Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
Caldasia
Print version ISSN 0366-5232On-line version ISSN 2357-3759
Caldasia vol.28 no.2 Bogotá Dec. 2006
HAEMATOZOA IN BIRDS FROM LA MACARENA NATIONAL NATURAL PARK (COLOMBIA)
Hematozoarios en aves del Parque Nacional Natural La Macarena (Colombia)
NATALIA BASTO
OSCAR A. RODRÍGUEZ
CORNELIS J. MARINKELLE
RAFAEL GUTIÉRREZ
NUBIA E. MATTA
Universidad del Valle, Cali, Colombia. nathaliabg@yahoo.com
Departamento de Biología. Universidad Nacional de Colombia, Bogotá D.C., Colombia. oscanrodrig@hotmail.com
Centro de investigación en Parasitología Tropical (CIMPAT), Universidad de Los Andes, Bogotá, D.C., Colombia. cmarinke@uniandes.edu.co
Departamento de Biología. Universidad Nacional de Colombia. Bogotá D.C., Colombia. rafaelgutierrez@latinmail.com; nemattac@unal.edu.co
ABSTRACT
Birds from 69 species in 25 families were collected from La Macarena National Natural Park in Colombia between June and November 2000 and examined for haematozoa. Eighty-two of the 342 birds (24%) were positive for one or more taxon. Microfilariae were the most commonly seen parasites (10.5%) and Leucocytozoon the least common (0.3%). Other parasites were species of the genera Plasmodium (4.4%), Trypanosoma (3.5%), Hepatozoon (3.5%) and Haemoproteus (3.2%). The low intensity of haemosporidian parasites agreed with other records from the Neotropics. Parasite prevalence in this Neotropical region was higher than levels found in other surveys in the Neotropics, but lower than levels found for the Nearctic area. A new host-parasite association is reported here, as well as avian species examined for haematozoa for the first time.
Key words. Birds, Colombia, haematozoa, haemoparasite, infection, Neotropics, prevalence.
RESUMEN
Se recolectaron aves pertenecientes a 69 especies y 25 familias en el parque nacional natural Sierra de La Macarena (Colombia), de junio a noviembre del año 2000, y se examinaron para hematozoarios. Ochenta y dos de las 342 aves (24%) fueron positivos para uno o más hematozoarios. En los preparados sanguíneos de aves positivas, las microfilarias fueron los parásitos más comunes (10,5%), y Leucocytozoon el mas escaso (0,3%). Otros parásitos observados pertenecieron a los géneros de Plasmodium (4,4%), Trypanosoma (3,5%), Hepatozoon (3,5%) y Haemoproteus (3,2%). Las bajas intensidades de parásitos hemosporidianos están de acuerdo con otros registros en el neotrópico. La prevalencia de parásitos en esta región es mayor que los niveles encontrados en otros estudios en la región neotropical, pero menores que los niveles encontrados para el área neártica. Nosotros reportamos aquí nuevas asociaciones parásito-hospedero, también como especies aviares examinadas por primera vez para hematozoarios.
Palabras clave. Aves, Colombia, hematozoarios, hemoparásitos, infección, neotrópico, prevalencia.
INTRODUCTION
Protozoan knowledge is currently linked to their clinical and economic importance; there is detailed information regarding some coccidian or haemoflagellate species which infect mammals whilst research on avian and reptilian Haemosporidia is more limited. Avian haematozoa infection has been related to behavioural changes, sexual selection and the reduction and extinction of native bird populations (VanRiper III et al. 1986, Garvin & Remsen 1997, Buchanan et al. 1999). However, the paucity of research concerning the distribution, prevalence and pathogenicity of these parasites has limited recognition of their importance in tropical bird demography and veterinary wildlife management. Despite great avian diversity in Colombia and the strong influence of migrant birds, this kind of study is scarce (Renjifo et al. 1952, Bennett & Borrero, 1976, Valkiünas et al. 2003, Matta et al. 2004).
