Remark
| 1) Why was this study conducted? |
| This study stemmed from concerns among the clinical staff at the institution, who observed that patients—particularly younger women and those affiliated with Private Insurance—were not adhering to hormonal therapy, despite the benefits associated with this treatment. Upon reviewing the literature, we identified a knowledge gap regarding this specific population, both in sociodemographic and clinical terms. |
| 2) What were the most relevant results of the study? |
| Con nuestro estudio encontramos una alta prevalencia en presentación de efectos adversos del tratamiento, principalmente en los grupos de baja y mediana adherencia terapéutica. También observamos que las pacientes con condiciones sociodemográficas más favorecedoras mostraron una mejor adherencia terapéutica. También pudo confirmarse que el olvido es la principal causa por la cual las pacientes dejan de tomar el medicamento. |
| 3) What do these results contribute? |
| These findings provide valuable clinical and sociodemographic insights about this population and relevant evidence regarding behaviors and barriers that impact adherence to hormonal therapy. This knowledge is essential for designing and implementing strategies, interventions, or programs aimed at improving adherence to treatment. |
Introduction
The World Health Organization highlights that adherence involves the patient's active participation and cooperation in the treatment process. Adherence can be defined as the extent to which a patient follows the prescribed treatment regimen. It also notes that adherence is a dynamic phenomenon influenced by five interrelated dimensions, refuting the common notion that patients alone are responsible for taking their medication. Many factors influence individuals’ behavior and their capacity to adhere to treatment. These dimensions include socioeconomic factors, healthcare team-related factors, disease-related factors, treatment-related factors, and patient-related factors 1.
For decades, hormonal therapy has demonstrated significant benefits in the management of hormone-sensitive breast cancer, particularly its ability to significantly hinder the spread of malignant cells. However, specialized literature reveals an alarming lack of adherence to this type of therapy, directly associated with increased relapse rates, disease progression, and long-term mortality 2. A systematic review showed that women with breast cancer who adhere to hormonal therapy have a 5-year survival rate of 93.2%, compared to 75.9% to 83.4% among women who do not adhere 3.
Abandonment of hormonal therapy in this population has been documented to range from 31% to 73% within the first five years 4, which directly impacts mortality rates. According to Ministerio de Salud y Protección Social, breast cancer was the leading cause of death among women in Colombia in 2022, accounting for 4,725 deaths 4.
Although it is well recognized that multiple factors influence treatment adherence, there are no established strategies in Colombia specifically designed to address this issue. Furthermore, the available literature remains limited in characterizing or explains the determinants, behavioral patterns, or barriers impacting adherence within this population. It is necessary to generate scientific evidence that supports further research and the development of strategies to improve adherence and, consequently, survival rates. This study aims to identify sociodemographic, clinical, and pharmacological factors associated with adherence to hormonal therapy.
Materials and Methods
A cross-sectional study was conducted using retrospective data. This study included women aged ≥18 years who were alive, had been diagnosed with hormone receptor-positive breast cancer between 2019 and 2023, had been prescribed hormonal therapy for at least three months prior to providing oral agreement, and were treated at the Clínica de Oncología Astorga in Medellín, Colombia. Women with a second active primary tumor or those who had initiated treatment at another institution were excluded.
Sample size was calculated based on an expected adherence rate of 73.0%, as reported in the literature 3. Using a single population proportion formula, at least 178 patients were required for the study. A simple random probabilistic sampling was conducted using Epidat 4.2 and Microsoft Excel.
The dependent variable was adherence to hormonal therapy. The 8-item Morisky Medication Adherence Scale (MMAS-8) is used to assess adherence to treatment. It was administered via telephone to identify behaviors or barriers that interfered with adherence. Based on the score obtained, patients were categorized into high, medium, and low adherence levels 5. For this study, these levels were defined as follows:
High adherence: Patients were considered adherent, meaning they followed medical instructions and treatment consistently without behaviors that negatively impacted adherence.
Medium adherence: These patients generally followed medical instructions but occasionally engaged in behaviors that negatively affected adherence.
Low adherence: Patients were considered non-adherent, consistently failing to follow prescribed instructions, demonstrating minimal or partial adherence to treatment.
Collected variables included sociodemographic data (age, educational level, socioeconomic status, type of health insurance, marital status, etc.), clinical data (stage at diagnosis, histology, etc.), and pharmacological data (medication, adverse effects, etc.). In Colombia, socioeconomic status is officially categorized from level 1 to 6, with 1 being the lowest and 6 the highest. For analysis, strata 1 and 2 were grouped as low, 3 and 4 as medium, and 5 and 6 as high. Health insurance is classified according to the method of access to the healthcare system: Contributive Regime for those able to pay; Subsidized Regime for those covered by the State due to limited resources; Special Regime for certain population groups such as police or university staff; and Private Insurance for those opting into higher-tier services.
