INTRODUCTION
Fish are the most representative vertebrates in aquatic systems because they present morphological characteristics that confer a successful performance in these systems (Acero & Polanco, 2017). Fish have a great taxonomic richness and in Colombia, 1692 species are recorded exclusively in freshwater systems (DoNascimiento et al., 2024) distributed in the different lotic and lentic ecosystems that comprise 10% of the national area (WWF, 2018). This great species richness places Colombia as the country with the second highest number of freshwater fish species in the world, only behind Brazil (SiB, 2022).
The Orinoco River macrobasin has an extension of 981,446 km2, 35% of which is part of the Colombian territory (Bustamante, 2019). In Colombia, the Orinoco macrobasin is made up of eight sub-basins: Orinoco, Inírida, Guaviare, Vichada, Tomo, Meta, Casanare and Arauca rivers, and 95 sub-basins (IDEAM, 2004), where 767 native fish species have been recorded (DoNascimiento et al., 2024). Some of these tributaries originate in the Eastern Cordillera of the Andes, including the Meta River, which receives several tributaries from the eastern flank of the Eastern Cordillera, such as the Cravo Sur, Guanapalo, Ariporo, Pauto, Casanare, Guachiría, Cusiana and Upía rivers (Cala-Cala, 2019). The latter is formed by the Guavio, Lengupá and Garagoa rivers, where 103 fish species have been recorded, represented in 20 families and 53 genera (Urbano-Bonilla et al., 2021). Specifically in the Garagoa river basin, 32 species have been recorded (Urbano-Bonilla et al., 2021). However, this information comes from the lower part of the basin, with the headwaters and smaller streams little studied, making part of their taxonomic richness still unknown, a pattern common to the headwater rivers of the Orinoco river basin (Cala-Cala, 2019).
The Garagoa river basin has an approximate area of 250,000 ha, where 93.24% corresponds to mountain landscape and the remaining portion a high plateau landscape (Corpochivor, 2018). The types of land use present in the watershed are: silvopastoral (48.64%), semi-intensive transient crops (22.41%), protective forest (11.59%), conservation and recovery (4.87%), agrosilvopastoral (4.83%), extensive grazing (3.72%), intensive transient crops (3.60%) and other uses (0.43%) (Corpochivor, 2018). This lower percentage of land use includes an alteration in fluvial connectivity, which is given by the presence of the "La Esmeralda" reservoir of the Chivor Hydroelectric Power Plant (Tobar-Guzmán, 1977). The objectives of this study are to list the fish species of the Garagoa river basin and to elaborate a taxonomic key for their identification based on primary (field visits) and secondary information (species recorded in previous studies). The identification key represents a relevant tool in the promotion of ecotourism projects, in the monitoring of the regional ichthyofauna and, consequently, contributes to the integrated and sustainable management of this watershed.
MATERIALS AND METHODS
The Garagoa River watershed (CRG) is located on the eastern slope of the Eastern Cordillera of the Andes and is made up of 33 municipalities, 28 in the department of Boyacá and five in the department of Cundinamarca, covering an area of 2506.6 km2 or 250,660 ha (Corpochivor, 2018). According to the Horton - Strahler methodology (Horton, 1945; Strahler, 1957), the Garagoa River is classified as an order seven drainage, divided into ten hydrographic sub-basins and 26 micro-basins that supply the surrounding urban settlements and are located in different altitudinal zones, covering vegetation formations from paramo and sub-paramo, to high Andean forest and Andean forest (Corpochivor, 2018).
