Artículo de investigación original
Biochemical Approach of Acid-Base Disturbances: Diagnosis Algorithm
Algoritmo diagnóstico para el abordaje bioquímico de trastornos ácido-base
1DiabetCentro Institute, Guatemala City, Guatemala
Abstract
Context:
The biochemical approach to acid-base balance classifies disturbances in two categories based on cellular mechanisms. The first one being cellular respiration (ATP), which presents high anion gap and changes in carbon dioxide levels, and the second one, cellular metabolism (ion transport through membrane channels), which evidences normal anion gap and changes in other cations and anions.
Objective:
To present a new diagnostic algorithm for acid-base disturbances, based on the biochemical approach.
Method:
Original research with systematic analysis and data organization, following biochemical processes that affect acid-base balance.
Results:
A simple, three-step algorithm which classifies information in a diagnostic table that connects blood gas analysis results (pH, anion gap or base excess, bicarbonate, carbon dioxide and unmeasured anions) with the biochemical cause and most probable clinical findings.
Conclusions:
The biochemical approach is considered the new model to understand, explain and diagnose acid-base balance and disturbances. All high anion gap disturbances occur in cellular respiration, while all normal anion gap disturbances occur in membrane channel function. The diagnosis algorithm simplifies and organizes the information to describe medical conditions with data from a blood gas analysis. This approach is the first model to establish a linear correlation between lab results, biochemical cause, and clinical findings of acid-base disturbances.
Keywords: Acid-base balance; Acid-base disturbance; Metabolic acidosis; Metabolic alkalosis; Respiratory acidosis; Respiratory alkalosis; Urea cycle disorders; Salicylate poisoning; Respiratory chain; ATP; Renal tubular acidosis; Hyperkalemia; Hypercapnia; Hypocapnia; Critical care; Emergency; Point of care; Biochemical approach
Resumen
Contexto:
El abordaje bioquímico de trastornos ácido-base clasifica las alteraciones en dos categorías basándose en mecanismos celulares. La primera categoría es la respiración celular (ATP), que presenta una brecha aniónica elevada y cambios en niveles de dióxido de carbono. La segunda, es el metabolismo celular (transporte de iones a través de canales de membrana), el cual presenta una brecha aniónica normal con cambios en otros cationes y aniones.
Objetivo:
Presentar un nuevo algoritmo diagnóstico para trastornos ácido-base con base en el abordaje bioquímico.
Método:
Investigación original con análisis sistemático y organización de datos siguiendo procesos bioquímicos que afectan el balance ácido-base.
Resultados:
Un algoritmo simple de tres pasos que clasifica la información en una tabla diagnóstica que conecta los resultados de gasometrías sanguíneas (pH, brecha aniónica o exceso de base, bicarbonato, dióxido de carbono y aniones no medidos) con la causa bioquímica y hallazgos clínicos más probables.
Conclusiones:
El abordaje bioquímico es el nuevo modelo para entender, explicar y diagnosticar el equilibrio ácido base y sus trastornos. Todas las alteraciones que presentan brecha aniónica elevada se originan en la respiración celular, mientras que todas las que presentan brecha aniónica normal se originan en canales de membrana. El algoritmo diagnóstico simplifica y organiza la información para describir condiciones médicas desde la información de una gasometría sanguínea. El abordaje bioquímico es el primer modelo que conecta los resultados gasométricos con la causa bioquímica y los hallazgos clínicos más probables de los trastornos ácido-base.
Palabras clave: Equilibrio ácido-base; Trastorno ácido-base; acidosis metabólica; alcalosis metabólica; acidosis respiratoria; alcalosis respiratoria; trastorno del ciclo de la urea; intoxicación por salicilato; cadena respiratoria; ATP; acidosis tubular renal; hiperkalemia; hipercapnia; hipocapnia; cuidados críticos; punto de cuidado; abordaje bioquímico
Introduction
All acid-base disorders with high anion gap relate to disturbances in cellular respiratory processes such as glycolysis, citric acid (Krebs) cycle, and respiratory chain in the inner mitochondrial membrane. Conversely, acid-base disorders with normal to low anion gap are related to disturbances in ion concentration (cations and anions) and are generally caused by changes in membrane channel function, ion gradients, and passive or active transport mechanisms. Some of these disorders are congenital and some are iatrogenic in nature [1].
Methodology
Original research article with systematic analysis and classification of data following a pattern based on biochemical processes that affect acid-base balance.
How to use the diagnostic algorithm (Table 1)
Step 1: Determine pH.
