Serviços Personalizados
Journal
Artigo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Acessos
Acessos
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares em
SciELO
Similares em
SciELO -
 Similares em Google
Similares em Google
Compartilhar
DYNA
versão impressa ISSN 0012-7353versão On-line ISSN 2346-2183
Dyna rev.fac.nac.minas v.78 n.170 Medellín dez. 2011
AN INTEGRATED REMEDIATION SYSTEM USING SYNTHETIC AND NATURAL ZEOLITES FOR TREATMENT OF WASTEWATER AND CONTAMINATED SEDIMENTS
UN SISTEMA DE REMEDIACIÓN INTEGRADO USANDO ZEOLITAS SINTETICAS Y NATURALES PARA EL TRATAMIENTO DE AGUAS RESIDUALES Y SEDIMENTOS CONTAMINADOS
CARLOS RIOS REYES
Geólogo PhD, Profesor Titular, Escuela de Geología, Universidad Industrial de Santander, Bucaramanga, Colombia, carios@uis.edu.co
DANEN APPASAMY
PhD student, School of Applied Sciences, University of Wolverhampton, Wulfruna Street, Wolverhampton WV1 1SB, England, d.appasamy@wlv.ac.uk
CLIVE ROBERTS
Geologist PhD, School of Applied Sciences, University of Wolverhampton, Wulfruna Street, Wolverhampton WV1 1SB, England, c.l.roberts@wlv.ac.uk
Received for review June 15th, 2010, accepted March 3th, 2011, final version March, 22th, 2011
ABSTRACT: The major sources of water pollution can be classified as municipal, industrial, and agricultural. Different types of polluted aqueous effluents and sediments may be produced, which contain relatively high levels of heavy metals. During the 1990s, the large-scale development of constructed wetlands around the world drew much attention from public and environmental groups. The present study looks at the use of an integrated remediation system using zeolites for the treatment of wastewater and sediments. Zeolites have been widely studied in the past 10 years due to their attractive properties such as molecular-sieving, high cation exchange capacities, and their affinity for heavy metals. Coal industry by-products-based zeolites (faujasite type) have been tested as an effective and low-cost novel alternative for wastewater treatment, particularly their removing of heavy metals. On the other hand, a preliminary laboratory-scale experiment was conducted on the use of natural zeolites (clinoptilolite type) for the retention of heavy metals from canal sediments. Experimental work revealed promising results, which could be replicated on a bigger scale. Although this has been developed for canal sediments, the remediation strategy can be adapted to different waterways such as rivers. The development of the proposed remediation system in a specific experimental site as the major part of an innovation park can provide great benefits to a population living near contaminated effluents. It provides not only opportunities for the mitigation of environmental impact, improving water quality and landscape amenity, but also allows for several recreational opportunities.
KEYWORDS: constructed wetlands, aqueous effluents, sediments, integrated remediation system, zeolites, heavy metalsRESUMEN: Las principales fuentes de contaminación del agua pueden ser clasificadas como urbanas, industriales y agrícolas. Diferentes tipos de efluentes acuosos y sedimentos contaminados pueden ser producidos, los cuales contienen niveles relativamente altos de metales pesados. Durante la década de los 90s, el desarrollo a gran escala global de humedales construidos dirigió la atención de grupos públicos y ambientalistas. El presente estudio examina el uso de un sistema de remediación integrado usando zeolitas para el tratamiento de aguas residuales y sedimentos. Las zeolitas han sido ampliamente estudiadas en la última década debido a sus propiedades atractivas como tamizado molecular, alta capacidad de intercambio catiónico y afinidad por metales pesados. Zeolitas (tipo faujasita) sintetizadas a partir de sub-productos de la industria del carbón han sido probadas como una novedosa alternativa eficaz y de bajo costo para el tratamiento de aguas residuales, en particular en la eliminación de metales pesados. Por otra parte, una experimentación preliminar a escala de laboratorio se llevó a cabo sobre el uso de las zeolitas naturales (tipo clinoptilolita) para la eliminación de metales pesados de sedimentos de canal. El trabajo experimental revelo resultados prometedores, que podrían reproducirse en una mayor escala. Aunque esto se ha desarrollado para los sedimentos de canal, la estrategia de remediación se puede adaptar a diferentes cursos de agua tales como ríos. El desarrollo del sistema de remediación propuesto en un sitio experimental específico, representando la parte más importante de un parque de innovación, puede proporcionar grandes beneficios a la población que vive cerca de efluentes contaminados. Ofrece no sólo oportunidades para la mitigación del impacto ambiental, la mejora de la calidad del agua y la amenidad del paisaje, sino también varias oportunidades de recreación.