It has been demonstrated that the distribution patterns and frequency of avian haemoparasite infections from the Neartic region differ from those found in Central and South America (Greiner et al. 1975, White et al. 1978). The main aim of this research was to determine the prevalence of haemotropic parasites of birds from La Macarena national natural park (NNP) (located in Meta, Colombia) and compare the results with previous studies carried out in this region and the neotropics.
MATERIALS AND METHODS
This work was carried out at El Cerrillo station in the La Macarena NNP, Colombia, between June and November 2000. The study area lies at 400-500m and has vegetation which includes gallery forest and savannah. The area has a mean annual temperature of 24oC with a 2,973 mm annual rainfall occurring mainly between June and August. Birds were mist netted and bled by clipping their claws to produce three thin smears on glass slides. The smears were fixed with 100% methanol and stained with Giemsa (7.2) for 40 min. The stained blood smears were examined by light microscope. Each smear was scanned double-blind at low magnification (100x) (10 min) for establishing the presence of Leucocytozoon, Trypanosoma and microfilariae and using oil immersion (1,000x) (20 min) for Plasmodium, Haemoproteus and Hepatozoon. Intensity of infection for microfilariae, Plasmodium, Haemoproteus and Leucocytozoon were established by counting 100 fields at 1,000 magnification by moving the slide in areas where the blood cells formed a single monolayer field at 100x oil objective containing around 100 erythrocytes. The intensity of infection was thus determined as a percentage of infected cells or the number of parasites/10,000 erythrocytes (Muñoz et al.1999).
RESULTS
Results of microscopic examination of blood smears are shown in table 1.These birds represented 69 species classified into in 25 families and nine orders. Eighty-two birds from 31 species were infected with either single or mixed infections (93 infections altogether) of parasites from six genera. Overall prevalence of infection was 24%. Microfilariae were the most common parasites encountered, occurring in 36 of the samples (10.5%), followed by Plasmodium in 15 birds (4.4%), Trypanosoma and Hepatozoon (3.5%), Haemoproteus (3.2%), Leucocytozoon (0.3%) and others (1.7%). The 69 avian host species examined included 10 species which were examined for blood parasites for the first time. 10 new host-parasite associations were also recorded.
Table I. Blood parasites found in birds from La Macarena NNP (Colombia).
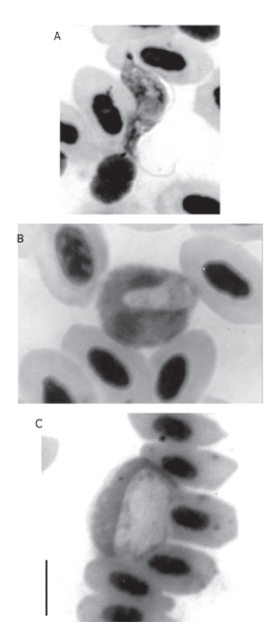
Most parasitaemias were low (0.02%-0.04%), suggesting chronic rather than acute disease. Haemoproteus columbae were only found in columbids; Haemoproteus caprimulgi occurred in the nightjar Caprimulgus nigrescens. Other haemoproteids found resembled Haemoproteus thraupi in the tanager Ramphocelus carbo, Haemoproteus nisi in the hawk Buteo magnirostris and Haemoproteus vireonis in Vireo olivaceus. The trypanosomatids found were similar to descriptions of Trypanosoma everetti (Molineux 1973) in Tyrannidae and T. paddae in Manacus manacus. Unidentified species of Trypanosoma were observed in Ramphocelus carbo, Thraupis episcopus, Arremonops conirostris, Pipra erithrocephala (Fig. 1a), Pachyramphus polychopterus and Vireo olivaceus. Hepatozoon atticorae were found in the swallow Neochelidon tibialis (Fig. 1b); unidentified species of this genus were seen in Dendrocincla fuliginosa and Glyphorhynchus spirurus and in Manacus manacus. Plasmodium infection included the subgenus Novyella in the species Psarocolius decumanus (possibly P. columbae), Saltator maximus, Arremonops conirostris and Arremon taciturnus (possibly P. nucleophilum). The subgenus Haemamoeba occurred in Mionectes oleaginea and Myiarchus ferox (possibly P. relictum) and Arremonops conirostris. Plasmodium subgenus Giovannolaia shizonts were found in Ramphocelus carbo. Leucocytozoon (probably fringillinarum) was detected in Dendroica discolor (Fig. 1c). Rounded gametocytes were observed in a male Pipra erythrocephala, as well as an unidentified member of the genus Haemoproteus or Plasmodium was seen in Arremon taciturnus; however, in the absence of mature and schizogonic forms, even a generic diagnosis was not possible.
