INTRODUCTION
The anthropogenic landscape contributes to the formation of new forms of bird behavior as a response to different environmental pressures (Kuchar et al., 2018). Habitat selection is the result of an evolutionary trade-off between different selection pressures that maximize survival or reproductive success (Krebs & Davies, 1987). How most rural and forest bird species adapt and respond to urbanization remains poorly understood (Han et al., 2019). However, it has been shown that species abundance, seasonality, and distribution during breeding are strongly determined by environmental factors such as the availability of suitable nest sites, food, climatic and hydrological conditions, flooding level, distance to other colonies, risk to disturbance, as well as species-specific interactions and life histories (Kazantzidis et al., 2013; Kelly et al., 1993; Squalli et al., 2022).
Nest site selection in birds can be influenced by a variety of factors, such as food availability, vegetation cover, tree height and orientation, habitat quality, and the presence of predators (Johnson & Grier, 1988). Birds rapidly adjust their reproductive strategies and optimize their reproductive expenditure to adapt to anthropogenic habitats and improve their fitness (Sheng et al., 2022). The choice of ecological aspects such as home range, suitable nest location and the use of trees for nesting is a decision that ensures survival in many species (Iskandar et al., 2022). The above, coupled with the effects of the urban environment, food resources, predation pressure, pollution, and human impact, are considered critical points in research on bird ecology in anthropogenic ecosystems (Lan et al., 2021). In the breeding season, birds may need more environmental resources for themselves and their offspring. In addition, the area where nests are located is important for the development of the young. Therefore, birds may be sensitive to overlapping nest sites and competition during the breeding season (Ye et al., 2019).
Over the past two decades, there has been a growing interest in investigating the influence of transformed habitats on fundamental aspects of avian biology, such as life history strategies, survival, reproductive performance, behavior, and individual health (Reynolds et al., 2019). In this context, there is an increase in the number of studies that provide information on how species use crops as breeding habitat. Areas of research address aspects related to nest site selection, nest predation, reproductive biology and the impact of management practices on the reproductive efficiency of the species involved (Brambilla et al., 2013; Chmielewski, 2019; Hua et al., 2014; Jokimäki et al., 2005; Kajtoch, 2017). Particularly, in nest site selection, some studies address particular species (Mounir et al., 2023); others focus on heterogeneous structures that maximize the diversity of species present in communities (Kajtoch, 2017; Xie et al., 2019), or analyze how invasive and predatory species generate antagonistic interactions (Squalli et al., 2022), with few studies focusing on the selection made by species at nest sites (Brambilla et al., 2013; Castro-Caro et al., 2022; Squalli et al., 2021).
In orange groves, research has been carried out addressing various aspects. Hanane (2016) explored nesting density, while Barba and Gil-Delgado (1990) delved into the colonization process and interspecific competition, focusing on artificial structures such as nest boxes. In addition, it has been suggested that non-random patterns of nest occupancy, in relation to sources of human disturbance, could be linked to the different tolerance levels of each species (Remacha & Delgado, 2009). In the specific case of the European Turtle Dove (Streptopelia turtur), Hanane and Baamal (2011) documented the selection of vertical and distal parts of olive and orange trees for nesting, finding changes in the position of nests in relation to the type of crop (Hanane, 2016).
The nesting sites of birds in transformed environments are quite different from those of their natural habitats, hence research on the impact on nesting can help to better understand the adjustment of birds' reproductive strategies (Lan et al., 2021). Based on the general hypothesis that segregation has been an ecological behavior that has determined the evolution and coexistence of species at the community level, the purpose of this work was to identify the level of overlap in the selection of nesting sites of some bird species in orange groves, and the nature of the differentiation of such sites among the species that could explain their coexistence, for which we propose the hypothesis that the six species of birds studied in the orange grove will present distinctive patterns in the vertical distribution of their nests during the reproductive season.
MATERIALS AND METHODS
Study area
The study was carried out in the province of Valencia, Comunidad Valenciana, located in the east of the Iberian Peninsula. The dominant climate in the Valencian Community is Mediterranean, one of the temperate climates characterized by an intense and sometimes long arid summer period (Urios et al., 1991). This climate presents a marked seasonality in the distribution of temperature and precipitation, as well as a high intra- and inter-annual variability. In general, summers are hot and dry, which generates a notable and very characteristic water and thermal stress in the species that make up the Mediterranean ecosystems (Valladares, 2007). The area is characterized by an average annual temperature between 14 and 18 °C. In the months of January to July, there is an isotherm of 6 ºC and an average annual precipitation between 600 and 800 mm (Castro et al. 2005).
