Definitions
Active TB: |
Disease caused by Mycobacterium tuberculosis. |
Adherence/Compliance with Treatment: |
Defined as taking at least 80% of the prescribed doses within the time frame specified by the treatment protocol. |
Adverse Reactions: |
Any signs or symptoms possibly indicating physiological or organic alterations in the child resulting from the medication at prescribed doses. May be identified through patient/caregiver interviews, clinical evaluations, or laboratory testing |
Child or Adolescent in Contact with a Person with Drug-Sensitive Pulmonary TB: |
Any child or adolescent under 18 years of age who has been in contact with a patient with pulmonary TB caused by a microorganism with no documented antimicrobial resistance. |
Complications of Active TB: |
Any complication associated with tuberculosis (TB) disease or resulting from the administered treatment. |
Contact with TB: |
Close contact with TB is defined as any person with daily close exposure for more than 6 hours in the household, community, or institutional setting. For this protocol, TB contact is defined as any child or adolescent under 18 years of age who has been in contact with a patient with pulmonary tuberculosis. In children, any TB contact is typically considered high risk. |
Drug-Sensitive TB: |
TB caused by M. tuberculosis with no documented resistance to antimycobacterial drugs (isoniazid, rifampin, fluoroquinolones, second-line injectables). |
Drug-Resistant Pulmonary TB: |
Any child or adolescent under 18 years of age who has been in contact with a patient with pulmonary TB resistant to one or more anti-TB drugs. |
Extensively Drug-Resistant TB (XDR): |
TB caused by a microorganism resistant to isoniazid and rifampin, all fluoroquinolones, and at least one of the second-line injectable drugs (e.g., amikacin, kanamycin, or capreomycin). |
False Negatives: |
Individuals with the disease who test negative. |
False Positives: |
Individuals without the disease who test positive. |
HIV Infection: |
Infection with the human immunodeficiency virus (HIV), regardless of clinical stage (including advanced disease or AIDS), confirmed according to laboratory criteria as per the current protocol. |
Immunosuppression and Other Comorbidities: |
Defined according to the criteria used by the authors of the studies reviewed. |
Interferon-Gamma Release Assay (IGRA): |
A test that quantifies interferon-gamma produced by sensitized T cells in response to M. tuberculosis-specific antigens, used to support the diagnosis of LTBI. (Test details available at: https://acortar.link/BwjFI) |
Latent TB (LTBI): |
A persistent immune response to M. tuberculosis antigens without clinical manifestations of active TB. No reference standard exists for directly detecting M. tuberculosis infection in humans. Most infected individuals are asymptomatic but remain at risk of developing active TB (WHO). In recent WHO documents, this is also called tuberculosis infection (TBI). |
LR+ and LR− (Positive and Negative Likelihood Ratios): |
Defined as the likelihood of a specific test result in patients with the condition compared to those without. LR+ indicates how much the probability increases following a positive test result, and LR− suggests the change in likelihood following a negative result. |
Mono-resistant TB: |
TB caused by a microorganism resistant to one first-line anti-TB drug. |
Multidrug-Resistant TB (MDR): |
TB is caused by a microorganism that is resistant to at least isoniazid and rifampin. |
Periodic Clinical Monitoring: |
Follow-up conducted by healthcare professionals (nurses, doctors, or pediatricians) at defined intervals to detect early signs and symptoms of adverse drug reactions such as nausea, vomiting, abdominal pain, anorexia, fatigue, dark urine, pale stools, jaundice, mucocutaneous lesions, or neurological symptoms. |
Preventive Treatment: |
Treatment offered to individuals at risk of developing TB to reduce their risk. Also refers to treatment for LTBI, TB preventive therapy, or treatment of TB infection. |
Progression to Active TB: |
Transition from latent TB infection (LTBI) to active TB disease. |
Radiological Findings of Active TB: |
A chest X-ray is considered consistent with TB if one or more of the following are observed: airway compression and/or tracheal displacement, soft tissue density suggestive of lymphadenopathy, airspace opacification, micronodular pattern (miliary or more widespread and bilateral), pleural effusion, cavities, vertebral spondylitis, or pericardial effusion. |
Sensitivity: |
The probability that a test is positive when the condition is present (i.e., in diseased individuals). Also defined as the proportion of true positives among all affected individuals. |
Signs and Symptoms of Active TB: |
Include one or more of the following: |
- Persistent cough: |
Lasting more than 2 weeks and not resolving. |
- Weight loss or poor weight gain: |
Weight-for-age below -3Z or -2Z; confirmed weight loss (>5%) since the last visit; or no weight gain over two consecutive visits. |
- Unexplained persistent fever: |
>38ºC for over a week, reported by caregiver or objectively recorded. |
- Persistent unexplained lethargy: |
Decreased activity or play, reported by the caregiver. |
- Additional signs in infants 0-60 days: |
Neonatal pneumonia, unexplained hepatosplenomegaly, or sepsis-like illness not responding to appropriate treatment. |
- Lymphadenopathy: |
Axillary (>1.0 cm), supraclavicular (>0.5 cm), cervical (>1.0 cm), or inguinal (>1.5 cm) nodes; or adenitis not self-resolving or unresponsive to antibiotics for common pyogenic bacteria. |
- Sweating: |
Night sweats. |
- Other signs and symptoms: |
Depend on the affected organ and may include acute/subacute/chronic meningitis with hydrocephalus, complicated pneumonia, hepatosplenomegaly, generalized adenitis, septic arthritis, hematuria, or sterile pyuria. |
Specificity: |
The probability that a test is negative if the disease is absent (i.e., in healthy individuals or those with a negative reference standard). Also defined as the proportion of true negatives among all healthy individuals. |
Standard Care: |
Defined as the weekly supervised delivery of medication at a healthcare facility by healthcare professionals (in the case of the rifapentine and isoniazid regimen), or monthly delivery with medical and nursing evaluation in the case of the unsupervised regimen. |
True Negatives: |
Individuals without the disease who test negative. |
True Positives: |
Individuals with the disease who test positive. |
Tuberculin Skin Test (TST): |
A standard method to determine whether a person is infected with Mycobacterium tuberculosis. (Test details available at: https://acortar.link/p3fLo). |
Introduction
Human tuberculosis (TB) is an infection caused by Mycobacterium tuberculosis. Pulmonary TB, the most common clinical manifestation, results in a high rate of fatal cases among untreated or inadequately treated patients 1-3. Approximately 5-10% of infected individuals develop progressive primary disease, most frequently within the first two years following infection 1-3. It is estimated that around one-third of the global population is infected with M. tuberculosis, representing the primary reservoir for post-primary TB 2-5.
The World Health Organization (WHO) estimated that in 2022, there were 10.6 million incident cases of TB worldwide, 12% of them in children under 15 years of age, and that 1.3 million people died from the disease. TB is the second leading cause of death by a single infectious agent, surpassed only by the COVID-19 pandemic, after being the leading cause since 2012, surpassing even HIV 6. In Colombia, 17,595 TB cases were reported in 2022 (an incidence of 31.95 cases per 100,000 inhabitants), 3.4% of which were in children under 15 years 7.
Although TB treatment has been available since the mid-20th century, the disease continues to claim lives globally 1,2. The WHO has issued management guidelines and recommendations to reduce TB-related morbidity and mortality 3. In recent decades, the WHO has launched various strategies to combat TB, including the “Stop TB Strategy” in 2005 3 and, more recently, the “End TB Strategy” in 2014 4. The goals of this latter strategy include a 95% reduction in mortality and a 90% reduction in incidence, along with the elimination of catastrophic costs for TB-affected families between 2015 and 2035. Colombia has adopted these strategies and updated its national TB control plans accordingly 5,6. However, before the development of the guideline presented in this article, the treatment of latent tuberculosis infection (LTBI) was only generally addressed for both children and adults in the technical appendix of Resolution 227 of 2020 issued by the Ministry of Health and Social Protection (MSPS) 7, and no specific clinical practice guideline (CPG) for managing children in contact with TB was available.
Resolution 412 from the Ministry of Health 8 introduced guidelines in Colombia for disease prevention and care in 2000, including the Guideline for the Management of Pulmonary and Extrapulmonary Tuberculosis, which was updated in 2007 9. The national TB regulations distribute responsibilities within the TB program among various actors and levels. At the national level, the program is led by the MSPS, which establishes rules for public health control actions. It is advised by a national advisory committee and subcommittee, supporting regional and municipal committees. The MSPS is also responsible for supplying free TB treatment by distributing medications through departmental health authorities, which then distribute them to municipal authorities. Additionally, the MSPS oversees the implementation of public health and surveillance actions through these territorial entities 10-12.