Most of the birds captured (309) were Passeriformes, followed by Apodiformes (15) and Columbiformes (11); all other orders were represented by only a few individuals. Frequency of infection varied markedly amongst the 15 parasitized families. Hepatozoon was found in 5 out of 8 (62 %) woodcreepers (Dendrocolaptidae) and all of 5 swallows (Hirundinidae). The Fringillidae (56%, primarily infected with Plasmodium), Turdidae (53% infected with microfilariae) and Columbidae (36% primarily infected with Haemoproteus) showed the highest prevalence of infection with haematozoa. Other families, such as Pipridae (12%) and Tyrannidae (11%), had the lowest prevalence. All other families were represented by small samples (Table I).
DISCUSSION
Our results concerning parasitaemia, when compared to other surveys carried out in Neotropical countries were similar to those obtained by Renjifo et al (1952) in eastern Colombia, Sousa & Herman (1982) in Panama, and Rodríguez & Matta (2001) also in eastern Colombia (27%, 18% and 16%, respectively); these were, however, higher than those found by White et al. (1978) in Neotropical area (10.5%), Bennett et al. (1991a) in Bolivia (5%) and Valkiünas et al (2003) in Colombia (8%). These values were lower than those recorded for the Nearctic area (Greiner et al. 1975).
The parasite index so found could also indicate higher vector potential in this area (1.13) when compared to other surveys in Neotropical regions such as Rodríguez & Matta (2001) in Colombia (1.08) and Bennett et al. (1991a) in Bolivia (1.03). This phenomenon was probably due to differences in the study areas’ ecological conditions since it is difficult to compare the prevalence of parasites in a broad range of bird species sampled in different ecological systems at different times of the year. However, when specifically compared to studies carried out in Colombia (Renjifo et al.1952, Rodríguez & Matta 2001), in similar geographical areas, avian host species sampled and the same climatic season, our results suggest that the prevalence of blood parasites could be related to current conservation status and land use during the past fifty years. The differences in the prevalence of blood parasites compared to those obtained from birds from La Macarena NNP possibly reflects slower reduction of natural biotypes through intensive agriculture and cattle ranching which are more intense practised in areas closer to Villavicencio, situated on the edge of Colombia’s eastern plains (Rodríguez & Matta 2001). A similar hypothesis has been proposed for European birds (Bennett et al.1982, Tella et al. 1999).
An interesting aspect of this survey was the high prevalence observed for certain blood parasites like Hepatozoon, Trypanosoma and microfilariae. The genus Hepatozoon was mainly found to be associated with families such as Hirundinidae and Dendrocolaptidae which usually occupy cavity or colony nests (Hilty & Brown 1986). Such nests frequently support large populations of mites, ticks, fleas and other relatively sedentary nest-occupying blood-feeding arthropods which could serve as potential vectors for this haematozoan genus (Kirkpatrick & Smith 1988, Bennett & Peirce 1989). The presence of microfilarial worms and trypanosomes was higher than that reported by Bennett et al. (1991a) in Bolivia. The microfilaries were not identified to the species level because only peripheral blood were collected, were the nematode adult is absent. Conversely, Leucocytozoon was restricted to a North-American migrant bird (Dendroica discolor) and was not found in resident birds. The apparent absence of leucocytozoids suggests a lack of suitable ornithophilic simuliid vectors, since North-American migrants harbouring Leucocytozoon over-winter in the neotropics and could serve as a leucocytozoid reservoir for infecting resident birds (Herman 1968, Bennett et al. 1991b).
