Introduction
The liver is crucial for maintaining body homeostasis and for the metabolism of carbohydrates, proteins, and fats 1,2. Nowadays, public health institutions consider hepatic diseases a priority due to the connection between their high prevalence and mortality. In developing countries, the primary causes of liver diseases include hepatitis viruses, environmental toxins, and drug and alcohol consumption. Medications such as antibiotics and other chemotherapeutics also contribute to acetaminophen (N-acetyl-p-aminophenol; APAP). APAP is widely used as an antipyretic and analgesic, and it is considered safe at the recommended therapeutic levels. A high dose of APAP, however, can cause significant liver damage and result in acute liver failure 3. In mice, as in humans, an overdose of APAP also results in acute liver failure. In contrast to most animal models of acute liver injury, the APAP-induced liver injury model in mice directly reflects the pathogenesis of a major and potentially fatal human disease 4.
The kidneys are essential for removing toxic materials from the blood, maintaining electrolyte balance, and regulating blood volume, blood pressure, and red blood cell production. The kidneys play a crucial role in regulating blood volume and pressure. They achieve this by secreting renin, activating vitamin D, producing erythropoietin (a hormone that controls red blood cell production), and performing gluconeogenesis during extended periods of fasting 5. However, the kidneys can be damaged or become inactive due to several factors, including oxidative stress, inflammation, ischemia/reperfusion injury, diabetes, and exposure to nephrotoxic agents, which are often found in various modern medications. For example, gentamicin, an aminoglycoside antibiotic, can produce nephrotoxicity in chronic treatments 6,7. An important mechanism of nephrotoxicity is increasing the production of mitochondrial reactive oxygen species (ROS) can cause a deficiency in intrinsic antioxidant enzymes. High ROS levels can damage key cellular molecules such as proteins, lipids, and nucleic acids, disrupt cell function, and contribute to cell death 8,9.
Natural products have significantly contributed to the identification of various drug targets for treating human ailments. They also offer several compounds that can help prevent toxicity and organ damage. Most of the protective effects of natural products are linked to their antioxidant and anti-inflammatory properties. Various natural products, particularly those derived from higher plants such as angiosperms, gymnosperms, and pteridophytes, have been shown to exhibit hepatoprotective and nephroprotective effects 10. Many medicinal plants have demonstrated protective effects in liver and kidney damage induced by paracetamol, gentamicin, profenofos, D-galactosamine, chronic stress, cytotoxic drugs, diabetic nephropathy, and chemically induced nephrolithiasis, due to the action of inflammatory mediators and oxidative stress 11. Polyphenolic compounds typically show strong radical-scavenging and antioxidant activity, which contributes to their protective functions. For example, the ethanolic extract of Prosopis ruscifolia leaves, rich in alkaloids 11, exhibits hepatoprotective and nephroprotective effects against acetaminophen and gentamicin, respectively, in mice 12. Phenolic and flavonoid compounds extracted from Daphne mucronata Royle have demonstrated hepatoprotective and nephroprotective effects in various in vivo animal studies 13.
Sida rhombifolia L., which is popularly called in Paraguay as ¨typycha hũ¨, is a semi-evergreen shrub approximately one meter tall, belonging to the Malvaceae family. 14,15. S. rhombifolia, according to its ethnopharmacological properties, is customarily used against several types of inflammation, as well as a booster for immunity, helping with well-being and vitality 16,17,18. In Brazil, it serves as a versatile remedy, prized for its ability to enhance digestion, promote bile flow, and act as a diuretic. Additionally, it is celebrated for its potential to lower cholesterol and blood sugar levels, support healthy blood pressure, and provide anti-inflammatory benefits 19,20. Leaf decoctions are used in Honduras for diarrhea, kidney ailments, fever, headaches, and stimulants. In Argentina, the infusion of the aerial parts is used to relieve menstrual pain and liver conditions. In Paraguay, a decoction made from leaves is used as a diuretic and for vaginal washes. It is also applied externally to relieve conditions such as rheumatism, gout, arthritis, lumbago, sciatica, and muscle pain 21. In the Central Chaco region, the root is used to treat kidney pain and promote wound healing 22. Various experimental models were used to validate the pharmacological effects of S. rhombifolia, demonstrating its anti-inflammatory 23-27 and antioxidant activity 28,29; as well as its antiarthritic capacity 23,30; and hypoglycemic, hypocholesterolemic, and cardioprotective potentials 29,31-33. The in vitro antimicrobial activity of different extracts of S. rhombifolia L. has also been described 18,34. In addition, a hepatoprotective effect of the ethanolic extract of S. rhombifolia L. has been found against a paracetamol-induced hepatotoxicity model in albino Wistar rats 35. Studies have demonstrated that the hydroethanolic extract of S. rhombifolia showed no significant toxic effects when administered orally to male and female rats in both acute and repeated toxicity models 36. Finally, methanolic extracts from the aerial parts of the plant showed low acute toxicity and antispasmodic activity in the intestinal and stomach regions of mice 37.
Some scientific studies revealed the presence of different bioactive molecules extracted from the leaves and roots of S. rhombifolia. The compounds include polyphenols, flavonoids, steroids, porphyrins, alkaloids, coumarins, and derivatives of ferulic acid. 18,28,31,32,38. The antioxidant properties could be responsible for most of the biological effects of this species.