• <7.38 = acidosis
• >7.42 = alkalosis
Step 2: Determine anion gap (AG)/base excess (BE)
• High AG (Low BE) means disturbance in cellular respiration.
o Conditions that affect oxygen supply or activate anaerobic respiration lead to elevated lactate with low CO2
о Conditions that affect substrates for citric acid cycle lower the production of CO2, leading to low CO2 and low HCO3-
о Conditions that affect the mitochondrial respiratory chain lower the production of H2 O, leading to high CO2 (hypercapnia) and low HCO3-
о In severe MRC blockage, cells resort to anaerobic respiration, leading to elevated CO2, low HCO3- and elevated lactate. Therefore, hyperlactatemia with hypercapnia indicates mitochondrial respiratory chain dysfunction.
• Normal AG (neutral BE) means disturbance in ion transport across membranes
о Most cases affect sodium, potassium and chloride levels.
о If lactate is elevated, consider hypovolemia, and refer to step 2.1 high AG [2].
Step 3: Determine the byproduct or metabolite.
Past and recent medical history, clinical examination, and biochemical analysis play an important role. Metabolites analyzed with CO2 and HCO3- levels will explain the underlying process.
• Lactate
Low CO2: tissular hypoxia - ventilation: alveolo-capillary membrane, cardiac output, hemoglobin alterations, hypovolemia, ischemia
о High CO2: mitochondrial respiratory chain blockage - uncouplers: alcohols, poisons, anesthetics
• Ketones - citric acid cycle with fatty acids instead of pyruvate - diabetic ketoacidosis, starvation, infections, alcohol abuse.
• Organic anions - citric acid cycle with amino acids instead of pyruvate - inborn errors of metabolism, severe catabolic state.
• Nitrogen derivates: Urea, ammonium - urea cycle disorders and generally related to citric acid cycle blockage in the alpha-ketoglutarate dehydrogenase enzyme.
• Salicylates - citric acid cycle blockage in succinic acid dehydrogenase and alpha-ketoglutarate dehydrogenase enzymes, coupled with ketoacid production [3].
• Hypercapnia (high CO2) - buildup from lack of H2O production in respiratory chain due to blockers or uncouplers (anesthetics, metformin, remdesivir, organophosphates, coenzyme deficiency, etc.) [4].
Once the disturbance is identified, proper treatment can be initiated [5].
Some anions, such as organic anions and respiratory chain blockers, are not routinely measured but can be inferred and then ordered to confirm and establish the correct diagnosis.
Discussion
Alterations in cellular respiratory (i.e. ATP) processes yield certain byproducts in weak acid form, which later dissociate, releasing hydronium ions and an anion [6]. Hydronium ions are responsible for changes in pH and cellular function (depolarization or hyperpolarization), leading to clinical manifestations. The dissociated anions elevate the anion gap, and their identification can help clinicians determine which step of the process is altered.
Most disturbances that present with high anion gap manifest as acidosis (pH <7.38). However, there are two exceptions: Urea cycle disorders and salicylate toxicity, which manifest as alkalosis with high anion gap. The most plausible explanation is that the byproducts of urea cycle and salicylates have a very high pH (9.8 and 13, respectively). Their buildup in plasma exerts an alkalinizing effect on serum pH (alkalemia) and masks the real acidosis occurring due to the disturbance of cellular respiration [7].
Table 1 Diagnostic Algorithm for Biochemical Approach of Acid-Base Disturbances
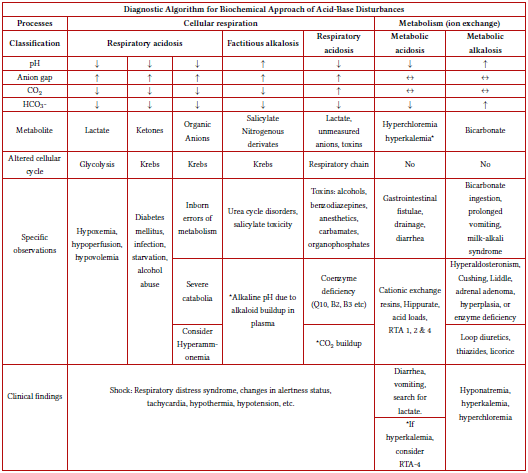
Source: Author's elaboration with information provided in the text.