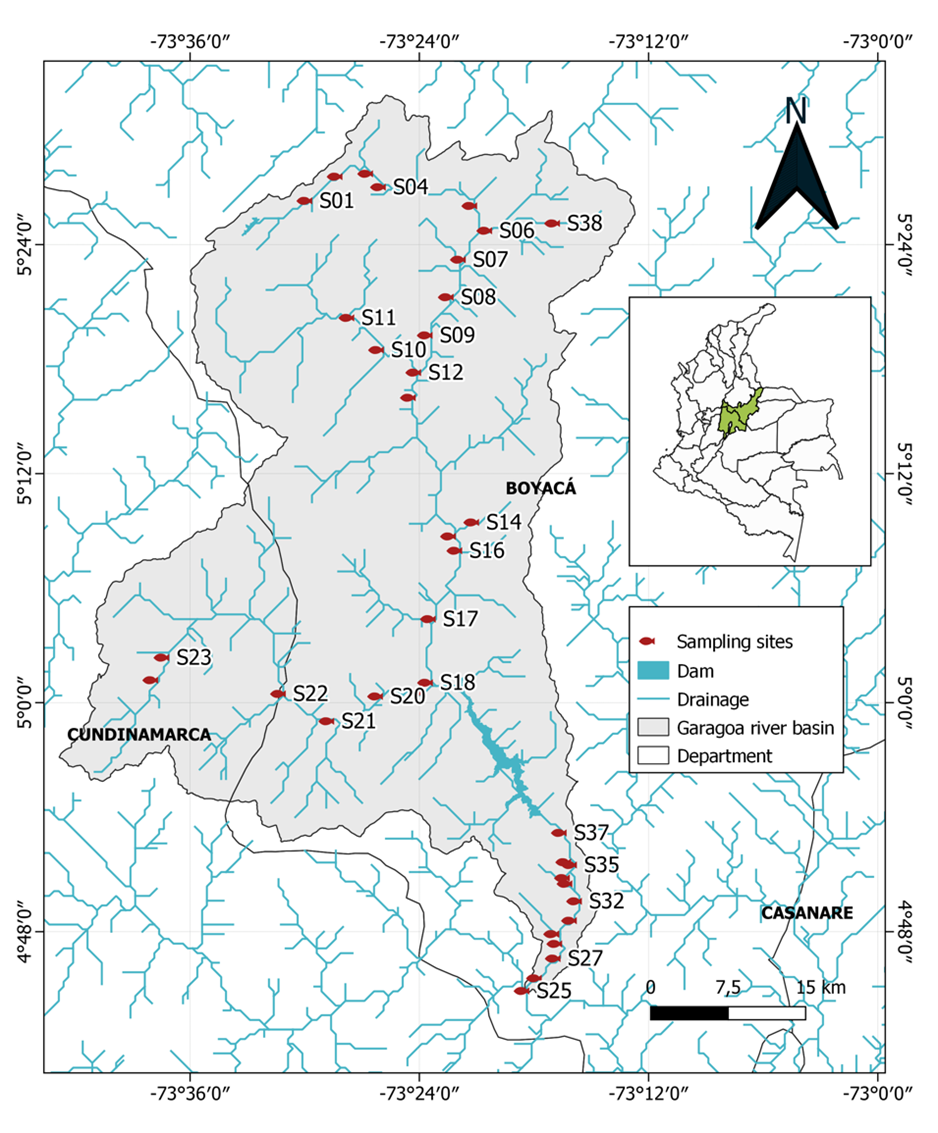
Thirty-eight sampling sites were established in the CRG covering both the upper (Teatinos River) and lower (Batá River to the junction with the Guavio River), with an approximate distance of 5 km between each sampling site following the drainage network (Table 1, Figure 1). At each site, a 100 m transect was established, where ichthyofauna collection was carried out by electrofishing with Smith Root LR-24 equipment and using electrical power between 500 and 990 V, varying according to water conductivity. Two passes were made per transect in countercurrent in a zigzag trajectory, for a total of 27 min per site. Electrofishing was complemented with the use of a trawl net (200 x 150 cm, 0.5 mm mesh eye) for 50 min at each site. Sampling was carried out during the day, between 06:00 and 17:00 hours.