PALABRAS CLAVE: aguas residuales, sedimentos, humedales construidos, sistema de remediación integrado, zeolitas, metales pesados
1. INTRODUCTION
The Oro River acts as the major site for the disposing of industrial wastes from the industrial zone and municipal wastes of Bucaramanga's metropolitan area. At the same time, it is the main source of water supply for the entire region. Weathering products of the Santander massif rocks constitute the lithogenic component of the bed load (solid and dissolved). Several companies operating along the Bucaramanga Industrial Park S.A., Santander (Colombia), discharge different types of contaminants into the Oro River. The main pollution is of an organic nature, coming from industries such as food, petrochemistry, and car parts, which contribute organic matter, biochemical oxygen demand, suspended solids, phosphorus, and nitrogen. Vehicle repair workshops contribute oils, grease, and heavy metals. Furthermore, remnants of pollutants, which include heavy metals from leather tanning and metal industries, pose potential environmental and health hazards. Heavy metals can be toxic to human health and the environment at concentrations deemed unacceptable by regulations. Therefore, pollutants comprise a broad range of organic and inorganic compounds, their sources are equally diverse, and effective interventions are consequently difficult to target.
Constructed wetlands (CWs) are increasingly being employed around the world since the early 1970s for recycling wastewater to return it to the environment. It is the last line of defense against water pollution and the need to quantify the degree to which the pollution has actually impacted the environment. These systems have been recognized as providing many benefits including water supply and control, mining, the use of plants, wildlife, fish and invertebrates, integrated systems and aquaculture, erosion control, gene pools and diversity, energy, education and training, recreation, and reclamation [1-6]. They are low construction, operation and maintenance costs compared with conventional advanced treatment technologies. Therefore, it is very important to develop new treatment technologies as a contribution for the improvement of the environmental impact. In this study, we have integrated the concept of CWs coupled with remediation technologies, using in situ channels (connected to the polluted river/canal) using zeolites as substrates.
Globally, the worldwide coal industry produces millions of metric tons per year of coal combustion wastes. They include fly ash, bottom ash, boiler slag, flue gas desulfurization sludge, and low-volume wastes. In general, these wastes are used beneficially in small portions as solid wastes in landfills, although their disposal poses major challenges and serious environmental problems. The spontaneous combustion of coal beds is a natural phenomenon that has occurred in carboniferous basins during the recent geologic past (mainly during the Pleistocene Period), which is a common geologic feature that has been reported throughout the world. Intense heating of adjacent rock units (primarily those overlying coal beds), produces a variety of thermally altered rock types collectively called natural clinker.
Recently, due to the shortage of landfill sites and stricter environmental regulations, there has been a shift in societal attitudes, resulting in a strong interest in developing the beneficial recycling of coal by-products, which is now permissible in various applications, such as the synthesis of zeolites [7]. The use of these by-products in the synthesis of zeolites means that the technological transformation of such industrial wastes can be used as a remediation alternative. Therefore, it will contribute to considerably reduce the waste load on landfills. A number of studies have attempted to evaluate the suitability of fly ash in the synthesis of zeolites for different applications [8-16]. Intense research on the potential industrial applications of fly ash-based zeolites has been developed mainly in the removal of heavy metals from wastewater [17-26]. Ríos and co-workers [23-24,27-28] have introduced the use of natural clinker in the synthesis of zeolites with potential applications in the treatment of polluted effluents. Several procedures for wastewater treatment, including chemical precipitation, electrodeposition, ion exchange, membrane separation, and adsorption have been developed [29-43]. However, adsorption has been the preferred method for heavy metal removal, because it is considered to be a particularly effective technique.
In principle, any solid material with a microporous structure can be used as an adsorbent (e.g., bone and coal char, clays, iron oxides, synthetic and natural zeolites, molecular sieves, and activated carbon [44]).
Remediation of contaminated sediment sites often proves technically difficult and costly [45-49]. On the other hand, the remediation techniques on contaminated sediments are generally much more limited than for most other solid waste materials. The widely diverse contamination sources in larger catchment areas usually produce a complex mixture of contaminants that is very difficult to treat [46]. Multiple efforts have been made during the last few decades to develop alternative non-removal remediation techniques. In-situ capping uses the sub-aqueous placement of a covering layer over contaminated sediment in order to prevent contaminants being released into the surface water. The inhibition of contaminant release is based on the (1) stabilisation of the sediment, (2) physical isolation, and (3) chemical isolation of the sediment from the overlying water body [47]. Different reactive materials (e.g., activated carbon, apatite, organoclay, zeolite, zero-valent iron) that are used for wastewater treatment can be applied to in-situ capping [48].
Appasamy and co-workers have used a zeolite mat at experimental scale for the treatment of contaminated sediments [50-51]. The main purpose of this research was to develop an experimental work on laboratory-scale to show the efficiency of synthetic faujasite and natural clinoptilolite in the removal of heavy metals from wastewater and contaminated sediments, respectively. A replicated work on a bigger scale (integrated remediation system) can be performed in a specific experimental site as the major part of an innovation park (Bucaramanga Industrial Park S.A.). This can provide water quality and landscape amenity, recreational opportunities, the creation of fauna and flora habitat, opportunities for storm water harvesting, sometimes combined with aquifer storage and recovery system, a degree of flood retention function and community education designed to promote increased knowledge, improved skills, and more positive attitudes about the environment.