The sampled area, located in the municipality of Algemesí, Ribera Alta region, Comunidad Valenciana-Spain, at 17 m.a.s.l., covered eight hectares of intensive orange tree cultivation, which were surrounded by an extensive matrix of crops with similar characteristics. The height of the trees ranged from 1.75 to 3.32 meters, with a distance of 3 meters between trees, which were arranged in rows in a uniform manner, forming a quadrangular grid, with spaces of four meters between each row. Among the orange trees, juvenile mandarin (Citrus reticulata) and kaki (Diospyros kaki) trees were found. In 80% of the area, irrigation, fertilization, weed control and pruning were permanent during the sampling, generating soils devoid of vegetation, as opposed to 20% where herbaceous vegetation was observed.
Data collection
Sampling focused on breeding bird communities (passerines). The analysis was adjusted to six species that nested in orange trees: Silvia melanocephala, Turdus merula, Serinus serinus, Chloris chloris, Turdus philomelos and Muscicapa striata.
The intensive search for nests was conducted during the breeding season, in the period from March to July 2016. Once a nest was found, the observers (Eyda Annier Moreno Mosquera-EM, José Alexander Bonilla Flores-JB, Kary Yiseth Minnota Sánchez-KM and Leider Palacios Palacios-LP) followed up on it every five days, to avoid disturbing the incubation period in the case of active nests. A total of ten samplings were carried out, distributed in two sessions in April and May, and six sessions in June. The frequency of the sampling was adjusted according to the discovery of new nests during the surveys in the selected area. This method of searching for nests consisted of checking all the orange trees present in the planting plots (Gil-Delgado et al., 2005; Gil-Delgado and Lacort, 1996). The orange trees were visually inspected by carefully walking around each tree to find nests located on the outside and by walking under the canopy next to the trunk to find nests located on the inside of the trees.
A nest site was defined as the place where a breeding pair built a nest. Active nests were geo-referenced with a GPS (Garmin e Trex), to be subsequently measured, in order to avoid disturbing the incubation process. The nests found were measured in-situ considering the following variables: tree height, crown cover, height of the nest in the tree, distance from the nest to the edge, distance from the nest to the center of the tree, and the number of branches that supported the nest (Schaefer and Barkow, 2004; Vera et al., 2009) (Table 1). The unit of measurement of the variables was in meters, using a flexometer.
Table 1 Description of the variables evaluated in the characterization of the nesting microsites of the identified bird species
| Variable | Acronym | Description |
|---|---|---|
| Tree height | H | Distance from the base of the host tree to the top of the tree |
| Coverage of the cup | C | Horizontal distance from the top of the host tree |
| Nest height | Nh | Distance from the base of the host tree to where the nest was constructed |
| Distance from nest to edge | Dne | Horizontal distance from the nest location to the outer edge of the canopy |
| Distance from nest to center | Dnc | Horizontal distance from the nest location to the center of the plant (stem) |
| Number of branches supporting the nest | Nb | Number of branches of the host tree in contact with the nest |
Information analysis
To characterize the nesting sites of each species, the mean (X̅) and standard deviation (±) of each variable were calculated. Consistent with the nature of the data collected, both parametric (one-way ANOVA) and non-parametric (Kruskal-Wallis) tests were conducted in order to identify possible significant changes in nesting variables among the various species studied. To identify the grouping of bird species according to nesting site variables, a discrimination analysis (LDA) was performed using the Bray-Curtis distance. Subsequently, a multivariate permutational PERMANOVA analysis (Anderson, 2017) was performed to identify differences between species groups in relation to nesting variables; 9999 permutations were applied in the analyses and the Bonferroni correction was used. All calculations were performed with the statistical program Past version 4.0.
RESULTS
A total of 98 nests belonging to six bird species were found (Table 2), and simultaneous nests with a maximum of three species were observed in the same host. The number of nests varied among species (t=4.169; p=0.001). Turdus philomelos, Silvia melanocephala and Turdus merula were the species that built the highest number of nests, while Chloris chloris and Muscicapa striata built the fewest, with a percentage of 5.1% and 4.1%, respectively (Figure 1). The six bird species presented different nesting strategies (Figure 1). T. philomelos increased the number of nests as sampling time increased, while C. chloris and M. striata maintained a constant number of nests. S. melanocephala, T. merula and S. serinus decreased the number of nests as sampling time increased (Figure 1).