Table 1 Location, Elevation and coordinates of the sampling sites (CRS: WGS84) in the Garagoa river basin.
| Sampling site | Municipality | Elevation (m) | Latitude | Longitude |
|---|---|---|---|---|
| S01 | Samacá | 2887 | 5°26'19''N | 73°29'41''W |
| S02 | Samacá | 2885 | 5°27'35''N | 73°28'06''W |
| S03 | Samacá | 2743 | 5°27'44''N | 73°26'30''W |
| S04 | Ventaquemada | 2731 | 5°27'02''N | 73°25'50''W |
| S05 | Boyacá | 2165 | 5°26'03''N | 73°21'03''W |
| S06 | Ramiriquí | 2112 | 5°24'45''N | 73°20'16''W |
| S07 | Jenesano | 2083 | 5°23'14''N | 73°21'39''W |
| S08 | Jenesano | 2059 | 5°21'16''N | 73°22'17''W |
| S09 | Tibaná | 2013 | 5°19'16''N | 73°23'23''W |
| S10 | Tibaná | 2106 | 5°18'30''N | 73°25'56''W |
| S11 | Turmequé | 2082 | 5°20'11''N | 73°27'29''W |
| S12 | Tibaná | 1960 | 5°17'19''N | 73°23'59''W |
| S13 | Chinavita | 1921 | 5°16'00''N | 73°24'16''W |
| S14 | Chinavita | 1732 | 5°09'28''N | 73°20'57''W |
| S15 | Chinavita | 1492 | 5°08'44''N | 73°22'10''W |
| S16 | Garagoa | 1473 | 5°07'59''N | 73°21'50''W |
| S17 | Garagoa | 1340 | 5°04'24''N | 73°23'13''W |
| S18 | Garagoa | 1269 | 5°01'04''N | 73°23'22''W |
| S19 | Garagoa | 1275 | 5°00'34''N | 73°23'48''W |
| S20 | Somondoco | 1307 | 5°00'21''N | 73°25'59''W |
| S21 | Guateque | 1370 | 4°59'03''N | 73°28'32''W |
| S22 | Guateque | 1424 | 5°00'29''N | 73°31'04''W |
| S23 | Machetá | 2046 | 5°02'23''N | 73°37'10''W |
| S24 | Machetá | 2790 | 5°01'12''N | 73°37'45''W |
| S25 | Santa María | 500 | 4°44'55''N | 73°18'19''W |
| S26 | Santa María | 523 | 4°45'35''N | 73°17'40''W |
| S27 | Santa María | 559 | 4°46'37''N | 73°16'41''W |
| S28 | Santa María | 573 | 4°47'23''N | 73°16'36''W |
| S29 | Santa María | 620 | 4°47'54''N | 73°16'45''W |
| S30 | Santa María | 572 | 4°47'53.8"N | 73°16'46 "W |
| S31 | Santa María | 659 | 4°48'36''N | 73°15'50''W |
| S32 | Santa María | 711 | 4°49'37''N | 73°15'34''W |
| S33 | Santa María | 958 | 4°50'32''N | 73°16'04''W |
| S34 | Santa María | 861 | 4°50'50''N | 73°16'12''W |
| S35 | Santa María | 1016 | 4°51'31 "N | 73°15'49.7 "W |
| S36 | Santa María | 1022 | 4°51'40''N | 73°16'08''W |
| S37 | Santa Maria | 1030 | 4°53'12''N | 73°16'22''W |
| S38 | Ciénega | 2267 | 5°25'08''N | 73°16'43''W |
The collected fish were anesthetized with Eugenol (Zahl et al.,2012) and then fixed with a 10% formaldehyde solution. After 72 h, they were preserved in a 70% ethanol solution. Taxonomic keys and specialized literature (e.g., Eigenmann, 1922; Maldonado-Ocampo et al., 2005; Taphorn, 2003; Urbano-Bonilla et al., 2018; van der Sleen & Albert, 2018) were used to identify the material. In addition, a visit was made to the freshwater fish collection of the Alexander von Humboldt Institute (IAvH-P) to verify the taxonomic identification. The collected material was deposited at the "Luis Gonzalo Andrade" Natural History Museum of the Universidad Pedagógica y Tecnológica de Colombia, with catalog numbers UPTC-Pe-00167 to UPTC-Pe-00198 (Annex 1). The species list for the CRG was enriched with information from previous work for the study area (Urbano-Bonilla et al., 2021) and with the review of datasets available in the Colombian Biodiversity Information System (Andrade-López et al., 2021; Cárdenas-Hincapié, 2023; Jiménez-Segura et al., 2023; Marchant et al., 2023; Mojica & Agudelo, 2023; Molina & Cano-Cobos, 2023; Prada-Pedreros et al., 2018; Ramírez-Chaves & Henao-Osorio, 2022; Ríos-Pulgarín et al., 2015; SiB Colombia, 2022; Villa-Navarro, 2017).
