Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
Boletín Científico. Centro de Museos. Museo de Historia Natural
Print version ISSN 0123-3068
Bol. Cient. Mus. Hist. Nat. Univ. Caldas vol.17 no.2 Manizales July/Dec. 2013
COLOMBIAN BUTTERFLIES (LEPIDOPTERA: PAPILIONOIDEA) ATTRACTED TO TREE EXUDATES*
MARIPOSAS COLOMBIANAS ATRAÍDAS POR EXUDACIONES DE CORTEZAS DE ÁRBOLES (LEPIDOPTERA: PAPILIONOIDEA)
Julián A. Salazar-E.1
* FR: 26-IV-2013. FA: 4-X-2013.
1 Facultad de Ciencias Exactas y Naturales, Programa de Biología, Universidad de Caldas, julianadolfoster@gmail.com
Resumen
A preliminary checklist is presented of fruit-feeding butterflies that visit the exudation of seven species of trees observed in several zones of Colombia. Additional data on secretion types, composition, butterfly behavior and the other insects sharing the same phenomena are included.
Palabras clave: feeding, Colombia, exudations, Coleoptera, Hymenoptera, food guild, proboscis.
Abstract
En este trabajo se publica una lista preliminar de especies de mariposas frugívoras, que se alimentan de exudados segregados por las cortezas de siete especies de árboles existentes en varias regiones de Colombia. Adicionalmente, se suministran datos de los tipos de secreciones, composición, hábitos de las mariposas y de otros insectos que comparten dicho fenómeno.
Key words: alimentación, Colombia, exudación, Coleoptera, Himenoptera, gremios alimenticios, proboscis.
INTRODUCTION
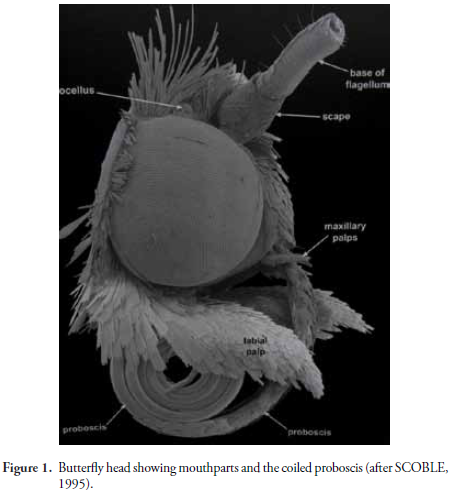
The Lepidoptera with their variety of colors and hyper diverse wing patterns are the most conspicuous species within the class Insecta. Almost all species have specialized eating habits using a proboscis, the tubular mouth parts evolved for the absorption of liquids or diluted foods (Fig. 1) (SCOBLE, 1995). The singular morphology of the proboscis is related to the feeding habits of adult Lepidoptera and the substrates on which they feed and is evident in both Rhopalocera (Glossata) and Heterocera (MOLLEMAN et al., 2005, 2010; KRENN, 2010; ZENKER et al., 2010). The last author details the proboscis of several butterfly species with electron microscopy images, illustrating the differences of those that feed on the nectar of flowers, pollen feeders and those that feed on rotting fruit, finding significant differences in the structures of the different groups studied. Likewise there are studies of sections of the functional mechanism and related muscles that act during the feeding process.
Other related works have treated, in addition to the morphological characters, such aspects as habits the communities of Lepidoptera have adapted and the use of certain food resources for adult diet benefits (NORRIS, 1935; HALL & WILLMOTT, 2000; BOGGS & DAU, 2004; MOLLEMAN et al., 2005; RIBEIRO et al., 2010). However what is not well known are those Neotropical species that have the predilection, in addition to feeding on fermented fruits, of imbibing the exudates or secretions from tree bark. These exudates are a resource of additional energy, rich in nutrients and are highly appreciated by insects, and include representative species of Lepidoptera. The objective of this work is to identify some of these butterflies in Colombia that specialize in this type of substrate and also the trees that have the property of secreting surface bark exudates.