Additionally, a 13-item survey was developed by the research team to collect missing sociodemographic data not available in medical records. Medical charts were reviewed for the remaining data. Qualitative variables were summarized using relative frequencies and prevalence, presented in tables and bar charts. The quantitative variables did not follow normal distribution. Therefore, they were reported as medians and interquartile ranges. Associations between variables were assessed using Chi-square and Kruskal-Wallis tests. Statistical significance was defined as a p-value <0.05.
A multinomial logistic regression was conducted. The model selection was performed based on clinical plausibility and using criteria for statistical significance. Odds ratios (crude and adjusted) were used as epidemiological association measures.
The study was approved by the ethics committee of Universidad CES and the Clínica de Oncología Astorga. It was classified as minimal risk and did not require written informed consent. All participants had previously authorized access to their data and medical records through the institution’s research unit, in accordance with institutional data protection mechanisms. Verbal informed consent was obtained during the telephone survey. The study adhered to ethical principles for research involving human participants, national regulations, and good clinical practice guidelines.
Results
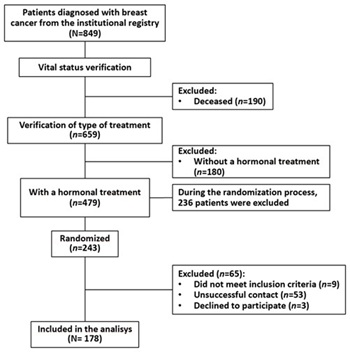
A total of 178 women were included (Figure 1), ranging in age from 33 to 92 years, with a median age of 64 years (IQR: 64.0-74.0). Over half were homemakers. A total of 56.7% had an educational level of high school or less, and 72.5% lived in the Valle de Aburrá, with 41.6% residing in the municipality of Medellín. About 49.4% were married or in a common-law marriage and most women belonged to socioeconomic strata 2 and 3. Approximately 48.3% lived with a partner and children or only with a partner, while 11.2% lived alone. The majority (70.2%) were affiliated with the contributory health regime. About 6.7% reported lacking a support network, whereas 92.1% indicated having one, primarily composed of family and friends (Table 1).
Table 1 Sociodemographic characteristics of breast cancer patients treated with hormonal therapy at the Clínica de Oncología Astorga in Medellín, Colombia.
| Variable | n (N=178) | % | |
|---|---|---|---|
| Age at the time of the survey - Median (IQR) | 64.0 years (55.0-74.0) | ||
| Occupation | Homemaker | 95 | 53.4 |
| Employed/Self-employed | 45 | 25.3 | |
| Retired | 38 | 21.4 | |
| Educational level | High school or less | 101 | 56.7 |
| Technical/Technological | 25 | 14.0 | |
| University/Postgraduate | 52 | 29.2 | |
| Municipality City of residence | Valle de Aburrá | 129 | 72.5 |
| Other* | 49 | 27.5 | |
| Socioeconomic status | Low | 66 | 37.1 |
| Middle | 77 | 42.1 | |
| High | 35 | 19.7 | |
| Affiliation type to the healthcare system | Subsidized | 5 | 2.8 |
| Contributive | 122 | 68.5 | |
| Special | 2 | 1.1 | |
| Prepaid | 49 | 27.5 | |
| Area of residence | Rural | 15 | 8.4 |
| Urban | 163 | 91.6 | |
| People the patient lives with | Lives alone | 20 | 11.2 |
| Partner with or without children or other† | 86 | 48.3 | |
| Children and/or other | 47 | 26.4 | |
| Siblings; other relatives; other | 25 | 14.0 | |
| Marital status | Single | 42 | 23.6 |
| Common-law/Married | 88 | 49.4 | |
| Divorced | 19 | 10.7 | |
| Widowed | 29 | 16.3 | |
| Type of support network | No support network | 12 | 6.7 |
| Formal support network | 2 | 1.1 | |
| Informal support network | 164 | 92.1 | |
*Other municipalities/cities of residence outside the Valle de Aburrá, such as Apartadó, Carmen de Viboral, Puerto Berrío, Rionegro, Chocó, Venecia, among others.