Considering the properties of S. rhombifolia, as illustrated by numerous pharmacological studies and traditional applications, the present research study was designed to evaluate the hepatoprotective and nephroprotective effects of the methanolic extract of the aerial parts in models of hepatotoxicity and nephrotoxicity induced by acetaminophen and gentamicin, respectively, in mice.
Materials and Methods
Chemical and reagent kits
Chemicals used, including APAP and silymarin, were purchased from Sigma Chemical Company (MO, USA). Gentamicin from LASCA Pharmaceutical (San Lorenzo, Paraguay), and sodium pentobarbital (Nembutal) from Abbott (Tokyo, Japan) were used; propylene glycol and ethanol for pharmaceutical use were bought locally. Methanol was purchased from Biopack®, (Buenos Aires, Argentina). Kits for liver biomarkers, alkaline phosphatase (ALP), aspartate aminotransferase (AST), alanine aminotransferase (ALT), total proteins, albumin, creatinine, urea, uric acid, total cholesterol, triglyceride and glycaemia were purchased from Human Diagnostics Worldwide reagent (Magdeburg, Germany) measurements were conducted using a semi-automatic analyzer from Biosystem BTS 350 from Biotec (Asunción, Paraguay).
Preparation of S. rhombifolia extract
Aerial parts of Sida rhombifolia L. were collected from Aguaity city (Cordillera, Paraguay; 25° 20´ 24´´ S, 57° 02´ 04´´ O) in October 2019. A specimen was identified by botanical specialists and deposited in the Herbarium of the School of Chemical Sciences, National University of Asunción (identification code: M. González 45 FCQ).
Aerial parts were dried at room temperature and protected from light, then they were reduced to powder with the help of a mill and macerated in methanol (24h x three times) and concentrated to dryness under reduced pressure using a rotary evaporator. The methanolic extract of S. rhombifolia L. (EMSr) was carefully preserved at room temperature within a desiccator, awaiting its application in various biological tests. In the experiment, the extract was diluted in water with 10% Tween 80.
Animal handling
The in vivo experiments were conducted using healthy adult Swiss albino male and female mice, weighing 25-35 g. The wide range of weights is due to the species' unique physiology, where male mice tend to be larger and heavier than females. The animals were housed in a plastic cage with a 12:12 hour light-dark cycle at a constant room temperature of approximately 23-25°C and a relative humidity of 50-60%. Five to six mice were kept per cage. The animals were fed commercial food and then fasted overnight, with free access to drinking water throughout the trials. The mice come from the Bioterio of the Department of Pharmacology of the National University of Asunción. All animal procedures were carried out by the ARRIVE guidelines (Animals in Research: Reporting In Vivo Experiments) and the Animal Scientific Procedure UK (1986), and the principles of the 3Rs 39,40. Additionally, the current experiment was authorized by the Bioethical Committee of the School of Chemical Sciences (National University of Asunción) (CEI 545/2019).
Acute oral toxicity and general behavior assay of methanolic extract of S. rhombifolia L. in mice
Because a limit test can efficiently identify substances with low toxicity, we conducted the OECD (Organization for Economic Comparition and Development) 425 acute oral toxicity test as a standard procedure to assess the safety profile and determine appropriate extract doses for the main experiments in mice 41. The doses used in the experiment were selected as described in the OECD guideline (50, 300, and 2000 mg/kg, administered by gavage). The assay was performed sequentially with 4 randomly selected groups of 5 mice per dose. One group received the vehicle 10% Tween 80 solution (control group), and the other 3 groups received doses of 50, 300, and 2000 mg/kg of EMSr. Male and female mice (30-40 g, b.w.) were treated to determine the acute oral toxicity. After treatments, each animal was kept under deep observation for lethal signals for 24 hours and the following 14 days. After the treatment, the animals were euthanized, and the liver, kidneys, heart, lungs, and spleen were evaluated for the presence of pathological damage at the macroscopic level.
On the other hand, the general behavioral effect of oral administration was evaluated according to Irwin´s test 42. The impact on behavior was assessed in 4 groups (n = 8 per group): a control group (10% Tween 80) and three groups that received 50, 300, and 2,000 mg/kg of EMSr, respectively. The selected doses were chosen according to OECD test number 425. According to the test, the general behavior of the animals was evaluated by placing them individually in the center of a transparent acrylic box (40 cm x 40 cm x 20 cm) immediately after oral administration. Animals were evaluated at different time points 0, 15, 30, 60, 120, and 240 minutes; then at 24, 48, 72 hours, and up to 7 days, for register alterations in behavioral parameters such as spontaneous mobility, piloerection and exophthalmia, pupillary dilation, stereotyped movements, tremor, tail erection, respiratory frequency, sedation, analgesia, anesthesia, loss of coordination reflexes, grooming, clonic seizures, tonic seizures, sialorrhea, fasciculation, mydriasis, catatonia, palpebral ptosis, ataxia, dyspnea, passivity and dorsal tone decrease. Each parameter was conveniently recorded and tabulated according to intensity from zero to +4 40.
