Introduction
Community-acquired pneumonia (CAP) has become the leading cause of infectious deaths worldwide1. In the United States, the estimated incidence of CAP ranges from 106 to 164 cases per 10,000 inhabitants. In Latin America, this figure can reach 294 cases per 10,000 inhabitants1-3. Despite advances in vaccination, CAP accounts for 5-12% of lower respiratory tract infections requiring hospitalization2-5. The mortality associated with CAP varies from 5% to 25% in hospital settings and can reach up to 50% in intensive care units (ICUs)2-4. Among hospitalized patients requiring ICU care, CAP mortality at 3 months post-discharge can increase to 28%, particularly in older adults, those with comorbidities, multi-organ impairment, and low blood oxygen levels6-8.
The coronavirus disease (COVID-19) pandemic has adversely affected the global healthcare system. Approximately 20% of individuals infected with Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) require hospitalization. Of these, up to 81% may need intensive care, with mortality rates varying widely from 40% to 85%9-11. A study by Cangemi et al.13 reported that the overall mortality risk in COVID-19 patients is five times higher than that in patients with CAP. These differences in mortality rates may be related to disease severity, comorbidities, and patient age, highlighting the need to stratify and analyze patient groups with CAP and COVID-19 pneumonia9-13.
To date, medical literature has provided limited information on long-term survival when comparing CAP with SARS-CoV-2 infection, an essential aspect for a comprehensive understanding of both diseases in the context of acute respiratory infections13,14. The objective of our research was to compare the survival of patients with CAP and SARS-CoV-2 pneumonia to that of a control group and to describe the risk factors associated with mortality in these groups.
Materials and methods
A retrospective cohort study on respiratory conditions was conducted on patients diagnosed with CAP, COVID-19, and a control group admitted to the emergency and intensive care services from January 2006 to December 2021. The primary objective of this study was to describe the differences in the survival and clinical characteristics associated with mortality in this population.
Selection Criteria
Inclusion criteria for the study were: age > 18 years, hospitalization for a diagnosis of CAP and COVID-19, and a complete medical history including paraclinical tests, chest radiography, and chest computed tomography (CT) at the time of admission. Patients admitted to palliative care and those who developed nosocomial infections.
CAP is defined as an acute illness associated with at least one of the following signs or symptoms: fever, new cough with or without sputum production, pleuritic chest pain, dyspnea, or altered breath sounds on auscultation2,8. This was in addition to a chest radiograph showing alveolar or interstitial infiltrates, consolidation, or cavitations with or without pleural effusion appearing within the first 48 h after hospitalization2,8. COVID-19 diagnosis was defined by a positive polymerase chain reaction (PCR) test15. Patients hospitalized with respiratory symptoms who did not meet the criteria for CAP or CO-VID-19 were classified as non-CAP/COVID-19. This selection process was carried out by trained healthcare professionals, including doctors and specialized nursing staff, who assessed the patients at the time of their admission to the study center.
Variables
Described variables included age, sex, comorbidities, and previous or current tobacco use, complete blood count, oxygen partial pressure, carbon dioxide partial pressure, bicarbonate, corrected bicarbonate, base excess, arterial oxygen saturation, lactate dehydrogenase, fractional inspired oxygen, oxygen partial pressure/fractional inspired oxygen, creatinine, blood urea nitrogen, chest X-ray, and chest CT; these data were obtained from the medical record at the time of hospital admission. Additionally, the need for ICU care, invasive mechanical ventilation (IMV), and/or vasopressor support was considered. The dependent variable was mortality assessed 12 months after the diagnosis of CAP, COVID-19, or in the control group.
Follow-up and survival information was obtained through phone calls to patients or their relatives. To minimize possible errors in the classification of the outcomes studied, the research team that recorded the clinical data had medical expertise for diagnosing the study conditions. To reduce data entry bias, the information was reviewed by at least two team members.
Sample Size
The sample size was calculated using the data from the studies by Uranga et al.16, Moreno-Torres et al.17, and Vestbo et al.18 reported 12-month survival rates of 88%, 89%, and 93% for patients with CAP, COVID-19, and other respiratory causes, respectively. With an expected loss rate of 5%, confidence level of 95%, and power of 80%, 240 patients per intervention group were required.