Municipal authorities are responsible for conducting field epidemiological visits, often subcontracted to municipal State Social Enterprises (In Spanish, ESEs) or other institutions. However, suppose a TB-symptomatic case or a person in a priority group for LTBI diagnosis and treatment is identified during a field visit. In that case, the municipal authority’s role is limited to referring the individual to the designated Health Care Provider (IPS) under their corresponding Health Promoting Entity (In Spanish, EPS), where further actions must be taken according to established regulations. Municipal authorities are also responsible for providing technical assistance and surveillance of EPS and IPS regarding TB management, but they do not have enforcement powers to sanction noncompliance 13.
EPSs are responsible, through subcontracted IPSs, for timely diagnosis of TB patients, clinical and paraclinical evaluation of contacts, delivery of treatment, and follow-up, including microbiological monitoring. As mentioned, the medication is supplied by the MSPS through territorial authorities but dispensed to patients by the EPS. This excludes drugs for drug-resistant TB not available from the MSPS.
Children with TB differ from adults in their response to the disease, which has important implications for prevention, diagnosis, and treatment. Diagnosis is particularly challenging due to: i) less specific signs and symptoms in younger children compared to older children and adults; ii) higher risk in young children of presenting with other infectious diseases; and iii) difficulty obtaining sputum samples for smear and/or culture, with samples tending to be paucibacillary 8. Moreover, children who live in close contact with a pulmonary TB source case with positive smear microscopy are at much higher risk of developing disease after infection with M. tuberculosis, particularly infants and children under five years of age 14-18. Among contacts under five years of age of TB patients, 8.5% develop active TB, and over 50% of household contacts have LTBI 15,19-23. The WHO has developed several high-quality TB CPGs, but it published specific recommendations for individuals with TB or LTBI in 2020 9.
This guideline, supplemented by other WHO CPGs 10, has informed global management of these patients. However, these guidelines provide recommendations for all age groups without separately assessing the evidence in children, do not include the most recent evidence, and fail to account for the specific context of Colombia. Given the unique characteristics of the pediatric population, the need to assess child-specific evidence and to develop exclusive recommendations for this age group is a priority. Therefore, as part of a research program funded by the Ministry of Science, Technology, and Innovation (Minciencias), an evidence-based CPG was developed to support clinical decision-making in the evaluation, treatment, and follow-up of Colombian children under 18 who are in contact with patients with pulmonary TB (Table 1).
Table 1 Summary of Recommendations
| 1 | We recommend performing chest X-rays to screen for active TB in all children and adolescents under 18 years of age who have been in contact with patients with pulmonary TB, rather than relying solely on the presence of TB-suspect signs and symptoms (moderate certainty. Strong recommendation). |
| 2 | We recommend using interferon-gamma release assays (IGRA), specifically the QuantiFERON-TB® test, as the diagnostic test for TB infection in children and adolescents aged 2 to 18 years who have been in contact with patients with pulmonary TB. For children under 2 years of age, and while the test is being fully implemented in Colombia, we recommend using the tuberculin skin test (low certainty. Strong recommendation). |
| 3 | 1. We recommend treating TBI with short-course regimens in children under 5 years of age without HIV infection who have been in contact with drug-sensitive pulmonary TB (low certainty. Strong recommendation). |
| 2. We suggest treating TBI with short-course regimens in children and adolescents aged 5 to 18 years, without HIV infection, who have been in contact with drug-sensitive pulmonary TB (low certainty. Conditional recommendation). | |
| 3. We suggest treating TBI with 6 months of isoniazid only when short-course regimens are not accessible, in children and adolescents under 18 years of age without HIV infection who have been in contact with drug-sensitive pulmonary TB (low certainty. Conditional recommendation). | |
| 4. We do not recommend treating TBI in children and adolescents under 18 years of age without HIV infection who have been in contact with drug-sensitive pulmonary TB using a 9-month daily isoniazid regimen (low certainty. Strong recommendation against). | |
| 4 | 1. We recommend treating TBI with short-course regimens in children under 18 years of age with HIV infection who have been in contact with drug-sensitive pulmonary TB (low certainty. Strong recommendation). |
| 2. We suggest treating TBI with 6 months of isoniazid only when short-course regimens are not accessible, in children and adolescents under 18 years of age with HIV infection who have been in contact with drug-sensitive pulmonary TB (low certainty. Conditional recommendation). | |
| 3. We do not recommend treating TBI in children and adolescents under 18 years of age with HIV infection who have been in contact with drug-sensitive pulmonary TB using a 9-month daily isoniazid regimen (low certainty. Strong recommendation against). | |
| 5 | 1. We suggest using the 12-dose weekly isoniazid-rifapentine short regimen instead of the long isoniazid-only regimen in children and adolescents aged 2 to 18 years with TBI and non-HIV-related special immunosuppressive conditions (very low certainty. Conditional recommendation). |
| 2. We suggest using isoniazid for 6 months in children under 2 years of age with TBI and non-HIV-related special immunosuppressive conditions, until rifapentine is approved for this age group or new evidence emerges (very low certainty. Conditional recommendation). | |
| 3. We do not suggest using regimens other than isoniazid and rifapentine due to lack of efficacy or safety evidence in children or adults with TBI and non-HIV-related immunosuppression (very low certainty. Conditional recommendation). | |
| 6 | 1. We suggest treating TBI with levofloxacin for 6 months in children under 5 years of age without HIV infection who have been in contact with rifampicin-resistant (RR) or multidrug-resistant (MDR) pulmonary TB. In cases of contact with levofloxacin-resistant TB, treatment should follow the drug susceptibility profile of the index case (moderate certainty. Conditional recommendation). |
| 2. We suggest treating TBI with levofloxacin for 6 months in children and adolescents aged 5 to 18 years without HIV infection who have been in contact with RR or MDR pulmonary TB, based on their risk factors for developing active TB. In cases of contact with levofloxacin-resistant TB, treatment should follow the drug susceptibility profile of the index case (moderate certainty. Conditional recommendation). | |
| 7 | 1. We believe that close clinical observation alone is not sufficient to manage cases that would benefit from treatment; therefore, we recommend treating TBI in children under 5 years of age who have been in contact with drug-sensitive pulmonary TB when the infection occurred more than one year ago. The time elapsed since exposure is irrelevant as long as the child is under 5 (low certainty. Strong recommendation). |
| 2. We believe that close clinical observation alone is not sufficient to manage cases that would benefit from treatment; therefore, we suggest treating TBI in children and adolescents aged 5 to 18 years who have been in contact with drug-sensitive pulmonary TB more than a year ago, based on their risk factors for developing active TB (moderate certainty. Conditional recommendation). | |
| 3. We do not suggest treating TBI in children and adolescents under 18 years of age who have been in contact with drug-resistant pulmonary TB when the infection occurred more than a year ago (expert consensus. Conditional recommendation). | |
| 8 | 1. We recommend providing preventive TB treatment to children under 5 years of age who have been exposed to TB and are in the immunological window period (very low certainty. Strong recommendation). |
| 2. We suggest providing preventive TB treatment to children and adolescents aged 5 to 18 years who have been exposed to TB and are in the immunological window period, based on their risk of developing active TB (very low certainty. Conditional recommendation). | |
| GPS Good Practice Statement: The panel considers it appropriate to repeat the immunological test (TST or IGRA, based on the availability and recommendations in Question 2) between 8 and 12 weeks after treatment initiation to determine whether to continue. If the second test is negative, treatment should be stopped; if positive, it should be completed. | |
| 9 | 1. We recommend monthly clinical monitoring and follow-up by primary care professionals trained in TB and medication management (physician or nurse) for children and adolescents under 18 years of age receiving treatment for TBI (expert consensus. Strong recommendation). |
| 2. We suggest not performing routine laboratory testing in children and adolescents under 18 years of age who are receiving or about to receive TBI treatment, except in patients with liver disease or on hepatotoxic medication, who should undergo monthly lab monitoring (expert consensus. Conditional recommendation). GPS Good Practice Statement. The panel considers that monthly clinical monitoring for children on TBI treatment should be carried out by a trained physician. | |
| 10 | 1. We recommend using short-course regimens over long-course regimens to improve adherence to TBI treatment in children and adolescents under 18 years of age (low certainty. Strong recommendation). |
| 2. We recommend implementing comprehensive approaches (including psychological, social work, and nutritional evaluations; patient-centered care; direct contact with healthcare personnel; multipurpose visits; among others) to identify and address barriers and promote adherence to TBI treatment in children and adolescents under 18 years of age (very low certainty. Strong recommendation). | |
| 3. We suggest considering interventions studied in caregivers and children, such as financial incentives for transportation, nutritional support, psychosocial assistance, and SMS reminders, based on the identified barriers, for TBI treatment in children and adolescents under 18 years of age (low certainty. Conditional recommendation). |
Objectives, scope, and target population of the guideline
Definition
Latent tuberculosis infection (LTBI) is a measurable immune response to M. tuberculosis without clinical signs of active disease. This response is measured using the tuberculin skin test (TST) and/or interferon-gamma release assay (IGRA). However, this definition does not address the duration or activity of the latent focus, which is not a homogeneous entity and may vary between individuals depending on host and bacterial factors 24. As a result, the terminology for treating individuals in contact with TB has evolved-ranging from "prophylaxis" and "preventive TB treatment" to the current terms "TB infection (TBI)" and "preventive TB treatment." In line with the terminology used in the latest WHO Global TB Report, this document adopts the term tuberculosis infection (TBI)25. Nevertheless, as the concept of LTBI was still in use during the evidence search for this guideline, it was employed in those searches.