Sub-chronic toxicity assay of methanolic extract of S. rhombifolia L. in mice
A sub-chronic toxicity assay was performed in mice according to Test Number 407 of the OECD Guideline, with repeated doses administered over 28 days 43. Twenty-eight male and female mice were randomly divided into four groups (n = 8). Three of them received different doses of EMSr respectively (50, 100, and 200 mg/kg, resuspended in 10% Tween 80 solution), and the control group was treated with 10% Tween 80 (0.1 mL/g b.w.) solution daily for 28 days by oral gavage. Animal body weight was recorded daily, and changes in the skin and fur, eyes and mucous membranes, as well as the respiratory, circulatory, autonomic, and central nervous systems, somatomotor activity, and behavior were monitored for symptoms of toxicity or lethality. On day 28, one hour after the treatment, the animals were anesthetized using sodium pentobarbital at a dosage of 40 mg/kg via intraperitoneal injection, and blood samples were collected by intracardiac puncture. As part of the evaluation of signs of toxicity, biochemical parameters (ALT, AST, creatinine, urea, and uric acid, total proteins, albumin, total cholesterol, triglycerides, and glucose) were measured in the serum obtained by centrifugation of the sample blood from the animals at 5000 rpm for 10 minutes. Biochemical parameters were determined by spectrophotometry using the Biosystems BTS 350 semi-automatic analyzer and the Human reagent kit. The organs, including the liver, kidneys, heart, lungs, and spleen, were evaluated macroscopically.
Acetaminophen-induced hepatotoxicity assay
The hepatoprotective activity of EMSr was evaluated using a model of induced hepatotoxicity in mice, as described by Mossanen and F Tacke 3. The animals were randomly divided into seven distinct groups (n=8) and treated orally as follows: control (tween 80 at 10%, p.o.; EMSr vehicle); vehicle (a mixture of 2,5% ethanol: 40% propylene glycol: 57,5% distilled water, p.o.; APAP and silymarin vehicle); acetaminophen (water, p.o., pathological group, APAP); silymarin (150 mg/kg of silymarin, p.o., standard hepatoprotective agent); and extracts EMSr50, EMSr100 and EMSr200 (50, 100 and 200 mg/kg of EMSr, p.o.). On day 4 of treatment, two hours after treatment, acute hepatotoxicity was induced with APAP (300 mg/kg, i.p.). All animals, except the control and vehicle groups, received APAP 3. Three hours after administering APAP, a blood sample was collected via cardiac puncture following anesthesia with sodium pentobarbital (40 mg/kg, i.p.) for measurement of ALT, AST, total protein, and albumin in the serum. The euthanasia by cervical dislocation was immediately performed after sample extraction.
Gentamicin-induced nephrotoxicity assay
The nephroprotective activity of EMSr was evaluated using a model of nephrotoxicity induced by gentamicin in mice 6,7,44. The animals were randomly divided into seven groups (n=8) and treated orally for 9 days the following treatments: control (Tween 80 at 10%, p.o.; EMSr vehicle); vehicle (2,5% ethanol: 40% propylene glycol: 57,5% distilled water, p.o., gentamicin and silymarin vehicle); gentamicin (water, p.o., pathological group); silymarin (150 mg/kg of silymarin, p.o., standard nephroprotective agent); and extract groups EMSr (50, 100 and 200 mg/kg of S. rhombifolia L. methanolic extract, p.o). Every day, all animals, except for the control and vehicle groups, received gentamicin at a dosage of 135 mg/kg body weight via intraperitoneal injection, one hour after the oral administration of the various treatments. On the 10th day, 24 hours after the last dose of gentamicin, the animals were anesthetized with sodium pentobarbital at a dose of 40 mg/kg via intraperitoneal injection. Blood was then extracted through cardiac puncture. The serum levels of creatinine, urea, and uric acid were measured. The euthanasia by cervical dislocation was immediately performed after sample extraction.
Determination of biochemical parameters
Liver and kidney profile parameters, including ALT, AST, ALP, creatinine, urea, and uric acid, were determined spectrophotometrically from the serum using a commercially available kit (Human Diagnostic Worldwide, Germany). Additionally, the total protein and albumin levels were determined. After obtaining the blood and allowing it to clot, it was centrifuged at 3,000 rpm for 15 minutes, and the serum was processed immediately. The blood volume obtained from each animal was sufficient to perform all the tests, resulting in eight replicates per group.
Statistical analysis
Data were analyzed using GRAPHPAD PRISM 9.0 software (GraphPad Software, Inc., San Diego, CA, USA). All data are expressed as mean ± SD (standard deviation) of the number of animals. Data distribution was determined after the Shapiro-Wilk normality test. Even for toxicity and hepato- and nephroprotection assays, the differences between groups were determined by one-way ANOVA and verified by Tukey´s post-hoc test, assuming the normal distribution of the samples. Values of p<0.05 were considered statistically significant. In specific experiments, such as the subchronic toxicity assay evaluating biochemical parameters (blood glucose, triglycerides, and total cholesterol), group sizes became unequal during the study owing to biological loss, technical failure, or the presence of outliers that were excluded using predefined criteria.