Statistical Analysis
Data were transcribed into Research Electronic Data Capture (REDCap) software19,20and analyzed using SPSS version 25. Quantitative variables were summarized using means and standard deviations (SD) for normal distributions and medians with interquartile ranges for non-normal distributions. Normality was assessed using the Shapiro-Wilk test. Qualitative variables were summarized as frequencies and percentages. To compare quantitative variables, ANOVA and Kruskal-Wallis tests were used based on distribution, and for qualitative variables, the chi-square test was used21,22. Twelve-month survival was evaluated graphically using the Kaplan-Meier method, and the log-rank test was used to assess statistical differences between diseases. The data were then stratified by age, sex, CURB-65, and Charlson index. Time to event was used as the dependent variable in the Cox regression model, with variables selected for inclusion through bivariate analysis using a significance level of p < 0.2 and biological plausibility related to mortality21,22. Hazard ratios (HR) for each variable were determined with a significance level of p < 0.05. Missing data imputation analysis was performed for variables with less than 10% loss, applying weighted mean imputation for quantitative variables and logistic regression for qualitative variables. Variables with > 10% data loss were excluded.
Results
General Characteristics of the Population
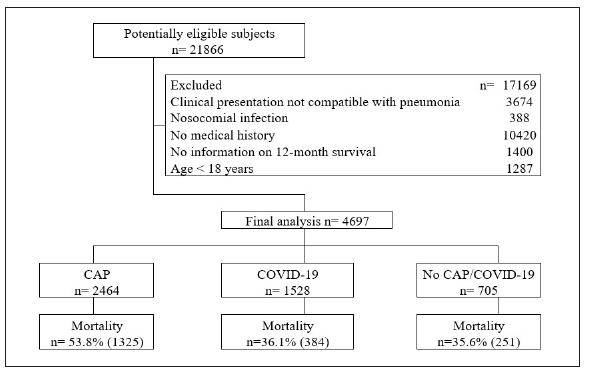
A total of 4,697 patients were included, with 52.5% having CAP (2,464/4,697), 32.5% having COVID-19 (1,528/4,697), and 15% having neither CAP nor COVID-19 (705/4,697) (Figure 1). The average age of the overall age was 63.9 years (SD: 19.65), and 59.5% of the patients were male (Table 1). Among patients diagnosed with CAP, 48.8% (1,201/2,464) had a history of systemic hypertension, 28.4% (700/2,464) had Chronic Obstructive Pulmonary Disease (COPD), and 12.1% (297/2,464) had diabetes mellitus. Cough was present in 77.8% (1,916/2,462) of patients with CAP compared to 18.3% (280/1,528) with COVID-19 and 44.3% (312/705) in the non-CAP/COVID-19 group. Headache was present in 24.2% (370/1,528) of the COVID-19 patients compared to 7.9% (194/2,462) of the CAP patients and 11.5% (81/705) of the non-CAP/COVID-19 group.
Table 1 General characteristics and comorbidities of the population
| Total population | CAP | Covid-19 | No CAP/COVID-19 | P value | |
|---|---|---|---|---|---|
| n= 4697 | n= 2464 | n= 1528 | n= 705 | ||
| Age years, m(sd) | 63.9 (19.65) | 67 (20.72) | 60.4 (16.23) | 60.7 (20.78) | <0.001 |
| Male sex n(%) | 2796 (59.5) | 1466 (59.5) | 917 (60) | 413 (58.6) | 0.813 |
| Dyspnea n(%) | 3038 (64.7) | 1747 (70.9) | 935 (61.2) | 356 (50.5) | <0.001 |
| Cough n(%) | 2508 (53.4) | 1916 (77.8) | 280 (18.3) | 312 (44.3) | <0.001 |
| Diarrhea n(%) | 526 (11.2) | 159 (6.5) | 296 (19.4) | 71 (10.1) | <0.001 |
| Fever n(%) | 2436 (51.9) | 1274 (51.7) | 849 (55.6) | 313 (44.4) | <0.001 |
| Headache n(%) | 645 (13.7) | 194 (7.9) | 370 (24.2) | 81 (11.5) | <0.001 |
| Altered consciousness n(%) | 467 (9.9) | 367 (14.9) | 34 (2.2) | 66 (9.4) | <0.001 |