Fish collections were covered by the Framework Permit to Collect Specimens of Wild Species of Biological Diversity for Non-Commercial Scientific Research Purposes granted to the Universidad Pedagógica y Tecnológica de Colombia (UPTC) by the National Environmental Licensing Authority (ANLA) through Resolution 0724 of July 4, 2014.
RESULTS
A total of 41 fish species were recorded in the Garagoa river basin. Of these, 24 were captured in the 38 sampling sites and belong to 19 genera, 12 families and seven orders (Figures 2 and 3). Nine of these 24 species correspond to new records for the basin, including two exotic species (Table 2). The remaining 17 species have been reported in previous studies and have material deposited in biological collections: Ichthyology Collection of the Institute of Natural Sciences of the National University (ICN-MHN-Ic), Fish Collection of the Museum of Natural History of the Pontificia Universidad Javeriana (MPUJ), Ichthyological Collection of the University of Antioquia (CIUA) and Ichthyological Collection of the Alexander von Humboldt Institute (IAvH-P).

Figure 2 Fish of the orders Salmoniformes, Siluriformes and Synbranchyformes from the Garagoa river basin. Scale bars = 1 cm.

Figure 3 Fish of the orders Characiformes, Cypriniformes, Cyprinodontiformes and Gymnotiformes of the Garagoa river basin. Scale bars = 1 cm.
Table 2 Fish species of the Garagoa river basin.
| Species | No. of specimens observed | This work | MPUJ (2021) | IAvH-P (2021) | CIUA (2023) | ICN-MHN-Ic (2023) |
|---|---|---|---|---|---|---|
| Family Characidae | ||||||
| Astyanax integer Myers, 1930 | X | |||||
| Corynopoma sp. | X | |||||
| Corynopoma riisei Gill, 1858 | 8 | X | X | |||
| Creagrutus sp. | X | X | ||||
| Creagrutus atratusVari, 2001 | 167 | X | X | |||
| Creagrutus calaiVari, 2001 | X | |||||
| Creagrutus taphorniVari, 2001 | X | |||||
| Grundulus bogotensis (Humboldt, 1821)* | 11 | X | ||||
| Hemibrycon metae Myers, 1930 | 12 | X | X | X | ||
| Knodus sp. | X | |||||
| Knodus alpha (Eigenmann, 1914) | 30 | X | X | X | ||
| Knodus cismontanus (Eigenmann, 1914) | 16 | X | X | X | ||
| Knodus deuterodonoides (Eigenmann, 1914) | 560 | X | X | |||
| Family Crenuchidae | ||||||
| Characidium chupa Schultz, 1944* | 5 | X | ||||
| Family Cyprinidae | ||||||
| Cyprinus carpio Linnaeus, 1758* Cyprinus carpio Linnaeus, 1758* | 1 | X | ||||
| Family Poeciliidae | ||||||
| Poecilia sp. | X | |||||
| Poecilia caucana (Steindachner, 1880) | 42 | X | X | |||
| Poecilia reticulata Peters, 1859* | 54 | X | ||||
| Family Apteronotidae | ||||||
| Apteronotus galvisi de Santana, Maldonado-Ocampo & Crampton, 2007 | 1 | X | X | |||
| Family Salmonidae | ||||||
| Oncorhynchus mykiss (Walbaum, 1792) | 69 | X | X | |||
| Family Astroblepidae | ||||||
| Astroblepus sp. | X | X | X | X | ||
| Astroblepus latidens Eigenmann, 1918 | 549 | X | X | |||
| Astroblepus mariae (Fowler, 1919) | 3 | X | X | |||
| Family Callichthyidae | ||||||
| Hoplosternum littorale Hancock, 1828* | 1 | X | ||||
| Family Cetopsidae | ||||||
| Cetopsis umbrosa Vari, Ferraris & de Pinna, 2005 | X | |||||
| Family Heptapteridae | ||||||
| Cetopsorhamdia orinoco Schultz, 1944 | X | |||||
| Chasmocranus rosaeEigenmann, 1922* | 25 | X | ||||
| Rhamdia quelen (Quoy & Gaimard, 1824) | 2 | X | X | X | X | |
| Rhamdia laukidi Bleeker, 1858 | X | |||||
| Family Loricariidae | ||||||
| Chaetostoma sp. | X | X | ||||
| Chaetostoma chimu Urban-Bonilla and Ballen, 2020 | X | |||||
| Chaetostoma dorsaleEigenmann, 1922 | 192 | X | X | X | ||
| Chaetostoma joropo Ballen, Urbano-Bonilla and Maldonado-Ocampo, 2016 | 22 | X | X | |||
| Dolichancistrus fuesslii (Steindachner, 1911) | 1635 | X | X | X | X | X |
| Farlowella sp. | X | |||||
| Farlowella colombiensis Retzer and Page, 1997* | 51 | X | ||||
| Farlowella mitoupibo Ballen, Urbano-Bonilla & Zamudio, 2016 | X | |||||
| Farlowella vittata Myers, 1942 | X | |||||
| Family Trichomycteridae | ||||||
| Trichomycterus sp. | X | X | X | X | ||
| Trichomycterus knerii Steindachner, 1882* | 17 | X | ||||
| Family Synbranchidae | ||||||
| Synbranchus marmoratus Bloch, 1795* | 1 | X | ||||
An asterisk (*) indicates that it is a new record for the basin.
Taxonomic key for the identification of the species currently recorded in the Garagoa river basin, upper Orinoco river.
