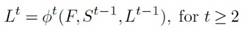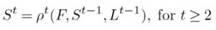1. Introduction
Parkinson’s Disease (PD) is the second most common neuro degenerative disorder, affecting more than 6.2 million people worldwide (Dorsey et al., 2018, Feigin et al., 2021). Moreover, there has been a significant increase in prevalence over the last three decades, reaching up to 5 times people suffering PD (Tolosa et al., 2021). Currently, the diagnosis of PD is based on the observation and analysis of progressive gait motor disorders, such as rigidity, slowness of movement (bradykinesia), postural instability, among many other related symptoms (Rovini et al., 2017).
Nowadays, standard support for gait analysis characterization is based on marker-based systems, which capture dynamics of key joints by using invasive methodologies based on special markers placed on specific anatomical positions (Baker, 2006). This methodology is nonethe less invasive and alters the natural gesture of movements, which for Parkinson disease can include limitations on the normal development of locomotion. Besides, some of these protocols are strongly dedicated to capture lower-limb kinematics, losing important markers of PD, such as postural instability and coordination.
In the literature, these limitations have been tackled from video analysis alternatives that include markerless setups, achieving remarkable results on the characterization of Parkinson movements. Much of these strategies are based on the training and modeling of video descrip tors to classify and differentiate Parkinson’s from other motions (Lancet, 2017). A main limitation on these approaches is the poor adaptation in the clinical context, offering alternatives that are difficult to implement in ob servational setups. In fact, much of the support of these strategies are based on probability scores about malignancy, but losing regional information of affected regions. Hence, many of these strategies may biased for artifacts in the sequences, losing relevance to characterize anatomical and physiological during a locomotion process.
This work introduces a convolutional network that learns spatio-temporal patterns from intermediate postural representations. The proposed approach is based on markerless setups, avoiding additional artifacts to al ter the patient’s gestures. This work starts by adapting an OpenPose architecture to return the bank of intermediate activations related to knowledge about joint fields and probability joint maps. Later, this intermediate representation is projected to a convolutional network, which is there after minimized to discriminate between control and parkinsonian patterns. The results evidence sufficient support to characterize Parkinson from classification scores, but also the capability to explain results from postural information.
2. Proposed Approach
This work introduces a computational strategy for characterizing motor patterns associated with Parkinson’s disease based on joint interest points calculated without the use of markers. Inspired in OpenPose(Cao et al., 2021), we generate the poses and identify key body points from video sequences. Then a spatio-temporal convolutional network is trained to discriminate these key points, regarding if the patient is control or Parkinson. This network was trained and adjusted from intermediate pose representations: the Joint Confidence Maps (JCM) and Part Affinity Fields (PAF) elements. The general pipeline of the proposed approach is illustrated in Figure 1.
2.1 A Deep Architecture for Pose Estimation (OpenPose)
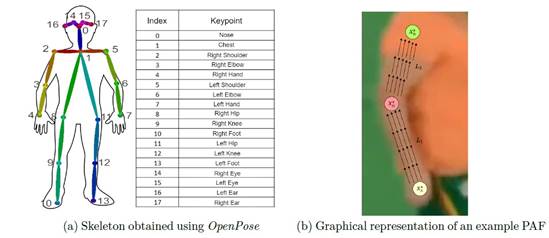
A main contribution of this work is to use markerless setups to avoid the limitation of marker-based configurations. Hence, OpenPose(Cao et al., 2021) architecture was used as a pose estimator from gait videos to extract intermediate features. This network is widely used in the literature for estimating key joint points during movement and actions for one or several persons in a scene. Formally, OpenPose architecture use an image IG R wxh where a posture will be extracted (P E N J and J E {ji, j-2, • • •, jn} represents the set of n body joints). This network use only 18 articular points (| J\ = 18), as showed in the Figure 2a. These joint points allow summarize the dynamics of a particular subject during locomotion. Specifically, each input image I is processed through convolutional layers ’J'(I) to obtain a set of deep activations F, which are further processed through two branches: Part Affinity Fields (L) and Joint Confidence Maps (S). These maps and vector fields are processed through bipartite matching resulting in the association of body joints with articular locations giving us the skele ton shown in Figure 2a.
2.2 Part Affinity Fields
The Part Affinity Fields (PAFs) are sets of 2D vector maps used to model spatial and anatomical relationships between pairs of body joints. They are formally described as a set L = {L¡} c , where L¡ E R wxhx2 j and C is a hyperparameter determining the number of PAFs to be found. Each pixel within the PAF contains a vector representing the direction and strength of the connec tion between the corresponding joint pair. The vector’s direction indicates the orientation of the connection, and the vector’s magnitude represents the confidence in that connection, as illustrated in Figure 2b.