2. EXPERIMENTAL PROCEDURE
2.1 Treatment of acid mine drainage with synthetic zeolite
A sample of acid mine drainage (AMD) from the Parys Mountain copper-lead-zinc deposit of Anglesey (North Wales) was collected for this study. It is strongly acidic (pH < 2) and metal-rich due to the oxidation of sulphide minerals, and its orange-brown colour is due to the very high concentrations of ferric iron in solution. The by-products of the coal industry used in the synthesis of zeolites were: (1) fly ash: supplied by the Rugeley Power Station, West Midlands (England), which is generated by pulverized coal combustion and classified as Class F fly ash, and (2) natural clinker: collected in an open pit of the Cerrejón coal mining site, La Guajira (Colombia), which is produced by the spontaneous combustion of coal beds. Clinoptilolite was provided by RS Minerals Ltd. (England). The synthesis of zeolites was conducted following the alkaline fusion method as reported by Ríos et al. [24]. Faujasite-type zeolites were prepared by grinding with a SpectroMill Ball Pestle Impact grinder and sieved on 200 mesh. Then, they were ground and particles of < 150 µm were selected for sorption experiments. Batch sorption reactions were carried out at room temperature using faujasite as a sorbent material. The sorption of heavy metals onto faujasite was studied in laboratory batch experiments, which were carried out at room temperature to investigate the efficiency of sorbents for removing heavy metals and ammonium from AMD. A weighted amount of sorbent (0.25 and 1 g) was introduced in 100 ml PVC plastic bottles. Then, a volume of 20 ml of AMD with pH 1.96 was added. The resultant faujasite:AMD mixtures were continuously shaken between 5 min and 24 h, and the temporal evolution of the solution pH and EC was monitored. At each scheduled reaction time, the PVC plastic bottles were removed from the shaker. The adsorbent (faujasite) was separated by filtration, and the filtrates were stored at 4 oC in a refrigerator for chemical analyses. All measurements were done according to the Standard Methods for the Examination of Water and Wastewater. Analysis of untreated and treated AMD and leachates obtained after batch experiments were performed measuring pH (pH 211 auto-calibration bench pH/mV meter, Hanna Instruments) and electrical conductivity (conductivity meter, version Cond 315i, WTW). The metal concentrations were determined using a Thermo Jarrell-Ash ICAP-9000 ICP-AE spectrometer. Figure 1 illustrates the experimental procedure followed with regard to the synthesis of faujasite and its application as a sorbent material for the treatment of AMD.
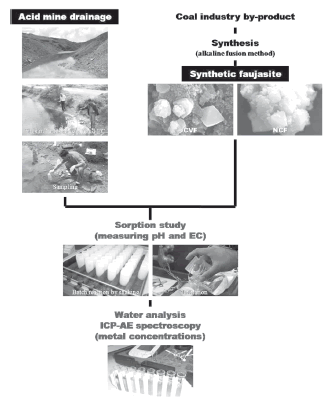
Figure 1. Chart flow showing the experimental procedure used during the synthesis of zeolites from coal by-products for the treatment of acid mine drainage
2.2 Treatment of contaminated sediments with natural zeolite
Preliminary research on the use of natural clinoptilolite for the treatment of canal sediments was conducted on a small scale laboratory setup (Fig. 2) as suggested by Appasamy and co-workers [50-51]. To determine the potential remediation of contaminated sediments by natural clinoptilolite, the zeolite was used in pillow-case like voile mats and placed over canal sediments in A4 size tanks (25.5 x 39.5 x 15.5 cm). The volume ratio of clinoptilolite to sediment was 1:7 and this setup was left undisturbed for 7 days. Sediment samples were taken from the tank at scheduled times and analyzed by X-ray fluorescence (XRF) spectroscopy.

Figure 2. Lab-scale experimental setup for pilot study
2.3 Design of the integrated remediation system
According to preliminary research on the use of zeolites as a remediation tool for the treatment of contaminated water and sediments, it was necessary to identify the potential sites in order to locate a specific experimental site, to estimate how to connect the proposed integrated remediation system to that specific site, to select types of canals (CWs without vegetation) to be used, to determine their required volume, to integrate them into site arrangement, and to determine their inlet and outlet features.