PRELIMINARY DATA
Studies on the attraction of butterflies and other insects to tree exudates are scarce, and appear sporadically cited or illustrated in informative works about butterflies (HOLLAND, 1922; KLOTS, 1960; OWEN, 1971; SCOBLE, 1995; FRANCINI, 2010). For example, NORRIS (1935) mentions that sap exudates from wounded tree bark attracts several species of Nymphalid butterflies, and by feeding on those liquids become completely intoxicated and unable to fly. In this same context is the pioneer work of FOX-WILSON & HORT (1926) which is a detailed study of the insects that visit sap exudates from trees in Europe. They note that the most common species with this habit is Vanessa atalanta (L.), besides other records for Vanessa antiopa (L.) and Vanessa polychloros (L.) (BENSON, 1877; PEACHELL, 1900). Other species with the same known behavior are Vanessa c-album (l.), Limenitis camilla (Scudder), Argynnis pandora Schiff., Apatura iris (l.), Satyrus sp. and also the Lycaenidae Zepyrus quercus (L.) (FOX-WILSON & HORT, 1926; NORRIS, 1935).
In tropical environments of Africa the butterflies of the genus Charaxes are strongly attracted to these exudates, as documented in KNOLL (1921), TRIMEN (1862-1866), SCHULTZE (1916) and more recently VANE-WRIGHT (2003) it. KRENN (2008) found in an area of Costa Rica and Panamá a wide variety of butterfly feeding habits and discriminated them into categories of species that feed on the nectar of flowers, pollen, blood, sweat, rotten fruit, carrion, excrement and tree sap exudates. The author states that there are representatives of the Nymphalidae family which never consume nectar in the course of their life, but only fruit juices, fungi and plant exudates. This specialized group constitutes about 40 to 55% of all tropical forest Nymphalidae butterflies DE VRIES (1987), those which have a proboscis morphology particularly adapted for this purpose. Most of them can uptake liquids by rubbing the proboscis on wet surfaces (Fig. 2), while the Charaxinae butterflies can drill through the skin of decomposed fruits (KRENN et al., 2001; MOLLEMAN et al., 2005). This guild in tropical areas is mainly comprised of the Charaxinae, Morphinae, Brassolinae, Satyrinae, Biblidinae and Limenitidinae, all subfamilies of nymphalids butterflies (DE VRIES, 1988; SHAHABUDDIN & TERBORGH, 1999; DE VRIES & WALLA, 2001; FREITAS & BROWN, 2004; RIBEIRO et al., 2010; DE VRIES et al., 2012).

BACKGROUND IN COLOMBIA
The author's first experience of this phenomena was in March of 1987 during a visit to the biological station of the Organization for Tropical Studies (OTS) in Costa Rica. A Panacea prola lysimache (G. & S.) female, males of Prepona laertes octavia (Hbn) and Opsiphanes spp., were seen feeding on exudates of a unknown tree. Specific observations in Colombia are rare; VÉLEZ & SALAZAR (1991) and ANDRADE (1998) refer to the feeding habits of butterflies according to substrate or food guild groups but do not mention those that feed on tree sap. URIBE & SALAZAR (1998) note some species sighted at Caño Limón, Arauca, such as Archaeoprepona demophon (L.), Colobura dirce (L.), and Opsiphanes cassina ssp. feeding on exudates of small trees. SALAZAR et al. (2003) mentioned that certain species of trees secrete a sweet fluid from the bark that may be more effective than traps to capture butterflies, as it happened in a region of the Eastern Cordillera. Also detailed in this paper was the observation of Agrias amydon amaryllis Mich., A. amydon athenais Fruhst., and Prepona pylene jordani Fruhst., attracted to exudates in the Western Cordillera. OROZCO et al. (2009) found 9 species of fruit feeding butterflies in the southwest of Antioquia, one of them (Colobura dirce L.) was cited feeding on decomposed fruit, bark exudates and the droppings of mammals.