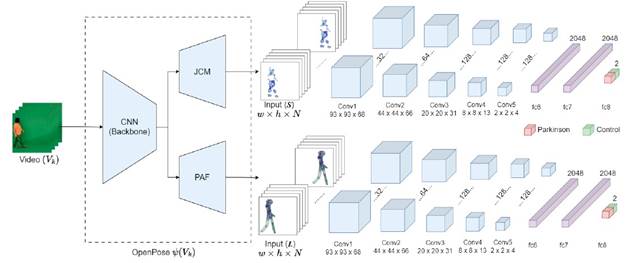
Figure 1 Proposed Architecture for Generating OpenPose Activations, Featuring a 3D Convolutional Net-Work for Patient Classification Based on Gait
Formally, OpenPose generates a set of PAF , represented as Lt =1 = ÿt =1 (F), where fit =1 refers to the convolutional layers used for PAF calculation at t = 1. For each subsequent refinement stage, the predicted PAF from the previous stage, the original features F , and a set of joint confidence maps (St-1) are combined and used to generate refined predictions:
This approach allows progressive refinement of the P AF, contributing to the process of detecting and associ ating various body parts. In Figure 3, you can observe the resulting activations of these fields, which are potential intermediate representations of the locomotion process, including predominant directions during locomotion.
2.3 Joint Confidence Maps
In a parallel branch of processing, another bank of con volutional filters serves as input to generate a probabilistic representation of poses. Specifically, a Joint Confidence Map (JCM) is a two-dimensional representation that reflects the probability of a specific body part be ing located at a particular pixel. They are formally described as a set S = {Sj} J , with Sj € R wxh , where J € {j 1, j2,... ,j n } is the number of body parts (joints). Each pixel in Sj contains a value representing the probability that joint j is located at that position in the image. In other words, the JCM s indicate how confident the model is that a specific joint is located at each pixel of the image.

Figure 3 Visualization of all Part Affinity Fields (PAF and all Joint Confidence Maps (JCMs) Obtained during the Gait of a Control Subject
Similar to the generation of PAF s, the network generates a set of JCMs, represented as St=1 = pt=1(F), where pt =1 refers to the convolutional module used for map calculation at t = 1. At each subsequent refinement stage, the JCMs and PAFs from the previous stage, along with the features F, are combined to generate refined predictions (see Figure 3):
2.4 3D Convolutional Network for Classifying Parkin sonian Patterns
Once the OpenPose architecture is fine-tuned with videos of both control subjects and Parkinson’s patients, intermediate representations can be obtained: the PAF (L) and the Joint Confidence Maps (S). These intermediate representations are activations from OpenPose that contain relevant information about kinematics during locomotion (L) and joint importance in each frame (S).
In this work, we use these intermediate deep representations (L and S) for characterizing spatio-temporal motor patterns related to Parkinson’s disease. To achieve this, a 3D convolutional network was designed and tuned, allowing for the learning of deep relationships while considering the volumetric nature of the information. The 3D convolutional architecture specializes in extracting spatiotemporal features from complete videos, capturing long-term temporal patterns in sequential data. This is crucial in the analysis of parkinsonian gait, as several cardinal symptoms of Parkinson’s disease require pro longed observation throughout the gait cycle for accurate identification. In fact, such volumetric representations with 3D convolutions have been proposed in the literature for video analysis in various tasks, such as action recognition (Varol et al., 2017). In this work, the PAFs L were obtained for each frame In of gait video during the last refinement stage (t = T), i.e., L = {L T (In)} N , where N corresponds to the total number of video frames. Then, the set of PAFs for all frames, denoted as L, was fed into the 3D convolutional architecture. This architecture incorporates spatiotempo ral convolutions to identify different patterns that may occur during a gait process, determining the probabil ity of whether these patterns correspond to a Parkinson’s patient or a control subject, as shown in Figure 1. Formally, the network’s operation can be expressed as P (Parkinsons| L) = 1 - P (Control| L) = ^(L), where ^ represents the set of kernels, layers, functions, and operations that make up the model.
It is worth noting that the intermediate representa tion of the PAF s contains directional information about the positioning of the joints. Therefore, through 3D convolutions, the architecture is expected to learn kinematics with greater discriminatory power between control subjects and parkinsonian patterns. On the other hand, from Joint Confidence Maps, it is expected to learn co herence between structural activations, which can also be discriminative.
3. Experimental Setup
3.1 Database Description
The database used in this work consists of a series of markerless RGB videos captured during a locomotion exercise. In this study, 30 subjects were invited to participate, including 16 control subjects and 14 who had been diagnosed with Parkinson’s disease (PD). The PD patients were in stages of the disease ranging from 1.0 to 4.0 on the Hoehn and Yahr scale. In total, 8 patients were diagnosed with a score of less than or equal to 2.5, and 6 patients scored between 2.5 and 4.0. Each subject in the study was recorded on eight occasions while performing markerless natural walking, four times to the left and four times to the right, resulting in a total of 240 video sequences. This dataset is balanced by age, with an average age of 70.4±5.38 years for control patients and 73±7.45 years for PD patients. All videos were recorded indoors, with a static camera and a uniform background color. The average duration of the videos is 2 seconds. All participants provided informed consent and the research was approved by the ethics committee of the Industrial University of Santander.