Key of orders of fish present in the CRG
1a. Dorsal and pelvic fins absent ................................................................................ 2
1b. Dorsal and pelvic fins present.............................................................................. 3
2a. Two gill slits; body compressed; anal fin present, long and extending over most of ventral margin.....................GYMNOTIFORMES (Apteronotidae: Apteronotus galvisi)
2b. One ventral gill slit; body anguilliform; anal fin absent.........SYNBRANCHIFORMES (Synbranchidae: Synbranchus marmoratus)
3a. Body naked or covered by bony plates; barbicels usually present; first ray of the pectoral fin thicker and ossified than the rest, sometimes modified in the manner of a spine......................................................................................................SILURIFORMES
3b. Body covered by scales; barbicels absent; first radius of pectoral fin normal, never modified into a spine..............................................................................................................4
4a. Caudal fin usually bifurcated or emarginate.....................................................................5
4b. Truncated or rounded caudal fin.......................................CYPRINODONTIFORMES
5a. Short dorsal fin; mouth not protractile.............................................................................6
5b. Extensive dorsal fin; protractile mouth............... CYPRINIFORMES (Cyprinidae: Cyprinus carpio)
6a. Pelvic fins without axillary process, less than 70 scales on lateral line ............... .................................................................................CHARACIFORMES
6b. Pelvic fins with axillary process, more than 100 scales on the lateral line; dorsum grayish green with iridescent purple and belly white or gray .............................. ..................................................SALMONIFORMES (Salmonidae: Oncorhynchus mykiss)
SILURIFORMES
1a. Naked body............................ .................................................................................2
1b. Body with bony plates, at least in most of its body......... .......................................9
2a. A patch of odontodes on the operculum...............................Trichomycterus knerii
2b. Ophthalmus without odontodes patch............. ........................................................3
3a. Ventral mouth; broad lower lip, forming a suction disk together with the upper lip .......................................................................................................................................4
3b. Unmodified suction disk-shaped mouth................................................................. 5
4a. Adipose fin present.................................................................. Astroblepus latidens
4b. Absence of adipose fin............................................................. Astroblepus mariae
5a. Absence of adipose fin..................................................................Cetopsis umbrosa
5b. Presence of adipose fin .......................................................................................... 6
6a. Maxillary barbicels not exceeding the base of the dorsal fin...................................7
6b. Maxillary barbicels surpassing the base of the dorsal fin .......................................8
7a. Anal fin origin in vertical line with adipose fin origin; adipose fin longer than ¼ of standard length.............................................................................. Chasmocranus rosae
7b. Anal fin origin anterior to adipose fin origin; adipose fin shorter than ¼ of standard length.........................................................................................Cetopsorhamdia orinoco
8a. Adipose fin 27%-35.6% of standard length, with or without multiple sensory pores on head (Silfvergrip, 1996); caudal fin moderately bifurcated, with equal lobes .............................................................................................................. Rhamdia quelen
8b. Adipose fin 44.6% - 51.3% of standard length, sensory pores of head invariably multiple (Silfvergrip, 1996); caudal fin deeply bifurcated, dissimilar lobes, with the upper one larger than the lower one ............................................................... Rhamdia laukidi
