Serviços Personalizados
Journal
Artigo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Acessos
Acessos
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares em
SciELO
Similares em
SciELO -
 Similares em Google
Similares em Google
Compartilhar
DYNA
versão impressa ISSN 0012-7353versão On-line ISSN 2346-2183
Dyna rev.fac.nac.minas v.72 n.158 Medellín maio/ago. 2009
AMMONIA: A REVIEW OF CONCENTRATION AND EMISSION MODELS IN LIVESTOCK STRUCTURES
AMONIO: REVISIÓN ACERCA DE LOS MODELOS DE CONCENTRACIÓN Y EMISIÓN EN ESTRUCTURAS PARA ANIMALES
JAIRO ALEXANDER OSORIO
Profesor Facultad de Ciencias Agropecuarias, Universidad Nacional de Colombia, Sede Medellín, aosorio@unal.edu.co
ILDA FERREIRA TINOCO
Profesora Departamento de Engenharia Agricolas, Universidade Federal de Viçosa, Brasil, iftinoco@ufv.br
HECTOR JOSE CIRO
Profesor Facultad de Ciencias Agropecuarias, Universidad Nacional de Colombia, Sede Medellín, hjciro@unal.edu.co
Recibido para revisar junio 10 de 2008, aceptado noviembre 13 de 2008, versión final noviembre 18 de 2008
ABSTRACT: Air quality in animal confinements is directly related to animal metabolisms, releasing heat, humidity and carbon dioxide (CO2) to the air from respiration and gases from digestion and feces such as NH3, CH4, H2S and dust. Heating systems produce CO and NO2 due to incomplete combustion which produces concentrations greater than those allowed by Colombian legislation (Decreto 948, 1995), and international countries such as the USA (National Organization for Security and Health NIOSH, 1994) and Brazil (NR-15, 1978), which represent some of the largest livestock development industries in the world. Among these gases, ammonia is the pollutant most often found in air inside animal confinements which deteriorates the health and productivity of animals and people. Hence, the study of ammonia has attracted the attention of researchers for years in various different countries. The objective of this work was to discover important aspects which must be taken into account in animal confinements, such as permissible ammonia concentrations. The developed models calculate concentrations and emissions, which can be used in countries such as in Colombia , where works and knowledge on environmental control and the production of gases in livestock confinements are few.
KEYWORDS: Ammonia, air pollution, environment, livestock confinements.
RESUMEN: La calidad del aire en los sistemas de crianza está directamente relacionada con el metabolismo de los animales, liberando directamente a la atmosfera calor, humedad y dióxido de carbono CO2, a través de la respiración y los gases de la digestión y excretas como NH3, CH4, H2S, material particulado, y gases de la combustión incompleta de los sistemas de calefacción como el CO, NO2, con concentraciones mayores a las permitidas por las normas Colombianas (Decreto 948 de 1995), así como con otros países como EE.UU. (Organización Nacional para la Seguridad y la Salud de NIOSH, 1994) y Brasil (NR-15, 1978), que tienen un importante desarrollo en la industria pecuaria a nivel mundial. Entre estos gases, el amonio es el gas más tóxico y encontrado con mayor frecuencia en el aire, dentro de la instalaciones de cría de animales que causa daños a la salud y la productividad de los animales y de las personas. Por lo tanto, el estudio de amonio ha llamado la atención de los investigadores durante años en diferentes países. El objetivo de este trabajo es dar a conocer aspectos importantes que deben tenerse en cuenta en las estructuras para animales, como las concentraciones admisibles de amonio, y los modelos desarrollados para calcular las concentraciones y las emisiones, que pueden ser utilizados en países como Colombia, donde los trabajos sobre el control ambiental y el conocimiento de la producción de gases en las estructuras pecuarias son pocos.
PALABRAS CLAVE: Amonio, polución del aire, ambiente, instalaciones pecuarias.
1. INTRODUTION
In livestock confinements, air contaminants principally originate from organic material. The inhalation of particulates and organic vapors can lead to respiratory tract diseases in humans and animals. In this sense, air quality problems in animal confinements must consider the following two aspects:
First, pollutants can directly cause alterations in the animal as a result of agent-body interactions (mechanical irritation, local inflammation etc.), harming the very animal which produces the pollutants, as well as making the attacked tissue susceptible to new diseases. Secondly, the excess of certain components may increase stress in the animal leading to a decline in its immune system, and consequent vulnerability to diseases and decrease in productive and reproductive performance.
The emission of NH3 from agricultural activities in Europe, excluding the former USSR, doubled between 1950 and 1986 [5, 41], while during the same time period in the Netherlands , NH3 emissions increased by a factor of 2.5. This increase in NH3 emissions has contributed significantly to the deposit of critical levels of nitrogen (N) in soil in many European countries, leading to the eutrophication and acidification of soils ]16, 17]. In Netherlands , for example, approximately 46% of the potential acid deposition is caused by emissions of NH3, mainly from agriculture [3].
