener. 32, 1063-1084. [ Links ]
Achten, W.M.J., W.H. Maes, R. Aerts, L. Verchot, A. Trabucco, E. Mathijs, V.P. Singh, and B. Muys. 2010. Jatropha: from global hype to local opportunity. J. Arid. Environ.74, 164-165. [ Links ]
Akbar, E., Y. Zahira, K. Siti Kartom, I. Manal, and S. Jumat. 2009. Characteristic and composition of Jatropha curcas oil seed from Malaysia and its potential as biodiesel feedstock. Eur. J. Sci. Res. 29, 396-403. [ Links ]
Akbarian, M.M., N. Modafebehzadia, and M.A. Bagheripoura. 2010. Study of fertilizer (NPK) effects on yield and triglycerids in jatropha (Jatropha curcas). Plant Ecophysiol. 2, 169-172. [ Links ]
Aregheore, E.M., K. Becker, and H.P.S. Makkar. 2003. Detoxification of a toxic variety of Jatropha curcas using heat and chemical treatments, and preliminary nutritional evaluation with rats. South Pac. J. Nat. Sci. 21, 50-56. [ Links ]
Azam, M.M., A. Waris, and N.M. Nahar. 2005. Prospects and potential of fatty acid methyl esters of some non-traditional seed oils for use as biodiesel in India. Biomass Bioener. 29, 293-302. [ Links ]
Bankovic-Ilic, I.B., S.O. Stamenkovic, and V.B. Veljkovic. 2012. Biodiesel production from non-edible plant oils. Renew. Sustain. Ener. Rev. 16, 3621-3647. [ Links ]
Bhagat, R.B., S.P. Taware, A.M. Chavan, and D.K. Kulkarni. 2011. Physico-chemical analysis, NM R spectroscopy and gas chromatographic studies of Jatropha curcas L. germplasm. J. Amer. Oil Chem. Soc. 88, 337-340. [ Links ]
Brittaine, R. and N. Lutaladio. 2010. Jatropha: a small-holder bioenergy crop. The potential for pro-poor development. Integrated Crop Management No. 8. FAO, Rome. [ Links ]
Campuzano, F. 2008. Programa Jatropha Colombia. Genética, agronomía, poscosecha y desarrollo agroindustrial del piñon en Colombia (2008-2012). Rev. Innov. Cambio Tecnol. 7, 64-68. [ Links ]
Chhetri, A.B., M.S. Tango, S.M. Budge, K.C. Watts, and M.R. Islam. 2008. Non-edible plant oils as new sources for biodiesel production. Int. J. Molec. Sci. 9, 169-180. [ Links ]
Chikpah, S.K. and B. Demuyakor. 2013. Evaluation of nutritional and antinutritional composition of meals of Jatropha curcas seeds/kernels obtained from four different agro-climatic areas of Ghana. Scient. J. Agric. 2, 36-46. [ Links ]
Emil, A., Z. Yaakob, M.N.S. Kumar, J.M. Jahim, and J. Salimon. 2010. Comparative evaluation of physicochemical properties of Jatropha seed oil from Malaysia, Indonesia and Thailand. J. Amer. Oil Chem. Soc. 87, 689-695. [ Links ]
Francis, G., R. Edinger, and K. Becker. 2005. A concept for simultaneous wasteland reclamation, fuel production, and socioeconomic development in degraded areas in India: need, potential and perspectives of Jatropha plantations. Nat. Resour. Forum 29, 12-24. [ Links ]
Foidl, N., G. Foid, M. Sanchez, M. Mittelbach, and S. Hackel. 1996. Jatropha curcas L. as a source for the production of biofuel in Nicaragua. Bioresour. Technol. 58, 77-82. [ Links ]
Furoc-Paelmo, R., R.G. Visco, W.M. Carandang, A.S.A. Castillo, and R.V.O. Cruz. 2012. Relationship of Jatropha curcas L. growth parameters with soil and microclimatic factors in varying short rotation-based agroforestry system. Philip. J. Crop Sci. 37, 76-87. [ Links ]
Goel, G., H.P.S. Makkar, G. Francis, and K. Becker. 2007. Phorbol esters: structure, bio-logical activity, and toxicity in animals. Int. J. Toxicol. 26, 279-288. [ Links ]
Habib, F. 2000. Kernel-specific DNA clones encoding three different isoforms of seed storage protein glutelin from oil palm Elaeis guineensis. Plant Sci. 160, 913-923. [ Links ]
Harinder, P.S.M. and K. Becker. 2009. Jatropha curcas, a promising crop for the generation of biodiesel and value-added coproducts. Eur. J. Lipid Sci. Technol.111, 773-787. [ Links ]
Holdridge, L.R. 1967. Ecología basada en zonas de vida. Tropical Science Center, San Jose. [ Links ]
ICA, Instituto Colombiano Agropecuario. 1992. Fertilización en diversos cultivos. Quinta aproximación. Manual de Asistencia Técnica No. 25. Bogota. [ Links ]
Islam, A.K.M.A., Z. Yaakob, N. Anuar, S.R.P. Primandari, and M. Osman. 2012. Physiochemical properties of Jatropha curcas seed oil from different origins and candidate plus plants (CPPs). J. Amer. Oil Chem. Soc. 89, 293-300. [ Links ]