Objective
To develop an evidence-based clinical practice guideline (CPG) for evaluating, treating, and following children in contact with pulmonary TB patients. The recommendations from this CPG aim to bridge the gap between evidence and clinical practice in managing children with TBI in Colombia. This guideline is intended to inform healthcare professionals, decision-makers, policymakers, patients, and caregivers.
Population
These recommendations target all children and adolescents under 18 who have been in contact with pulmonary TB patients in Colombia. This includes immunocompetent and immunosuppressed children (e.g., those with HIV infection or other causes of immunosuppression), as well as those in contact with drug-resistant TB cases. The guideline does not provide recommendations for children already diagnosed with active TB.
Users
This CPG is designed for use by the National TB Control and Prevention Program (PNCPT) at all levels (national, departmental, and municipal), Health Benefit Plan Administrators (EAPB), and Health Service Provider Institutions (IPS) across all levels of care. It is intended for all healthcare professionals caring for children in contact with TB, including general practitioners (inside or outside TB programs), family physicians, nurses (professional or auxiliary), pediatricians, and pediatric pulmonology and infectious diseases subspecialists. The guideline also includes educational materials for parents and proposes a national implementation strategy aligned with the General System of Social Health Insurance.
Materials and Methods
Registration and reporting
This guideline was registered on the PREPARE platform (PREPARE-2022CN411: http://www.guidelines-registry.org/guid/1787), and progress updates were published on the following website: https://www.uniremington.edu.co/facultades/facultad-de-ciencias-de-la-salud/investigacion/. This report follows the recommendations from the AGREE (Appraisal of Guidelines for Research and Evaluation) and RIGHT (Reporting Items for Practice Guidelines in HealThcare) checklists 26-28. Table 1 presents a summary of recommendations.
Panel composition, conflict of interest disclosure, and question development
The panel included clinical experts (general practitioners from TB programs, pediatricians, pediatric infectious disease specialists and pulmonologists, a pediatric radiologist, and a psychologist), field experts (national, departmental, and municipal TB program leads; experts in nursing, bacteriology, and microbiology), researchers, decision-makers, and delegates from related scientific societies. The appendix presents the full list of participating institutions, panel members, and their expertise, and roles.
Conflicts of interest were disclosed using a modified version of the Colombian methodological guide 29, which included the monetary value of any benefits received, as recommended by the Guidelines International Network 30. Declarations were reviewed by an internal committee (methodological and thematic leaders), which decided on full participation, partial participation, or exclusion (see appendix ). Using the GRADE methodology, clinical questions were developed, and outcomes were prioritized as critical or important but non-critical for decision-making 31,32. A list of generic questions is provided in Table 2. Each question was formatted in PICO format, detailed later in the document along with the results, and included in the appendix .
Table 2 Generic Questions
| 1. What is the diagnostic accuracy of detecting signs and symptoms and performing chest X-rays in all children and adolescents under 18 years of age to rule out active tuberculosis in contacts of pulmonary TB cases? |
| 2. What is the diagnostic accuracy of the interferon-gamma release assay (IGRA) compared to the tuberculin skin test (TST) for diagnosing latent TB infection in children and adolescents under 18 years of age who are contacts of active pulmonary TB cases (including children with or without HIV or other immunocompromised conditions)? |
| 3. What is the effectiveness and safety of the available preventive TB treatment regimens for children and adolescents under 18 years of age without HIV infection in contact with drug-sensitive TB? |
| 4. What is the efficacy and safety of the available regimens for treating latent TB infection in children and adolescents under 18 years of age with HIV infection who are contacts of drug-sensitive TB cases? |
| 5. What is the efficacy and safety of the available regimens for treating latent TB infection in children and adolescents under 18 years of age with special conditions (immunocompromise -solid organ transplant, oncology, primary immunodeficiency [PID], severe malnutrition-, liver disease, kidney disease, or diabetes)? |
| 6. What is the efficacy and safety of the available regimens for treating latent TB in children and adolescents under 18 years of age diagnosed with latent TB infection and who are contacts of drug-resistant TB cases (monoresistant, multidrug-resistant [MDR], or extensively drug-resistant [XDR])? |
| 7. What is the utility of close clinical observation without pharmacological treatment in TB contacts when the infection period exceeds one year, depending on age? |
| 8. During the immunological window period following TB exposure, should preventive treatment be provided to children and adolescents under 18 years of age in contact with TB? |
| 9. What monitoring tests should be used for adverse effects during latent TB treatment follow-up in children and adolescents under 18 years of age? |
| 10. What interventions can be implemented to improve treatment adherence for latent TB in children and adolescents under 18 years of age? |
Literature search and evidence selection criteria
Systematic searches were conducted up to February 14, 2023, updated on September 1st, 2024, for questions 3 and 6. For primary studies and systematic reviews, the following databases were searched: MEDLINE (Ovid), Embase (Ovid), the Cochrane Central Register of Controlled Trials, and the Database of Systematic Reviews (Ovid). To identify CPGs, we also searched guideline repositories and developers' websites (see appendix ). For CPGs and systematic reviews on TB infection (TBI) in children, two independent reviewers screened all identified references and selected eligible ones. Full texts were then independently reviewed for inclusion. Discrepancies were resolved by consensus or a third reviewer.
The quality of included CPGs and systematic reviews was assessed independently and in duplicate using the AGREE-II 33,34 and AMSTAR-2 35 tools, respectively. If low-quality, outdated, or non-aligned reviews/guidelines were found, de novo systematic reviews (SRs) were conducted. For diagnostic questions, we considered cross-sectional, case-control, and cohort studies providing data for diagnostic accuracy. For intervention (treatment and follow-up) questions, we prioritized randomized controlled trials (RCTs), followed by prospective observational studies. Title/abstract screening, full-text selection, and risk of bias assessment (using ROB-2 for RCTs and ROBINS-I for observational studies) 36,37 were conducted independently by two or three reviewers. Disagreements were resolved by consensus or third-party adjudication. Lastly, we also searched for studies on patient/caregiver values and preferences (qualitative or quantitative), cost-effectiveness, feasibility, applicability, and equity (with no design limitations). Results were summarized using GRADE’s Evidence-to-Decision (EtD) framework 38,39 and are detailed in the appendix .
Evidence synthesis and certainty assessment
All included studies were summarized narratively. When existing SRs were low-quality or did not match our PICO questions, de novo SRs and meta-analyses were performed. We updated SRs with search dates older than two years. GRADE Summary of Findings tables were constructed for SRs that lacked them.
For diagnostic questions (1 and 2) requiring de novo SRs and meta-analyses, MetaDisc® 40 was used following the Cochrane Handbook. Protocols were registered in PROSPERO (CRD42022355211, CRD42021271512). For intervention questions (3 and 5), we used RevMan (version 5.3, Nordic Cochrane Centre, Cochrane Collaboration, 2014) for direct comparisons and R software (https://www.R-project.org) for network meta-analyses (PROSPERO: CRD42021271512, CRD42021237928). We assessed heterogeneity with the χ² test (diagnostics) and I² statistic (interventions) 41. Lastly, certainty of evidence was assessed using the GRADE approach, considering risk of bias, imprecision, inconsistency, indirectness, and publication bias for interventions 42-46, diagnostics 47,48, and network meta-analyses 49-52. Certainty was rated as very low, low, moderate, or high 53. Table 3 explains certainty levels.
Table 3 GRADE Certainty of the Evidence and Its Interpretation
| Certainty Level | Interpretation |
|---|---|
| High | The authors have high confidence that the true effect is close to the estimated effect. |
| Moderate | The authors believe that the true effect is likely to be close to the estimated effect. |
| Low | The true effect may be substantially different from the estimated effect. |
| Very Low | The true effect is likely to be substantially different from the estimated effect. |
Inclusion of values and preferences
A panelist who was both a patient with a history of TB and the mother of a child with TBI participated with clinicians and methodologists in all discussions, voting, and decisions. Additionally, we conducted a SR on values and preferences of children with TBI and their caregivers 54, and a survey was administered to parents/caregivers of children with TBI in three Colombian cities. These inputs informed the development of the recommendations.