The presence of haemoproteids in some avian hosts indicates that suitable ceratopogonid and hippoboscid vectors are present in the area; however, the low number of infections recorded suggests that vector densities are low. The frequency of Plasmodium was lower than that found by Renjifo et al. (1952), but higher than that encountered in other Colombian surveys (Bennett & Borrero 1976, Rodriguez & Matta 2001). A possible explanation for the paucity of haematozoa in Colombian birds has been recently proposed by Valkiünas et al. (2003), suggesting that neotropical strains of avian hematozoa may be more virulent and kill more birds than other strains elsewhere, leading to low prevalence in mist-netted birds. While this is an interesting hypothesis, we have no data to support it.
The low intensity of parasitaemia obtained in the sampled birds has also been recorded previously (Bennett & Borrero 1976, Rodriguez & Matta 2001, Valkiünas et al. 2003), factors associated with the state of infection (acute or chronic) (Valkiünas et al. 2003), type of diagnostic test used (Kirkpatrick & Smith 1988), vector density and evolutionary relationship with their host (Bennett et al. 1980, Young et al.1993), could be affecting this result.
It is interesting to note that the origin of bird families could be related to the prevalence of haemoparasites. Bennett et al. (1980) suggested that Neartic avian families (i.e. Turdidae, Columbidae) have a higher prevalence of blood parasites than those from presumed Neotropical origins (i.e. Tyrannidae, Trochilidae) and our data are in accordance with this suggestion. Certain families have thus evolved with neoarctic and neotropical parasites, being thereby inherently more susceptible to infection by different blood parasites (Bennett et al. 1980). Behavioural aspects of various avian host families may also bring them into more frequent contact with suitable ornithophilic vectors (Kirkpatrick & Smith 1988). Trochillidae family members are rarely examined due to their difficult to catch, also the low prevalence detected in this birds, might be associated with their flight which could minimise host-vector contact (White et al. 1979)
The prevalence and intensity of haemoparasites in birds (as in other parasitic infections) is a result of a dynamic process involving the vector-host-environment. We have tried to demonstrate that the ecological conservation of an area is directly associated with the number of infections detected; however, the intensity of infection has continued to be low. Specific studies aimed at determining the pathogenicity of neotropical strains of avian hematozoa are therefore desirable, as would be studies on vectors of these parasites.
ACKNOWLEDGEMENTS
The authors would like to thank the staff of Unidad Administrativa Especial Sistema Nacional de Parques Nacionales Naturales (UAESPNN) and La Macarena PNN for granting permission to carry out the study and cooperating in the sampling. They would also like to thank FG Styles for ornithological assistance, encouragement and reviewing the manuscript, along with Jason Garry. Financial support was provided by the Universidad Nacional de Colombia’s Division de Investigación, Bogotá (DIB).