3.2 Proposed Method Setup
From each video, we selected N = 70 intermediate frames (to cover approximately one gait cycle). Each video record ensures a complete gait cycle that fully exposes the kinematics during locomotion. The videos were resized to a size of 95 x 95 pixels (w x h). Each frame was individually passed to the OpenPose network for pose estimation. The VGG19 net was used to compute convolutional features (Simonyan and Zisserman, 2014). These deep features were then used to generate PAF s and JCMs, with the number of stages t set to 6.
The proposed convolutional architecture for classifying parkinsonian patterns from PAF and JCM sequences was fine-tuned considering different convolutional and embedding levels. In this particular work, the following configurations were validated: 5 Conv3D 3 dense layers, 5 Conv3D 1 dense layer, 3 Conv3D 3 dense layers, and 3 Conv3D 1 dense layer. For our models, we used 10 training epochs, a learning rate of 1 x 10 -4 , an Adam op timizer, and a cross-entropy loss function. To evaluate each of the configurations, a leave-one-patient-out cross validation scheme was followed, in which a model was trained for each patient, with the other samples used for model training. Additionally, classification metrics such as accuracy, precision, sensitivity, F2-score, and the area under the curve (AU C ) were used for validation.
4. Evaluation and Results
To validate the capability of intermediate pose represen tations, we projected the PAF and JCM activations ex tracted from the OpenPose architecture. This block of activations was fed into a volumetric convolutional archi tecture to learn discriminative representations between Parkinson’s disease and a control population. Simultane ously, we assessed the convolutional representation that yielded the best performance in the discrimination task, determining the various 3D convolutional layers and as sociated embedded vectors. Figure 4 shows the results obtained for the entire set of 22 patients, with accuracy as the measurement basis for the classification process.
As observed in Figure 4, the intermediate PAF rep resentation yields consistent and robust results across different configurations of the trained architecture. PAF vector maps can influence the pose orientations, which may serve as a characteristic pattern allowing the net work to discriminate between the two study populations. On the other hand, the J CM representation exhibits a limited performance in the architecture with 3 dense layers. This could be attributed to the limited train ing data, particularly in the case of JCM maps, which provide reduced information regarding attention maps around the joints.
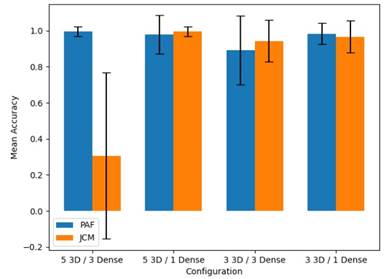
Figure 4 Comparison of Patient Classification Accuracy for Control and Parkinson’s Disease using Different Configurations, Using PAF vs JCM as Inputs
In a second experiment, we conducted a compari son with a state-of-the-art proposal that advocates volumetric representation but uses projections from raw videos or maps the response of an optical flow algorithm (Guayacán & Martinez, 2021). Table 1 summarizes the results obtained by the state-of-the-art method, employ ing an architecture with similar characteristics (3D con volutions) on both RGB and flow sequences (OF). We also included the projections using intermediate repre sentations based on PAF and JCM maps.
Table 1 Comparison of Classification Metrics for Control and Parkinson’s Patients between our Proposal and the State of the Art
| Method | Accuracy | Precision | Sensitivity | F2-Score | AUC |
|---|---|---|---|---|---|
| Guayacán (FOB) | .949 | .910 | 1.0 | .780 | .950 |
| Guayacán (OF) | .847 | .870 | .780 | .700 | .910 |
| Ours (PAF) | .994 | 1.0 | .989 | .991 | .999 |
| Ours (J CM} | .994 | .989 | 1.0 | .997 | 1.0 |
As observed, in general, all the projections exhibit a notable performance in classification metrics. This re sult could be attributed to the limited dataset or the stages of Parkinson’s patients within the population. It is worth noting that the intermediate projections pro vide a better representation of the information, correctly classifying the samples from the mapped videos (achiev ing perfect precision, sensitivity, and AUC in one of the two configurations). Furthermore, the reported AUG for the intermediate maps not only makes it robust for binary classification but also demonstrates a marked class separation. This can be crucial when extending the analysis to more comprehensive studies with addi tional cases. Additionally, these maps can offer greater explanatory power, breaking down the kinematic infor mation into postural components.
5. Conclusions and Perspectives
This work introduced a novel markerless strategy to characterize spatiotemporal parkinsonian patterns from pose intermediate representations. In this work, firstly an Openpose architecture is tuned to learn locomotion from Parkinson disease and control subjects. From such pose generator is taken the intermedia bank of activa tions related to probability maps of joints and vector file maps of pose structure. These intermediate representa tions are mapped to a 3D convolutional net, adjusted to learn discriminative patterns among two considered populations. The results showed a high capacity in this task, with these indices being potential indicators of ab normalities associated with the disease during locomo tion tasks. Future works include the analysis on extra datasets with a larger cohort of patients with different degrees of the disease.