Cost and resource considerations
We consulted the Ministry of Health (In Spanish, Ministerio de Salud y Protección Social, MSPS) for treatment regimen costs and contacted specific diagnostic laboratories and other institutions for information on IGRA and chest X-ray costs. We also conducted an economic evaluation to determine the cost-effectiveness of IGRA in Colombia 55.
Recommendation formulation and strength
We used the GRADE-EtD framework 38,39, which compiles evidence on effectiveness and safety (GRADE profile), benefit-risk balance (beneficial vs. harmful outcomes, including adverse events or hepatotoxicity), values and preferences, resource use, cost-effectiveness, impact on health equity, acceptability, and feasibility. The panel's judgments on each factor were recorded, followed by voting and consensus. We defined agreement when ≥70% of votes were achieved in any EtD judgement and for the final recommendations. Recommendations were classified as strong or conditional (either for or against), depending on the overall GRADE-EtD judgment. Table 4 outlines these definitions and implications. We initially used the GRADEpro GDT software (https://www.gradepro.org/) and later received a grant from the MAGIC Foundation to finalize the CPG and publish it on the MAGICApp platform (https://app.magicapp.org/#/guideline/7189).
Table 4 Implications of the Strength of Recommendations According to Different Guideline Users
| Strength of the Recommendation | Strong | Conditional |
|---|---|---|
| Clinicians | Most individuals should receive the recommended course of action. Adherence to this recommendation could be used as a quality criterion or performance indicator. Formal decision aids are not likely to be needed to help individuals make decisions aligned with their values and preferences. | Different choices may be appropriate for different patients, and clinicians must assist each patient in making a management decision consistent with their values and preferences. Decision aids may be useful to help individuals make choices aligned with their values and preferences. Clinicians should expect to spend more time in shared decision-making. |
| Patients | Most people in this situation would want the recommended course of action, and only a small proportion would not. | While many would prefer the suggested course of action, a substantial number would not. |
| Policymakers / Health System Decision-Makers | The recommendation can be adopted as policy in most situations, including as a basis for performance indicators. | Policymaking will require extensive deliberation and stakeholder engagement. Policies are more likely to vary across regions. Performance indicators should reflect the need for proper deliberation on available management options. |
Available at: https://gdt.gradepro.org/app/handbook/handbook.html
Updating the guideline
Recommendations should be updated considering new evidence. appendix presents a prioritization exercise for updates using the UpPriority tool 56. Questions rated as high-priority should be updated no later than three years from publication.
Implementation and patient/caregiver guide
The appendix outlines an implementation strategy, including prioritized recommendations, follow-up indicators, and barrier analysis. A management algorithm summarizes evaluation, diagnosis, and follow-up recommendations (Figure 1), and treatment regimens are summarized in Table 5. We also developed a plain-language guide for patients and caregivers, outlining key recommendations. It is available on the Corporación Universitaria Remington website (https://www.uniremington.edu.co/facultades/facultad-de-ciencias-de-la-salud/investigacion/) and the Evidence and Deliberation Unit for Decision-Making (UNED) of the University of Antioquia (https://uned.medicinaudea.co/uned/productos-y-servicios-2/).
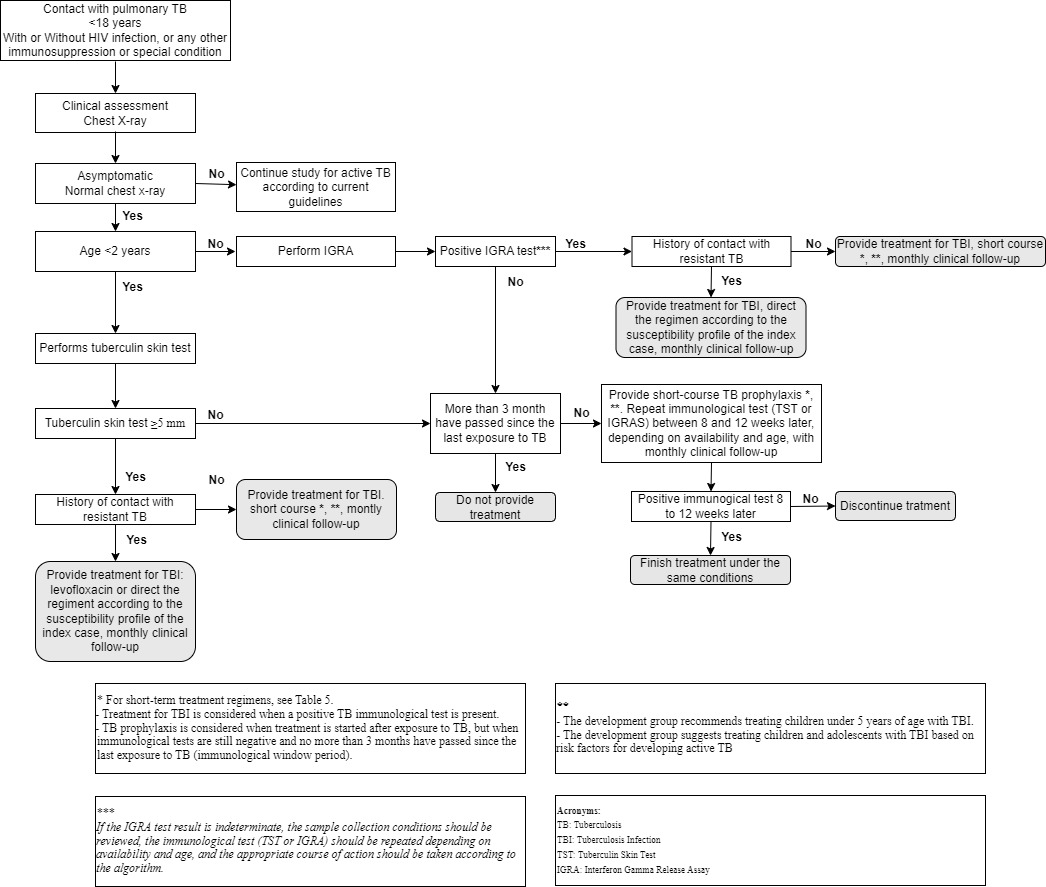
Figure 1 Algorithm for the Evaluation, Treatment, and Follow-up of Children in Contact with TB. TB: tuberculosis, TBI: tuberculosis infection, TST: tuberculin skin test, IGRA: Interferon gamma release assay. * For short-term treatment regimens see Table 5 (-treatment for TBI is considered when a positive TB immunological test is present. -TB prophylaxis is considered when treatment is started after exposure to TB, but when immunological tests are still negative and no more than three months have passed since the last exposure to TB (immunological window period). ** the development group recommended treating children under five years of age with TBI and the development group suggests treating children and adolescents with TBI based on risk factors for developing active TB. *** if the IGRA test result is indeterminate, the sample collection conditions should be reviewed, the immunological test (TST or IGRA) should be repeated depending on availability and age, and the appropriate course of action should be taken according to the algorithm.
Table 5 Treatment Regimens by Population.
| Population | Treatment Regimen | |
|---|---|---|
| TBI in individuals under 18 years without immunosuppression, in contact with drug-sensitive pulmonary TB | Under 2 years | • *Isoniazid/rifampicin, once daily for 3 months |
| • *Rifampicin, once daily for 4 months | ||
| • Isoniazid, once daily for 6 months (if short regimens are unavailable) | ||
| Aged 2 years or older | • *Isoniazid/rifapentine, once weekly for 3 months | |
| • *Isoniazid/rifampicin, once daily for 3 months | ||
| • *Rifampicin, once daily for 4 months | ||
| • Isoniazid, once daily for 6 months (if short regimen are unavailable) | ||
| TBI in individuals under 18 years with HIV infection, in contact with drug-sensitive pulmonary TB | • *Isoniazid/rifampicin, once daily for 3 months | |
| • *Isoniazid/rifapentine, once weekly for 3 months | ||
| • *Rifampicin, once daily for 4 months | ||
| • Isoniazid, once daily for 6 months (if short regimen are unavailable) | ||
| TBI in individuals under 18 years with non-HIV immunosuppression** in contact with drug-sensitive pulmonary TB | • Weight <10 kg: Isoniazid, once daily for 6 months | |
| • Weight ≥10 kg: Isoniazid/rifapentine, once weekly for 3 months | ||
| TBI in individuals under 18 years in contact with drug-resistant pulmonary TB | • Levofloxacin, once daily for 6 months | |
| • Regimen should be tailored according to the drug susceptibility profile of the index case. | ||
* Short-term treatment regimens
** Non-HIV immunosuppression conditions include solid organ transplant recipients, oncology patients, severe malnutrition, primary immunodeficiency, renal or hepatic disease, and diabetes, among others.