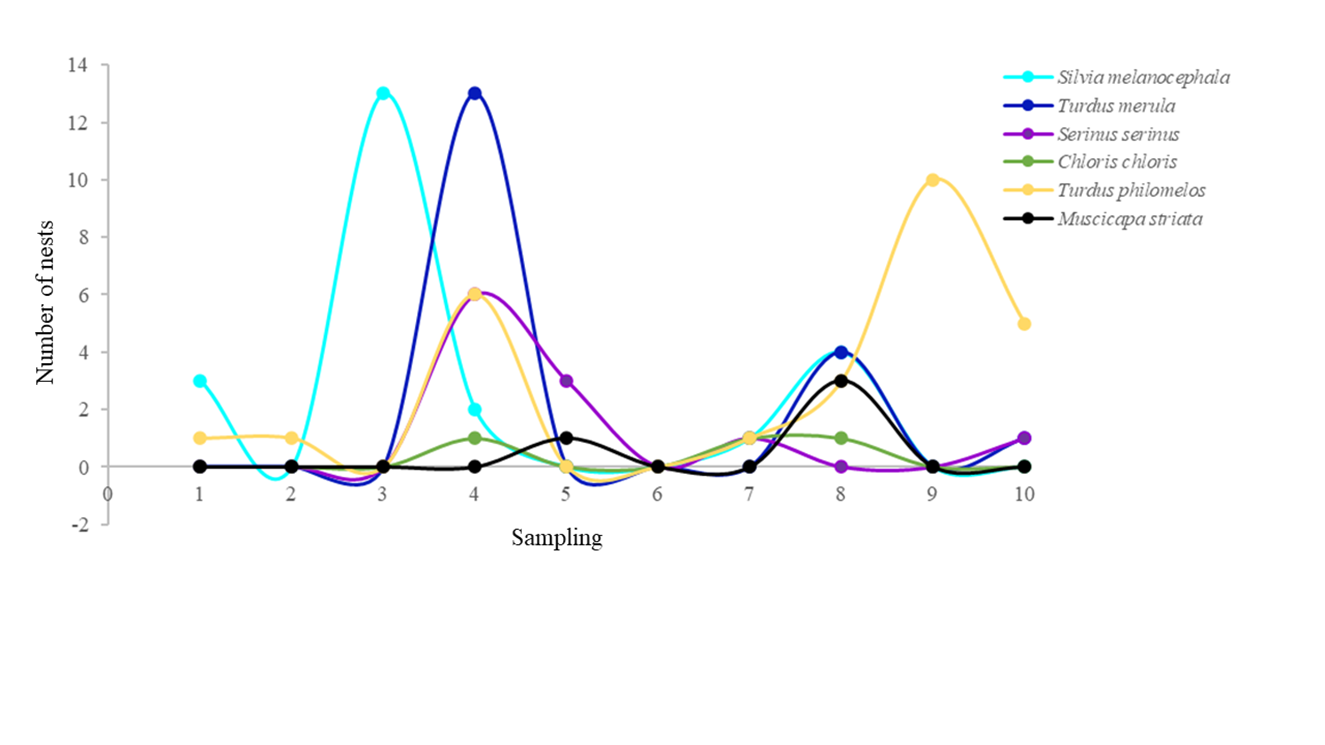
Figure 1 Breeding phenology of the six bird species cohabiting in orange agroecosystems, Algemesí, Comunidad Valenciana-Spain.
The bird species analyzed in this study built their nests depending on the height (H=42.2; p>0.001) and cover (F=98.1; df=97; p>0.001) of the host tree, and variations in the vertical distribution of nests were observed among species (Figure 2). Nest height and distance from the nest to the edge were the variables that showed the greatest differences. Turdus philomelos and C. chloris built their nests in the upper parts of the trees, while S. melanocephala and S. serinus built them in the middle part, and T. merula and M. striata in the lower part (Table 2). In addition, T. philomelos was the species that built its nests farthest from the edge, while M. striata built them closer. Silvia melanocephala, T. merula and C. chloris located their nests in intermediate areas (Table 2, Figure 2).