From the given information, livestock producers are current seeking to guarantee animal welfare conditions, improve the well-being of people working directly in production systems and to minimize environmental impacts. All these propositions have a direct relationship with air quality and concentration and emission of harmful gases in livestock confinements as well as the atmosphere.
Ammonia (NH3)
Ammonia is a colored gas with a penetrating odor; it is lighter than air and soluble in water. Once emitted, NH3 can be quickly converted into ammonium (NH4), through reactions with nitric or sulfuric acid easily found in these environments. A ammonia is a toxic pollutant most often found in air within livestock confinements, and produced from the microbial decomposition of uric acid found in manure. High ammonia concentrations are usually found in closed buildings. This gas can act as an irritant to the eyes and respiratory membranes, developing a chronic stress that can affect the course of infectious diseases and also directly influence the growth of young animals. The maximum concentrations, suggested by reference [31], indicate that humans may be exposed to 25 ppm for duration of 8 hours, 35 ppm for 15 minutes and 50 ppm for 5 minutes.
CONCENTRATIONS AND AMMONIA EMISSIONS ON ANIMAL PRODUCTION
Poultry houses
In birds, the excess of ammonia causes various disorders and problems such as the reduction of appetite and respiratory rate, lesions in the respiratory tract, conjunctivitis and larger susceptibility to viral infections. To reference [46], in an experiment conducted in poultry houses in the United States, produced in isolated buildings with tunnel type negative ventilation systems, detected ammonia concentrations ranging from 10 to 50 ppm, with an average of 24.2 ppm. [47], recommend a maximum ammonia level of 20 ppm in poultry buildings.
To determine the concentration of ammonia in industrial poultry houses, and given that determination and control helps to increase production and reduction of levels of disease, permanent monitoring of ammonia production of ammonia is performed by control systems. (Worley. J.W (2002) cited by [45].
Given that knowledge of ammonia emissions are important to determine the local and regional impacts of industries on regional air quality, a reference [15] found that poultry houses with new and re-used bedding contribute to 8.8 and 11.7 kT/year of ammonia, respectively, in the states of Kentucky, USA, and 240 and 324 kT/year of ammonia emissions for new and re-used bedding respectively on the national level. To achieve these results, the authors used the equation (1), which has an error Standard (SEER) of 1.1 mg NH3 bird-1 d-1, and obtaining the average annual production of NH3 by the bird in each period of growth .
Birds are grown to different weights, and thus by equation (1), the total emission per bird varies by market weight.
ER = 0:03 - x (1)
Where, ER is the rate of ammonia emission (g NH3 bird-1 d-1),
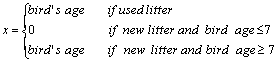
Studies evaluating different facilities and ventilation systems for the production of poultry (ducks, turkeys, breeders and broiler chickens), found a direct correlation between high rates of emission of ammonia and odors (OR, Odour Unit or Unit of Odor) in production of ducks (376 OR / LU and 40.6 g / h.LU) and turkey (138 OR / LU and 11.3 g / h.LU), in naturally ventilated facilities with high currents of air. For broilers, were found averages of 3.1 g / h.LU, of ammonia and 79 OR / Lu to smell with measurements of short duration, and the emission of ammonia has grown rapidly in the final stages of production when the mass of animal and the amount of manure produced increases [28]. In tropical climates several studies have been done to determine concentrations in poultry, but no emissions [6]. Among these works has been done by the reference [24], where they found the distribution of ammonia in a shed type tunnel
Hog buildings
To references [21, 22], reported that average ammonia concentrations measured in different hog farms in Northern Europe were between 5 and 28 ppm, and registering momentary increases of 50 ppm. According to references [20, 37], conducting research in HRHB (High Rise-Hog Buildings) systems in the USA, concentrations of ammonia during the summer are less than those seen during the winter, the period in which the facilities are kept closed ventilation is minimal. Sun et al. (2002), found ammonia concentrations in excess of 30 ppm in enclosed hog buildings during winter when ventilation was minimal.
Studies conducted by [38], on high-growth HRHB swine production systems, demonstrated that in structures in which manure pits are found directly below the building, the ammonia concentrations are below 20 ppm in the animal environment, and below 40 ppm in the pits where manure is deposited. This behavior was observed for most of the year. Ammonia levels greater than 25 ppm present health risks to both pigs and workers. To reference [36], encountered reductions of 12, 30 and 29.9% in pig weight gains ( 8.5 kg body weight) when they were exposed to concentrations of 50, 100 and 150 ppm of NH3, respectively, for four weeks.