Results
Acute oral toxicity and general behavior assay of methanolic extract of S. rhombifolia L. in mice
The acute toxicity test conducted with a single dose of EMSr did not reveal any signs of toxicity, morbidity, or mortality in the treated animals for the 24-hour observation period or throughout the 14 days of the experiment. The test indicated that there were no adverse effects from the extract at doses of up to 2,000 mg/kg body weight in mice. In comparison to the control group, all animals exhibited normal behaviors, including postural reflexes, grooming, responses to painful stimuli, and consistent water and food intake (data not shown). All the animals survived until the scheduled euthanasia. No differences were observed in organ weights or macroscopic observations (data not shown).
Sub-chronic toxicity assay of methanolic extract of S. rhombifolia L. in mice
Oral administration of EMSr at doses of 50, 100, and 200 mg/kg over 28 days did not result in any deaths among the animals. There were no statistically significant differences in daily body weight between the treatment groups for both male and female animals (Figure 1). Additionally, no effects on the general behavior of either male or female mice exposed to EMSr were observed (data not shown). All animals survived until the scheduled euthanasia, and no differences were noted in organ weight or macroscopic observation when compared to the control group (data not shown).
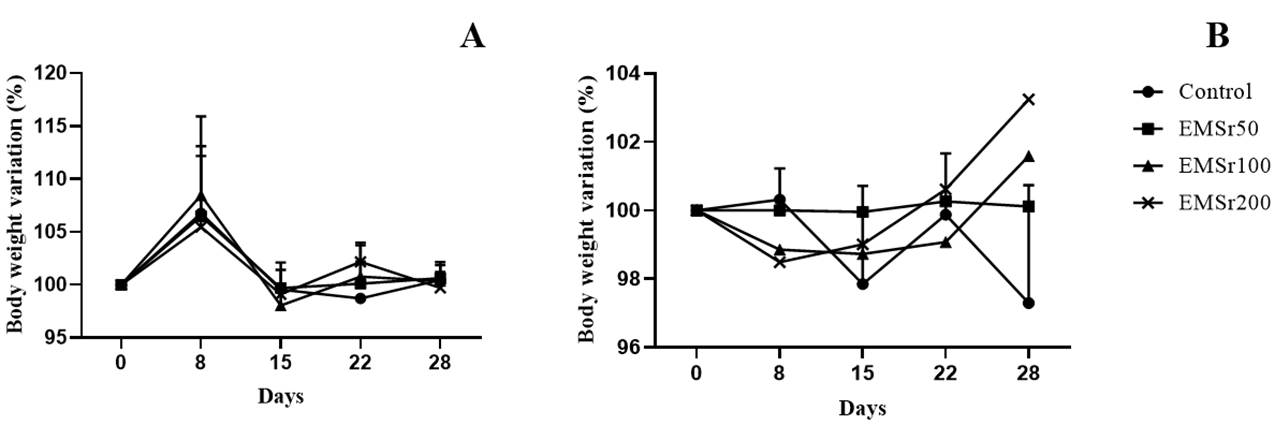
Figure 1 Body weight of male (A) and female (B) mice during the treatment period (28 days). Each point represents the mean ± SD (n = 8). Statistical analysis was performed using a linear regression Test.
Serum uric acid levels significantly decreased in male groups receiving EMSr at three different doses (EMSr50, EMSr100 and EMSr200), while in females, only the higher doses (EMSr100 and EMSr200) decreased the serum level (Table 1).
Table 1 Effect of sub-chronic oral administration of S. rhombifolia L. on different serum biochemical parameters.
| Experimental groups | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Control | EMSr50 | EMSr100 | EMSr200 | |||||||
| Parameters | Units | Male ♂ | Female ♀ | Male ♂ | Female ♀ | Male ♂ | Female♀ | Male ♂ | Female ♀ | |
| ALT | U/L | 32.57±3.74 | 53.00±21.00 | 40.57±7.72 | 47.29±19.90 | 38.00±3.37 | 38.50±7.68 | 33.29±8.48 | 33.29±8.85 | |
| AST | U/L | 154.57±38.13 | 260.0±135.120 | 180.40±78.19 | 171.86±69.28 | 186.71±43.56 | 195.67±64.23 | 149.29±62.78 | 184.50±81.15 | |
| ALP | U/L | 159.57±37.29 | 194.29±41.94 | 162.57±37.40 | 170.38±58.61 | 187.43±41.52 | 190.25±18.41 | 171.43±23.78 | 179.57±17.74 | |
| Creatinine | mg/dL | 0.38±0.09 | 0.44±0.12 | 0.43±0.10 | 0.38±0.05 | 0.46±0.12 | 0.40±0.05 | 0.41±0.06 | 0.41±0.04 | |
| Urea | mg/dL | 37.50±4.75 | 51.00±8.19 | 39.25±6.25 | 46.00±3.30 | 35.43±5.95 | 43.63±5.95 | 38.00±4.78 | 40.13±4.91** | |
| Uric acid | mg/dL | 4.05±0.91 | 4.75±0.98 | 2.87±0.47** | 4.41±1.63 | 3.10±0.51* | 3.12±0.52* | 2.61±0.24*** | 2.64±0.24** | |
| Total proteins | g/dL | 5.07±0.25 | 5.37±0.22 | 5.32±0.40 | 5.62±0.16 | 5.20±0.43 | 5.29±0.21 | 4.91±0.28 | 5.53±0.22 | |
| Albumin | g/dL | 2.75±0.11 | 2.83±0.13 | 2.79±0.08 | 2.86±0.06 | 2.83±0.12 | 2.77±0,09 | 2.78±0.14 | 2.80±0.09 | |
| Total Cholesterol | mg/dL | 129.00±10.77 | 120.67±17.32 | 122.86±11.77 | 108.63±18.78 | 129.43±17.51 | 99.29±7.52 | 123.00±19.60 | 118.50±16.97 | |
| Triglyceride | mg/dL | 72.80±20.89 | 110.00±13.55 | 59.33±14.50 | 93.17±20.34 | 72.00±11.43 | 101.29±22.48 | 61.14±17.30 | 89.86±19.81 | |
| Glycemia | mg/dL | 162.25±13.33 | 150.00±8.49 | 171.67±20.11 | 125.25±14.86 | 173.00±26.85 | 145.67±33.38 | 167.75±11.24 | 139.75±16.38 | |
| ALT: alanine aminotransferase, AST: aspartate aminotransferase; ALP: alkaline phosphatase | Data represent the mean ± SD of each group/gender (n=8). ANOVA of one factor, followed by the Tukey test, was used; p < 0.05 was considered statistically significant (* p < 0.05, ** p < 0.01, *** p < 0.001; significantly different from the control group). | |||||||||
No other parameters were affected by the different repeated oral doses of the extract.