3. RESULTS AND DISCUSSION
3.1 Heavy metal removal from acid mine drainage by faujasite
The sorption process considered here refers to a sorbent (zeolite of the faujasite type) and a polluted aqueous media (AMD) containing dissolved heavy metals. The pH and EC trends for the neutralization reactions between the investigated sorbent and AMD are shown in Fig. 3. The starting pH (1.96) changed during a monitoring time of 24 h to 4.42 (fly ash-based faujasite, FAF) and 4.29 (natural clinker-based faujasite, NCF), although for the monitoring time NCF generally produced slight higher pH values than those obtained with the addition of FAF. According to Genç-Fuhrman et al. [52], pH increases mainly due to the dissolution of the sorbent in the process of shaking. The final pH values were observed in the batches due to the hydrolysis of the faujasite as well as cationic exchange. Similar results are reported elsewhere with a remark that the pH increase is almost unavoidable in a faujasite heavy metal system [53]. A similar behaviour was observed in the EC trends.
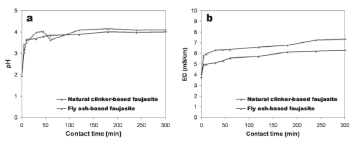
Figure 3. Variation of (a) pH and (b) EC as a function of time during the adsorption batch experiments with starting pH of 1.96 and EC of 3.77 mS/cm, sorbent (faujasite) dose of 0.25 g and volume of AMD of 20 ml
The immobilization of heavy metals from aqueous solutions is quite a complicated process, which consists of ion exchange and adsorption. However, it is likely to be accompanied by the precipitation of metal hydroxide complexes on active sites of the particle surface [54]. The removal of heavy metals from AMD using the synthetic faujasite is illustrated in Fig. 4, indicating that, in spite of the starting material used for the synthesis of faujasite, the zeolite showed a similar behaviour in the removal of heavy metals. However, the NCF produced the lowest residual concentrations of all metal ions, except for Cu and Zn, compared with results obtained using FAF. Faujasite has a strong affinity for heavy metals, which are attracted into the sorbent and bound there by different mechanisms. It is well known that the main mechanism for metal uptake is the precipitation of metal complexes on the surface of the sorbent and not sorption. We are in agreement with Peric et al. [54], with regard to how the removal of heavy metals from aqueous media is not only controlled by ion exchange and adsorption but also by precipitation of metal hydroxide complexes on active sites of the particle surface. On the other hand, the addition of an alkaline material such as faujasite to the AMD increased the pH, and heavy metals can be hydrolyzed and precipitated as suggested by Evangelou and Zhang [55]. To investigate the effect of sorbent dose, NCF was selected to develop an additional batch reaction test (results not shown), taking into account that this sorbent produced the highest pH value (9.43) after a contact time of 24 h. The pH is strongly affected by the sorbent material rather than the AMD composition and particularly with a higher sorbent dose.
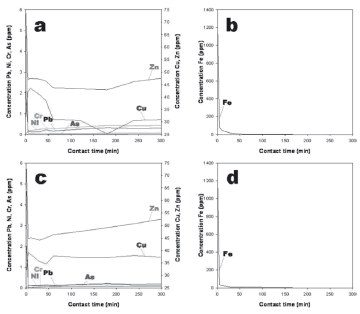
Figure 4. Heavy metal concentration variation as a function of time during the adsorption batch experiments with a starting pH of 1.96, an EC of 3.77 mS/cm, and a sorbent (faujasite):AMD mixture of 0.25 g / 20 ml; (a-b) FAF; (c-d) NCF
3.2 Heavy metal removal from contaminated sediments by clinoptilolite
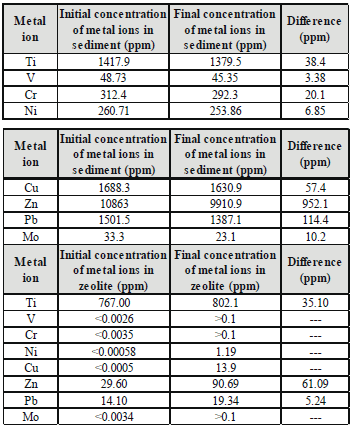
In the remediating of the contaminated sediments, a reactive mat composed by clinoptilopite was used to eliminate the need for a thick cap and subsequent ex situ treatment of sediments. Contaminants may be trapped in the zeolite mat. Results from the remediation experiments show that most of the heavy metals were removed from the sediments and the concentration of heavy metals considerably increased in the zeolite mat. Table 1 summarizes the concentration of metal ions of untreated and treated sediments with the clinoptilolite mat. Zn is the element with the highest concentration loss from sediments and highest adsorption by the clinoptilolite followed by Pb and Cu. In general, other heavy metals concentrations also decreased but in very low concentrations to be considered as positive results. Results reveal that there is a mass balance difference between the concentration of heavy metals being released from the sediment and that being absorbed by the natural clinoptilolite. This can be explained by the fact that heavy metals went into solution but were not in contact with the clinoptilolite as the mat is only in contact with the top layer of sediments. It can also be due to the saturation of the zeolite as the zeolite:sediment ratio used in the experiments was only 1:7. Further research is needed on secondary processes occurring in sediments, such as oxidation, adsorption and complexation. These processes are important, especially in anoxic sediments as they regulate the availability of the heavy metals and therefore the efficiency of the remediation process. Other waterways, such as rivers, have different secondary processes, which would have to be considered before designing the remediation strategy to achieve maximum efficiency. Results demonstrated the effectiveness of clinoptilolite in reducing the mobility of heavy metals, which makes in-place immobilization an effective remediation alternative.