To estimate and control ammonia emissions in hog buildings, To references [7, 29], developed a simulation from a mathematical model which considered temperature, air speed and pH. It is known that ammonia is produced from hog manure. Of the nitrogen consumed by animals, between 50% and 20% is excreted as urea and organic nitrogen components (feces). Urea is hydrolyzed into NH3 and CO2, and this reaction is catalyzed by the urease enzyme, which is produced by bacteria found in feces. When simulating ammonia emissions with the proposed control system, emissions were reduced by about 8% on an annual basis. This reduction was mainly due to the low rate of ventilation during certain periods of the year, maintaining more ammonia inside the building. The resulting concentration of ammonia inside was higher with the new algorithm (13.2 versus 9.1 ppm), but never exceeded the maximum tolerable level of 25 ppm.
Cattle confinements
Ammonia in closed buildings for dairy cattle produces a large quantity of urea hydrolysis in urine [27]. The amount of urea in relation to the total nitrogen content in the urine of dairy cattle depends on physiological factors such as diet and levels of composition and production. A growing surplus of dietary degradable proteins results in an increase in the rate of urine excretion [43], while the volume of urine produced is related to the intake of nitrogen (N), potassium (K) and sodium (Na) [44].
Permissible ammonia concentration levels as stated by the American Conference of Governmental Industrial Hygienists [2] are 15 ppm in confinements. According to reference [25], ammonia emissions for dairy cattle range from 20- 45 g dia–1 cow-1.
To reference [34], found that ammonia emissions per cow in the USA ranged from 13.1 to 55.5 kg of NH3 cow-1 years-1, with a mean of 23.9 kg NH3 cow-1 years -1, depending on weather and geographic region. To determine emissions produced by each animal per day, To reference [34], used the following equation:
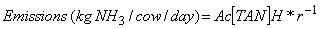 (2)
(2)
To reference [34], found that the greatest ammonia emissions in the United States were found in the American south and east, in states such as Arizona and California. The authors attributed this to higher temperatures recorded in the mentioned states, and the intense production of cattle in these places .
Models for the evaluation of ammonia emissions in animal production facilities
Ammonia emissions estimates for facilities that have been based on as-designed animals numbers or animal units (1AU = 500 kg), or maximum AU capacity, can also impact the estimate of annual emissions [15]. Emissions rate is approximately the product of ammonia concentration and ventilation exhaust airflow rate. While this calculation is simple in concept, in practice, both concentration and ventilation are difficult to measure accurately under commercial animal house conditions [49]. These measures are more difficult in naturally ventilated animal house in tropicals countries such as in Colombia .
To identify and quantify the uncertainties of existing to reference [33], suggested some methods for measuring ammonia emissions from naturally ventilated animal houses and waste stores. Four basic approaches were identified: I. feed and manure nitrogen balance (ammonia by difference); II. Summation of local ammonia sources; III. determining ammonia fluxes, either directly or indirectly, using an envelope more or less remote fro m the ammonia source; IV. measurement of ammonia sources to air e.g. by a tracer ratio method. A ranking exercise was carried out to help identify the most promising of these approaches for development to provide a robust method for use on farms. Overall, the approach of measuring ammonia sources to air, by a tracer ratio method or similar, achieved the top ranking. The second ranking was achieved by the approach of determining ammonia fluxes, either directly or indirectly, using an envelope more or less remote from the ammonia source.
Similarly, a reference [14], did a comparative demonstrations of three methods (flux sampling, external tracer ratio, and internal tracer ratio), were mounted in four real farm situations. A flux sampling method was demonstrated at a commercial dairy cow house (slurry-based), at a commercial piggery (straw-based), at a full-scale above-ground cylindrical slurry store (dairy cow slurry) and a full-scale earth-bank lagoon (pig slurry). An external tracer ratio method was demonstrated, in parallel with the flux sampling method, at the dairy cow house and at the above-ground slurry store. An internal tracer ratio method was demonstrated at the dairy cow house only. At the dairy cow house, the corrected emission rates from the flux sampling method and from the external tracer ratio method agreed to within the estimated experimental range, while the emission rate from the internal tracer ratio method was significantly lower. The overall conclusion of the study is that all three methods can have a useful role, the choice of which to deploy depending on the particular measurements needed in each case, for measure the emission of ammonia from naturally ventilated animal houses and wastes stores.
Although, these methodologies, still are difficult for use in tropicals countries, and needed of particularly conditions for their used, because they are thus naturally ventilated, which makes the flux of NH3 from them difficult to determine, as the ventilation rate (the number of air mass exchanges per unit time) varies according to temperature, wind speed, building design, orientation to the wind and animal occupancy. Conversely, for countries such as in Europe and U,S, there are several methodologies that has been using in many investigation, whit a good accuracy such as did for references [4, 8,11, 13, 15, 19, 30, 34] and others.