External review and dissemination
Two stakeholder engagement events were held (see appendix ). Per the usual journal editorial process, this manuscript was peer-reviewed by two external reviewers and revised accordingly.
Results (recommendations)
1. Clinical evaluation and chest X-ray for identifying active TB
1. We recommend performing a chest X-ray to screen for active TB in all children and adolescents under 18 years of age who have been in contact with pulmonary TB patients, instead of relying solely on the presence of clinical signs and symptoms suggestive of TB (Moderate Certainty. Strong recommendation)
Considerations: The guideline panel considers that, for implementation of this recommendation, it is essential to ensure that the Ministry of Health and Social Protection (MSPS) makes every effort to provide communities with adequate access to this technology, which is included in the benefits plan, also covering the transportation costs it may entail. Alternatives such as telemedicine for image interpretation or results delivery may be explored to reduce the access gap to chest X-rays, particularly in rural and remote areas.
Summary of the evidence
A de novo systematic review and meta-analysis of diagnostic accuracy studies of signs/symptoms and chest X-ray for detecting active TB was conducted. Four studies were included 57-60. The meta-analysis showed that signs and symptoms (cough, fever, and poor weight gain) had a combined sensitivity of 98% (95% CI: 36 to 100%) and combined specificity of 65% (95% CI: 58 to 71%), with a positive likelihood ratio (LR+) of 2.83 (95% CI: 2.33 to 3.45) and a negative likelihood ratio (LR-) of 0.02 (95% CI: 0.00 to 2.51; 3 studies; 724 children).
In contrast, chest X-ray showed a combined sensitivity of 91% (95% CI: 62 to 99%) and combined specificity of 96% (95% CI: 83 to 99%), with an LR+ of 28.04 (95% CI: 4.03 to 195.13) and LR- of 0.09 (95% CI: 0.01 to 0.47) (see appendix ). Lastly, one study including 186 children showed that a combination of cough, sweating, weight loss, fever, anorexia, fatigue, abnormal auscultation, and abnormal chest X-ray yielded a sensitivity of 60% and a specificity of 99.4% for active TB.
From evidence to recommendation Users
The panel considered achieving an accurate diagnosis of TBI in the Colombian pediatric population a priority. Chest X-ray is a diagnostic method with significant desirable effects and minimal undesirable effects that can support this goal. The evidence for chest X-ray accuracy was judged to be of moderate quality for the target population. Moreover, the analysis of patients' values and preferences, healthcare professionals, parents, caregivers, and the health system showed no significant uncertainty or variability regarding the use of this technology. Resource requirements were considered moderate, with high certainty. Routine chest X-ray implementation in the Colombian context was judged likely to improve equity, with high acceptability and feasibility. Given all the above, the overall balance of effects favors chest X-ray, supporting a strong recommendation.
2. Interferon-Gamma Release Assays (IGRAs) and Tuberculin Skin Test (TST)
2. We recommend the use of Interferon-Gamma Release Assays (IGRAs), specifically the QuantiFERON-TB® test, as the diagnostic test for tuberculosis infection in children and adolescents aged 2 to 18 years who have been in contact with patients with pulmonary TB. In children under 2 years of age, and while full implementation of the test is underway in Colombia, we recommend using the tuberculin skin test (TST). (Low certainty. Strong recommendation)
Considerations:
1. The available and evaluated evidence pertains entirely to the QuantiFERON-TB® test.
2. The development panel considers it essential that the Ministry of Health and Social Protection (MSPS) makes every effort to implement this test nationwide so that all communities can have adequate access to this technology.
Summary of the evidence
A de novo systematic review and meta-analysis of Interferon-Gamma Release Assays (IGRAs) diagnostic accuracy studies and the Tuberculin Skin Test (TST) were conducted. Twelve studies on QuantiFERON-TB® test (QFT) 63-72 and eight 61,62,64-66,68,70,71 on TST were included, respectively. We performed a random-effects meta-analysis for diagnostic tests to evaluate the combined accuracy of the IGRA and TST ≥5 mm or >5 mm for detecting progression to TB disease.
The QFT, using development of TB as the gold standard (12 comparisons, 9,738 cases), showed a combined sensitivity of 0.81 (95% CI: 0.56 to 0.94) and a combined specificity of 0.84 (95% CI: 0.75 to 0.90). The combined positive likelihood ratio (LR+) was 5.11 (95% CI: 2.91 to 8.95), and the negative likelihood ratio (LR-) was 0.22 (95% CI: 0.08 to 0.60). The TST > or ≥5 mm, also using TB development as the gold standard (8 comparisons, 6,430 cases), showed a combined sensitivity of 0.76 (95% CI: 0.60 to 0.86), specificity of 0.72 (95% CI: 0.55 to 0.84), LR+ of 2.67 (95% CI: 1.64 to 4.32), and LR- of 0.34 (95% CI: 0.20 to 0.56).
Cost-effectiveness study of QFT
We conducted a cost-effectiveness analysis (CEA) comparing QFT and TST in the Colombian context. The QFT test was cost-effective with an incremental cost-effectiveness ratio (ICER) of USD 576 per correctly diagnosed case, below the cost-effectiveness threshold for Colombia of USD 6,666. The complete CEA has been published and is available elsewhere 55.
From evidence to recommendation
The development panel considered an accurate diagnosis of TB in the pediatric population in Colombia a priority. QFT likely has a high diagnostic performance, and the risk-benefit balance of its use is favorable. The certainty of evidence was judged to be very low to low for diagnostic accuracy but high overall for the test’s effects. No significant uncertainty or variability was noted regarding values and preferences among patients, healthcare professionals, parents, caregivers, and the health system. Required resources were considered moderate based on cost data from various regions of Colombia. Moreover, our group’s CEA showed that QFT is likely to be dominant over TST in the Colombian context. Finally, implementing QFT nationwide would likely improve health equity, with high acceptability and feasibility. Therefore, the panel considered the balance of effects favors using the QFT test, supporting a strong recommendation.
3. Treatment of children without HIV in contact with drug-sensitive TB cases
3.1 We recommend treating tuberculosis infection (TBI) with short-course regimens in children under 5 years of age without HIV infection who have been in contact with drug-sensitive pulmonary TB. (Low certainty. Strong recommendation)
3.2 We suggest treating TBI with short-course regimens in children and adolescents aged 5 to 18 years without HIV infection who have been in contact with drug-sensitive pulmonary TB. (Low certainty. Conditional recommendation)
3.3 We suggest treating TBI with isoniazid for 6 months only when short-course regimens are not accessible, in children and adolescents under 18 years of age without HIV infection who have been in contact with drug-sensitive pulmonary TB. (Low certainty. Conditional recommendation)
3.4 We do not recommend treating TBI in children and adolescents under 18 years of age without HIV infection who have been in contact with drug-sensitive pulmonary TB using the 9-month daily isoniazid regimen. (Low certainty. Strong recommendation against)
Considerations
- The recommended short-course regimens are:
○ 3 months of isoniazid plus rifampin
○ 3 months of isoniazid plus rifapentine (12 weekly dose; only in children > 2 years of age)
○ 4 months of rifampin monotherapy.
- The administration of these regimens depends on the child’s ability to take the available pharmaceutical forms.
Summary of the evidence
We conducted a de novo systematic review with network meta-analysis of randomized controlled trials (RCTS) in children who had been in contact with drug-sensitive TB. Seven RCTs were included 73-79, evaluating isoniazid plus rifapentine for 3 months (3HP), isoniazid plus rifampin for 3 (3HR) and 4 months (4HR), rifampin for 4 months (4R), isoniazid for 6 months (6H), 9 months (9H), and 12 months (12H), and no treatment or placebo (NT/PB). We found that the 4HR regimen may be superior to 9H monotherapy (RR 0.49; 95% CI: 0.32 to 0.76; 1 study; certainty: low).
For adherence, 3HP was likely superior to 9H (RR 1.09; 95% CI: 1.03 to 1.15; 1 study; certainty: low), 4HR (RR 1.07; 95% CI: 1.01 to 1.14; 1 study; certainty: moderate), and 4R (RR 1.12; 95% CI: 1.05 to 1.20; 1 study; certainty: moderate). For adverse events, the NMA suggests that 3HP increases the likelihood of any adverse event compared to 4HR (OR 4.56; 95% CI: 1.22 to 16.96; certainty: very low), and 4R (OR 6.37; 95% CI: 2.11 to 19.19; certainty: very low). The NMA also suggests that 4R is associated with fewer adverse events than 9H (OR 0.34; 95% CI: 0.20 to 0.58; certainty: moderate). Risk differences were not estimable or not significant for hepatotoxicity, treatment discontinuation due to adverse events, and 5-year mortality.