Table 1. Metal ion concentration of untreated and treated sediments
3.3 Large scale remediation setup for the treatment of wastewater and contaminated sediments
The intended IRS (Fig. 5) can be used to improve the water quality and landscape amenity, combined with recreational opportunities for people of different ages. This system consists in four long narrow canals with the following dimensions: 10 m long, 0.4 m wide, and 0.3 m deep, which provide 4.0 m2 of surface area. Zeolite is used as a substrate. However, in order to compare the performance of each canal, it should be interesting to use different types of substrates as suggested by Ríos et al. [56]. These channels are interconnected to each other. They are connected to the canal system through a pump which will pump both water and sediments into the system. Once they settle (in the static case), and after zeolites have adsorbed the contaminants, the water can then be drained and sediments removed and used for other purposes (since they will not be contaminated). Static and dynamic experiments should be conducted. In static experiments, water will enter from one side of the reactive mat until it became full of water. It should be allowed to stand in the reactive mat for 24 h, and then out flowed from the system. In static experiments, the zeolite mat can work mainly on two mechanisms: (1) the absorption of the substrate due to their large ratio surface, (2) the biological action of abundant microbes in the zeolite mat. In dynamic experiments, oxygen with recycling water strengthened the biological function.
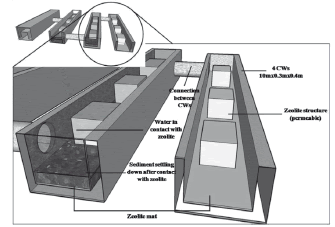
Figure 5. Large scale remediation setup
The IRS would be very important focusing on advanced remediation and treatment system for the treatment of wastewater and contaminated sediments in a specific experimental site (Fig. 6) as the major part of an innovation park, providing several benefits for potential users (tourism industry, governmental departments, private entrepreneurs, private residences, aquaculture industries, and agro-industries) for water quality improvement and pollutant removal.
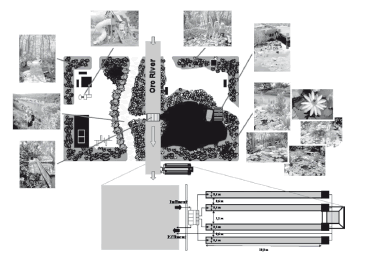
Figure 6. General sketch of an experimental site, showing the different alternatives of recreation and tourism attractions to be considered around a specific IRS
The benefits of the IRS applied to the treatment of wastewater and contaminated sediments would include improvement of water quality and landscape amenity, recreational opportunities, the creation of fauna and flora habitat, opportunities for storm water harvesting, sometimes combined with aquifer storage and recovery system, a degree of flood retention function, and community education designed to promote increased knowledge, improved skills and more positive attitudes about the environment (Fig. 7).
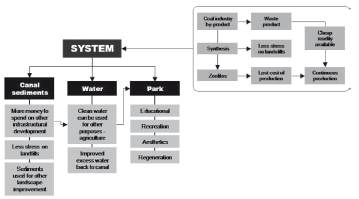
Figure 7. Sustainable water and sediment management
Figure 8 is an example of the regeneration process of a degraded area, similar to several areas observed along the Bucaramanga Industrial Park S.A. However, the intended idea aims to encourage tourism business to operate in an environmentally friendly way, taking into account that it is necessary to make efforts to reduce the environmental impact, but keeping in mind that in spite of its benefits the environment or nature should not be used as a marketing tool. Therefore, the use of the IRS proposed in this study would provide not only opportunities for mitigation of the environmental impact, improving water quality and landscape amenity, but also several recreational opportunities along the area of interest.

Figure 8. Regeneration process of a degraded area
4. CONCLUSIONS
Results reveal that the efficiency of the faujasite-type zeolite for the removal of heavy metals from AMD depends on the sorbent material and applied dose. Selectivity of faujasite for metal removal was in decreasing order: Fe > As > Pb > Zn > Cu > Ni > Cr. In addition to cation exchange reactions, other reactions commonly occur. Precipitation of hydroxide species played an important role in the co-precipitation and adsorption, and, therefore, in the immobilization of metals. The sorption study suggests that natural clinker-based faujasite represents a beneficial product, which can be used in future as an ion exchanger in removing acidity and heavy metals from AMDs. On the other hand, the natural clinoptilolite showed to be effective with up to 75 % absorption efficiency in some cases. The selectivity for this natural zeolite was Pb > Cu > Cd > Zn > Cr > Co > Ni. Its absorption efficiency ranged from 75 % for Pb to approximately 45 % for Ni.