Models for the evaluation of ammonia concentration in animal production facilities
Hog building
To reference [48] applied two models for assessing the concentration of ammonia in hog confinements using two different diets (HPC and HPSBP), and the monitored results were which were compared with the predictions based on models of mass transfer. Model 1 was developed by reference [1,7] and Model 2 by the same researchers. Mass balances were performed using different mass transfer coefficients and different concentration gradients.
The release of ammonia from the surface of manure pits to the environment, in a process that typically occurs inside the confinement, can be modeled using the general mass transfer equation (equation 3):
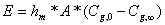 (3)
(3)
Several methods have been used to determine the mass transfer coefficient used in equation 3. Haslam et al. (1924), cited by [48], developed a set of four equations to describe the mass transfer coefficient using gas (equation 4) liquid boundary layer (equation 5) and the overall volume transfer using a coefficient for the partial pressure gradient (equation 6) or a concentration gradient (equation 7)
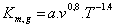 (4)
(4)
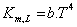 (5)
(5)
The coefficients a and b in equations 4 and 5, were converted in some cases using the ideal gas law [1] to express km,g and km,l in SI units
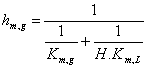 (6)
(6)
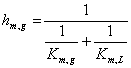 (7)
(7)
According to reference [48], many empirical relationships have been developed to determine the convective mass transfer coefficient of equation 3 for different flows, regimes and surface geometry. The variable hm can be described by one of the following equations, as a function of the Reynolds number (Re) and the Schmidt number (Sc) [18].
For laminar flow:
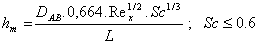 (8)
(8)
For turbulent flow:
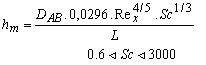 (9)
(9)
Using equation 11 and a measured value of total amoniacal nitrogen (TAN), equation 3 can be reorganized and solved for ammonia gas concentration in the boundary layer. Equation 3 can then be specified in a more useful form, according to reference [48]:
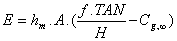 (10)
(10)
To reference [48] reported that, after the review of much ammonia emission models [29] supported the use of the equation (11) to determine the Henry constant (H):
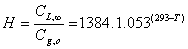 (11)
(11)
The concentration of ammonia in urine (CL, ∞) is determined by the fraction of total amoniacal nitrogen (TAN), that is un-ionized. The fraction of un-ionized is calculated from the ammonia pH and the acid ionization of ammonia constant, shown in equation 12, according to Zhang et al. (1994) and cited by reference [48]:
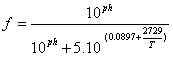 (12)
(12)
The main difference in the use of two models is that the Model 1 uses equation 4 to determine the mass transfer coefficient, while the Model 2 uses equations 8 and 9.
Interior gas concentration for Model 1 can be estimated with the following equation:
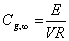 (13)
(13)
The variable E can be obtained using equation 4 as the mass transfer coefficient in which a is set equal to constant 50.1 [1, 49],
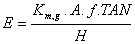 (14)
(14)
The internal gas concentration for the Model 2 was based on a dynamic balance of control volumes of the equation 15. Equations 11 and 12 were used in both models to determine H and F.
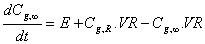 (15)
(15)
Both models represent the ammonia concentration behavior in environments of pigs subjected to two different diets (HPC and HPSBP), however, Model 2 was chosen determined to be better because it is nearer to the actual values. The equations used in the model tested are capable of simulating the concentration of ammonia and a emission of pollutants when TAN, pH, wind speed and temperature are measured accurately.
Cattle building
A combination of the reference [34] and [25, 26], models, combining natural and mechanical ventilation, was used to predict concentrations of ammonia from the solutions of the four differential equations (equations 4, 5, 6 and 7), proposaled by Haslam et al. (1924) and referenced by [34],, to model ammonia in closed hog systems and cited by Welford et al. (2003).
It was assumed that air within the facility is well mixed as well as the chemical constituents in urine present in a separate pool on the floor The equations proposed cited by [25] to find concentrations of ammonia in cattle confinements where the unknown variables can be obtained with the equations of 10 to 12 are:
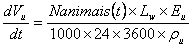 (16)
(16)
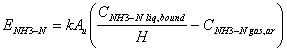 (17)
(17)
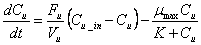 (18)
(18)
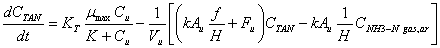 (19)
(19)
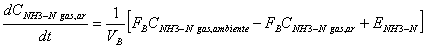 (20)
(20)
The use of the previously shown equations in computer programs such as Matlab provide simulations results of ammonia behavior in dairy cattle structures that do not deviate from the results measured in field conditions.