Table 2 Descriptive statistics of the microsite variables evaluated for each of the species.
| Species | N | H | C | Nh | Dne | Dnc | Nb |
|---|---|---|---|---|---|---|---|
| Silvia melanocephala | 23 | 2.290±0.576 | 2.589±0.630 | 1.332±0.429 | 0.718±0.350 | 0.706±0.375 | 0.706±0.375 |
| Turdus Merula | 22 | 2.542±0.369 | 2.782±0.606 | 1.191±0.235 | 0.849±0.209 | 0.700±0.375 | 1.843±0.394 |
| Muscicapa Striata | 4 | 2.500±0.336 | 2.790±0.615 | 1.197±0.286 | 0.560±0.510 | 0.630±0.233 | 1.786±0.278 |
| Serinus serinus | 17 | 2.526±0.268 | 2.635±0.541 | 1.528±0.291 | 0.672±0.204 | 0.812±0.381 | 1.958±0.416 |
| Chloris chloris | 5 | 2.442±0.1972 | 2.820±0.476 | 1.636±0.242 | 0.844±0.191 | 0.674±0.311 | 2.040±0.209 |
| Turdus Philomelos | 27 | 2.671 ±0.387 | 2.994±0.601 | 1.647±0.335 | 1.050±0.406 | 0.585±0.226 | 1.947±0.369 |
| Test | 42.2 | 98.12 | 50.51 | 159.3 | 171 | 41.98 | |
| P | 8.24E-11 | 2.03E-16 | 1.18E-12 | 2.52E-22 | 2.49E-23 | 9.21E-11 |
N: number of nests. H: height. C: cover. Nh: nest height Dne: distance from nest to edge. Dnc: distance from the nest to the center of the plant. Nb: number of branches supporting the nest.
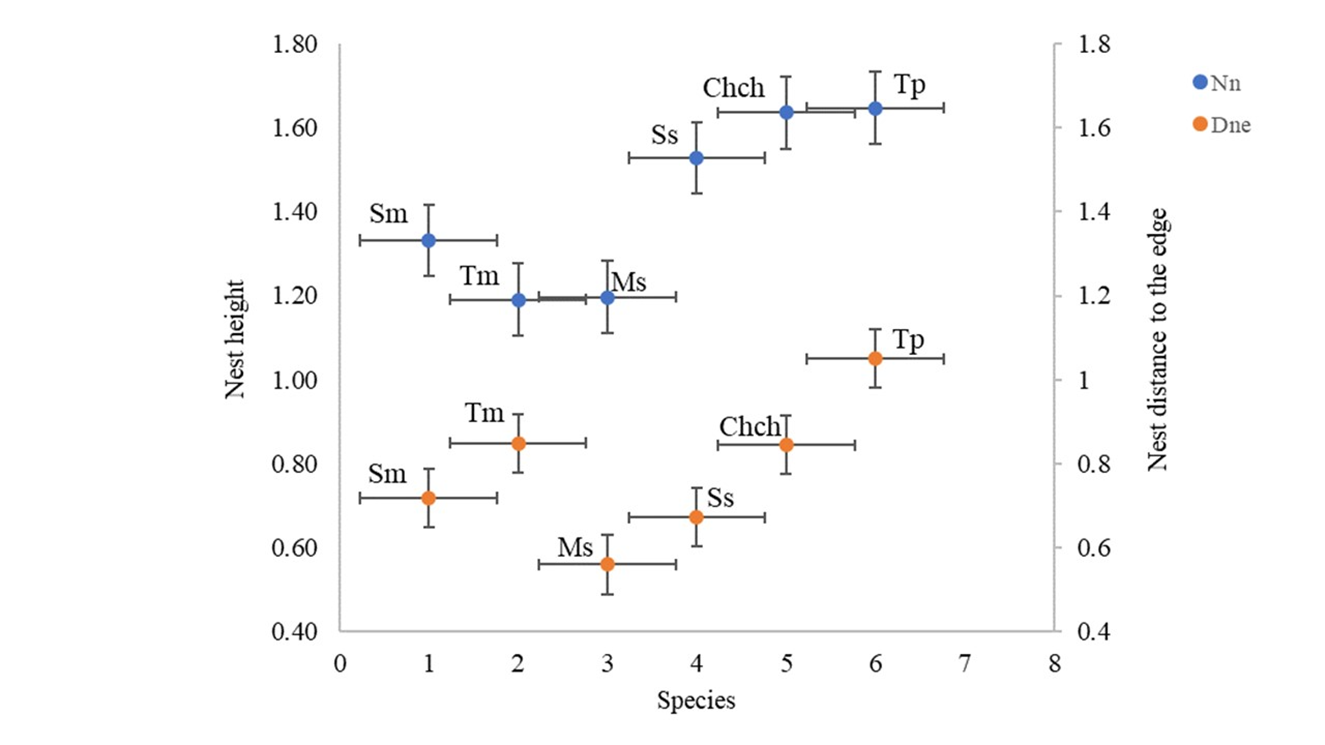
Figure 2 Vertical distribution of nests of the six bird species in relation to height and distance from nest to edge in orange trees. Nh: nest height; Dne: distance from nest to edge. Sm= Silvia melanocephala, Tm=Turdus merula, Ss=Serinus serinus, Cch=Chloris chloris, Tp=Turdus philomelos, Ms=Muscicapa striata.