†Other refers to third- or fourth-degree relatives or individuals with no family ties
Most women (78.7%) had a diagnosis of infiltrating ductal carcinoma, and 77.5% were at stage I or II. A total of 168 women (94.4%) were on a treatment plan with curative intent. Half were prescribed tamoxifen, while nearly the other half (47.8%) received letrozole or anastrozole. Half began treatment within 117 days (IQR 61-175) of diagnosis. Of the total, 139 had between one and six comorbidities (median 2.0). Overweight and obesity were the most common (81.3%), followed by hypertension (64.8%) and dyslipidemia (33.1%) (Table 2).
Table 2 Clinical characteristics of breast cancer patients treated with hormonal therapy at the Clínica de Oncología Astorga in Medellín, Colombia.
| Variable | n (N=178) | % | |
|---|---|---|---|
| Histology | Ductal | 140 | 78.7 |
| Lobular | 20 | 11.2 | |
| Mixed | 2 | 1.1 | |
| NOS (Not Otherwise Specified) | 8 | 4.5 | |
| Other | 6 | 3.4 | |
| Unknown | 2 | 1.1 | |
| Treatment intent | Curative | 168 | 94.4 |
| Palliative | 10 | 5.6 | |
| Stage at diagnosis | I | 70 | 39.3 |
| II | 68 | 38.2 | |
| III | 34 | 19.1 | |
| IV | 5 | 2.81 | |
| Unknown | 1 | 0.6 | |
| Comorbidities | Yes | 139 | 78.1 |
| No | 38 | 21.4 | |
| Unknown | 1 | 0.6 | |
| n=139* | % | ||
| Arterial hypertension | 90 | 64.8 | |
| Dyslipidemia | 46 | 33.1 | |
| Diabetes/Prediabetes | 29 | 20.9 | |
| Thyroid gland disorders | 38 | 27.3 | |
| Overweight | 71 | 51.1 | |
| Obesity (Class I, II, III) | 42 | 30.2 | |
| Number of comorbidities - Median (IQR) | 2.00 (1.00 - 3.00) | ||
| Time (days) from diagnosis to start of treatment - Median (IQR) | 117 (61.00 - 175.90) | ||
| Medication | Tamoxifen | 89 | 50.0 |
| Letrozole | 51 | 28.7 | |
| Anastrozole | 34 | 19.1 | |
| Other† | 4 | 2.3 | |
| Years since diagnosis - Median (IQR) | 4.0 (3.0 - 5.0) | ||
| >Years on treatment- Median (RIQ) | 4.0 (3.0 - 5.0) | ||
* Patients reported having between 1 and 6 comorbidities; thus, each one may or may not be represented in the different categories of this variable.
†Fulvestrant y Exemestan.
More than 75.0% of those who had stopped taking their medication at least once cited forgetfulness as the main reason (Table 3).
Table 3 Reasons for discontinuing medication among breast cancer patients treated with hormonal therapy at the Astorga Oncology Clinic in Medellín, Colombia
| Variable | n (N=178) | % | |
|---|---|---|---|
| Has stopped taking the medication at least once | No | 94 | 52.8 |
| Yes | 84 | 47.2 | |
| Reasons | n=84* | % | |
| Forgetfulness | 64 | 76.2 | |
| Medical indication/Event | 9 | 10.7 | |
| Delay in delivery/Administrative issues | 21 | 25.0 | |
| Other | 9 | 10.7 | |
| Unknown | 1 | 1.2 | |
*The total number of reasons does not match n (84) because some patients reported more than one reason for discontinuing medication
Regarding adverse effects, 126 women reported experiencing some side effects after initiating hormonal therapy (Table 4). The most prevalent symptoms were hot flashes and fatigue. Sixty-one women also reported pain, dizziness, and skin dryness.