However, further research is needed to estimate the efficiency of the tested absorbents for the removal of other heavy metals. Heavy metals are often associated with stable phases and thus have to be made soluble first so that zeolites can achieve maximum adsorption efficiency.
Constructed wetlands can be an effective, environmentally friendly means of treating liquid and solid wastes. However, we have adopted in this study the principles of CWs, although without the use of vegetation, as an IRS for wastewater and contaminated sediment treatment. Zeolites represent an inexpensive and environmentally friendly solution to improve the effectiveness for treatment purposes. Furthermore, the intended IRS can provide several benefits.
ACKNOWLEDGMENTS
This research was supported by the Programme Alban, The European Union Programme of High Level Scholarships for Latin America, scholarship No. E05D060429CO, and the Universidad Industrial de Santander (a remunerated commission) for funding C. Ríos. Authors have benefitted from research facilities provided by the School of Applied Sciences at the University of Wolverhampton.
REFERENCES
[1] Peterson, H., Use of constructed wetlands to process agricultural wastewater. Can. J. Plant Sci. 78, pp. 199-210, 1998. [ Links ]
[2] Drizo, A., Frost, C., Smith, K., Grace, J., Phosphate and ammonium removal by constructed wetlands with horizontal subsurface flow, using shale as a substrate. Water Sci. Technol. 35,pp. 95-102, 1997. [ Links ]
[3] Richardson, C., Qian, S., Craft, C. and Qualls, R. Predictive models for phosphorus retention in wetlands. Wetlands Ecol. Manag. 4,pp. 159-175, 1997. [ Links ]
[4] Vymazal, J., Cooper, P., Harberl, R., Perfler, R., Laber, J., Removal mechanism and types of constructed wetlands. Backhus Publishers, Leiden, 1995. [ Links ]
[5] Crites, R., Design criteria and practice for constructed wetlands. Water Sci. Technol. 26, pp. 1-6, 1994. [ Links ]
[6] Brix, H., Use of Constructed Wetlands in Water Pollution Control: Historical Development, Present Status, and Future Perspectives. Water Sci. Technol. 30, pp. 209-223, 1994. [ Links ]
[7] Ríos, C.A., Synthesis of zeolites from geological materials and industrial wastes for potential application in environmental problems: Doctoral thesis, School of Applied Sciences, University of Wolverhampton, England, 233P. [ Links ]
[8] Höller, H., Barth-Wirsching, U. Zeolite formation from fly ash. Fortschr. Miner. 63, pp. 21-43, 1985. [ Links ]
[9] Mondragón, F., Rincón, F., Sierra, L., Escobar, J., Ramírez, J., Fernández, J., New perspectives for coal ash utilization: Synthesis of zeolitic materials. Fuel 69, pp. 263-266, 1990. [ Links ]
[10] Moreno, N., Querol, X., Andrés, J.M., López-Soler, A., Janssen-Jurkovicová, M., Nugteren, H., Towler, M., Stanton, K., Determining suitability of a fly ash for silica extraction and zeolite synthesis. J. Chem. Technol. Biotechnol. 79, pp. 1009-1018, 2004. [ Links ]
[11] Shigemoto, N., Shirakami, S., Hirano, S., Hayashi, H., Preparation and characterisation of zeolites from coal ash. Nip. Kag. Kai. 5, pp. 484-492, 1992. [ Links ]
[12] Shigemoto, N., Sugiyama, S., Hayashi, H., Miyaura, K., Characterization of Na-X, Na-A, and fly ash zeolites and their amorphous precursors by IR, MAS NMR and XPS. J. Mater. Sci. 30, pp. 5777-5783, 1995. [ Links ]
[13] Park, M., Choi, C.L., Lim, W.T., Kim, M.C., Choi, J., Heo, N.H., Molten-salt method for the synthesis of zeolitic materials: I. Zeolite formation in alkaline molten-salt system. Microporous and Mesoporous Mater. 37, pp 81-89, 2000a. [ Links ]
[14] Park, M., Choi, C.L., Lim, W.T., Kim, M.C., Choi, J., Heo, N.H. Molten-salt method for the synthesis of zeolitic materials: II. Characterization of zeolitic maerials. Microporous and Mesoporous Mater. 37, pp. 91-98, 2000b. [ Links ]
[15] Querol, X., Umaña, J.C., Plana, F., Alastuey, A., Lopez-Soler, A., Medinaceli, A., Valero, A., Domingo, M.J., Gracia-Rojo, E. Synthesis of Na zeolites from fly ash in a pilot plant scale: Examples of potential environmental applications. Fuel 80, pp. 857-865, 2001. [ Links ]