Effect of oral administration of methanolic extract of S. rhombifolia L. on liver profile parameters in male mice
Treatment of male mice with acetaminophen led to severe liver damage, evidenced by elevated serum levels of ALT, AST, and ALP as shown in Figure 2 (A-C).
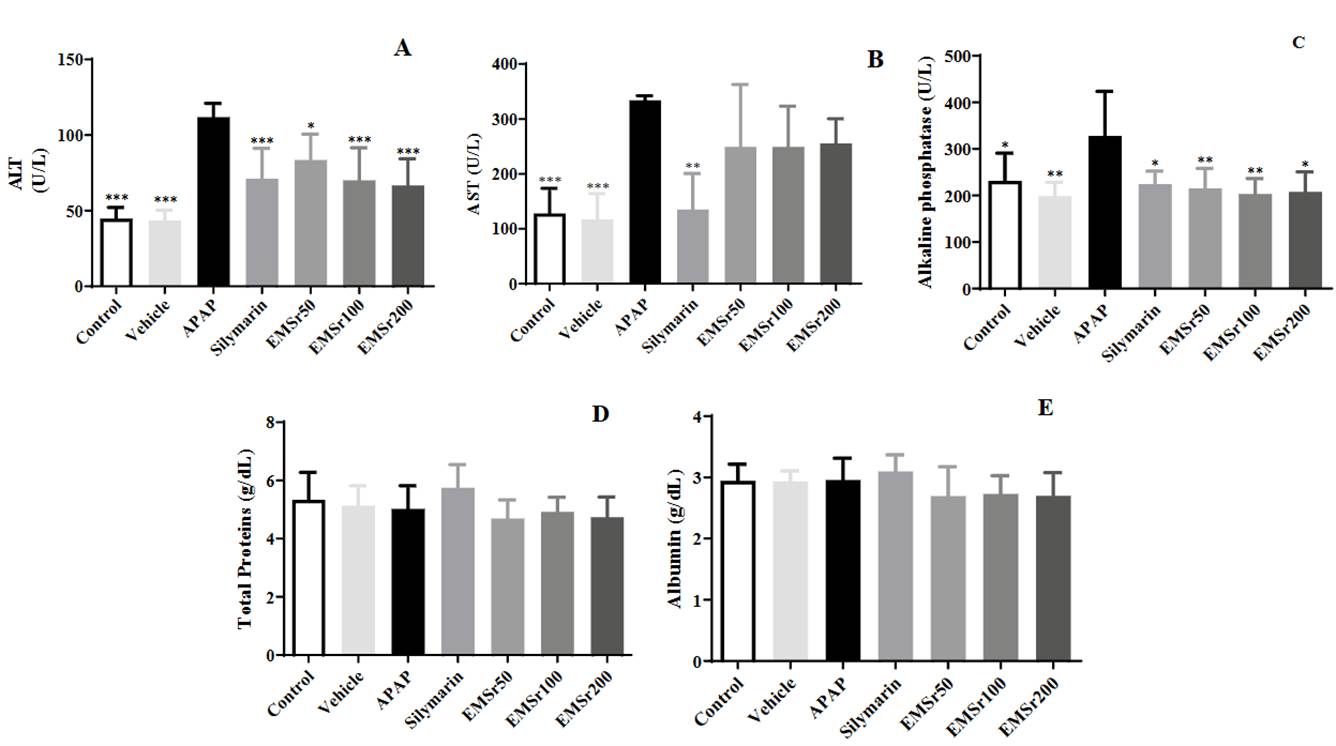
Figure 2 Serum levels of ALT (A), AST (B), Alkaline phosphatase (C), Total Proteins (D), and Albumin (E) of mice (n = 8) treated with S. rhombifolia L. leaves extract. The data are plotted as mean ± SD. ANOVA of one factor, followed by the Tukey test was used, p < 0.05 was considered statistically significant (*p < 0.05, ** p < 0.01,*** p < 0.001, **** p < 0.0001; significantly different from APAP).