Under ideal conditions, the carbon:hydrogen:oxygen ratio and the amount of released CO2 and H2O remain constant [8]. This is known as the respiratory quotient (Q). The body maintains constant equilibrium between CO2 and HCO3- levels, as HCO3- formation depends on that ratio. HCO3- levels are a key component of base excess determination. Since changes in substrates or enzymatic activity of cellular respiration that alter the anion gap will also alter HCO3- levels, it is logical to consider anion gap and base excess as mirror parameters of severity. The higher anion gap, the lower (more negative) the base excess. This is very useful in certain settings where the blood gas analyzer lacks the capacity to measure cations for the AG formula.
All disturbances in cellular respiration lower the respiratory quotient, resulting in increased oxygen demand, which is expressed clinically as tachypnea. It is also logical to consider tachypnea as a response mechanism to maintain adequate oxygen supply to ensure ATP production, rather than to eliminate CO2 to "compensate" the "metabolic acidosis", as previous models state [9-11].
Conclusion
The biochemical approach is the new model to understand, explain and diagnose acid-base balance and disturbances. All high anion gap disturbances occur in cellular respiration, while all normal anion gap disturbances occur in membrane channel function. The diagnosis algorithm simplifies and organizes information to describe medical conditions with data from a blood gas analysis. The biochemical approach is the first model to establish a linear correlation between lab results, biochemical cause, and clinical findings of acid-base disturbances.
Acknowledgements
To Mario Alejandro Nitsch-Montiel, Fabiola Prado-Barragan, Juan Andrés Nitsch-Prado, and Deborah Abigail Nitsch-Prado for their continuous support. To the staff and Editor in Chief at Revista Colombiana de Nefrología for considering, reviewing, and accepting this manuscript. "Let everything that has breath praise the LORD" - Psalm 150:6
References
[1] Nitsch-Prado A. Biochemical Approach to Acid-Base Disturbances. Rev. Colomb. Nefrol. 2023 Jul;10(3). https://doi.org/10.22265/acnef.10.3.612 ↑See page 2
[ Links ]
[2] Berend K. Diagnostic Use of Base Excess in Acid-Base Disorders. NEJM. 2018 April 12; 378(15):1419-1428. https//doi.org/10.1056/NEJMra1711860 ↑See page 3
[ Links ]
[3] Dzierba AL, Abraham P. A Practical Approach to Understanding Acid-Base Abnormalities in Critical illness. J Pharm Practice. 2011 March; 24(1):17-26. https://doi.org/10.1177/0897190010388153 ↑See page 4
[ Links ]
[4] Ahmad M, Wolberg A, Kahwaji CI. Biochemistry, Electron Transport Chain. [Updated 2023 Sep 4] . In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2024 Jan-. Available from: https:/www.ncbi.nlm.nih.gov/books/NBK526105/ ↑See page 4
[ Links ]
[5] Kaufman DA. Interpretation of ABgs. [Internet]. Thoracic. [cited 2023]. Available from: https:/www.thoracic.org/professionals/clinical-resources/critical-care/clinical-education/abgs.php ↑See page 4
[ Links ]
[6] Anesthesia Education Website. Acid-base physiology. [Internet]. Anesthesia Education Website [cited 2023]. Available from: https://www.anaesthesiamcq.com/AcidBaseBook/ab2_1.php ↑See page 4
[ Links ]
[7] Palmer BF, Clegg DJ. Salicylate Toxicity. NEJM. 2020 June; 382:2544-2555. https://doi.org/10.1056/NEJMra2010852 ↑See page 4
[ Links ]
[8] United states environmental Protection Agency. Technical Overview of Volatile Organic Compounds. [Internet]. 2021[cited 2023]. Available from: Available from: https/www.epa.gov/indoor-air-quality-iaq/technical-overview-volatile- organic- compounds#conclusion
↑See page 6
[ Links ]
[9] Saha A, Ranjan-Sharma A, Bhattacharya M, Sharma G, Lee S, Chakraborty C. Probable Molecular Mechanism of Remdesivir for the Treatment of COVID-19: Need to Know More. J. ARCMED. 2020 August; 51(6):585-586. https:/doi.org/10.1016/j.arcmed.2020.05.001 ↑See page 6
[ Links ]
[10] Blough B, Moreland A, Mora A. Metformin-induced lactic acidosis with emphasis on the anion gap. Proc (BaylUniv Md Cent). 2015 Jan; 28(1):31-33. https//doi.org/10.1080/08998280.2015.11929178 ↑See page 6
[ Links ]
[11] DuBose TD. Metabolic and respiratory acidosis. In Wilcox CS. Therapy in Nephrology and Hypertension. Saunders Elsevier; 2008:368-380. ↑See page 6
[ Links ]
