[16] Rayalu, S., Meshram, S.U., Hasan, M.Z. Highly crystalline faujasitic zeolites from fly ash. J. Hazard. Mater. 77, pp. 123-131, 2000. [ Links ]
[17] Catalfamo, P., Corigliano, F., Patrizia, P., DI Pascuale, S., Study of the pre-crystallization stage of hydrothermally treated amorphous aluminosilicates through the composition of the aqueous phase. J. Chem. Soc., Faraday Trans. 89, pp. 171-175, 1993. [ Links ]
[18] Singer, A., Berkgaut, V., Cation exchange properties of hydrothermally treated fly ash. Environ. Sci. Technol. 29, pp. 1748-1753, 1995. [ Links ]
[19] Kolousek, D., Seidl, V., Prochazkova, E., Obsasnikova, J., Kubelkova, L., Svetlik, L., Ecological utilization of power-plant fly ashes by their alteration to phillipsite: Hydrothermal alteration, application. Acta Univ. Geol. 37,pp. 167-178, 1993. [ Links ]
[20] Moreno, N., Querol, X., Ayora, C., Pereira, C.F., Janssen-Jurkovicová, M., Utilization of zeolites synthesized from fly ash for the purification of acid mine waters. Environ. Sci. Technol. 35, pp. 3526-3534, 2001a. [ Links ]
[21] Moreno, N., Querol, X., Ayora, C., Alastuey, A., Fernández-Pereira, C., Janssen-Jurkovicová, M., Potential environmental applications of pure zeolitic material synthesized from fly ash. J. Environ. Eng. pp.127, 994-1002, 2001b. [ Links ]
[22] Querol, X., Alastuey, A., Moreno, N., Alvarez-Ayuso, E., García-Sánchez, A., Cama, J., Ayora, C., Simón, M., Immobilization of heavy metals in polluted soils by the addition of zeolitic material synthesized from fly ash. Chemosphere 62, pp. 171-180, 2006. [ Links ]
[23] Ríos, C.A., Williams, C.D., Castellanos, O.M. Síntesis y caracterización de zeolitas a partir de la activación alcalina de caolinita y subproductos industriales (cenizas volantes y clinker natural) en soluciones alcalinas. Bistua 4, pp. 60-71, 2006. [ Links ]
[24] Ríos, C.A., Williams, C.D., Roberts, C.L., Removal of heavy metals from acid mine drainage (AMD) using coal fly ash, natural clinker and synthetic zeolites. J. Hazard. Mater. 156, pp. 23-35, 2008. [ Links ]
[25] Ríos, C.A., Williams, C.D., Roberts, C.L., A comparative study of two methods for the synthesis of fly ash-based sodium and potassium type zeolites with potential use in the purification of wastewaters. Fuel 88, pp. 1403-1416, 2009. [ Links ]
[26] Prasad, B., Sangita, K., Tewary, B.K., Reducing the hardness of mine water using transformed fly ash. Mine Water Environ. 30, pp. 61-66, 2011. [ Links ]
[27] Ríos, C.A., Williams, C.D. Synthesis of zeolitic materials from natural clinker: a new alternative for recycling coal combustion by-products. Fuel 87, pp. 2482-2492, 2008. [ Links ]
[28] Sandoval, M.V., Henao, J.A., Ríos, C.A., Williams C.D., Apperley, D.C., Synthesis and characterization of zeotype ANA framework by hydrothermal reaction of natural clinker. Fuel 88, pp. 272-281, 2009. [ Links ]
[29] Diz, H.R., Novak, J.T., Fluidized bed for the removing of iron and acidity from acid mine drainage. J. Environ. Eng. 124, pp. 701-708, 1998. [ Links ]
[30] Webster, J.G., Swedlund, P.J., Webster, K.S., Trace metal adsorption onto an acid mine drainage iron (III) oxy hydroxy sulphate. Environ. Sci. Technol. 32, pp. 1361-1368, 1998. [ Links ]
[31] Feng, D., Aldrich, C., Tan, H., Treatment of acid mine water by use of heavy metal precipitation and ion exchange. Miner. Eng. 13, pp. 623-642, 2000. [ Links ]
[32] Chartrand, M.M.G., Bunce, N.J., Electrochemical remediation of acid mine drainage. J. Appl. Electrochem. 33, 259-264, 2003. [ Links ]
[33] Santos, S., Machado, R., Correia, M.J.N. Treatment of acid mining waters. Miner. Eng. 17, pp. 225-232, 2004. [ Links ]
[34] Mohan, D., Chander, S., Removal and recovery of metal ions from acid mine drainage using lignite - A low cost sorbent. J. Hazard. Mater. B137, pp. 1545-1553, 2006. [ Links ]
[35] Gibert, O., De Pablo, J., Cortina, J.L., Ayora, C., Municipal compost-based mixture for acid mine drainage bioremediation: Metal retention mechanisms. Appl. Geochem. 20, pp. 1648-1657, 2005a. [ Links ]
[36] Gibert, O., De Pablo, J., Cortina, J.L., Ayora, C., Sorption studies of Zn(II) and Cu(II) onto vegetal compost used on reactive mixtures for in situ treatment of acid mine drainage. Water Res. 39, pp. 2827-2838, 2005b. [ Links ]