GARCÍA et al. (2002) states that adults in the subfamily Charaxinae inhabit the forest canopy and feed on fermented fruits, decomposing organic material, mammal excreta and tree exudates that produce acetyl ferments from the wounds caused by boring insect. Finally VALENCIA et al. (2005) cited the following species that feed on exudates of unidentified trees: Archaeoprepona amphimachus (L.), Fountainea glycerium (Dbl), Memphis philumena indigotica (Salv.), Colobura dirce (L.), Historis orion dious (Lam.), Panacea prola (Dbl.), Consul fabius cecrops (Dbl.) and Opsiphanes quiteria cauca (Rob). The authors also assert that Prepona philipponi (LM.) feeds on exudates, visits bee (Meliponini) honeycombs and imbibes the honey spilled on the crevices of logs, and that Siderone galanthis (Cr.) adults feed on exudates from wounds of the bark of "Yarumo" (Cecropia spp.), Cecropiaceae trees.
MATERIALS AND METHODS
Observations were carried out in the Municipality of Riosucio, Caldas (Vereda Aguacatal, La Iberia and Loma Las Brujas) located between 1510 and 1750 masl. Additionally in other locations the phenomena was observed in places such as Bajo Tablazo-Manizales, El Arroyo-Villamaría, and in the Eastern Cordillera at Chirajara located in the Municipality of Guayabetal at 1200 masl. Dates include: April 2, 21-30, May 1, 7 and 15, July 5, November 6, 7,13, 14, and 15 of 2011, March 26 and April 20, 2013. Butterflies feeding on trees exudates were photographed with a Canon Power-Shot 480A of 10.0 megapixels and a Canon Eos 3000 N. Observed species were identified visually but were not captured. The reference material is deposited at the Museum of Natural History, University of Caldas (MHN-UCa).
Secretions were collected in 2 cm plastic vials and preserved on ice. The samples were analyzed at the biochemistry laboratory at the University of Caldas by thin layer chromatography. The samples were diluted in 1.5 ml of ethanol and then placed in a silica chromatographic plate (2.5 cm wide x 5 cm. high) and wet with a mixture of hexane and ethyl acetate for 4 minutes. A UV lamp was also used to reveal any additional composition. Butterfly taxonomy follows the classification of LAMAS (2004) except for some recently described species (SALAZAR & CONSTANTINO, 2001; ORTIZ-ACEVEDO & WILLMOTT, 2013; MAIA-SILVA, 2013). SARMIENTO (1994), MARTÍNEZ (2000) and SUÁREZ & AMAT (2007) papers were consulted for the other groups of arthropods. The trees were identified with the help of PÉREZ-ARBELÁEZ (1996), MURILLO (2000), CAMPOS et al. (2008), CENICAFÉ (2010) and the Herbarium at the University of Caldas.
RESULTS
1) Trees that secrete exudates
Exudates found on certain tree bark are produced as a response to mechanical wounds caused by animal and human actions or pathological processes (BAXTER, 1952). In the first case an example is the well-known extraction process of latex from the bark of the rubber trees Hevea brasiliensis M.R. (Euphorbiaceae) (DOLIWA, 2008). Pathogens or diseases are recognized by various symptoms that show on the bark and cause fluids to leak from the tissues, according to MARSHALL-WARD (1901) this reveals the abnormal state of the tree, often due to bacteria present in the wound caused by mechanical damage, insects and fungi (Phytophthora spp.). The author includes in this treatise on the diseases of plants a special chapter on exudation and rot, including the phenomena of tumescence, decomposition, humidity, acidification, dew, gomosis and the emergence of diverse workflows that facilitate a profuse or permanent exudation in the barks. In Colombia eight species of trees have been registered as producers of bark secretions whose fermentation was attractive to butterflies and other orders of insects (Figs. 3-4).