From evidence to recommendation
The panel judged that short-course regimens are preferred over long-course regimens due to their desirable effects (lower rates of active TB at 2 years, greater treatment adherence), despite the low certainty of evidence. The benefit-risk balance was more favorable in children under 5, who are at higher risk of developing TB, while the balance was considered less clear in older children and adolescents. The panel judged that the costs of short-course treatments would be moderate when rifampin is used, and potentially lower when using rifapentine. Although no cost-effectiveness studies of short-course regimens in children in contact with TB were found, evidence supports that implementing evaluation programs and strengthening TB prevention efforts is likely cost-effective in low- and middle-income countries-even if additional costs are incurred, they are low relative to the expected benefit. Furthermore, the panel found that implementing short-course regimens will likely improve health equity and considered these regimens acceptable and feasible. Based on this, and the more favorable risk-benefit balance in children under 5, the panel issued a strong recommendation for short-course regimens in children under five and a conditional recommendation for older children and adolescents, depending on clinician judgment and discussion with patients and caregivers.
Research recommendations
Head-to-head RCTs comparing short-course regimens (3HR, 3HP, 4R) are needed. Trials in different age populations (<5 and ≥5 years) are also necessary to reassess the conditional recommendation. Further research is required on cost-effectiveness, patient/caregiver values and preferences related to these treatments, and relevant outcomes.
Implementation needs
Access barriers to rifampin and rifapentine in the country must be eliminated. For instance, rifapentine availability is subject to centralized procurement in Colombia, while rifampin has been challenging to obtain in child-appropriate pharmaceutical forms (dispersible tablets or syrup). It is suggested that the MSPS ensure procurement of dispersible tablets for the recommended regimens.
4. Treatment of Children with HIV Infection in Contact with Drug-Sensitive TB Cases
4.1 Recommendations for Question Four. Treatment of children with HIV infection in contact with drug-sensitive TB cases
4.2 We recommend treating tuberculosis infection (TBI) with short-course regimens in children under 18 years of age with HIV infection who have been in contact with drug-sensitive pulmonary TB. ( Low certainty. Strong recommendation )
4.3 We suggest treating TBI with isoniazid for 6 months only when short-course regimens are not accessible, in children and adolescents under 18 years of age with HIV infection who have been in contact with drug-sensitive pulmonary TB . ( Low certainty. Conditional recommendation )
4.4 We do not recommend treating TBI in children and adolescents under 18 years of age with HIV infection who have been in contact with drug-sensitive pulmonary TB using the 9-month daily isoniazid regimen. (Low certainty. Strong recommendation against)
Considerations:
- The recommended short-course regimens are:
● 3 months of isoniazid plus rifampin
● 3 months of isoniazid plus rifapentine (12 weekly doses; only in children over 2 years)
● 4 months of rifampin monotherapy
- Administration of these regimens depends on the child’s ability to take the available pharmaceutical forms.
- In children with HIV infection receiving antiretroviral therapy (ART) available in Colombia (zidovudine, lamivudine, nevirapine, raltegravir, abacavir, lopinavir, ritonavir), pediatric infectious disease consultation is advised to determine the optimal regimen, considering potential drug interactions. (See the national pediatric HIV guideline: https://www.minsalud.gov.co/sites/rid/Lists/BibliotecaDigital/RIDE/VS/PP/ET/gpc-atencion-infeccion-vih-ninos-ninas-adolescentes-para-padres-cuidadores.pdf)
Summary of the evidence
We identified three potentially eligible systematic reviews (SRs). Two were excluded: one for very low methodological quality and lack of age stratification 80 and one for outdated searches (over 10 years) 81. The review by Zunza 82, rated high methodological quality, included three RCTs 83-85. A later published study was excluded due to clinical heterogeneity 86. The meta-analysis showed that in children with HIV infection not receiving ART, isoniazid prophylaxis compared to placebo reduced the incidence of active TB (HR: 0.31; 95% CI: 0.11 to 0.87; 1 study; 240 participants; low certainty) and mortality (HR: 0.46; 95% CI: 0.22 to 0.95; 1 study; 240 children; low certainty). No differences were found in grade 3 or higher laboratory adverse events (RR: 0.83; 95% CI: 0.26 to 2.63; 1 study; 182 children; very low certainty), and no clinical grade 3 or higher adverse events were reported. In children receiving ART, isoniazid prophylaxis versus placebo showed no differences in incidence of active TB, mortality, clinical adverse events, or laboratory adverse events of grade 3 or higher.
From evidence to recommendation
The panel considered that treatment is superior to placebo in children without ART; however, this scenario is unlikely in Colombia, where access to ART is relatively good. Additionally, the available evidence does not explicitly address children on ART in contact with TB cases. Therefore, the panel chose to use the evidence for HIV-negative children (Question 3) as indirect evidence, supporting that short-course regimens appear superior to longer ones, and both are better than no treatment or placebo. The panel considered this question a priority. Given the variability in adverse event reporting by regimen type, the desirable effects of short-course preventive TB regimens were moderate, and the undesirable effects were minor. No studies were found on values and preferences, but the panel assumed similar preferences favoring short regimens, without significant uncertainty or variability, as in HIV-negative children. Despite the low certainty of the evidence, the balance of effects likely favors short-course regimens. Moreover, treatment costs were judged moderate for regimens including rifampin and lower for those with rifapentine. No cost-effective studies of short-course regimens were found. Evidence from question 3 was used to support the idea that implementing short regimens likely improves health equity. Finally, the panel judged short-course regimens acceptable and feasible. Thus, in summary, given the greater risk-benefit balance in children with HIV, the panel is strongly recommended to treat TBI in HIV-infected children and adolescents under 18 years in contact with drug-sensitive TB.
Research recommendations
RCTs comparing short-course (3HR, 3HP, 4R) vs. long-course (9H) regimens in HIV-infected children and adolescents in contact with drug-sensitive TB and receiving ART are needed. Cost-effectiveness studies, values and preferences studies in the Colombian context, and pharmacokinetic studies of rifapentine and rifampin when co-administered with ART in children are also required.
Implementation needs
Access barriers to rifampin and rifapentine in Colombia must be removed. Rifapentine is subject to centralized procurement, while rifampin has been challenging to obtain in child-appropriate pharmaceutical forms (dispersible tablets or syrup). The Ministry of Health and Social Protection (MSPS) should ensure the acquisition of dispersible tablets for the recommended regimens.
5. Treatment of Children with Special Conditions (Non-HIV Immunosuppression)
5.1 We suggest using the short-course regimen of isoniazid plus rifapentine in 12 weekly doses, instead of the long-course regimen of isoniazid alone, in children and adolescents aged 2 to 18 years with TBI and any special non-HIV immunosuppressive condition. (Very low certainty. Conditional recommendation)
5.2 We suggest using isoniazid for 6 months in children under 2 years of age with TBI and any special non-HIV immunosuppressive condition, while awaiting approval of rifapentine for this age group or emergence of new evidence for other regimens. (Very low certainty. Conditional recommendation)
5.3 We do not suggest using regimens other than isoniazid and rifapentine, due to the lack of evidence on efficacy or safety in children or adults with TBI and non-HIV immunosuppression. (Very low certainty. Conditional recommendation)
Considerations:
1. Special conditions include immunosuppressive treatment, solid organ transplant, primary immunodeficiency, diabetes, or other related conditions.
2. The available evidence is indirect, derived exclusively from adult populations.
3. The panel recommends conducting clinical trials in children to confirm efficacy in this population.
Summary of the evidence
No clinical practice guidelines or systematic reviews were found to address this question. A review of primary studies identified three prospective cohort studies in adults receiving biological therapy for rheumatoid arthritis 87, poorly controlled diabetes mellitus 88, and chronic kidney disease on dialysis 89. All studies compared isoniazid plus rifapentine with the 9-month isoniazid monotherapy regimen (9H). Pooled analysis of these studies showed increased treatment adherence with short-course regimens compared to long-course treatment (RR 1.13; 95% CI: 1.00 to 1.28; 3 studies; very low certainty). Although the authors measured active TB and hepatotoxicity, no cases of these outcomes were reported.
From evidence to recommendation
This question is a priority since the number of children with non-HIV-related immunosuppression is expected to grow, and guidance is needed. The certainty of evidence was judged very low due to its indirectness (adult data) and risk of bias (all studies were observational). Despite this, the panel considered the risk-benefit balance to favor short-course regimens (isoniazid plus rifapentine, 3HP) over long-course regimens (9H). The panel also thought the same factors related to resource use, feasibility, acceptability, values, and cost-effectiveness as those applied to short-course treatment in children without immunosuppression. No evidence was found on cost-effectiveness or values and preferences.