REFERENCES
1. Bennett, G.F. & J.I. Borrero. 1976. Blood parasites of some birds from Colombia. Journal of Wildlife Disease 12: 454-458. [ Links ]
2. Bennett, G.F. & M.A. Peirce.1989. Hepatozoon parus n. sp. from the Paridae and redescription of H. atticorae (de Beaurepaire Aragão) Hoare, 1924 from the Hirundinidae. Canadian Journal of Zoology 67: 2859-2863. [ Links ]
3. Bennett, G.F., H. Whitt & H. White. 1980. Blood parasites of some Jamaican birds. Journal of Wildlife Disease 16: 29-38. [ Links ]
4. Bennett, G.f., F. Thommes, J. Blancou & M. Artois. 1982. Blood parasites of some birds from Lorraine region, France. Journal of Wildlife Disease 18: 81-88. [ Links ]
5. Bennett, G.F., M. Garvin & J.M. Bates. 1991a. Avian hematozoa from west-central Bolivia. Journal of Parasitology 77: 207-211. [ Links ]
6. Bennett, G.F., A.A. Aguirre & R.S. Cook. 1991b. Blood parasites of some birds from northeastern Mexico. Journal of Parasitology 77: 38-41. [ Links ]
7. Buchanan, K.L., J.W. Catchpole & A. Lodge. 1999. Song an indicator of parasitism in the sedger warbler. Animal Behaviour 57: 307-314. [ Links ]
8. Herman, C.M. 1968. Blood protozoa in free-living birds. Symposium of the Zoological Society of London. 24: 177-195. [ Links ]
9. Garvin, M.C. & J. Jr. Remsen. 1997. An alternative hypothesis for heavier parasite loads or brightly colored birds: exposure at the nest. The Auk 114: 179-191. [ Links ]
10. Greiner, E.C., G.F. Bennett, E.,. White & R.F. Coombs. 1975. Distribution of the avian haematozoa of North America. Canadian Journal of Zoology 53: 1762-1787. [ Links ]
11. Hilty, S.L. & W.L. Brown. 1986. A guide to the birds of Colombia. Princeton University Press. Princeton. [ Links ]
12. Kirkpatrick, C.E. & T.B. Smith. 1988. Blood parasites of birds in Cameroon. Journal of Parasitology 74: 1009-1013. [ Links ]
13. Matta, N.E., N. Basto, R. Gutierrez, O. Rodriguez & E.C. Greiner. 2004. Prevalence of Blood Parasites In Tyrannidae (Flycatchers) in the Eastern Plains of Colombia Memorias Instituto Oswaldo Cruz. 9: 271-274. [ Links ]
14. Muñoz E., D. Ferrer, R. Molina & R.D. Adlard.1999. Prevalence of haematozoa in birds of prey in Catalonia, north-eastern Spain. Veterinary Record 144: 632-636 [ Links ]
15. Renjifo, S., C. Sanmartin & J. Zulueta. 1952. A survey of the blood parasites of vertebrates in eastern Colombia. Acta Tropica 9: 151-169. [ Links ]
16. Rodríguez, O. & N. Matta. 2001. Blood parasites in some birds from eastern plains of Colombia. Memorias Instituto Oswaldo Cruz 96: 1173-1176. [ Links ]
17. Sousa, O.E. & C.M Herman. 1982. Blood parasites of birds from Chiriqui and Panama provinces in the republic of Panama. Journal of Wildlife Disease 18: 205-221. [ Links ]
18. Tella, J.L., G. Blanco, M.G. Forero, A. Gajon, J. Donazar & F. Hiraldo. 1999. Habitat, world geographic range, and embryonic development of hosts explain the prevalence of avian hematozoa at small spatial and phylogenetic scales. Proceedings of the National Academy of Sciences 96: 1785-1789. [ Links ]
19. Valkiünas, G., P. Salaman & T.A. Lezhova. 2003. Paucity of hematozoa in Colombia birds. Journal of Wildlife Disease 39: 445-448. [ Links ]
20. Van Riper, III. C., S. Van Riper, L. Goff & M. Laird. 1986. The Epizootiology and ecological significance of malaria in Hawaiian land birds. Ecological Monographs 56: 327-344. [ Links ]
21. White, E.M., E.C. Greiner, G.F.bennett & C.M. Herman.1978. Distribution of the hematozoa of neotropical birds. Revista de Biología Tropical 26: 43-102. [ Links ]
22. Young, B.E., M.C. Garvin & D.B. Mcdonald D. B. 1993. Blood parasites in birds from Monteverde, Costa Rica. Journal of Wildlife Disease 29: 555-560. [ Links ]
23. White, E.M., G.F. Bennett & N.A.williams. 1979. Avian Haemoproteidae.11.The haemoproteids of the hummingbird family Trochilidae. Canadian Journal of Zoology 57: 908-913. [ Links ]
Recibido: 07/03/2006
Aceptado: 07/09/2006