Chagas is a disease with acute, subacute, and chronic phases caused by Trypanosoma cruzi (T. cruzi) infection 1. T. cruzi, is a pleomorphic hemoflagellate protozoan belonging to the class Mastigophora 2-4. It can be harbored by more than 100 animal species including wild and domestics animals. In Colombia, the main vector of T. Cruzi is a triatomine, Rhodnius Prolixus, but it could also be transmitted by blood transfusions (Figure 1).
In Colombia, Chagas infection is concentrated in the departments of Santander, Meta, Boyacá, and the Magdalena Valley.1 Chagas disease causes heart failure (AHF), which can be moderate, unilateral, acute, and chronic, depending on the involvement of the heart 5-7.
Primary strategies on Chagas disease prevention consist of eliminating the risk of infection and interrupting the chain of transmission through vector and non- vector control. Secondary prevention rests on screening and detection of individuals infected with T Cruzi, in the early stages of the disease, especially in acute cases. But for individuals in advanced stages of chronic diseases, tertiary prevention consists of clinical interventions to limit the morbidity and mortality of the disease and it may include surgical procedures 5. Recently, it has been proposed that implantation of devices that provide mechanical circulatory support may help Chagas' patients with advanced IC to improve its quality of life.
An exploratory search of the literature at a global and national level was carried out on the Hear mate 3 techno-logical device for the correction of chronic heart failure due to Chagas disease. The databases explored included Cochrane Library, LILACS, SciELO, MEDLINE, PubMed, and PubMed Central. We found no paper where a similar device had been used in a Chagas' patient.
Here, we present a case report on a patient with a chronic cardiopathy due to Trypanosoma cruzi infection who was implanted with Heart mate III, a new device providing mechanical circulatory support. This is likely the first of such reports in Colombia.
MATERIALS AND METHODS
Original case study of a patient with Chagas disease. The methods used for the correct diagnosis of chronic heart failure caused by Chagas disease were based on serological tests for the detection of IgG antibodies, which detected the presence of T Cruzi. Additionally, the patient underwent different diagnostic tests that detected the presence of the disease due to functional and structural failures of the heart (Table 1).
Table 1 Differential diagnoses were found during care, before the implantation of the technological device (Hearmate III)
| Diagnostic tests are performed before implantation of the technological device (Hearmate III) | |
|---|---|
| Type of examinations | Results Trans esophageal ultrasound, Doppler, trans thoracic echocardiogram, chest CT |
|
Transesophageal ultrasound Doppler Transthoracic Echocardiography Chest CT scan |
Steveson acute chronic heart failure. Chagasic dilated cardiomyopathy. Severely depressed LVEF 15% - 20%. Advanced biventricular Chagasic heart failure Severe pulmonary hypertension Moderate tricuspid and mitral insufficiency Atrial fibrillation and atrial flutter, unspecified Congestive heart failure. Cardiogenic Shock. Risk of right ventricular dysfunction CDI reprogramming. |
| Chest x-rays | Chest X-Ray Results |
|
Vascular pedicle without alteration Calcified atheromas on the aortic button. Prominence of the central pulmonary vasculature. Severe global cardiomegaly. A nodular image of a calcified appearance projected on the left lung base may correspond to residual nodules. Three-chamber synchronizing cardio. Right peripheral insertion catheter end in superior vena cava Risk of hemodynamic deterioration. Arterial hypertension. | |
RESULTS
The patient was a 54-year-old male patient, of mixed race, who worked as a bus driver in a coal mine in the department of La Guajira, but was retired from work, with benefits, since 2017. In 2017, he was diagnosed with Chagas disease after consulting for symptoms compatible with chronic heart failure - exhaustion, fatigue, and difficulty breathing. (Figure 2).
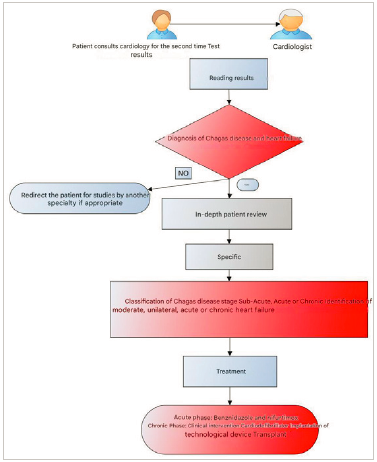
Source: Own elaboration.
Figure 2 Diagnostic and treatment algorithm for Chagas disease and heart failure
He was ordered a trans esophageal ultrasound doppler, a transthoracic echocardiography and a chest CT scan which reported a dilated cardiomyopathy, a severely depressed ejection fraction (15% - 20%), advanced biventricular heart failure, severe pulmonary hypertension, moderate tricuspid and mitral insufficiency, atrial fibrillation and atrial flutter. Chest X rays reported prominence of the central pulmonary vasculature and severe global cardiomegaly. (Figure 3).