| Rales n(%) | 2037 (43.4) | 1380 (56) | 436 (28.5) | 221 (31.3) | <0.001 |
| Wheezing n(%) | 643 (13.7) | 531 (21.6) | 60 (3.9) | 52 (7.4) | <0.001 |
| Acute myocardial infarction n(%) | 204 (4.3) | 129 (5.2) | 52 (3.4) | 23 (3.3) | 0.007 |
| Chronic heart failure n(%) | 524 (11.2) | 418 (17) | 50 (3.3) | 56 (7.9) | <0.001 |
| Dementia n(%) | 353 (7.5) | 267 (10.8) | 33 (2.2) | 53 (7.5) | <0.001 |
| COPD n(%) | 912 (19.4) | 700 (28.4) | 111 (7.3) | 101 (14.3) | <0.001 |
| Diabetes n(%) | 645 (13.7) | 297 (12.1) | 272 (17.8) | 76 (10.8) | <0.001 |
| Hypertension n(%) | 2066 (44) | 1201 (48.8) | 599 (39.2) | 266 (37.7) | <0.001 |
| Asthma n(%) | 101 (2.2) | 50 (2) | 35 (2.3) | 16 (2.3) | 0.836 |
| Active smoker n(%) | 38 (1.9) | 6 (3.2) | 25 (1.6) | 7 (2.3) | 0.286 |
| Ex-smoker n(%) | 138 (6.8) | 13 (6.9) | 110 (7.2) | 15 (4.9) | 0.334 |
| Charlson Comorbidity Index, m(sd) | 1.9 (2.15) | 1.7 (2.18) | 2.3 (2.03) | 1.8 (2.18) | <0.001 |
Notes: m: average; sd: standard deviation; CAP: Community-Acquired Pneumonia; COVID-19: coronavirus disease 2019; COPD: Chronic obstructive pulmonary disease.
Laboratory Tests and Medical Treatment
Among the general population, 13% (640/4,697) of the total population required vasopressor support, with 19.8% (303/1,528) in the COVID-19 group, 10.4% (257/2,464) in the CAP group, and 7.1% (50/705) in the non-CAP/COVID-19 group (Table 2). 21.5% (1,011/4,697) of the general population required ICU admission, with 35.6% (544/1,528) in the COVID-19 group, 15.1% (372/2,464) in the CAP group, and 13.5% (95/705) in the control group. Details of the laboratory tests and arterial gases are provided in the supplementary files (supplementary file).
Table 2 Corticosteroids, vasopressor support and mechanical ventilation
| Total population n= 4697 | CAP | Covid-19 | No CAP/ m\ti n 1Q | P value | |
|---|---|---|---|---|---|
| n= 2464 | n= 1528 | LUV 1 \J- 1 7 n= 705 | |||
| Dexamethasone n(%) | 1330 (65.7) | 117 (62.2) | 1041 (68.2) | 172 (55.8) | <0.001 |
| Septic shock n(%) | 640 (13.6) | 318 (12.9) | 264 (17.3) | 58 (8.2) | <0.001 |
| Vasopressor support n(%) | 610 (13) | 257 (10.4) | 303 (19.8) | 50 (7.1) | <0.001 |
| ICU n(%) | 1011 (21.5) | 372 (15.1) | 544 (35.6) | 95 (13.5) | <0.001 |
| Days in ICU, m(sd) | 9.4 (13.99) | 10.3 (19.25) | 8.9 (7.43) | 9.6 (18.92) | 0.200 |
| IMV n(%) | 638 (13.6) | 266 (10.8) | 320 (21) | 52 (7.4) | <0.001 |
| NIMV n(%) | 384 (8.2) | 158 (6.4) | 165 (10.8) | 61 (8.7) | <0.001 |
Notes: CAP: Community-Acquired Pneumonia; COVID-19: coronavirus disease 2019; n: number; m: average; sd: Standard deviation; ICU: intensive care unit; IMV: invasive mechanical ventilation; NIMV: Non-invasive mechanical ventilation.
One-Year Survival Analysis
The overall survival rate was 58.3% (Supplementary file). Survival among CAP patients was 46.2%, among COVID-19 patients was 74.9%, and among those with neither CAP nor COVID-19 was 64.4% (Figure 2). The survival rates for men and women were 46.2% and 47.2% for CAP (p < 0.001), 73.3% and 78.3% for COVID-19 (p < 0.001), and 64.9% and 63.9% for non-CAP/COVID-19 (p < 0.001), respectively.