Table 4 Reported adverse effects of hormonal therapy in the study population
| Variable | n (N=178) | % | |
|---|---|---|---|
| Experiences adverse effects from treatment | No | 52 | 29.2 |
| Yes | 126 | 70.8 | |
| Category | Adverse effects | n=126* | % |
| Gastrointestinal disorders | Nausea | 18 | 14.3 |
| Vomiting | 7 | 5.5 | |
| Other | 5 | 4.0 | |
| Nervous system disorders | Headache | 6 | 4.8 |
| Dizziness | 9 | 7.1 | |
| Other | 10 | 7.9 | |
| Musculoskeletal disorders | Pain | 14 | 11.1 |
| Other | 5 | 4.0 | |
| Skin and subcutaneous tissue disorders | Skin dryness | 11 | 8.7 |
| Other | 3 | 2.4 | |
| General disorders | Fatigue | 50 | 39.7 |
| General discomfort | 5 | 4.0 | |
| Vascular disorders | Hot flashes | 82 | 65.1 |
| Worsening of varicose veins | 1 | 0.8 | |
| Other disorders† | Emotional disturbances | 4 | 3.2 |
| Changes in weight and appetite | 5 | 4.0 | |
| Ocular or vision changes | 5 | 4.0 | |
| Cough | 4 | 3.2 | |
| Vaginal dryness | 1 | 0.8 | |
| Number of specific adverse effects - Median (IQR) | 2 (0.0 - 3.0) | ||
* The “n” corresponds to the total number of women who reported experiencing symptoms. Therefore, the sum of frequencies does not reflect this total, as each participant reported a different number of symptoms.
† Other disorders include: respiratory, thoracic, and mediastinal disorders; psychiatric disorders; additional examinations; metabolic and nutritional disorders; reproductive system and breast disorders; and ocular disorders
The prevalence of high adherence was 51.1% (95% CI: 43.5-58.7), medium adherence was 34.3% (95% CI: 27.0-51.5), and low adherence was 14.6% (95% CI: 9.1-20.1).
Among the independent variables, age was the only one significantly associated with adherence (p= 0.034) (Annex A).
In the explanatory model, women over 56 years of age were found to be 4.9 times more likely to have high adherence compared to those under 44 years, and this difference was of statistical significance (p= 0.04, 95% CI: 1.08-31.55) (Table 5). Additionally, women from medium and high socioeconomic strata tended to show better adherence than those from low strata, although this difference was not statistically significant.
Table 5 Multivariate analysis (multinomial regression) including variables selected based on clinical plausibility and statistical significance (p ≤0.05).
| Adherence level | Predictor | p | Crude Odds Ratio | Adjusted Odds Ratio | 95% Confidence Interval | |
|---|---|---|---|---|---|---|
| Lower | Upper | |||||
| Medium vs. Low | Age | |||||
| 45-55 vs. ≤44* | 0.9 | 1.06 | 0.91 | 0.19 | 4.37 | |
| ≥56 vs. ≤44* | 0.98 | 1.56 | 1.02 | 0.23 | 4.53 | |
| Educational level | ||||||
| Technical/Technologist vs. High school or lower* | 0.55 | 1.56 | 1.7 | 0.3 | 9.66 | |
| University/Postgraduate vs. High school or lower* | 0.98 | 1 | 1.02 | 0.26 | 4.06 | |
| Socioeconomic status | ||||||
| Medium vs. Low* | 0.47 | 1.44 | 1.52 | 0.48 | 4.76 | |
| High vs. Low* | 0.87 | 0.96 | 1.15 | 0.21 | 6.18 | |
| Support network | ||||||
| Yes vs. No* | 0.74 | 1.86 | 1.32 | 0.25 | 6.94 | |
| Adverse effects since the start of treatment | ||||||
| No vs. Yes* | 0.09 | 2.72 | 2.77 | 0.86 | 8.97 | |
| High vs. Low | Age | |||||
| 45-55 vs. ≤44* | 0.45 | 2.28 | 2.02 | 0.33 | 12.25 | |
| ≥56 vs. ≤44* | 0.04 | 6.75 | 5.84 | 1.08 | 31.55 | |
| Educational level | ||||||
| Technical/Technologist vs. High school or lower* | 0.24 | 2.61 | 2.73 | 0.51 | 14.48 | |
| University/Postgraduate vs. High school or lower* | 0.84 | 1.06 | 0.87 | 0.23 | 3.34 | |
| Socioeconomic status | ||||||
| Medium vs. Low* | 0.18 | 2.49 | 2.14 | 0.71 | 6.46 | |
| High vs. Low* | 0.29 | 1.85 | 2.38 | 0.48 | 11.91 | |
| Support network | ||||||
| Yes vs. No* | 0.83 | 2.24 | 1.2 | 0.23 | 6.18 | |
| Adverse effects since the start of treatment | ||||||
| No vs. Yes* | 0.73 | 1.42 | 1.22 | 0.38 | 3.9 | |
*Reference value. ≥56: Greater than or equal to 56 years. ≤44: Less than or equal to 44 years
Women without adverse effects also showed better adherence than those who experienced side effects, though this difference was not statistically significant (OR= 1.22; 95% CI: 0.38-3.90; p= 0.73). Finally, women with a support network had a 20.0% to 30.0% higher likelihood of having greater adherence compared to those without such support (Table 5).