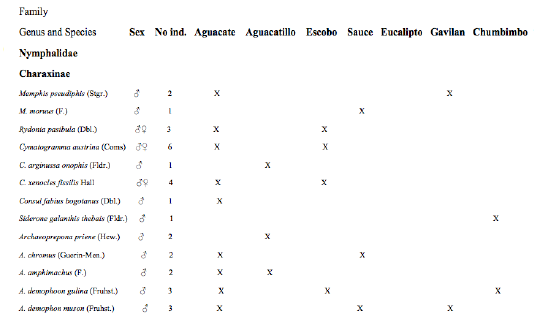
However this phenomena is common also in many species of trees that secrete fluids and do not attract any type of arthropod feeding (see Tabs. 1 and 2).
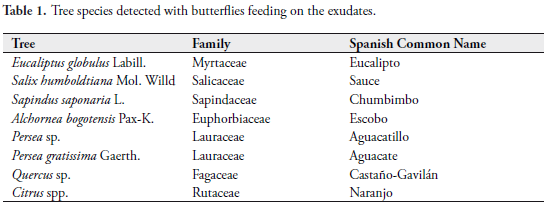
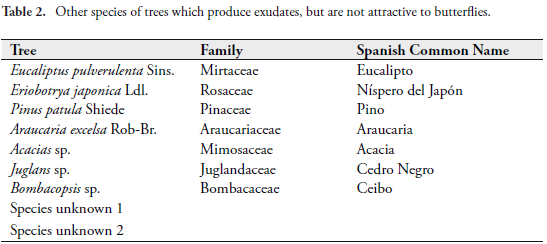
TYPES OF EXUDATES
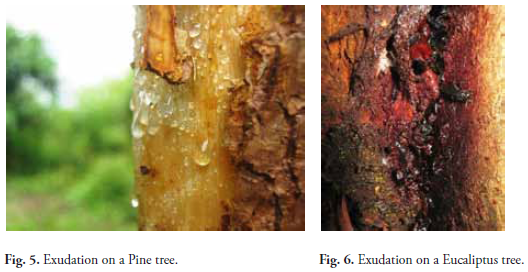
O'GILVIE (1924) observed several types of exudates on the bark of Elm trees, Chestnuts and Apples in Europe. He found mud red flows and another of a whitish color appearing in elms and willows. Bacteria and other organisms were detected in both types including Fusarium, Isaria, Torula, Alternalia etc. and yeasts (Oospora, Verticillium, Chlorella). In this work exudate types are categorized by their consistency: melosa (pines), resinous (eucalyptus, cedro, acacia), gummy (araucaria), gelatinous (unknown tree), viscose (aguacate, níspero, ceibo) and frothy (aguacate, escobo) (Figs. 5-10). The last, like some resins such as Eucalyptus and Willow, were attractive assets to butterflies.
Composition
According to FOX-WILSON & HORT (1926) the tree sap mostly contains variable amounts of sugar, which is easily fermented and produces in the company of several yeasts, alcoholic products. Sparkling exudates have been detected from reduced sugar and from that not reduced as fructose, glucose, dextrose and levulose (maltose) (OMURA et al., 2011) in an acidic reaction (ph 4-5), especially in the viscous exudate of Oak and also contained insoluble components composed of cellulose. A preliminary chromatographic analysis of the foamy secretion from the bark of the "Escobo" tree of revealed polyphenol compounds.


2) Communities of butterflies that attend the exudates. Some observations.