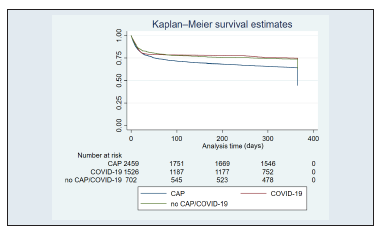
Figure 2 General survival by disease. CAP: Community-Acquired Pneumonia; COVID-19: coronavirus disease 2019.
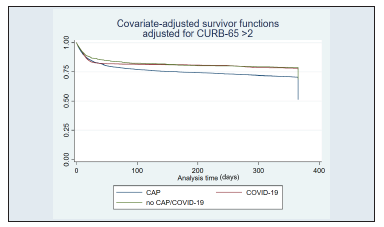
When stratified by age, patients aged > 65 years had a survi val rate of 28.2% for CAP (p < 0.001), 61.3% for COVID-19 (p < 0.001), and 43% for non-CAP/COVID-19 (p < 0.001) (Figure 3). When stratified by a Charlson score ≥3, survival was 17% for CAP (p < 0.001), 59.4% for COVID-19 (p < 0.001), and 49.7% for non-CAP/COVID-19 (p < 0.001) (Figure 4). Finally, when stratified by CURB-65 ≥2, survival rates were 15.4% for CAP (p < 0.001), 31.5% for COVID-19 (p < 0.001), and 20.2% for non-CAP/COVID-19 (p < 0.001) (Figure 5).
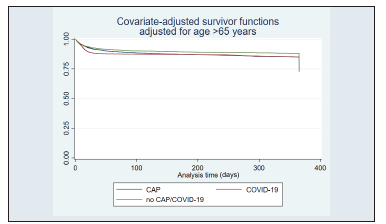
Figure 3 Age adjusted survival. CAP: Community-Acquired Pneumonia; COVID-19: coronavirus disease 2019.
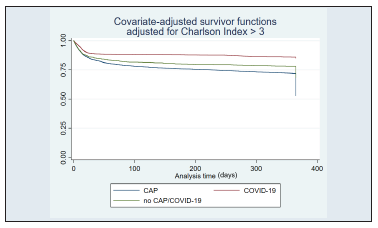
Figure 4 Charlson index adjusted survival. CAP: Community-Acquired Pneumonia; COVID-19: coronavirus disease 2019.
Cox Regression: Independently Associated Characteristics with Long-term Mortality
Cox regression analysis showed that being male (HR: 1.142; 95% CI: 1.042-1.252; p = 0.004), age over 65 years (HR: 2.622; 95% CI: 2.324-2.959; p < 0.001), Charlson >3 (HR: 1.770; 95% CI: 1.604-1.954; p < 0.001), CURB-65 >2 (HR: 2.081; 95% CI: 1.874-2.313; p < 0.001), and a history of CAP (HR: 1.569; 95% CI: 1.420-1.735; p < 0.001) were associated with increased mortality at one year (Table 3).
Table 3 Cox Regression: Independently Associated Characteristics with Mortality
| Variable | HR | CI 95% | p-value |
|---|---|---|---|
| Male sex | 1.142 | 1.042-1.252 | <0.001 |
| Charlson Comorbidity Index >3 | 1.770 | 1.604-1.954 | 0.004 |
| CURB-65 >2 | 2.810 | 1.874-2.313 | <0.001 |
| Age >65 years | 2.622 | 2.324-2.959 | <0.001 |
| History of CAP | 1.569 | 1.420-1.735 | <0.001 |
| LR chi2(5) = 1158.63 | |||
| p-value: <0.001 | |||
Notes: HR: Hazard Ratio; CAP: Community-Acquired Pneumonia.
Discussion
In this study, we evaluated the 12-month survival of patients admitted with CAP, COVID-19, and a control group. Clinical factors associated with reduced survival rates were also identified. Lower survival at one year was observed in patients with CAP, particularly in men, those older than 65 years, those with a Charlson score ≥3, CURB-65 ≥2, and a history of CAP. The need for vasopressor support and intensive care unit (ICU) admission was higher in patients with COVID-19. The most common symptoms were cough and headache in patients with CAP and COVID-19, respectively. Hypertension, COPD, and diabetes mellitus were more common in patients with CAP. Our analysis shows relevant differences in 12-month survival and clinical characteristics at admission between the two patient populations hospitalized for CAP or COVID-1923,24.