Significant differences in vertical nest distribution were found between species (PERMANOVA, F=3.001; p=0.002) (Table 3), which was also confirmed in the LDA discriminant analysis (Figure 3). Species pairwise tests in PERMANOVA showed that the vertical nest distribution of S. melanocephala and S. serinus differed significantly from T. merula. In addition, the distribution of T. philomelos differed significantly from S. melanocephala by selecting taller trees, from T. merula by placing nests higher and from M. striata by having a greater distance from the nest to the edge (Table 3).
Table 3 PERMANOVA analysis of nest site variables among six bird species
| S. melanocephala | T. merula | M. striata | S. serinu | C. Chloris | T. philomelos | |
| Silvia melanocephala | 2.794* | 0.791 | 2.427 | 1.648 | 7.464** | |
| Turdus merula | 1.095 | 3.050* | 2.122 | 6.209** | ||
| Muscicapa . striata | 1.391 | 1.928 | 2.856* | |||
| Serinus serinu | 0.613 | 2.451 | ||||
| Chloris chloris | 0.542 | |||||
| Turdus philomelos |
p<0.05*; p<0.001**.
DISCUSSION
The six bird species showed different nest location strategies and adaptation to the environment that could explain their coexistence in the orange agroecosystem. Turdus philomelos seems to be a species that finds opportunities to increase its number of nests over time, unlike C. chloris and M. striata that maintain a constant number of nests. In contrast, S. melanocephala, T. merula and S. serinus are species that seem to have a different reproductive strategy, since they decreased the number of nests as sampling time passed. The different strategies described, may be attributed to the ecological particularities of each of the species studied, as proposed by Brambilla et al. (2013) with Thrushes, who claim that multiple breeding bird species may show variable density and changing relationships between species and habitats during the breeding season, and their horizontal distribution in the habitat could change during the breeding period. In this regard, Squalli et al. (2022) found a strong relationship between the breeding chronology of Pica mauritanica and food availability, contrary to Streptopelia turtur arenicola and Streptopelia decaocto, species where clutches were more threatened by predation attacks and the development of management practices coinciding with breeding activities.
The variation observed in the maximum nesting peaks of the six bird species found throughout the reproductive season seems to have favored their coexistence (Figure 2), and may be attributed to their behavioral response to ecological factors such as food availability and interspecific competition (predation and space); this is based on the fact that in congeneric species, it has been proven that the existence of a degree of segregation in one or more aspects of the ecological niche is sufficient to avoid competition (Mikula et al., 2014; Navarro et al., 2013; Squalli et al., 2022). So too, reduced overlap during foraging would reduce interspecific competition, in this sense, overlap, reflected in reduced behavioral aspects such as foraging schedules and foraging time (Navarro et al., 2013). For example, the coexistence of congeneric species of Turdus spp. has been explained by changes at the level of nesting microsites, variation in food preferences and differences in reproductive peaks (Mikula et al., 2014). For the case of species of the genus Streptopelia sp. and Pica mauritanica dispersal in space and time, explain patterns of habitat use in relation to intraspecific competition, while horizontal and vertical segregation in the choice of nest sites reduces interspecific competition of these sympatric species within olive groves (Squalli et al., 2022). Mounir et al., (2023), reported similar strategy in breeding populations of Streptopelia turtur arenicola where the first pairs explore the orchard and select nesting trees near feeding resources, while the second wave of breeders colonize trees near successfully used exploration sites.