Therefore, despite the very low quality of evidence, the panel strongly recommends that children and adolescents under 18 years of age living with HIV who are in contact with drug-susceptible pulmonary tuberculosis receive the same preventive treatment as those without HIV infection.
6. Treatment in children without HIV infection in contact with drug-resistant TB cases
6.1 We suggest treating tuberculosis infection (TBI) with levofloxacin for 6 months in children under 5 years of age without HIV infection who have been in contact with rifampicin-resistant (RR) or multidrug-resistant (MDR) pulmonary TB. In cases of confirmed contact with levofloxacin-resistant TB, we suggest tailoring treatment based on the drug susceptibility profile of the index case. (Moderate certainty. Conditional recommendation)
6.2 We suggest treating TBI with levofloxacin for 6 months in children and adolescents aged 5 to 18 years without HIV infection who have been in contact with RR or MDR pulmonary TB, based on their individual risk factors for developing active TB. In cases of contact with levofloxacin-resistant TB, we suggest tailoring treatment according to the drug susceptibility profile of the index case. (Moderate certainty. Conditional recommendation)
Considerations:
- These recommendations are conditional on the publication of the available preliminary evidence and future results from ongoing studies evaluating the effectiveness of regimens for children in contact with drug-resistant TB.
- The panel noted barriers to treatment access, as fluoroquinolones-present in several regimens-are not registered by INVIMA (Instituto Nacional de Vigilancia de Medicamentos y Alimentos; the Colombian drug regulatory agency) for use in children under 12 years in Colombia. The panel urges the Ministry of Health and Social Protection (MSPS) and INVIMA to facilitate authorization for their use in children with TBI or drug-resistant active TB.
Summary of the evidence
We conducted a de novo SR due to the lack of recommendations in clinical practice guidelines (CPGs) and the poor quality of existing SRs. In the initial February 2023 search, no clinical trials were found; seven observational studies were included 90-96. Three studies included both adults and children, and four included only children. Only one study 96 compared treatment to no treatment. None conducted multivariable analyses to adjust for confounding. All had a very high risk of bias and used varied regimens, most of which included levofloxacin. At the time, three RCTs were ongoing (registered on clinicaltrials.gov). The study by Schaaf 96 reported 6 cases of TB development in the untreated group.
From evidence to recommendation
The panel judged that evidence on treatment regimens for children in contact with drug-resistant TB is essentially lacking. Limited available data suggest levofloxacin-based regimens may be beneficial, though certainty is very low, and significant uncertainty remains. No data were found on values, costs, applicability, or feasibility. Due to its low cost, levofloxacin was considered to have minimal budgetary impact, whereas rifampicin-based regimens had a moderate impact (as seen in previous questions). No cost-effectiveness studies were found for drug-resistant TB regimens. The panel judged that treating these children would likely improve health equity and that this intervention is acceptable and feasible. Based on this, the balance of effects slightly favors treatment over no treatment. Thus, the panel decided to issue a conditional recommendation for treatment. Since some relevant evidence is pending, treatment should be based on the index case's drug susceptibility profile and the patient's risk factors for TB. Evidence should be re-evaluated as trial results become available.
Update in response to new evidence
After the panel meeting and formulation of the initial recommendation (October 2023), and during the finalization of this guideline, preliminary results from two RCTS 97,98) were identified. The V-QUIN and TB CHAMP trials, summarized in the 2024 WHO 98, provided relevant findings. TB CHAMP randomized children in contact with drug-resistant TB to levofloxacin or placebo and found a reduction in confirmed TB cases (3 vs. 7) after one year. V-QUIN, conducted in adults, found a similar reduction (6 vs. 11 cases). WHO used this evidence in its 2024 guideline to recommend levofloxacin for all ages. We held an extraordinary meeting in October 2024 to assess this evidence. The TB-CHAMP study in children showed a possible benefit (RR 0.44; 95% CI: 0.11 to 1.70), while V-QUIN showed similar results (RR 0.54; 95% CI: 0.20 to 1.46). Meta-analysis of both studies yielded a pooled effect favoring levofloxacin (RR 0.50; 95% CI: 0.23 to 1.12), though the confidence interval crossed the line of no effect. The certainty of evidence was rated as moderate. No differences between levofloxacin and placebo were found in short-term adverse events (RR 1.01; 95% CI: 0.36 to 2.82).
The panel re-evaluated its original recommendation and maintained a conditional recommendation for levofloxacin in children exposed to drug-resistant TB based on moderate certainty evidence. A strong recommendation was not issued because the final publications were not yet available then, and the effect estimate still shows some imprecision. Levofloxacin may have feasibility and availability limitations; its long-term safety remains unknown (adverse events were measured only up to 30 days after treatment), and pediatric formulations require approval by INVIMA. Additionally, cost-effectiveness in the Colombian context is still unknown.
Research recommendations
Studies on the cost-effectiveness of levofloxacin for children in Colombia and on the values and preferences of patients and caregivers regarding treatment in this population are needed.
Implementation needs
Access barriers to fluoroquinolones in children must be removed, especially as these drugs are included in several regimens described in the literature and are restricted for children under 12 in Colombia (according to the INVIMA). The panel urges the Ministry of Health and Social Protection and the INVIMA to authorize their use in children with TBI or drug-resistant active TB. Levofloxacin is available in dispersible tablet form, which should be used in children unable to swallow tablets-provided it is available through the National TB Control Program (PNPCT). Daily dosing should follow national PNPCT guidelines.
7. Treatment in children presenting more than one year after contact
7.1 We consider that clinical observation alone is not sufficient to manage cases that may benefit from treatment; therefore, we recommend treating TBI in children under 5 years of age who have been in contact with drug-sensitive pulmonary TB, even when more than one year has passed since exposure. If the child is under 5 years of age, the time elapsed since exposure is not relevant. (Low certainty. Strong recommendation)
7.2 We consider that clinical observation alone is not sufficient to manage cases that may benefit from treatment; therefore, we suggest treating TBI in children and adolescents aged 5 to 18 years who have been in contact with drug-sensitive pulmonary TB more than one year earlier, based on their risk factors for developing active TB. (Moderate certainty. Conditional recommendation)
7.3 We suggest not treating TBI in children and adolescents under 18 years of age who have been in contact with drug-resistant pulmonary TB more than one year earlier. (Expert consensus. Conditional recommendation)
Summary of the evidence
Only one individual participant data meta-analysis 99 was included. This review assessed the risk of developing TB in children with close contact to a TB case and included 34 cohort studies from 34 countries (29 prospective and 17 retrospective), including studies from Colombia. Given the lack of RCTs comparing treatment vs. no treatment in children presenting one year after infection, the panel considered this systematic review an adequate source of indirect evidence. Martinez et al. 99, reported that in children with a positive TST or IGRA, the incidence of active TB per 100 person-years was 3.9 at 91-365 days, 1.2 at 366-730 days, and 1.1 after >730 days from initial infection. The 2-year cumulative risk of developing TB among untreated children varied by age and infection status. In untreated children, the risk followed a U-shaped pattern: 7.6% in children <5 years, 5.2% in children aged 5-9 years (p= 0.0027 vs <5 years), 5.6% in those aged 10-14 years (p= 0.0145 vs <5 years), and an increase to 6.7% in those aged ≥15 years (p= 0.3491 vs <5 years.
The panel also discussed the local study by Benjumea et al. 100, which found an annual infection risk of 17% (95% CI: 11.8-22.2), with a high risk of active TB persisting into the second-year post-exposure, especially in children under five and particularly those under 1 year. The incidence rate was 12.4 per 1,000 person-years. Risk of active TB increased with higher initial TST results (TST 5-9 mm: HR = 8.55, 95% CI: 2.5-29.2; TST ≥10 mm: HR = 8.16, 95% CI: 2.0-32.9). The certainty of evidence was judged as moderate.
From evidence to recommendation
The panel considers this a key issue. The common belief is that risk diminishes significantly after one year, making observation without treatment seem acceptable. However, the evidence shows that the risk remains meaningful. In addition, the panel considered the goals of the WHO “End TB” Strategy and Colombia's “Towards the End of TB” Strategic Plan 2016-2025, which aims to reduce incidence to fewer than 10 cases per 100,000 and provide complete TBI treatment to 90% or more of eligible cases by 2025. As a significant proportion of active TB cases result from latent TB infection (LTBI) reactivation or post-primary TB, proper treatment of LTBI is essential to meet these targets. Moreover, no evidence was found regarding values, cost-effectiveness, acceptability, equity, or feasibility specifically in children presenting more than a year after infection. However, the panel applied the same considerations from question 3, as the recommended treatment is the same for children presenting earlier. Overall, the panel judged that the balance of effects favors treating children, presenting more than a year after infection over clinical observation alone.