9a. Body covered by two rows of bony plates on each side, long maxillary barbicels, passing the operculum.................................................................. Hoplosternum littorale
9b. Body covered by more than two rows of bony plates on each side, short maxillary barbicels, never reaching the operculum........................................................................... 10
10a. Caudal peduncle strongly depressed; slender body ................................................... 11
10b. Caudal peduncle thick, not depressed; body stout.................................................... 13
11a. Ventral median line of discontinuous bony plates, exhibiting a row of three median plates in front of the pelvic fins........................................................ Farlowella mitoupibo
11b. Absence of ventral midline of bony plates or exhibiting two midplates in front of the pelvic fins........................................................................................................................... 12
12a. Length from tip of snout to start of mouth divided by length of pectoral fin < 0.6 ........................................................................................................ Farlowella colombiensis
12b. Length from tip of snout to start of mouth divided by length of pectoral fin > 1.0 .................................................................................................................... Farlowella vittata
13a. Hypertrophied operculum odontodes, passing or reaching the pectoral fin ........................................................................................................... Dolichancistrus fuesslii
13b. Non-hypertrophied operculum odontodes, never reaching the pectoral fin .................14
14a. Parieto-supraoccipital ridge absent ............................................... Chaetostoma dorsale
14b. Parieto-supraoccipital crest present..............................................................................15
15a. Dorsal region of pectoral spine with uniform black coloration; dot-like spots on head and body with the same density; background of body sides uniformly grayish to yellowish-green (Urbano-Bonilla & Ballen, 2021) ............................... ..................................................................................................................Chaetostoma joropo
15b. Main radius of pectoral fin with large dark spots, dot-like spots denser on head than on rest of body; lateral coloration of body dark brown to light brown (Urbano-Bonilla & Ballen, 2021) ..........................................................................................Chaetostoma chimú
CHARACIFORMES
1a. Pelvic and pectoral fins oriented horizontally ..................................Characidium chupa
1b. Vertically oriented pelvic and pectoral fins......................................................................2
2a. Absence of adipose fin .....................................................................................................3
2b. Presence of adipose fin ....................................................................................................4
3a. Body subfusiform; males with claviform prolongation starting from the operculum; anal fin with 27 rays ......................................................................................... Corynopoma riisei
3b. Robust body; males without claviform prolongation starting from the operculum; anal fin with 21 rays................................................................................... Grundulus bogotensis
4a. Upper jaw longer than the lower jaw and protruding ................................................... 5
4b. Upper jaw not protruding; 16 or more rays in the anal fin ........................................... 7
5a. Body somewhat tubular; branched anal fin rays 9 to 13; interorbital distance 34% - 37.9% of head length (Vari, 2001) .......................................................... Creagrutus atratus
5b. Body slightly compressed, not tubular; branched rays of anal fin 8 to 12; interorbital distance 26% - 35.4% of head length (Vari, 2001) ............................................................. 6