This work reports on the communities of butterflies that imbibe the exudates of seven species of trees. They belong to the guild of the fruit feeding species and are composed of representatives of the family Nymphalidae; subfamilies Charaxinae, Brassolinae, Satyrinae, Limenitidinae; Biblidinae and Nymphalinae. A total of 61 species in 35 genera of butterflies were registered. In addition other insects belonging to the orders Coleoptera and Hymenoptera that in some way interact with species of butterflies on the exudates and as regular visitors also were identified. Several butterflies that frequented the exudates of Aguacate, Aguacatillo, Gavilán and Escobo were seen especially between the hours of 8 a.m., to 3 p.m. It should be noted that certain species such as Pycina zamba (Dbl.), Opsiphanes spp. and Caligo spp. were observed only very early, surely because their crepuscular activity, after which they disappear from the exudates. Noted also is that the fermented product of the exudates can intoxicate the butterflies while feeding, to such an extent that they can be taken manually or be photographed with ease, even with species of the elusive genus Agrias (Figs. 11 and Figs. 12).

This irresistible attraction to butterflies is due to the release of odors by the breakdown and fermentation of the components of the exudates. In fact OMURA et al. (2000) in his studies on the secretions of oaks in Japan, found 14 volatile compounds such as ethanol and acetic acid which produced strong reactions in two species of Nymphalidae, Kaniska canace (l.) and Vanessa indica (Herbst), and attracted them to feed. This is probably the case with exudates observed in Colombia which exercised a strong attraction to different organisms, and more when the secretory activity in the bark is constant. An Aguacate tree observed produced a flow for 5 months (April-September, 2011) and in the case of an Escobo, for more than two years (November 2011 to June 2013). Long time exudation was observed from a single oak tree in a garden in Surrey, England which maintained this activity for up to five years FOX-WILSON & HORT (1926). This suggests that in old trees the exudation may be constant, as was found in trees of Aguacate on the Atlantic coast (F. Montero, pers. com.). From the analysis of the list of butterflies in Annex I, it is clear that most of the observed individuals arriving at exudates, are male (Figs. 13-17). However the females of some species like Epiphile epimenes (Hew.), Cymatogramma xenocles fissilis (Hall), C. perenna austrina (Comstock) and Manataria hercyna maculata (Hopffer) were quite common.


In terms of effectiveness, we can say that this natural method for the attraction of diurnal species may be more effective than those obtained with the use of artificially baited traps (Figs. 18-20), type Van Someren-Rydon, using fruit, fish or shrimp in decomposition. This phenomena can be utilized, in rainforest areas where colorful butterflies of the fruit feeding guild inhabit, as another alternative for the capture of rarities.



OTHER ASSOCIATED INSECTS
During the course of the observations made on tree exudate in this study, other arthropods were also present the flows. FOX-WILSON & HORT (1926) made a count of insects visiting an Oak tree, listing several species of Diptera, Hymenoptera and Coleoptera, many of them attracted by chemotropism, stimulated by the smell of the exudate.


In this study they found an commensal association similar to the one found by HEDSTROM & ELMQUIST (1984) in Costa Rica with the beetle Gymnetis liturata Olivier (Cetoniinae) and the charaxine butterfly Prepona laertes (Hbn). Here at least 4 species of Coleoptera: Gymnetis pantherina Blanchard, Amithao decemguttatum Watherhouse, Euphoria precaria Janson (Cetoniinae) and Trachyderes succinctus (Cerambycidae) and the wasp Polistes erythrocephalus L. (Hymenoptera) chew certain parts of the bark cortex which produces an exudation that is exploited by several species butterflies (eg. Epiphile epimenes Hewitson, Catonephele numilia esite Felder, Fountainea glycerium comstocki Witt, Cymatogramma xenocles fissilis Hall, Diaethria clymena marchallii Guer-Ménéville, and Rydonia pasibula Doubleday, among others) (Figs. 21-22).
ACKNOWLEDGEMENTS
Special thanks to José I. Vargas, Cristóbal Ríos-Málaver, Gabriel Rodríguez, Luis M. Constantino, Michel Dottax and Fredy Montero whom provided valuable information on the butterflies and trees studied in this work. Also to Frank Wieland (Dpt. Entomology, University of Göttingen, Germany), Dr. Keiichi Honda (College of Arts and Sciences, Hiroshima University, Japan), Dr. Harald Krenn (Dpt. Evolutionary Biology, University of Vienna), Dr. Olaf Mielke (Dpt. Entomology, University of Paraná, Brazil), Dr. Arthur H.B. Rydon (r.i.p.), Michael Dottax and Professor Fernando Vallejo (Dpt. Entomology, University of Caldas) for providing old works and additional identification, the staff of the laboratory of biochemistry at the University of Caldas and particularly to Mónica Obando for their support and Greg Nielsen for his kindly assistance in the revision of the English text.