Discussion
This study identified potential sociodemographic, clinical, and pharmacological factors associated with adherence to hormonal therapy in women with breast cancer treated at the Clínica de Oncología Astorga in Medellín, Colombia.
The World Health Organization recognizes that adherence is a dynamic process not solely dependent on the patient and that it is shaped by various dimensions including socioeconomic status, healthcare team, disease characteristics, treatment, and the individual patient 1. This study corroborates that framework by identifying an association between adherence and age. Other factors associated with greater adherence —though not statistically significant— included high socioeconomic level, higher education level, and absence of treatment-related adverse effects. These trends suggest the presence of both facilitating and limiting factors for adherence.
The sample size in this study is comparable to other studies conducted in China and Mexico, which included younger participants 6,7. No previous studies were found characterizing the sociodemographic profile of this population in Colombia.
Compared to prior studies reporting high adherence rates between 60.0% and 80.0% 8-11, the adherence rate in this study was slightly over 50.0%. However, when adherence was stratified into three levels using MMAS-8, our findings aligned with similar prevalence reported in other studies 6,7,12.
The medium adherence group was the second most prevalent and was characterized by a high frequency of adverse effects, low socioeconomic and educational levels, and forgetfulness as the main reason for non-adherence. These findings closely resemble those in the low adherence group, indicating a potential risk profile that could be used for early identification of patients at risk of non-adherence.
Age was significantly associated with adherence. Consistent with Brito’s study, women over 56 years of age had higher adherence compared to younger women 10. Regarding treatment-related adverse effects, Villareal et al. found a significant association between these symptoms and incomplete adherence to endocrine therapy 13. This supports our findings, particularly the high prevalence of side effects among patients in the medium and low adherence groups. Younger women showed the highest prevalence of adverse effects, possibly due to menopausal symptoms that are harder to manage at younger ages, thereby contributing to poor adherence 14. This suggests that adverse effects may represent a significant barrier to adherence, supporting findings from other studies linking side effects with incomplete adherence 12,15.
Interestingly, women with comorbidities had a higher prevalence of high adherence (53.0%), which aligns with Liu’s findings that the presence of at least one comorbidity increased the likelihood of adherence 16. Patients with multiple health conditions may be more aware of the consequences of non-adherence, leading to better compliance 17.
Socioeconomic status also showed a trend toward better adherence among women from medium and high strata. This is consistent with other studies indicating that low socioeconomic status is associated with poor adherence and worse treatment outcomes 11,18. Similarly, education level followed this trend, with better adherence among those with an education beyond high school. This may be explained by greater understanding of disease and treatment among more educated individuals. However, other social determinants —such as household workload, financial stress, and access barriers— could indirectly impact adherence but were beyond the scope of this study.
This study also suggests that having a support network positively impacts adherence. Prior studies confirm that lack of support increases the risk of poor adherence, while the presence of support networks is associated with better compliance 12,15,19. This may be because support systems provide emotional reinforcement, resilience tools, and help in understanding treatment instructions and disease management.
Another key finding was that forgetfulness was the most common reason for discontinuing medication in both the low and medium adherence groups. This is in line with findings from other studies, which also identified forgetfulness as the leading cause of non-adherence 7,8. Notably, patients in the high adherence group did not report forgetfulness as a reason for missed doses.
This study’s main limitations include the sample size, which, while homogeneous, limited the ability to detect significant associations in subgroups with imbalanced frequencies. Additionally, the cross-sectional design and single time-point measurement did not capture longitudinal changes in adherence. Telephone administration of the questionnaire and use of a single measurement tool could have led to over- or underestimation of adherence levels. Moreover, the underrepresentation of patients from the subsidized health regime limits the generalizability of findings to that population. Despite these limitations, the study provides reliable and valuable insights into factors affecting adherence in a population not previously described in this context.
Conclusion
This study provides previously unavailable prevalence data in Colombia and helps identify potentially modifiable factors that may positively influence adherence to hormonal therapy. It also reveals that younger patients have lower adherence. Other factors, such as low socioeconomic status, low educational level, absence of a support network, presence of disease-related symptoms, and forgetfulness -although not statistically significant- were highly prevalent among patients with low adherence.
Future studies should explore strategies to improve adherence among younger women, including close monitoring and direct contact with pharmacovigilance teams. Efforts to reduce forgetfulness, such as mobile phone alarms or pill organizers, should also be evaluated for their effectiveness in improving adherence.











 text in
text in