INTRODUCTION
Neurocysticercosis (NCC) is the most frequent helminth infection of the central nervous system and the main cause of acquired epilepsy worldwide (30%). 1-3 NCC occurs when an individual ingests the eggs of the Taenia solium tapeworm (carried by pigs), previously expelled in the feces of another individual with teniosis. 4
Currently, there are 50 million people affected by NCC around the world, which makes it an endemic disease in Colombia and other Latin American countries. 5 In 2010, the World Health Organization (WHO) listed NCC as an unattended zoonosis in permanent expansion, thus turning it into a serious public health issue in developed countries due to migratory phenomena. 3,6
Flórez-Sánchez et al.7 found that the prevalence of seropositivity for cysticercosis varies in the Colombian population (0.5340.19%) and that the Vaupés department has the highest seroprevalence rates.
The objective of this article is to promote knowledge about the heterogeneous manifestations of neuroinfection by T. solium and highlight the importance of the prevention measures against the disease.
CASE PRESENTATION
A 51-year-old male patient from the rural area of Caldono (Cauca, Colombia), farmer, of low socioeconomic status and with basic primary education was admitted to a hospital in Popayán in November 2015, describing a single NCC event successfully treated with albendazole 2 years before. The reason for consultation was the impossibility of walking by himself. During anamnesis, his relatives reported frequent consumption of undercooked pork, lack of sewage service and lack of knowledge of proper hand washing by the patient.
The patient presented with a clinical picture of 8 months of evolution consisting of progressive gait impairment, loss of sphincter control, left hemiparesis and headache. On admission, he did not obey orders, presented dromomania, inappropriate language, dysarthria, cognitive deterioration, space-time disorientation, verbal-motor automatisms (he constantly said "I am alone"), pathological palmomental reflexes and bilateral prehension.
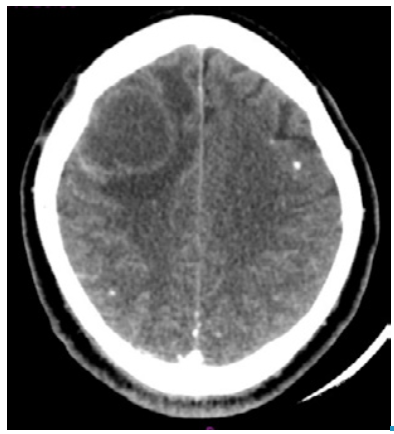
The neurology service requested a computed tomography (CT) that revealed a dilation of the supratentorial ventricular system and a right frontal subcortical cystic lesion that created a mass effect with midline shift. In addition, he presented perilesional edema and small residual bilateral frontoparietal calcifications, suggesting sequelae of NCC (Figure 1). Blood count, C-reactive protein (CRP) and renal function were normal. Pharmacological management was initiated with albendazole at an oral dose of 1600mg every 24 hours, dexamethasone 8mg IV every 8 hours, paracetamol at an oral dose of 1g every 8 hours and omeprazole at an oral dose of 20mg every 24 hours. The patient did not report any side effect caused by these drugs.
Source: Own elaboration based on the data obtained in the study.
Figure 1 Computed tomography with right frontal subcortical cystic lesion, perilesional edema and calcified nodules.
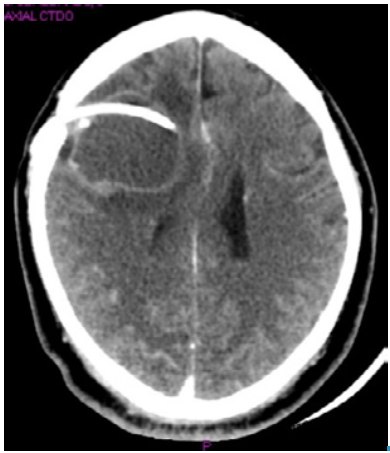
A week after admission, the patient presented with left hemiplegia and sialorrhea, and he did not have any verbal response. A craniotomy was performed to remove the cyst and conduct a histopathological study, while a ventriculoperitoneal shunt was arranged to reduce intracranial pressure (Figure 2). Macroscopically, neurosurgery reported a frontal cyst of greenish content with walls strongly adhered to the parenchyma and the frontal horn of the lateral ventricle. Histopathological analysis confirmed the suspicion of NCC and reported reactive gliosis. The patient evolved satisfactorily, did not present any type of sequelae and was discharged.
Source: Own elaboration based on the data obtained in the study.
Figure 2 Computed tomography with right frontal subcortical cystic lesion, midline shift and ventriculoperitoneal shunt.
Reinfection was suspected due to a previous history of NCC (a significant risk factor), the presence of calcified nodules in the imaging and regional epidemiology.