BIBLIOGRAPHY
ANDRADE, M.G., 1998.- Uso de las mariposas como bioindicadores del tipo de hábitat y su biodiversidad en Colombia. Rev. Ac. Col. Cienc. Ex. Fis. & Nat., 23 (4): 497-421 + figs. [ Links ]
BAXTER, D.W., 1952.- Pathology in Forest Practice: 601 pp. + figs. John Wiley & Sons, N.Y. [ Links ]
BENSON, F., 1877.- Entomologic notes, captures. The Entomologist, 10: 252. [ Links ]
BOGGS, C. & DAU, B., 2004.- Resource specialization in puddling Lepidoptera. Environm. Ent., 33 (4): 1020-1024. [ Links ]
CAMPOS, E., ESPÍNDOLA, M. & MIJARES OVIEDO, P., 2008.- Diversidad del género Persea y sus usos: 58 pp. + figs. ICAMEX-CONACYT, México. [ Links ]
CENICAFÉ, 2010.- Árboles encontrados en zonas cafetaleras: 149 pp. inf. Cenicafé, Chinchiná, Colombia. [ Links ]
DE VRIES, P.J., 1987.- The Butterflies of Costa Rica and Their Natural History, I: 327 pp. + figs. Princeton University Press, Princeton. [ Links ]
________., 1988.- Stratification of fruit feeding nymphalid butterflies in a Costa Rican rainforest. J. Res. Lepid., 26 (1-4): 98-108. [ Links ]
DE VRIES, P.J., & WALLA, TH., 2001.- Species diversity and community structure in Neotropical fruit-feeding butterflies. Biol. J. Linnean Soc., 74: 1-15 + figs. [ Links ]
DE VRIES, P.J., ALEXANDER, L., CHACÓN, I. & FORDYLE, J., 2012.- Similarity and differences among rainforest fruit-feeding butterflies communities in Central and Southamerica. J. Animal Ecology, 81: 472-482 + figs. [ Links ]
DOLIWA, P., 2008.- Látex, historia, proceso de elaboración de la leche del árbol llorón. Matrona Prof., 9 (1): 30-32. [ Links ]
FRANCINI, R.B., 2010.- Historia Natural das Borboletas do Vale do Quilombo. Santos, SP. (autor ed.): 550 pp. + figs. Sp., Brasil. [ Links ]
FREITAS, A.V. & BROWN, K.S., 2004.- Phylogeny of the Nymphalidae (Lepidoptera). Systematic Biol., 53(13): 363-383 + figs. [ Links ]
FOX-WILSON, G. & HORT, N.D., 1926.- Insect visitors to sap-exudations of trees. Trans. Ent. Soc. London, 74: 243-254 + figs. [ Links ]
GARCÍA, C., CONSTANTINO, L., HEREDIA, M.D. & KATTAN, G., 2002.- Mariposas Comunes de la Cordillera Central de Colombia: 130 pp. + figs. Wildlife Conserv. Soc., Colombia. [ Links ]
HALL, J.P. & WILLMOTT, K., 2000.- Patterns of feeding behavior in adult male riodinid butterflies and their relationship to morphology and ecology. Biol. J. Linnean Soc., 69: 1-23 + figs. [ Links ]
HEDSTROM, I. & ELMQVIST, TH., 1984.- Prepona butterflies (Nymphalidae) and Hoplopyga beetles (Cetoniinae) on the same food source during the Neotropical dry season - a case of commensalism? Rev. Biol. Trop., 32 (2): 313-315 + fig. [ Links ]
HOLLAND, W.J., 1922.- The Butterfly Book: 382 pp. + figs. Doubleday Page & Comp., N.Y. [ Links ]
KNOLL, F., 1921.- Insekten und Blumen. Verlag der Zool-Bot. Gesellf., 12: 375 pp. + tafs. [ Links ]