Pre-treatment of mice with silymarin, a standard control, significantly reduced the elevated levels of serum transaminases and alkaline phosphatase (ALP) induced by acetaminophen (APAP). Oral administration of all doses of EMSr (50, 100, and 200 mg/kg) also showed a significant effect in lowering serum ALT and ALP levels that were increased by acetaminophen when compared to the pathological group (APAP). On the other hand, serum AST levels were not affected by any dose of the extract. No dose-dependent effect was observed with the tested doses of extract. The significantly reduced liver profile parameters in the silymarin group confirm that the model of acute liver injury caused by acetaminophen was validated. Additionally, no significant differences were observed in serum levels of total protein and albumin among the different treatments (Figures 2D-E).
Effect of oral administration of methanolic extract of S. rhombifolia L. on renal profile parameters in female mice
Results of the nephroprotective effects of S. rhombifolia L. on gentamicin-intoxicated female mice are presented in Figure 3 (A-C).
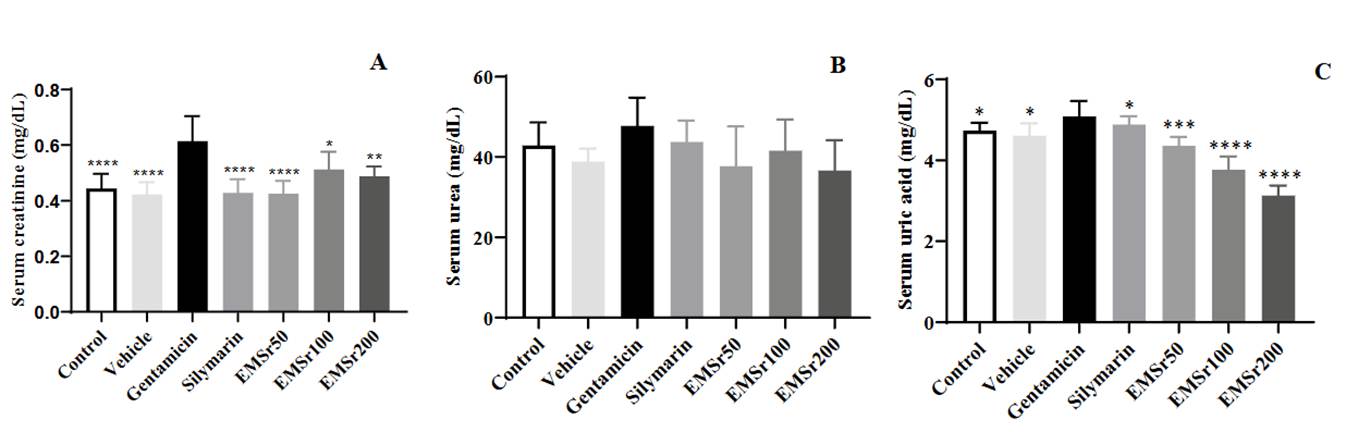
Figure 3 Serum levels (mg/dL) of creatinine (A), urea (B), and uric acid (C) of mice (n = 8) treated with S. rhombifolia L. leaves extract. The data are plotted as mean ± SD. ANOVA of one factor followed by the Tukey test was used; p < 0.05 was considered statistically significant (* p < 0.05,** p < 0.01, *** p < 0.001, **** p < 0.0001; significantly different from Gentamicin).
A significant increase in kidney biomarkers, such as serum creatinine and uric acid, was observed in animals exposed to gentamicin compared to the control and treated groups (silymarin/extract). Pretreatment with silymarin at a dose of 150 mg/kg body weight effectively reduced the elevated levels of creatinine and uric acid. Additionally, all doses of the extracts (50, 100, and 200 mg/kg) significantly lowered serum creatinine and uric acid levels when compared to the gentamicin group. Additionally, no significant differences were observed in serum urea levels between the different treatments. No dose-dependent effect was observed with the tested doses of extract.
Discussion
Medicinal plants and their secondary metabolites hold significant value in today's world and have been utilized for centuries. However, before they can be used as therapeutic agents, it is essential to assess their pharmacological and toxicological effects scientifically 45. Keeping in view the importance of medicinal plants, S. rhombifolia L., the current study evaluated the hepatoprotective and nephroprotective effects of S. rhombifolia L., in intoxicated mice. Studies in literature have shown that plants contain valuable phytoconstituents responsible for their hepatoprotective and nephroprotective effects 46-48. Furthermore, previous research on extracts obtained with different solvents from S. rhombifolia L. has reported similar biological activities, indicating that the active compounds in the plant may not vary significantly depending on the extraction method, as long as the plant material comes from the same geographical region. This strengthens the relevance of our findings with the hydroalcoholic extract, despite not using the methanolic extract as originally planned, which is consistent with the secondary metabolites reported for the hydroalcoholic extract of S. rhombifolia L., such as alkaloids and tannins 37.