DISCUSSION
The patient presented with a frontal syndrome characterized by left hemiparesis, disobedience of orders, dromomania, cognitive impairment, space-time disorientation and verbal-motor automatism, which are related to cysticercosis cysts in the right frontal lobe. According to anatomical distribution, 60-90% of cysticerci are located in the cerebral parenchyma 8, being less frequent in the ventricles, subarachnoid space, eyes, meninges and spinal cord. 9
When the T. solium larva is viable, initial neurological symptoms are caused by compression, in most cases. When the cysticercus dies, intense inflammation with exudate, peri-arteritis and endarteritis is usually observed, which can close the vascular lumen and impede the normal flow of cerebrospinal fluid, favoring the presence of hydrocephalus and intracranial hypertension. 10 Following the inflammatory response, the cysticercus usually reaches its final stage (nodular calcified), which has been associated with epilepsy. 10 However, the patient reported in this article did not develop hydrocephalus or epilepsy during his stay in the service.
Reinfection by T. solium in this patient was suspected due to the presence of calcified nodules and perilesional edema 11 -compatible with active cysticercosis- observed on the CT scan and based on the information provided by his relatives during anamnesis: history of NCC 2 years before, frequent consumption of undercooked pork, lack of drinking water and sewage services and lack of proper hand washing habits. 12,13 The patient and his relatives were given the pertinent recommendations and measures to prevent the disease: correct hand washing (soap and water or glycerinated alcohol) before and after consuming food and after contact with another person 4; adequate disposal of feces in the rural area; exclusive consumption of drinking water; environmental sanitation, and management of pigs under the current regulations. 9,14
The signs and symptoms of NCC depend on the location, number, dimensions, cysticercus stage (vesicular, colloidal, granular-nodular and calcified nodule), genotype and immune status of the host. 4 Headaches and seizures are the most frequent clinical manifestations; visual or psychiatric disorders, hydrocephalus and meningitis may also occur. 3,15
The prevalence of NCC is higher in rural areas, where people work with pigs and sanitary conditions are often deficient. 3,5,16 This disease compromises the health of those affected, the livelihood of agricultural communities and their economy due to absenteeism, increase in health costs and community stigmatization. 3,16 According to Flórez-Sánchez et al.7, the prevalence of cysticercosis in Colombia ranges between 0.53% and 40.19% in Caldas and Vaupés, respectively. In Cauca, Vásquez-Arteaga et al.14 found a seroprevalence of 55.2% in patients with neurological symptoms of five municipal hospitals. This information is very useful for the region, since the history of NCC and the neurological manifestations compatible with the disease make it necessary to discard it.
In southwestern Colombia, cysticercus is known as "granalla", "granizo", "pepa" or "pepita", and is considered as a part of the pig. Actually, the inhabitants of this region think that they add a taste to the meat. In addition, knowledge on the life cycle of the parasite is deficient, which leads to difficulties when making promotion and prevention campaigns. 17 Understanding the life cycle and behavior of the teniasis/ cysticercosis complex (TCC) is important when considering implementing public health policies and promotion and prevention campaigns.
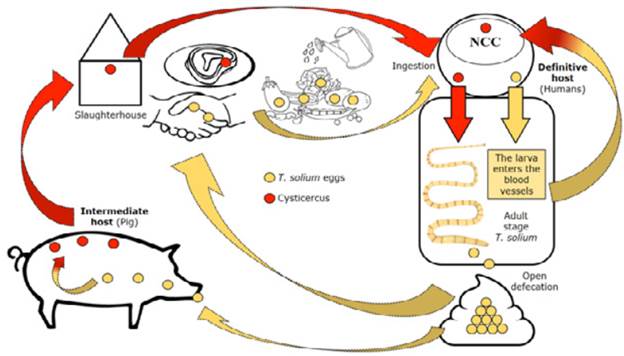
Depending on the development stage of T. solium, if the cysticercus or eggs are swallowed, teniosis or cysticercosis, respectively, will occur (Figure 3). It should be noted that both diseases could occur simultaneously in the same individual. 9
Source: Own elaboration based on the data obtained in the study.
Figure 3 Life cycle of the teniasis/cysticercosis complex.
As seen in Figure 3, humans develop teniosis when cysticerci are ingested. The larvae mature to the adult form of T. solium and release the eggs to the environment; then, pigs consume them and suffer from cysticercosis. When animals are slaughtered, if there are deficiencies in sanitary control, pork meat is commercialized and humans end up consuming cysticerci and developing teniosis. Similarly, the definitive host may present with cysticercosis when consuming food irrigated with water contaminated by the eggs of the adult tapeworm. Contact with sick individuals is an important way of contagion, being the main risk factor for TCC infection. 18
This case shows strength in diagnosis, epidemiology and clinical foundation. The importance of anamnesis should be highlighted, since it provides a guide for patient's approach. In the same way, intervention for promotion and prevention is highlighted as relevant. However, this case did not include a molecular test that identified IgM antibodies for T. Solium due to local limitations.
CONCLUSIONS
Making a timely diagnosis along the process (medical history, imaging and laboratory tests) is important when the history, signs and symptoms are compatible with NCC.
Providing comprehensive management to the patient, in this type of cases, is necessary, first, to carry out a complete cysticidal treatment and, second, to provide information to patients, relatives and the community in general about the prevention measures against NCC.