Kalannavar, V.N. 2009. Effect of major nutrients on growth and yield of Jatropha curcas L., Karnataka, J. Agric. Sci. 22, 1095-1096. [ Links ] Kumar, A. and S. Sharma. 2008. An evaluation of multipurpose oil seed crop for industrial uses (Jatropha curcas L.): a review. Indust. Crops Prod. 28, 1-10. [ Links ]
Laviola, B.G. and L.A.D. Dias. 2008. Teor e acúmulo de nutrientes em folhas e frutos de pinhão-manso. Rev. Bras. Ciênc. Solo, 32, 1969-1975. [ Links ]
Lieth, H. 1975. Primary productivity of the major vegetation units of the world. pp. 64-87. In: Lieth, H. and R.K. Whittaker (eds.). Primary productivity of the biosphere, Springer, Berlin. [ Links ]
Makkar, H.P.S. and K. Becker. 2009. Jatropha curcas L, a promising crop for the generation of biodiesel and value-added coproducts. Eur. J. Lipid Sci. Technol. 111, 773-787. [ Links ]
Malavolta, E. 1987. Fertilidade dos solos da Amazônia. p. 374-416. In: Vieira, L.S., P.C.T.C. Santos (eds.) Amazônia: seus solos e outros recuraos naturais. Agronômica Ceres, San Pablo, Brazil. [ Links ]
Marrone, J. 2009. Efecto de deficiencias nutricionales en el cultivo de piñón (Jatropha curcas). Undergraduate thesis. Escuela Agrícola Panamericana (Zamorano), Honduras. [ Links ]
Marschner, H. 1997. Mineral nutrition of higher plants. Hohenheim, Stuttgart, Germany. pp. 263-297. [ Links ]
Mengel, K. and E.A. Kirkby. 1987. Principles of plan nutrition. I.P.I. Bern, Switzerland. [ Links ]
Millard, P. and G. Grelet. 2010. Nitrogen storage and remobilization in trees: ecophysiological relevance in a changing world. Tree Physiol. 30, 1083-1095. [ Links ]
Nielsen, S. 2003. Food analysis laboratory manual. 2nd ed. Kluwer Academic; Plenum, Publishers, West Lafayette, IN. pp. 37-42. [ Links ]
Openshaw, K. 2000. A review of Jatropha curcas L: an oil plant of unfulfilled promise. Biomass Bioener. 19, 1-15. [ Links ]
Pal, A., V.S. Negi, S. Khanal, and D. Borthakur. 2012. Immunodetection of curcin in seed meal of Jatropha curcas using polyclonal antibody developed against curcin-L. Curr. Nutr. Food Sci. 8, 213-219. [ Links ]
Patil, S.J. and K.G. Parameshwarappa. 2007. Silvicultural management of jatropha (Jatropha curcas L.) under rainfed conditions. pp. 371-372. In: National Seminar on Changing Global Vegetable Oil Scenario: Issues and Challenges Before India, ISOR, Hyderabad, India. [ Links ]
Phasukarratchai, N., V. Tontayakom, and C. Tongcumpou. 2012. Reduction of phorbol esters in Jatropha curcas L. pressed meal by surfactant solutions extraction. Biom. Bioener. 45, 48-56. [ Links ]
Phengnuam, T. and W. Suntornsuk. 2013. Detoxification and antinutrients reduction of Jatropha curcas seed cake by Bacillus fermentation. J. Biosci. Bioeng. 115, 168-172. [ Links ]
Pedraza, E.A. and D.G. Cayón. 2010. Caracterización morfofisiológica de Jatropha curcas L. variedad Brasil cultivada en dos zonas de Colombia. Acta Agron. 59, 30-36. [ Links ]
Ramos, M.J., C.M. Fernández, A. Casas, L. Rodríguez, and Á. Pérez. 2009. Influence of fatty acid composition of raw materials on biodiesel properties. Biores. Technol. 100, 261-268. [ Links ]
Rodrigues, J., I. Miranda, J. Gominho, M. Vasconcelos, G. Barradas, H. Pereira, F. Bianchi-de-Aguiar, and S. Ferreira-Dias. 2013. Variability in oil content and composition and storage stability of seeds from Jatropha curcas L. grown in Mozambique. Indust. Crops Prod. 50, 828-837. [ Links ]
Sauerwein, M., F. Sporer, and M. Wink. 1993. Insect-toxicity of phorbol esters from Jatropha curcas seed oil. Planta Med. 59, A686. [ Links ]
Sengo, I., J. Gominho, L. Orey, M. Martins, E. Almeida-Duarte, H. Pereira, and S. Ferreira-Dias. 2010. Response surface modeling and optimization of biodiesel production from Cynara cardunculus oil. Eur. J. Lipid Sci. Technol. 112, 310-320. [ Links ]
Taiz, L. and E. Zeiger. 2006. Plant physiology. 5th ed. Sinauer Associates, Sunderland, MA . [ Links ]
Upchurch, R.G. 2008. Fatty acid unsaturation, mobilization, and regulation in the response of plants to stress. Biotechnol Lett. 30, 967-977. [ Links ]
Verrastro, F. and S. Ladislaw. 2007. Providing energy security in an interdependent world. Wash. Quart. 30, 95-104. [ Links ]
Zahawi, R.A. 2005. Establishment and growth of living fence species: an over-looked tool for the restoration of degraded areas in the tropics. Rev. Ecol. 13, 92-102. [ Links ]