[37] Johnson, D.B., Hallberg, K.B., Acid mine drainage remediation options: a review. Sci. Total Environ. 338, pp. 3-14, 2005a. [ Links ]
[38] Johnson, D.B., Hallberg, K.B., Biochemistry of the compost bioreactor components of a composite acid mine drainage passive remediation system. Sci. Total Environ, pp. 338, 81-93, 2005b. [ Links ]
[39] Wattena, B.J., Sibrella, P.L., Schwartzb, M.F., Acid neutralization within limestone sand reactors receiving coal mine drainage. Environ. Pollut. 137, pp. 295-304, 2005. [ Links ]
[40] Wei, X., Viadero JR., R.C., Buzby, K.M., Recovery of iron and aluminium from acid mine drainage by selective precipitation. Environ. Eng. Sci. 22, pp. 745-755, 2005. [ Links ]
[41] Kalin, M., Fyson, A., Wheeler, W.N., The chemistry of conventional and alternative treatment systems for the neutralization of acid mine drainage. Sci. Total Environ. 366, pp. 395-408, 2006. [ Links ]
[42] Fernánez, B., Ayala, J., Evaluation of fly as ashes for the removal of Cu, Ni, and Cd from acidic waters, Dyna 161, pp. 141147, 2010. [ Links ]
[43] Pinzón-Bedoya, M.L., Vera, L.S., Kinetic modeling biosortion of Cr(III) using orange shell, Dyna 160, pp. 96-106, 2009. [ Links ]
[44] Khraisheh, M.A.M., Al-Degs, Y.S., Mcminn, W.A.M., Remediation of wastewater containing heavy metals using raw and modified diatomite. Chem. Eng. J. 99, pp. 177-184, 2004. [ Links ]
[45] Jacobs, P.H., Förstner, U., Concept of sub-aqueous capping of contaminated sediments with active barrier systems using natural and modified zeolites. Water Res. 33, 2083-2087, 1999. [ Links ]
[46] Jacobs, P.H., Förstner, U., Managing contaminated sediments IV. Subaqueous storage and capping of dredged material. J. Soils Sediments 1, pp. 205-212, 2001. [ Links ]
[47] Azcue, J., Zeman, A., Förstner, U., International Review of application of sub-aqueous capping techniques for remediation of contaminated sediments. Proceedings of the 3rd International Congress on Environmental Geotechnics, Lisbon, September, pp. 7-11, 1998. [ Links ]
[48] Olsta, J.T., Darlington, J.W., Innovative systems for dredging, dewatering or for in-situ capping of contaminated sediments. Proceedings of the Annual International Conference on Soils, Sediments, Water and Energy: Vol. 11, Article 20, 2006. Available at: http://scholarworks.umass.edu/soilsproceedings/vol11/iss1/20 [ Links ]
[49] Murphy T., Moller A, and Brouwer H., In situ treatment of Hamilton Harbor sediment. J. Aquat. Ecosyst. Health 4, pp.195-203, 1995. [ Links ]
[50] Ríos, C.A., Appasamy, D., Roberts, C.L., Williams, C.D., An integrated canal remediation system for wastewater treatment in the Birmingham Canal Navigations (BCN) main line, West Midlands (England) with potential recreation, education and tourism alternatives. 10th International UFZ-Deltares/TNO Conference on Soil-Water Systems, Milano, Italy, pp. 3-6 June, 2008. [ Links ]
[51] Appasamy, D., Roberts, C.L., Williams, C.D., Physical and chemical characterisation of canal sediments and their potential remediation using zeolites. 11th International UFZ-Deltares/TNO Conference on Management of Soil, Groundwater & Sediment, Salzburg, Austria, pp. 22-24 September, 2010. [ Links ]
[52] Genç-Fuhrman, H., Mikkelsen, P.S., Ledin, A., Simultaneous removal of As, Cd, Cr, Cu, Ni and Zn from stormwater: Experimental comparison of 11 different sorbents. Water Res. 41, pp. 591-602, 2007. [ Links ]
[53] Pitcher, S.K., Slade, R.C.T., Wards, N.I., Heavy metal removal from motorway stormwater using zeolites. Sci. Total Environ. 334-335, 161-166, 2004. [ Links ]
[54] Peric, J., Trigo, M., Medvidovi´c, N.V., Removal of zinc, copper and lead by natural zeolite A comparison of adsorption isotherms. Water Res. 38, pp. 1893-1899, 2004. [ Links ]
[55] Evangelou, V.P., Zhang, Y.L., A review: Pyrite oxidation mechanisms and acid mine drainage prevention. Crit. Rev. Environ. Sci. Technol. 25,pp. 141-199, 1995. [ Links ]
[56] Ríos, C.A., Gutiérrez, L., Aizaki, M., A case study on the use of constructed wetlands for the treatment of wastewater as an alternative for petroleum industry. Bistua 5,pp. 25-41, 2007. [ Links ]