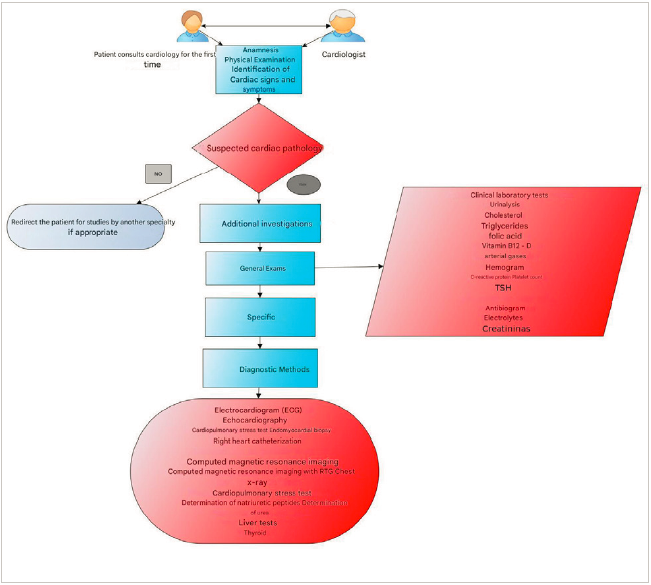
Source: Own elaboration.
Figure 3 Algorithm of diagnostic methods for the evaluation of patients with suspected heart disease
An IgG test for T Cruzi infection yield positive so the patient was confirmed as a case of advanced cardiac failure with severe cardiac damage due to Chagas disease in 2017.
In 2022, after 5 years of clinical management and visible increase in the clinical and hemodynamic compromise, a joint medical board agreed that the best option to correct the chronic heart failure, and improve the quality of life of the patient, was through the implantation of a left ventricular mechanical device (Heart mate 3) using a minimally invasive heart surgery. He was considered not adequate for heart replacement because of a high pulmonary pressure.
The surgical procedure consisted in a sternotomy incision to implant the left ventricular assist device -cardiac pump- at heart followed by a supraumbilical incision for the exit of the modular cable to the mini-console (Figure 4).
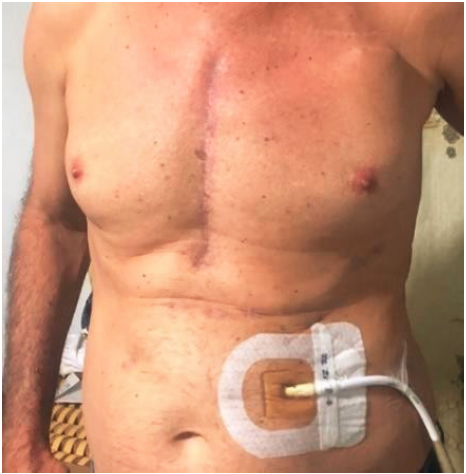
Figure 4 Sternotomy incision with technological device placement approach and lateral supraumbilical incision for the exit of the modular cable to connect to the mini-console
After 1 year follow up the patient has referred a substantial improvement in his quality of life and hemodynamic indicators.
DISCUSSION
In this study, the use of HeartMate III for the correction of chronic heart failure in a patient with untreated Chagas disease was described. It was unknown how this patient was infected but he was born and lived his whole life in a semi-rural area in the department of La Guajira. This place has not been identified previously as an endemic area for Chagas disease, but has all the conditions to be one, such as presence of houses with mud walls, palm tree roofs, earthy floor, and close to rural areas with numerous palm trees where Rhodnius prolixus is known to live.
The patient spent almost 5 years waiting for a clinical intervention that could improve his quality of life and ameliorate his symptoms. He went from one health care institution to another, begging for a solution, undertaking which Abadia Barrero has called "bureaucratic itineraries" 8. Bureaucratic itineraries" are caused by an endless number of administrative barriers that health insurers have raised to control health expenditures but disproportionately affect poor and rural populations. In this case, the untimely intervention of the disease caused an important increase in suffering plus higher costs in medical care, because the time lag between date of diagnosis and date when a solution was implanted, and because the patient had to be transferred from his home town to Bogota, 600 miles away, to solve his clinical problem 9.
Among patients not eligible for heart transplantation, Heart Mate's patients had an important reduction of mortality (48%) compared to those under optimal medical management 10.
HeartMate is a left ventricular support system, able to generate a blood flow of up to 10 liters per minute (lpm). It connects to the apex of the left ventricle and to the ascending segment of aorta, diverts blood from the weakened left ventricle and propels it towards the rest of the body. It is controlled through an internal computer that regulates the pump (Figure 5).

Source: taken and adapted from the Heartmate III Left Ventricular Device Patient Manual 12.
Figure 5 HeartMate III left ventricular assist
The implantation of the device generated a change in the patient's quality of life since it allowed him to live a seemingly normal life despite the extensive damage to his circulatory system. However, there are still very few experiences in Latin America and worldwide with the use of this technology in Chagas patients. Most evidence comes from case series, with few patients, or case reports. The Brazilian Society of Cardiology (SBC) asserts that "currently, the major limitations to its applicability are high cost, RV dysfunction, and need for a specialized team for device implantation and management. The SBC guideline on mechanical circulatory support recommends careful assessment of the RV function as mandatory before implantation, and, in the presence of moderate to severe dysfunction, one should be prepared for biventricular support implantation" 11.