KRENN, W.H., 2010.- Feeding mechanism of adult Lepidoptera: structure, function and evolution of the mouthparts. Ann. Rev. Ent., 55: 307-327 + figs. [ Links ]
__________., 2008.- Feeding behaviours of Neotropical butterflies (Lep. Papilionoidea). Stapfia, 80 (NS): 295-304 + figs. [ Links ]
KRENN, W.H., ZULKA, K. & GATSCHNEGG, T., 2001.- Proboscis morphology and food preferences in nymphalid butterflies (Lep. Nymphalidae). J. Zool. London, 254: 17-26. [ Links ]
KLOTS, A.B., 1960.- Vida y Costumbres de la Mariposas: 204 pp. + figs. Ed. Juventud, Barcelona. [ Links ]
LAMAS, G. (editor), 2004.- Atlas of Neotropical Lepidoptera. Checklist, part 4A Hesperioidea-Papilionoidea. ATL + Scient. Publ. Gainesville, USA. [ Links ]
MAIA-SILVA, F., 2013.- Taxonomic e análise filogénetica de Memphis (Hübner,1819) e géneros afins (Lep. Nymph. Charaxinae). Baseadaem caracteres morfológicos e moleculares: 284 pp. + figs. Teses. Universidade Federal do Paraná, Curitiba, Brasil. [ Links ]
MARSHALL-WARD, H., 1901.- Disease in Plants: 309 pp. Macmillan & Co. Ltd., London. [ Links ]
MARTÍNEZ, C., 2000.- Escarabajos longicornios (Col. Cerambycidae) de Colombia. Biota Col., 1(1): 76-105. [ Links ]
MOLLEMAN, F., WHITKER, M. & CAREY, J., 2010.- Rating palability of butterflies by measuring feeding behavior. Entomol. Berichten, 70(2): 52-62 + figs. [ Links ]
MOLLEMAN, F., KRENN, W.H., ALPHEN, M., BRAKEFIELD, P., DE VRIES, P. & ZWAAN, B., 2005.- Food intake of fruit-feeding butterlies: evidence for adaptative variation in proboscis morphology. Biol. J. Linnean Soc., 31 (90): 31-41+ figs. [ Links ]
MURILLO, J., 2000.- Las Euphorbiaceae de Colombia. Biota Col., 5 (2): 183-200. [ Links ]
NORRIS, M.J., 1935.- The feeding-habits of the adult Lepidoptera Heteroneura. Trans R. Ent. Soc. London, 85 (2): 61-89. [ Links ]
O'GILVIE, L., 1924.- Observations on the "Slime Flux" of trees. Trans British Mycol. Soc., 9 (3): 167-182 + fig. [ Links ]
OMURA, H., HONDA, K. & HAYASHI, N., 2000.- Identification of feeding attractants in Oak sap for adults of two nymphalid Butterflies, Kaniska canace and Vanessa indica. Physiol. Ent., 25: 281-287 + figs. [ Links ]
OMURA, H., HONDA, K., ASAOKA, K. & INOUE, J., 2011.- Divergent behavioral and electrophysiological taste response in the mid-legs of adult butterflies Vanessa indica and Argyreus hyperbius. J. Insect. Physiol., 57: 118-126. [ Links ]
OROZCO, S., MURIEL, S. & PALACIO, J., 2009.- Diversidad de Lepidópteros diurnos en un área de bosque seco tropical del occidente antioqueño. Actual. Biol., 31 (90): 31-41 + figs. [ Links ]
ORTIZ-ACEVEDO, E. & WILLMOTT, K., 2013.- Molecular systematics of the butterfly tribe Preponini (Nymph. Charaxinae). Systematic Ent., 38 (2): 440-44. [ Links ]