Models to simulate the flow and distribution of ammonia in the structures
Models which use computational fluid mechanics (CFD) have been developed to study the behavior of flows in greenhouses and in animal structures. To reference [39],, developed a model in two dimensions with CFD to analyze air quality and ventilation conditions and to predict the flow and concentration of ammonia in HRHB. The two dimensional CFD models are limited by their depictions of a real geometry of the structure, processes such as displacement of internal air, and distribution of ammonia through the horizontal section. To overcome these limitations, a three dimensional model was proposed by reference [40]. To develop the model, the system was assumed to be isothermal (no heat sources such as pigs, heaters or lights were considered) and conservation of mass, conservation of momentum, conservation of species, and the RNG κ-ε renormalization equations and the turbulence model were used.
When applying the previous equations, To reference [40] found that during summer conditions the maximum concentration of ammonia was found by less than 5 ppm, and under winter conditions reached 29 ppm. The model results based on equations were consistent with the measurements taken in real conditions.
Thus the researchers recommend using 3D models in livestock structures to predict the behavior of air quality and ventilation requirements, as in the example applied to a HRHB. Of the works presented above for the prediction of ammonia concentrations in livestock structures, control of contour conditions proved to be much easier in closed buildings. In the case of open structures found in tropical and subtropical climates in countries such as Colombia and Brazil , these types of work they have not yet been made, but are still considered important for the prediction of ammonia emissions produced in livestock buildings in these countries.
CONTROL OF AIR QUALITY
Given the harm caused by excessive ammonia, both in the confinement buildings and on the environment, industry groups along with the U.S. Federal Government established guidelines and regulations on the emission of ammonia. In 2005, the U.S. Union of Egg Producers stated that producers should maintain ammonia concentration levels below 50 ppm (at the height of chickens), so that the property is certified as "animal care". However, maintaining ammonia levels low is difficult for large producers, especially during the winter when ventilation is minimal [23]. Ammonia emission control in the structures is also corrected by some management techniques such as:
Animal Nutrition: When animals are fed a basic ration of corn and soybeans, studies show that the ammonia concentration limit of 50 ppm is
exceeded both in poultry buildings with and without waste management systems [23]. Recently studies cited by [9] showed that a diet using dried beans and corn dried distillers grains with solubles (DDGS), can reduce between 40 and 50% the ammonia emission in laying hen structures without affecting the production of eggs.
Chemical treatments: Research shows the potential of certain chemical compounds to reduce ammonia emissions in poultry buildings. More et al. (2003) cited by [9], found that the use of liquid aluminum sulfate on chicken bedding showed positive results for reducing ammonia emissions. Thus, the application of liquid aluminum sulfate is effective in reducing the emission of ammonia from poultry litter with a moisture content less than 20%, keeping the concentration of ammonia at 25 ppm during in the first three weeks of manure production. The high acidity of aluminum sulfate helps to reduce the existing pathogens, ensuring a more healthy environment [9, 10]. . To reference [21], evaluated and compared the effectiveness of currently utilized various additives, i.e. tap water, salt water, digested manure, microbial additive, soybean oil, artificial spice and essential oil, reducing odor emissions from the confinement pig building.
Ventilation: Natural or forced ventilation allows for alterations and control in the structures, providing oxygen and removing gases such as ammonia. The ventilation meets the thermal and hygienic requirements differently. In the winter when ventilation is minimal, dust and gas concentrations increase, including ammonia [35], Within poultry houses with natural ventilation there are three distinct layers of air: an upper level of hot air with high levels of acid hydrogen sulfide and ammonia, an middle layer of fresh air recently introduced, and a bottom layer of cold air which warms quickly upon contact with birds and is tainted with carbon dioxide from bird respiration [42].
CONCLUSIONS
The production of gases such as ammonia within structures should be considered when proposing industrial projects and environmental control policies.
The models presented to predict the ammonia concentrations and emissions in closed livestock structures showed positive results, similar to those obtained in real conditions. However, these models should be adapted to function in open structures, to be used in countries such as Colombia , where work regarding air pollution control in industries has been little developed.