Implementation needs
Efforts should be made to ensure that contact tracing is performed promptly upon TB case detection, that children exposed in the household are identified early, and that they are swiftly evaluated in health facilities to initiate treatment based on age and risk according to recommendations in question 3. Eliminating access barriers will prevent children from being diagnosed more than a year after infection.
8. Treatment During the Immunological Window Period
8.1 We recommend providing preventive TB treatment to children under 5 years of age who have been exposed to TB and are in the immunological window period. (Very low certainty. Strong recommendation)
8.2 We suggest providing preventive TB treatment to children and adolescents aged 5 to 18 years who have been exposed to TB and are in the immunological window period, depending on their risk of developing active TB. (Very low certainty. Conditional recommendation)
GPS Good Practice Statement: The guideline panel considers it advisable to repeat the immunological test, depending on availability (tuberculin skin test [TST] or IGRA, according to the recommendations in question 2), between 8 and 12 weeks after treatment initiation, to determine whether treatment should be continued. If the second test is negative, treatment should be discontinued; if positive, it should be completed.
Summary of the evidence
We searched for primary studies and included eight studies 101-108. Due to methodological and clinical heterogeneity, results were not statistically pooled-only one study 104 compared treated and untreated children under 5 years of age. All eight studies had a high risk of bias and were heterogeneous in outcomes and follow-up duration. Six studies reported no cases of active TB in children with negative TST. Five studies reported adverse events, three of which mainly described mild events in 1-11% of participants. Definitions of treatment adherence varied, with reported adherence rates ranging from 20% to 95%. Among the three studies that repeated TST, the conversion rate (positive second TST) ranged from 1% to 63%.
The most relevant study 101 assessed asymptomatic children under 6 months with recent TB contact-regardless of initial test results-and children aged 6 months to 5 years with initial IGRA or TST results, all of whom started prophylaxis during the window period (8-12 weeks post-exposure), totaling 752 children. Treatment was isoniazid (home-supervised); rifampin was used in cases of resistance or contraindication. No children developed active TB during the window period or follow-up (median: 5.7 years; IQR: 3.7-7.6). Adverse events occurred in 0.9% of participants. The second TST (performed at 8-12 weeks) was positive in 37 children (4.9%), of whom 35 (94.6%) completed treatment.
The panel also considered indirect evidence from studies informing question 7. The risk of TB in children with negative TST or IGRA after TB contact was reported in a meta-analysis 99, with a 2-year incidence of 0.8%, highest in the first 90 days and particularly among children under 5. Additionally, the study by Benjumea et al. 61, in Antioquia, Colombia, reported an annual infection risk of 17% (95% CI: 11.8-22.2) (see question 7). Preliminary findings from another local study in which children under 5 with negative TST and IGRA, and who were in the immunological window, received rifampin, were also considered. Of 26 children meeting criteria, 15 (57.7%) had a negative second TST and discontinued treatment, while the remainder continued treatment due to a positive second test. No cases of active TB were detected.
From evidence to recommendation
The panel considers that cases of children presenting during the immunological window are not uncommon and represent a critical opportunity for early intervention. Although there is no direct evidence from RCTs comparing treatment vs. observation in this setting, the panel judged that short-course treatments improve adherence and that evidence from local studies shows nearly half of children later tested positive on TST. Therefore, the benefit-risk balance favors treatment over observation. Moreover, while no formal evidence exists, the panel considers it good clinical practice to repeat the immunological test (TST or IGRA) between weeks 8 and 12. If the second test is negative, treatment may be discontinued; if positive, it should be completed. No studies were found on values, cost-effectiveness, acceptability, equity, or feasibility specifically for this population. Thus, the panel applied the same considerations discussed in question 3, as the treatment is identical to that used for children outside the window period. In conclusion, the balance of effects favors treatment over clinical observation alone in children in the immunological window period.
9. Clinical and laboratory follow-up for children undergoing treatment
9.1 We recommend monthly clinical monitoring and follow-up by trained primary healthcare professionals in TB and medication management (physician or nurse) for children and adolescents under 18 years of age receiving treatment for tuberculosis infection (TBI). (Expert consensus. Strong recommendation)
9.2 We suggest not performing routine laboratory tests in children and adolescents under 18 years of age who are initiating or undergoing TBI treatment, except in patients with liver disease or those receiving hepatotoxic medications, in whom monthly laboratory monitoring is recommended. (Expert consensus. Conditional recommendation)
GPS: Good Practice Statement: The panel considers that clinical follow-up for children receiving TBI treatment should be performed monthly by a trained physician.
Summary of the evidence
No CPGs, SRs, observational studies, or randomized clinical trials were identified that directly compared outcomes in patients undergoing laboratory monitoring versus exclusive clinical follow-up. The analysis in question 3 provided indirect evidence not derived from RCTs, including adverse events such as hepatotoxicity. None of the trials evaluating treatment regimens in children reported any cases of hepatotoxicity. Available reports suggest that hepatotoxicity is much rarer in children than in adults.
From evidence to recommendation
The evidence supporting this recommendation is indirect and of low quality. No data was found regarding clinical or laboratory follow-up strategies' cost-effectiveness, values, feasibility, or acceptability. The use of resources for clinical monitoring is negligible compared to laboratory testing, especially considering the indirect costs likely to be assumed by patients and families (e.g., additional visits to healthcare facilities for blood tests). The panel agreed that implementing clinical follow-up alone is feasible, unlikely to affect health equity negatively, and acceptable to stakeholders. Based on this, the balance favors clinical over monthly laboratory monitoring. However, the panel emphasizes that this approach is only appropriate for patients without a history of liver disease and not receiving hepatotoxic medications. Moreover, clinical monitoring should be performed by physicians trained in managing these treatments.
10. Interventions to improve treatment adherence
10.1 We recommend using short-course regimens over long-course regimens to improve adherence to TBI treatment in children and adolescents under 18 years of age. (Low certainty. Strong recommendation)
10.2 We recommend implementing comprehensive approaches (including psychological assessment, social work, nutrition support, patient-centered care, direct contact and access to healthcare staff, multipurpose visits, among others) to identify and address barriers and promote adherence to TBI treatment in children and adolescents under 18 years of age. (Very low certainty. Strong recommendation)
10.3 We suggest considering caregiver- and child-focused interventions such as transport-related financial incentives, nutritional support, psychosocial assistance, and SMS reminders, depending on the identified barriers, for TBI treatment in children and adolescents under 18 years of age. (Low certainty. Conditional recommendation)
Summary of the evidence
No CPGs or SRs directly addressing this question were identified. Six SRs were potentially eligible, of which four were of high quality 90,109-112. There was high variability in the interventions assessed (individual or combined strategies), most focused on adults and adherence to active TB treatment. The panel conducted a de novo SR, identifying 14 randomized trials potentially relevant to the question. Due to heterogeneity in the interventions and comparators, a meta-analysis was not feasible. Studies were analyzed by intervention type.
Eight studies assessed short-course treatments to improve adherence: three of these compared rifampin or rifapentine-based regimens versus nine months of isoniazid. The remaining five did not assess adherence directly, compared treatment to no treatment, focused on directly observed therapy, or used ultra-short regimens (1 month), and were excluded. The three included studies 76,77,80) -also cited in question 3-reported higher adherence with short regimens (3HP, 4R, and 4HR) versus 9H, with moderate certainty of evidence. Three studies evaluated educational strategies directed at adolescents, parents, or using community-based approaches 113-115. All showed adherence benefits, with relative risk increases between 1.05 and 1.55 (very low certainty). Two SMS reminders 116,117 studies were conducted in adults.
Additionally, as part of the research program funding this guideline, a quasi-experimental study evaluated the impact of an integrated care strategy on adherence in children in contact with TB 62. It compared a retrospective cohort receiving usual care (isoniazid) to a prospective cohort receiving 4 months of rifampin with a comprehensive, child- and family-centered care package. This included active follow-up, financial incentives (transport and food subsidy), diagnostic access without insurance authorization, and multidisciplinary team evaluation 62. Treatment completion was 59% in the isoniazid group and 90.7% in the intervention group (absolute difference: 31%; p <0.001). The certainty of evidence was low.
From evidence to recommendation
The panel concluded that short-course regimens improve adherence compared to longer regimens, which were already recommended under question 3. Additionally, low-quality evidence suggests that educational interventions may increase adherence. The balance of effects favors these strategies over inaction. The panel also considered that the factors discussed in question 3-such as resource use, values, acceptability, feasibility, and equity-also apply here and support short-course regimens. Considering the above and the favorable benefit-to-risk ratio, the panel issued a strong recommendation in favor of short-course regimens. Despite limited evidence in children and low certainty, the panel also believes educational, and support strategies should be considered to improve adherence.











 text in
text in 