S. rhombifolia L. is an herbaceous plant found worldwide, used to treat various medical conditions 14,15. However, there is currently insufficient information regarding the phytochemical composition of its crude extracts obtained from different solvents, as well as concerns about the safety of its consumption 36.
Toxicity testing on S. rhombifolia in our study aimed to confirm its safety based on locally collected specimens. Although there are previous reports supporting its safety, these studies have primarily examined plants from regions such as Africa and Asia. Variations in climate, soil composition, and other environmental factors in those regions may influence the plant’s secondary metabolite profile, potentially altering its pharmacological or toxicological effects. Therefore, evaluating the local variant was essential to ensure the relevance and accuracy of the safety data.
The use of animal models remains crucial in assessing the toxicity of new therapeutic agents, as it is an important component of the preclinical phase, which precedes their application to humans 49-51. To determine the acute oral toxicity of EMSr in male and female mice, the OECD 425 guideline 41 was used, which enables the identification of the median lethal dose (LD50) and undesirable effects at various doses. The results obtained in this study suggest that EMSr has low acute oral toxicity up to a maximum dose of 2,000 mg/kg, as no signs of toxicity were observed and no anatomical alterations were noted in the internal organs compared to the control group. These results are consistent with findings from studies conducted by Thounaojam et al., where they demonstrated the absence of signs of toxicity, and anatomical alterations of the internal organs with the oral administration of 5,000 mg/kg of an aqueous extract of roots of S. rhomboidea in rats 52; as with those reported by Heinichen et al., where they did not observe oral toxic effects with doses of 3,000 mg/kg of a hydroalcoholic extract of S. rhombifolia L. in mice 37. Although the extracts evaluated were of different natures and plant parts, they present similarities in terms of this parameter.
The effects of the different doses of the methanolic extract of S. rhombifolia L. on behavior, studied using the Irwin test 42, were not evident after oral administration of single doses of 50, 100 and 200 mg/kg of EMSr in mice of both genders, this indicates that EMSr does not affect the behavior or physiological function of the mice studied. Furthermore, it supports the findings of the acute and subchronic toxicity tests, since no adverse effects on general behavior were observed, nor was acute neurotoxicity observed in the murine model. However, diarrhea was observed in both sexes in the groups corresponding to the 200 mg/kg dose of EMSr (EMSr200), consistent with the prokinetic effect of the hydroalcoholic extract of S. rhombifolia L. in mice reported by Heinichen et al. 37.
Toxicity trials are classified based on their duration, which is typically aligned with clinical use. The sub-chronic toxicity test, following the 28-day protocol established in the OECD Guide 43, is a more applicable model to the domestic use of medicinal plants, since they are not generally consumed in a single high dose, but in smaller doses for a long time. The repeated dose toxicity test showed no signs of mortality or visible toxicity. There were no differences in daily body weight between the experimental groups. These results can be compared with those obtained in studies carried out by Da Costa et al., where they did not show signs of toxicity, nor differences in the appearance of the internal organs with a chronic treatment of varied doses of crude extract from aerial parts Sida rhombifolia L. in rats 36. Conversely, the various biochemical parameters remained unchanged by the end of the treatment period (day 28) with the different doses of the extract in mice of both sexes, except the serum levels of uric acid, which showed a significant decrease with the doses of EMsr50, EMSr100, and EMSr200 in males, and EMSr100 and EMSr200 in females.
The aim of evaluating the extract in the hepatocellular toxicity assay was to investigate its potential hepatoprotective effects in a model of paracetamol-induced liver injury. Silymarin was included as a reference hepatoprotective agent to serve as a positive control, allowing comparison and assessment of whether the extract exhibited similar protective activity.
A high dose of acetaminophen can lead to liver toxicity by forming a harmful metabolite, which is associated with membrane damage and elevated levels of ALT, AST, and ALP. The body attempts to detoxify this substance by developing a conjugate. However, nonmetabolized toxic products can bind covalently to proteins, resulting in further toxicity. Acetaminophen has been shown to cause significant inflammation in the liver tissue and surrounding areas 3.
In our research, hepatotoxicity is indicated by increased serum enzyme activities of ALT, AST, and ALP at a 300 mg/kg dose of acetaminophen (Figures 2A, 2 B, and 2C) compared to the control group. This has been linked to the compromised structural integrity of the liver and dysfunction of hepatocytes. The significantly reduced liver profile parameters in the silymarin group confirmed the validity of the acetaminophen acute liver injury model. Silymarin is known to be hepatoprotective due to its antioxidant properties, and its ability to promote cell regeneration is attributed to increased protein synthesis 53-55. Treatment with all doses of EMSr decreased the elevated serum activities of ALT and ALP compared with the APAP group. The restoration of deficient marker enzyme functioning implicates its cytoprotective role by stabilizing membrane actions towards normal liver cell function. In contrast, levels of total proteins and albumin were unaffected by any treatment, which was expected since the model used was for acute liver injury; thus, the secretory function of the liver remains intact (Figure 2 D and E).