In Colombia , it is important to begin performing studies on the contribution of livestock premises on global emissions of ammonia and producing air quality index maps
REFERENCES
[1] AARNINK, A.J.A. AND A. ELZING. 1998. Dynamic model for ammonia volatilization in housing with partially slatted floors, for fattening pigs. Livestock Production Science. 53(2): 153-69 [ Links ]
[2] ACGIH – AMERICAN CONFERENCE OF GOVERNMENTAL INDUSTRIAL HYGIENISTS. 2001 TLVs and BEIs – Handbook. ACGIH: Cincinnati, 2001. ISBN: 1-882417-40-2. [ Links ]
[3] ANONYMOUS, 1996. Environmental balance 96 (In Dutch). Samsom H.D. Tjeenk Willink bv, Alphen a/d Rijn, 142 pp. [ Links ]
[4] AROGO, J., WESTERMAN, P.W., HEBER, A.J., 2003. A review of ammonia emissions from confined animal feeding operations. Transactions of the ASAE 46 (3), 805–817. [ Links ]
[5] ASMAN, W.A.H., B. DRUKKER & A.J. JANSSEN, 1988. Modelled historical concentrations and depositions of ammonia and ammonium in Europe. Atmospheric Environment 22: 725–735. [ Links ]
[6] BAETA F.C; SOUZA, C.F. Ambiência em edificações rurais – conforto térmico animal. Viçosa: UFV, 1997. 246 p. [ Links ]
[7] BERCKMANS, D., DE MOOR, M., & DE MOOR, B. (1992). A new approach to modelling and control the energy and mass transfer in a three dimensional imperfectly mixed ventilated space. Proceedings of Roomvent '92: Air Distribution in rooms.\, Aalborg, Denmark , 1992, September 2 (Vol.1, pp.399-416). [ Links ]
[8] BLUNDEN JESSICA, VINEY P. ANEJA, PHILLIP W. WESTERMAN. Measurement and analysis of ammonia and hydrogen sulphide emissions from a mechanically ventilated swine confinement building in North Carolina. . Atmospheric Environment 42 (2008) 3315–3331 [ Links ]
[9] BURNS, R., MOORE,P.A., JR,MOODY, L. 2008. Using liquid aluminium sulphate to reduce poultry housing ammonia emissions. Mitigating air emissions from animal feeding operations conference, Iowa StateUniversity. [ Links ]
[10] CHANG C.W, CHUNG H, HUANG C.F, SUS H.J.J. Exposure assesment to airbone endotoxin, dust,ammonia, hydrogen, sulfudi and carbon dioxide in open style swine houses. Ann. Occup. Hyg, Vol 45, 6 (2001), 457-465. [ Links ]
[11] DALGAARD RANDI; HALBERG NIELS; KRISTENSEN IB S.; LARSEN. Inger.Modelling representative and coherent Danish farm types based on farm accountancy data for use in environmental assessments. Agriculture, ecosystems & environment. 2006,vol. 117, n 4, pp. 223-237 [ Links ]
[12] DECRETO 948.. Normas Colombianas sobre emisiones de gases por Fuentes fijas y moviles. 1995. [ Links ]
[13] DEMMERS T.G.M, PHILLIPS V.R, SHORT S, BURGESS LR, HOXEY R:P, WATHES C.M. SE-estructure and environment: validation of ventilation rate measurement methods and ammonia emission from naturally ventilated dayri and beef buildings in the United Kindom. Journal of Agricultural Engineering Research, volume 79, Issue , May 2001, Pages 107-116 [ Links ]
[14] DORE C.J., B.M.R. SCHOLTENS JONES R., J.W.H. HUIS INT VELD L.R. BURGESS, PHILLIPS V.R.. Measuring ammonia emission rates from livestock buildings and manure storespart 2: Comparative demonstrations of three methods on the farm. Atmospheric Environment 38 (2004) 3017–3024 [ Links ]
[15] GATES R.S, CASEY K.D, WHEELER E.F, XIN H, PESCATORE A.J. U.S. broiler housing ammonia emissions inventory. Atmosphery Environment (2007).9 p doi:10.1016/j.atmosenv.2007.06.057. [ Links ]
[16] HEIJ, G.J. & J.W. ERISMAN (EDS.), 1997. Acid atmospheric deposition and its effects on terrestrial ecosystems in the Netherlands . The third and final phase (1991–1995). Studies in Environmental Science 69, Elsevier, Amsterdam, 705 pp. [ Links ]
[17] HEIJ, G.J.& T. SCHNEIDER, 1991. Acidification research in the Netherlands . Studies in Environmental Science 46, Elsevier, Amsterdam, 771 pp. [ Links ]