OWEN, D.F., 1971.- Tropical Butterflies: 214 pp. + figs. Clarendon Press, Oxford. [ Links ]
PARRA, M.L., VARGAS, J.I. & TABARES, M., 2000.- Mariposas de Manizales: 117 pp. + figs. IPC-Tizán Ltd., Manizales. [ Links ]
PEACHELL, G., 1900.- Captures and field reports. The Entomologist, 33: 304. [ Links ]
PÉREZ-ARBELÁEZ, E., 1996.- Plantas útiles de Colombia: 831 pp. + figs. Fondo FEN, Bogotá, Colombia. [ Links ]
RIBEIRO, D., PRADO, P., BROWN. K.S. & FREITAS, A.V., 2010.- Temporal diversity patterns and phenology in fruit feeding Butterflies in the Atlantic forest. Biotropica, 42 (6): 710-716 + figs. [ Links ]
SALAZAR, J.A. CONSTANTINO, L.M., 2001.- Synthesis of the colombian Charaxidae and description of new genera for Southamerica: Rydonia, Annagrapha, Pseudocharaxes, Muyshondtia, Zikania. (Lep. Nymphaloidea). Lambillionea, 101 suppl. 2: 344-369 + figs. [ Links ]
SALAZAR, J.A., GIRALDO, M. & VARGAS, J.I., 2003.- Más observaciones sobre la concentración de mariposas territoriales en cumbres de cerros colombianos y especies residentes en el bosque de Bavaria (Villavicencio, Meta), su distribución y trofismo. Bol. Cient. Mus. Hist. Nat. U. de Caldas, 7: 255-317 + figs. [ Links ]
SARMIENTO, C., 1994.- Lista de las avispas sociales (Hym. Vespidae) de Colombia. Rev. Biol. Trop., 42 (1/2): 357-363. [ Links ]
SCHULTZE, A., 1916.- Die Charaxiden und Apaturiden der kolonie Kamerun. Arch. für Biont., 4 (1): 129 pp. + tafs. [ Links ]
SCOBLE, M., 1995.- The Lepidoptera: form, function and diversity: 404 pp. + figs. The Natural History Museum, Oxford University Press, Oxford. [ Links ]
SHAHABUDDIN, G. & TERBORGH, J., 1999.- Frugivorous butterflies in Venezuela forest fragments: abundance, diversity and the effects of isolation. J. Trop. Ecology, 15: 703-722 + figs. [ Links ]
SUÁREZ, M. & AMAT, G., 2007.- Lista de especies de los escarabajos fruteros de Colombia (Col. Cetoniinae). Biota Col., 8 (1): 69-76. [ Links ]
TRIMEN, R., 1862-1866.- Rhopalocera Africae Australis: 353 pp. + taf. Trubner & Co., Patternoster row, London. [ Links ]
URIBE, C. & SALAZAR, J.A., 1998..- Naturaleza de la Orinoquia, Mariposas del Llano: 99 pp. + figs. C. Uribe editores, Bogotá [ Links ].
VALENCIA, C.A; GIL, Z. & CONSTANTINO, L.M., 2005.- Mariposas diurnas de la zona cafetera colombiana: 244 pp + fihs. Cenicafé, Chinchiná, Colombia. [ Links ]
VANE-WRIGTH, D., 2003.- Butterflies: 112 pp + figs. Life Series. The natural History Museum, London publ [ Links ]
VELEZ, J. & SALAZAR, J.,1991.- Mariposas de Colombia: 26-27.Villegas eds, Bogotá [ Links ].
ZENKER, M., PENZ, C., PARIS, M. & SPECHT, A., 2010.- Proboscis morphology and its relationship to feeding habits in noctuid moths. J. Insect Science, 11 (42): 1-10 + figs. [ Links ]