6a. Third infraorbital bone well developed, its ventral margin approaching or in contact with the horizontal branch of the preoperculum; humeral spot clearly expanded anteriorly, posteriorly and dorsally of the lateral line, tapered or curved anterodorsally behind the head (Vari, 2001) ................................................................................................Creagrutus calai
6b. Third infraorbital bone small, with a wide space separating its ventral margin from the horizontal branch of the preoperculum; humeral patch in the shape of a uniform vertical bar or with a slight dorsal expansion (Vari, 2001) ..................................... Creagrutus taphorni
7a. Anal fin with less than 19 rays ........................................................................................ 8
7b. Anal fin with more than 23 rays ..................................................................................... 9
8a. Anal fin with 16 rays.......................................................................Knodus cismontanus
8b. Anal fin with 17-18 rays............................................................Knodus deuterodonoides
9a. 34-38 scales on the lateral line….................................................................Knodus alpha
9b. 41 or more scales above the lateral line................................................ ....................... 10
10a. 41-44 scales above lateral line ......................................................... Hemibrycon metae
10b. 46-50 scales above lateral line ............................................................ Astyanax integer
CYPRINODONTIFORMES
1a. Ventral fin with 11-13 rays; dorsal fin with a black spot surrounded by two yellow spots............................................................................................................ Poecilia caucana
1b. Ventral fin with 5 rays; dorsal fin without black spot........................ Poecilia reticulata
DISCUSSION
Prior to this study, 32 species belonging to ten families and five orders had been recorded for the Garagoa river basin; however, eleven of them are reported up to genus (Urbano-Bonilla et al., 2021). In the present study, seven orders, 12 families and 24 species are recorded, increasing the taxonomic reports for the basin by nine species, including two exotic species (Cyprinus carpio and Oncorhynchus mykiss), representing a 27% increase in the richness of fish species recorded for the basin.
Cyprinus carpio is a species that is cultivated in artificial systems near the Garagoa River, also having been introduced into the La Esmeralda reservoir for consumption and recreation (Valderrama, 1985). Cyprinus carpio individuals may have escaped from the crops and/or the reservoir which, being a lentic system and located in a temperate climate, favors the establishment of this opportunistic species (Kottelat & Freyhof, 2008; Valderrama, 1985). The presence of C. carpio represents a pressure on native species, as it modifies the habitat by consuming aquatic plants (Richardson et al., 2000) and has a high demand for food resources, including eggs of other fish species (Gutiérrez et al., 2012). These pressures could generate a population reduction, displacement and/or loss of native species in the CRG.
Poecilia reticulata has been introduced in different regions due to its ornamental use and for dipteran control (Kottelat & Whitten, 1996). Its occurrence in the CGR is attributed to one or several introductions and its wide tolerance to habitat modification, which allows it to establish and colonize tributaries (Roman-Valencia et al., 2018). For its part, we suspect that the occurrence of Hoplosternum littorale is due to an introduction event, since this species has preferences for marshes and warm, stagnant floodplain waters (Baensch & Riehl, 1985; Reis & van der Sleen, 2018), which reduces the probability of occurrence by migration and/or natural widening of its distribution at 2731 m.a.s.l. (S04), a locality in the upper part of the CRG where it was captured (see details in Annex 1). The above suspicion is supported, as this species has been introduced in other regions of America (Orfinger, 2015) and individuals of H. littorale are traded in pet stores in Tunja, Boyacá, a city close (~20 km) to the locality where the species was captured.