[18] INCROPERA, F.P. AND D.P. DEWITT. 1990. Fundamentals of Heat and Mass Transfer. Toronto, ON: John Wiley & Sons. [ Links ]
[19] JACOBSON, L.D., HETCHLER, B.P., JOHNSON, V.J., 2005. Spatial, diurnal, and seasonal variations of temperature, ammonia, and hydrogen sulfide concentrations in two tunnel ventilated sow gestation buildings in Minnesota. In: Livestock Environment VII, Proceedings of the Seventh International Symposium, May, 2005, Beijing, China, pp. 18–20. ASAE Publication No. 701P0205. St. Joseph, MI. [ Links ]
[20] KEENER, H. M., D. L. ELWELL, K. EKINCI, AND H. A. J. HOITINK. 2000. Composting and value-added utilization of manure from a High-Rise TM swine finishing facility. ASAE Paper No.004143. St. Joseph, Mich.: ASAE. [ Links ]
[21] KIM KI-YOUN, HAN-JONG KO, HYEON-TAE KIM, YOON-SHIN KIM, YOUNG-MAN ROH, CHEOL-MIN LEE AND CHI-NYON KIM. Odor reduction rate in the confinement pig building by spraying various additives. Bioresource Technology. Volume 99, Issue 17, November 2008, Pages 8464-8469. [ Links ]
[22] KOERKAMP, G. P. W. G., J. H. M. METZ, G. H. UENK, V. P. PHILIPS, M. R. HOLDEN, R. W. SNEATH, J. L. SHORT, R. P. WHITE, J. HARTUNG, J. SEEDORF, M. SCHRODER, K. H. LINKERT, S. PEDERSON, H. TAKAI, J. O. JOHNSEN, AND C. M. WATHES. 1998. Concentrations and emissions of ammonia in livestock building in North Europe. J. Agric. Eng. Res. 70: 79-95. [ Links ]
[23] LIANG, Y., H. XIN,E.F. WHEELER, R. S GATES, H. LI, J.S ZAJACZKOWSKI, P.A. TOPPER, K.D CASEY, B. R BEHRENDS, AND F.J ZAJACZKOWSKI. Ammonia emissions from U.S. Laying hen houses in Iowa and Pennsilvania. Trnsactions of the ASAE, 48: 1927-1941, 2005. [ Links ]
[24] MIRAGLIOTTA, M.Y. Avaliação dos níveis de amônia em dois sistemas de produção de frangos de corte com ventilação e densidade diferenciados. Campinas: Faculdade de Engenharia Agrícola, 2000. 122 p. (Dissertação, Mestrado em Construções Rurais e Ambiência). [ Links ]
[25] MONTENY G.J, ERISMAN J.W. Ammonia emission from dairy cow buildings:a review of measurement techniques, influencing factors and possibilities for reduction. Netherlands Journal of Agricultural Science 46 (1998) 225-247. [ Links ]
[26] MONTENY, G.J., D.D. SCHULTE, A. ELZING & E.J.J. LAMAKER. 1998. A conceptual mechanistic model for the ammonia emissions from free stall cubicle dairy cow houses. Transactions of the ASAE. 41(1):193-201. [ Links ]
[27] MUCK, R.E. & T.S. STEENHUIS, 1981. Nitrogen losses in free stall dairy barns. In: Anonymous (Ed.) LivestockWastes: A Renewable Source. American Society of Agricultural Engineers, St. Joseph, pp.406–409. [ Links ]
[28] MÜLLER, H.J.; REINER, B.; GÜNTER, H.; ANTONIN, J. Odour and Ammonia emissions from poultry houses with different keeping and ventilation systems. In: International Symposium on Gaseous and Odour Emissions from Animal Production Facilities, 2003, Denmark . Proceedings. Horsens: Danish Institute of Agricultural Sciences, 2003, p.172 – 179. [ Links ]
[29] NI, J. Q., BERCKMANS, D., VRANKEN, E., & COENEGRACHTS, J. 1994. A simulation model to evaluate the ammonia emission from pig house. European Simulation Symposium, Istanbul, Turkey , 1994, October 9 p.13-15. [ Links ]
[30] NICHOLSON F.A.; CHAMBERS B.J.; WALKER A.W. Ammonia Emissions from Broiler Litter and Laying Hen Manure Management Systems. . Biosystems Engineering (2004) 89 (2), 175–185. [ Links ]
[31] NIOSH – National Institute for occupational safety and health. http://www.cdc.gov/niosh/pel88/7664-41.html. Ontario Ministry of the Enviroment, 2001. Ontario Air Satandards for Ammonia. Ontario,47 p. [ Links ]
[32] NR15. Normas tecnicas Brasileiras para controle de gases. 1978. [ Links ]
[33] PHILLIPS, V.R., LEE, D.S., SCHOLTENS, R., GARLAND, J.A., SNEATH, R.W., 2001. A review of methods for measuring emission rates of ammonia from livestock buildings and slurry or manure storespart 2: Assessment of techniques for measuring flux rates, concentrations and air flow rates. Journal of Agricultural Engineering Research 78, 1–14 [ Links ]