The above results suggest that the methanolic extract of S. rhombifolia L. presents hepatoprotective activity. This result was similar to those reported by Ramadoss et al. the ethanolic extract of S. rhombifolia significantly reduces paracetamol-induced hepatotoxicity in rats 35. On the other hand, Poojari et al. demonstrated the hepatoprotective effect of the methanolic extract of S. rhombifolia seeds in rats in a model of hepatotoxicity induced by carbon tetrachloride. Also, they studied its potential chemopreventive effects associated with its ability to eliminate free radicals 56.
Gentamicin administration in high doses caused marked changes in the kidney tubules due to gentamicin reabsorption in the proximal convoluted tubules, resulting in degeneration and necrosis of the epithelial cells within the tubules. In some instances, this side effect can be so severe that the medication must be stopped. The effects are characterized by dilated tubules, loss of the brush border, significant infiltration of leukocytes, tubular degeneration, and the presence of tubular casts 6,7. Nephrotoxicity can be inferred from the increased levels of serum creatinine, urea, and uric acid (Figure 3 A, B, and C) at a 135 mg/kg dose of gentamicin compared with the control group. In silymarin-treated mice, those parameters were significantly attenuated when compared to the pathological group (gentamicin group). The significant reduction of the kidney profile parameters in the silymarin group confirms that the model of kidney injury caused by gentamicin was validated. Our results align with previous studies, which show that pre-treatment with silymarin significantly reduces the elevated serum creatinine and uric acid levels induced by gentamicin 13. Treatment with EMSr decreased the elevated serum levels of creatinine and uric acid compared to the gentamicin group. The result supports the nephroprotective effect of EMSr. It is important to note that serum uric acid levels showed a statistically significant decrease when different doses of the extract were administered together, compared to the control group. This decrease was similar to what was observed in the subchronic toxicity test. These findings have led to studies investigating the potential uricosuric activity of the extract.
There is increasing evidence implicating the mechanism of free radicals in the hepatotoxic and nephrotoxic effects of certain chemicals. Both acetaminophen and gentamicin, generate reactive oxygen species for which have a cytotoxic effect. It is important to note that serum uric acid levels showed a statistically significant decrease when different doses of the extract were administered together, compared to the control group. This decrease was similar to what was observed in the subchronic toxicity test. 3,6,7. Cell and tissue destruction leads to increased lipid peroxidation because antioxidants are diluted out, and transition metal ions that stimulate the peroxidation process are released from disrupted cells. Therefore, an increase in lipid conjugated dienes and hydroperoxides could significantly counteract treatment with EMSr.
Plants contain phenols, diterpenes, and coumarins, which are associated with significant antioxidant activities 57. A strong relationship exists between antioxidant activities and phenolic compounds 10,58. The antioxidant properties of plants depend on their content of phenols and flavonoids, which enable them to scavenge free radicals. This scavenging action may help protect against oxidative stress and related complications in animal models 13. Then, the hepatoprotective and nephroprotective effects of S. rhombifolia L. can be attributed to the antioxidant components present in the extract. Several studies have shown the antioxidant potential of extracts from the aerial parts of S. rhombifolia L., which demonstrate the presence of various bioactive molecules, including polyphenols, flavonoids, steroids, porphyrins, alkaloids, coumarins, and derivatives of ferulic acid (28, 31, 32, 36-38). In recent years, numerous plants have been tested in animal models for their potential to alleviate liver damage caused by paracetamol. Researchers have isolated the bioactive compounds that are responsible for reducing oxidative stress 59. Recently, Dhalwal et al. reported that extracts from the roots, leaves, whole plant, and stem of this plant demonstrated in vitro activities, including free radical scavenging, reducing power, superoxide anion scavenging, nitric oxide scavenging, and anti-lipid peroxidation 29.
In the current study, the extent of damage caused by acetaminophen and gentamicin was evident through increased serum levels of liver and kidney parameters, indicating hepatotoxicity and nephrotoxicity, respectively. On the other hand, the decreased level of EMSr extracts supports the hepatoprotective and nephroprotective properties of this plant species. Further studies will be necessary to identify the compound with antioxidant effects, quantify it, and elucidate the mechanism by which it exerts these potentially beneficial biological activities in clinical medicine. Many natural products exhibit significant antioxidant properties, as supported by numerous in vitro and in vivo studies, providing potential leads for the development of effective hepatoprotective and nephroprotective agents.
Conclusion
It was concluded that the methanolic extract of Sida rhombifolia L. aerial parts showed no significant toxic effects when administered orally to both male and female mice in acute and repeated dose toxicity models. A reduction in triglyceride levels was observed compared to the control group, which could suggest a possible improvement in metabolic function. These findings are consistent with the results of the Irwin test, which confirmed the physiological and neurological safety of the test. In addition, we demonstrate that the methanolic extract of S. rhombifolia L. aerial parts possess in vivo hepatoprotective and nephroprotective potential by reducing elevated levels of liver biochemical parameters, such as ALT and ALP, and kidney biomarkers, including serum creatinine and uric acid, in acetaminophen- and gentamicin-intoxicated mice, respectively. Literature review indicates that S. rhombifolia L. contains phenolic compounds and flavonoids, which may be responsible for its hepatoprotective and nephroprotective properties. However, the presence and contribution of these compounds in our extract will require further investigation to be confirmed.
