Regarding the occurrence of Grundulus bogotensis in the CRG, we propose two hypotheses. The first suggests an introduction, so that the species was extracted from a nearby tributary belonging to the Magdalena-Cauca basin and then introduced into the headwaters of the CRG (belonging to the Orinoco basin) (Maldonado-Ocampo et al., 2005). This is feasible, since in the 1950s G. bogotensis was transplanted to be used as food for humans and as a food resource for other commercially important fish species (e.g., O. mykiss) in different bodies of water in the Cundiboyacense highlands, located near the watershed between the Magdalena-Cauca basin and the CRG, Orinoco basin (Alvarado-Forero & Gutiérrez-Bonilla, 2002). In the second hypothesis, we suggest that the headwaters of the CRG on the border with the Magdalena-Cauca basin could be part of the natural distribution area of the species. This is supported by the uplift of the Eastern Andean mountain range, since this event produced the isolation of the different populations of Grundulus (Roman-Valencia et al., 2010) .Thus, due to the proximity between the headwaters of the Magdalena-Cauca basin with those of the Orinoco basin, the species could be naturally distributed in some of these tributaries located in the vicinity of the Cundiboyacense highlands.
Hernández et al. (2015) record the distribution of Rhamdia quelen only for the Amazon region and the Paraná River basin and position R. laukidi as the only species present in the Orinoco River basin. However, in this work we have recorded the collection of specimens with morphological characters coinciding with the description of R. quelen, such as the absence of multiple sensory pores on the head (Silfvergrip, 1996), which rules out that they belong to the species R. laukidi. Consequently, we suggest increasing morphological and molecular taxonomic studies to contribute to the knowledge of this group and its distribution. On the other hand, the presence of Characidium chupa, Farlowella colombiensis and Synbranchus marmoratus, is attributed to a natural extension of their distribution (i.e., the CRG is an area of natural distribution of these species), but they had not been recorded so far.
Relevance of the results for the management of the CRG
Although most of the municipalities that make up the CRG have an economy based on agricultural production, it is projected that this will diversify through alternatives such as ecotourism, adventure tourism and sports (Corpochivor, 2018), including aquatic sports such as rafting (Silvera, 2023). In this sense, our results can contribute to this transformation through the use of information to consolidate activities such as nature tourism (e.g., underwater observation), aquatics and/or commercial fishing (e.g., aquaculture; Morales & Lasso, 2022).
In addition, species lists, identification keys and information on the composition of aquatic communities provide crucial information for understanding the dynamics of river ecosystems; they are also the basis for identifying species sensitive to environmental changes, serve to detect early signs of ecosystem disturbance (Karr & Dudley, 1981), and contribute to the knowledge of biodiversity and ecological relationships between species (Costello, 2020; Lagomarsino & Frost, 2020). In the future, the results of this type of work could be integrated with other parameters for decision making, policy formulation and planning on natural resources. For example, through indices such as the index of biotic integrity (IBI), which allows the identification of trends in river health over time and enables effective management and conservation planning (Karr & Dudley, 1981). This is especially important in the context of the CRG, since being part of the Upía river basin, it has been identified as one of the priority areas for the conservation of the ichthyofauna of the Andean Orinoquia (Lasso et al., 2016; Zamudio & Maldonado-Ocampo, 2022).
In conclusion, an updated list of the ichthyofauna of the Garagoa river basin is presented, comprising a total of 41 species. Of these, nine correspond to new records for the basin, an increase of 27% with respect to the number of species previously recorded. This result, added to the high presence of endemisms at higher altitudes in Andean rivers (Maldonado-Ocampo et al., 2005), evidences the importance of continuing to explore the rivers and streams of the upper Andean watersheds. Likewise, a dichotomous key for taxonomic identification, the description of each of the species recorded in the basin (see Annex 1) and photographs of some of these are presented, tools that will facilitate the monitoring of the ichthyofauna and the generation of future research for this and other neighboring basins that lack this information, in addition to complementing the information already existing for the Garagoa River (Corpochivor, 2018; Urbano-Bonilla et al., 2021).