Other studies, have shown that, when environmental pressure is relatively large or conditions are more extreme, closely related species that share similar resource requirements coexist, resulting in phylogenetic clustering (Jarzyna et al., 2021; Si et al., 2017).
The six species studied seem to respond differently to the selection of nesting microsites in the orange plantations, being the height of the nest location and the distance from the nest to the edge, the most important variables in the differentiation. This corroborates the application of the segregation hypothesis to explain the conformation of the breeding bird community in the orange plantations studied.
Birds can also select nesting sites depending on the physical characteristics of the site, such as location and proximity to water sources (Martin, 1995). Similar results were recorded between Streptopelia decaocto and S. turtur, competing species that selected different nest sites in olive orchards (Squalli et al., 2021), demonstrating vertical segregation that reduces the competition recorded in nest location (Squalli et al., 2022). Regarding nest location height, deviation values were low, and were very close to the mean, corroborating the importance of this variable in nest location. Muscicapa striata was the species that showed relatively low values of average nest location height (1.2 m), compared to what was observed by Kuchar et al. (2018) who recorded for the same species and in anthropic environments, nest heights between 1.7-8.5 m. According to the same authors, this is a conditionally synanthropic bird species, which does not occur in the most urbanized parts of cities, preferring moderately modified habitats. For the case of S. serinus and C. chloris, tree cover and distance of the nest from the edge could allow differentiating their nesting microsites in the study area. Castro-Caro et al. (2022) found that in olive groves herbaceous cover was a differential factor between these species and related it to food availability. In our study, the orange groves had bare soils, so we associate the selection of C. chloris for trees with greater cover, since the species also selected a greater distance from the nest to the edge, and its low presence in the habitat (Table 1), may suggest a greater susceptibility to predation. Distance from nest to edge and nest height were greater in T. philomelos compared to the rest of the species (Mikula et al., 2014). Therefore, these variables can be considered as important in the selection of nesting microsites performed by the species. In other studies, it has also been observed that the presence of dense vegetation plays a significant role in this choice (Kelleher & O'Halloran, 2007) and the height of trees are important factors for the species when choosing a nest site (Brambilla et al., 2013).
The discriminant analysis shows a slight vertical overlap between species, suggesting differences in the selection of nesting microsites. It is possible that a finer analysis of the particularities of the nesting microsites, considering differential aspects of their life histories, such as altitude and nesting site (Chouteau, 2003), will help to understand the behavioral dynamics of the species that determine the ecological structuring of the communities of birds nesting in orange groves. In this regard, studies on the spatial distribution of species of the genus Coua (Family Cuculidae), endemic to Madagascar, infer that coexistence is possible due to the segregation of microsites, biotic interactions and abundance of resources (Chiatante, 2022). Birds are sensitive to habitat changes, the functional roles of birds can be described by morphological or life history traits related to their food preference, flight ability and habits, aspects necessary for the study of differences in community distribution and their ecological mechanisms (Pigot et al., 2016).
Nest habitat partitioning and differences in nesting habits in similar species has already been referenced in natural environments (MacArthur, 1958) and validated in transformed habitats (Mikula et al., 2014). Our results support the fact that ecological segregation between closely related species may also be common in a transformed environment such as orange groves. These species presented variations in the selection of nesting microsites, nesting heights and nest location in relation to the edge, features that, being distinctive of the life history of the species, may be acting as a mechanism to reduce competition and at the same time evidence a slight segregation of ecological niche in the studied community, relevant information in the design of management and conservation alternatives for species in ecosystems with constant stress and transformation dynamics such as crops.
Generally speaking, the bird species identified in orange groves have been previously recorded in transformed environments (Chiatante, 2022; Karpińska et al., 2022; Kuchar et al., 2018; Mansouri et al., 2021; Navarro et al., 2013; Squalli et al., 2021; Winarni et al., 2022; Zhang et al., 2023). However, it is important to indicate that the use of these habitats by the species is not similar, which could suggest differences in their adaptation to these types of environments, given that stressors may vary from one habitat to another. For this reason, we recommend carrying out studies that cover more than one reproductive season, since ecological processes will be influenced by the conditions of the moment, and according to Kuchar et al., (2018), landscapes modified by human activity contribute to the emergence of new behavioral patterns in birds as a response to their environment.
