The control group, consisting of patients hospitalized for respiratory symptoms but who did not meet the diagnostic criteria for CAP or COVID-19, provides a valuable baseline for comparison. Unlike the CAP and COVID-19 groups, the non-CAP/COVID-19 patients exhibited a distinct clinical profile, with moderate rates of symptoms, such as cough and headache, bridging the gap between the more severe presentations in the CAP and COVID-19 groups. Stratified analyses further revealed that age > 65 years, high Charlson scores, and CURB-65 severity correlated with decreased survival in this group, paralleling trends seen in CAP and COVID-19 populations. The inclusion of this control group highlights the spectrum of respiratory hospitalizations and emphasizes the heterogeneity of clinical outcomes, enriching the study's overall analysis.
In a population with a history of COPD, Sheikh et al.14 described a sevenfold higher risk of in-hospital mortality in patients with SARS-CoV-2 pneumonia than in a control group with CAP. Additionally, a median hospital stay of 15.5 days (IQR = 6.8-30.0) was recorded for patients with COVID-19 compared to 5 days (IQR = 3.0-9.0) for the group without SARS-CoV-2 infection14. In contrast, our results revealed lower survival rates and longer hospital stay in patients with CAP. This could be attributed to greater disease severity and frequency of respiratory failure, as well as impaired lung function and dysfunctional immunity caused by conditions such as COPD, diabetes mellitus, and smoking25-27.
Pathophysiological mechanisms involving chronic inflammation with higher systemic cytokine levels have been described in CAP than in COVID-19. These processes explain greater endothelial damage and platelet activation, which are associated with worse short- and long-term clinical outcomes28,29. However, our study was designed to evaluate the clinical differences and long-term survival between COVID-19 and CAP, and not to identify differences related to endothelial biomarker analysis and their association with clinical outcomes28,29.
A prospective observational study by Cangemi et al.13 reported a higher incidence of thromboembolic events and in-hospital mortality associated with thrombosis in patients with COVID-19 than in those with CAP. Although our results assessed long-term mortality, we observed lower survival rates in patients with CAP13. It is crucial to highlight that thromboembolic events in patients with SARS-CoV-2 infection significantly contribute to short-term mortality, underscoring the need to evaluate the long-term outcomes in patients with COVID-19 and CAP. However, both studies observed similarities in patients with CAP upon hospital admission. These patients were older, predominantly male, and had a higher burden of cardiovascular and pulmonary disease13.
In our study, patients with COVID-19 had higher long-term survival rates and a lower burden of associated comorbidities. At the time of hospital admission, the COVID-19 population was younger, predominantly male, and had a shorter ICU stay. Our findings highlight differences in comorbidity burden between patients with CAP and COVID-19, which may explain the lower 12-month survival rate in patients with CAP due to a higher incidence of hypertension, chronic obstructive pulmonary disease, and diabetes mellitus 23,24. It is important to note that a significant percentage of COVID-19 patients may remain asymptomatic or experience mild to moderate respiratory symptoms. However, in our study, a considerable number of patients with SARS-CoV-2 infection presented symptoms such as diarrhea and headache, which increased the risk of developing long-term COVID10-12. Nevertheless, more studies are needed to evaluate the relationship between comorbidity burden and the risk of developing "long COVID" with long-term fatal clinical outcomes.
The elderly population is at a higher risk of developing respiratory infections due to multiple comorbidities, alterations in the immune system, decreased cellular regeneration capacity, and reduced respiratory reserve30,31. Our study supports these data by identifying a significant association between reduced one-year survival, age over 65 years, and Charlson score ≥3. Additionally, factors such as reduced vaccine efficacy in older adults and those with comorbidities may also be linked to increased susceptibility to severe diseases and worse clinical outcomes30,31.
Limitations
The limitations of our study include its observational nature and the omission of information obtained from clinical records. It is worth noting that the staff responsible for data collection was trained in data transcription. Our study included sufficient subjects to test the proposed hypothesis. However, the cause of death at 12 months could not be determined, which might have provided important information. Future studies that corroborate our findings are warranted. Additionally, evaluating and comparing the effect of thrombosis and embolism events on 12-month mortality in patients with CAP and COVID-19 has been investigated13.
In conclusion, this study described lower survival in patients with CAP at 12 months of follow-up, identifying factors associated with reduced survival, such as being male, age > 65 years with comorbidities, and severe pneumonia. Patients with COVID-19 presented with a higher need for support and intensive care along with symptoms and comorbidities.