[34] PINDER ROBERT W, ANDERSON NATALIE J., STRADER ROSS, DAVIDSON CLIFF I, ADAMS PETER J. 2005. Ammonia Emissions from Dairy Farms: Development of a Farm Model and Estimation of Emissions from the United States . 15 p. [ Links ]
[35] RONCHI C: Principais práticas de manejo para aves recém nascidas. Editora Animal World. Numero 6 dezembro/janeiro de 2004. p 26-30. [ Links ]
[36] SMITH,L.F., J.F.PATIENCE, H.L.CLASSEN AND S.P.LEMAY. 2003. Manipulation of diet composition to alter the pattern of nitrogen excretion in growing pigs. Unpublished Undergraduate Thesis. Department of Animal and Poultry Science, University of Saskatchewan, Saskatoon, SK. [ Links ]
[37] STOWELL, R. R., D. ELWELL, B. STROBEL, H. KEENER, AND T. MENKE. 1999. Airflow and air quality within a High-Rise TM hog facility. ASAE Paper No. 994109. St. Joseph, Mich.: ASAE. [ Links ]
[38] STOWELL, R. R., H. KEENER, D. ELWELL, AND S. FOSTER. 2000. Gaseous emissions from a High-Rise TM hog facility. ASAE Paper No. 004080. St. Joseph, Mich.: ASAE. [ Links ]
[39] SUN, H., H. KEENER, R. R. STOWELL AND F. C. MICHEL, JR.. 2002. Two-dimensional computacional fluid dynamics (CFD) modeling of air and ammonia distribution in a High-RiseTM Hog Building (HRHB). Trans. ASAE 45(5): 1559-1568. [ Links ]
[40] SUN, H., H. KEENER, R. WEI DENG, AND F. C. MICHEL, JR.. 2004. Development and Validation of 3-D CFD Models to Simulate Airflow and Ammonia Distribution in a High-Rise Hog Building duringm Summer and Winter Conditions. [ Links ]
[41] TAKAI P, HARTUNG J, SEEDORF J, ; PEDERSEN S, JOHNSEN J, METZ J H,GROOT K, UENK G H, PHILLIPS V R, HOLDEM M R, SNEATH R W,WHITE R, LINKERT K H, WATHES C M.. Concentrations and emissions of airbone dust in livestock buildings in Northerm Europe. J agric. Engng Res. (1998) 70,59-70. [ Links ]
[42] TINOCO I.F.F. Avicultura industrial. Novos conceitos de materiais, concepções e técnicas construtivas disponíveis para galpões avícolas Brasileiros. Revista Brasilera de Ciência Avícola. ISSN 1516-635X. Vol 3 N.1. Campinas Jan/Apr. 2001. [ Links ]
[43] VAN VUUREN, A.M., C.J. VAN DER KOELEN C.J., H. VALK & H. DE VISSER, 1993. Effects of partial replacement of ryegrass by low protein feeds on rumen fermentation and nitrogen loss by dairy cows. Journal of Dairy Science 76: 2982–2993. [ Links ]
[44] VAN VUUREN, A.M. & M.C.J. SMITS, 1997. Effect of nitrogen and sodium chloride intake on production and composition of urine in dairy cows. In: S.C. Jarvis & B.F. Pain (Eds.), Gaseous Nitrogen Emissions from Grasslands, CAB International, Okehampton (in press). [ Links ]
[45] VRANKEN, E.. 2003. Reduction of ammonia emission from livestock buildings by the optimization of ventilation control settings. Proceedings of the Conference in Air Pollution from Agricultural Operations III. [ Links ]
[46] WATHES,C.M. Aerial emissions from poultry production..1998. World Poult Sci. 54: 241-251. [ Links ]
[47] WATHES,C.M, HOLDEM, M.R, SNEATH,R, WHITE,R.P, PHILIPS,V.R. 1997. Concentrations and emissions rates of aerial ammonia, nitrous-oxide, carbon-dioxide, dust and endotoxin in UK broiler and layer houses.Br Poul Sci.38: 14-28. [ Links ]
[48] WELFORD ERIN L., B.E, STÉPHANE P. LEMAY, P, ERNIE M. BARBER, STÉPHANE GODBOUT. Simulation of ammonia emissions from swine manure for various diet formulations. Written for presentation at the CSAE/SCGR 2003 Meeting Montréal, Québec July 6 - 9, 2003. [ Links ]
[49] WHEELER, E.F., CASEY, K.D., GATES, R.S., XIN, H., ZAJACZKOWSKI, J.L., TOPPER, P.A., LIANG, Y., PESCATORE, A.J., 2006. Ammonia emissions from twelve U.S.A. broiler chicken houses. Transactions of the ASABE 49 (5), 1495–1512. [ Links ]














