Serviços Personalizados
Journal
Artigo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Acessos
Acessos
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares em
SciELO
Similares em
SciELO -
 Similares em Google
Similares em Google
Compartilhar
DYNA
versão impressa ISSN 0012-7353versão On-line ISSN 2346-2183
Dyna rev.fac.nac.minas v.78 n.170 Medellín dez. 2011
PROMOTING EFFECT OF Mo ON Pd / g-Al2O3 SUPPORTED CATALYSTS IN THE OXIDATIVE DEHYDROGENATION OF PROPANE
EFECTO PROMOTOR DE Mo EN CATALIZADORES SOPORTADO DE Pd / g-Al2O3 EN LA DESHIDROGENACION OXIDATIVA DE PROPANO
HUGO ZEA
Ph.D. Departamento de Ingeniería Química y Ambiental, Universidad Nacional de Colombia, Bogotá, Colombia, hrzear@unal.edu.co
JOSE L. FIGUEIREDO
Ph.D. Laboratório de Catálise e Materiais, LSRE/LCM, Faculdade de Engenharia, Universidade do Porto, 4200-465 Porto, Portugal, jlfig@fe.up.pt
LUIS CARBALLO*
Ph.D. Departamento de Ingeniería Química y Ambiental, Universidad Nacional de Colombia, Bogotá, Colombia, lmcarballo@unal.edu.co *Present address: Durham, NC, USA
Received for review May 10th, 2010, accepted September 27th, 2010, final version October, 4th, 2010
ABSTRACT: Bimetallic catalysts of Pd-Mo supported on g-Al2O3 were prepared to study the interaction of Pd-Mo in terms of their structural properties and their catalytic activity for the reaction of oxidative dehydrogenation of propane (ODHP). The catalysts contain distributions of Pd-Mo loads between 0 and 5 wt %, and were characterized by total surface area (TSA), total pore volume (TPV), active surface area (ASA), XRD, and TEM-EDS. The results indicate that the addition of Mo to catalysts of Pd/g-Al2O3 modifies its catalytic activity for the ODHP reaction. The influence of the load relationship Pd-Mo in the catalytic activity was determined. Monometallic catalysts were prepared for purposes of comparison.
KEYWORDS: Bimetallic Pd-Mo catalyst, propane reactions, oxidative dehydrogenation
RESUMEN: Catalizadores bimetálicos de Paladio-Mo soportados en g - Al2O3 fueron preparados para estudiar la interacción bimetálica en términos de sus características estructurales y su actividad catalítica en la reacción de deshidrogenación oxidativa de propano (ODHP). Los catalizadores contienen distribuciones de las cargas Pd-Mo entre 0 y 5 % peso, y fueron caracterizados por superficie total, volumen total del poro, superficie activa, XRD y TEM-EDS. Los resultados indican que la adición de Mo a los catalizadores del Pd/g - Al2O3 modifica su actividad catalítica para la reacción ODHP. La influencia de la relación en porcentaje en peso de Pd-Mo en la actividad catalítica es evaluada. Catalizadores monometálicos fueron preparados para los propósitos de la comparación.
PALABRAS CLAVE: Catalizadores bimetálicos de Pd-Mo, reacciones con propano, deshidrogenación oxidativa
1. INTRODUCTION
There are strong economic incentives to develop catalysts for the selective conversion of alkanes, especially propane. Four different routes have been proposed to transform light alkanes into higher value hydrocarbons [1-8]: (a) carry out the alkylation over zeolites (although the alkylation of hydrocarbons of less than four carbons is thermodynamically unfavorable) [9], (b) transform propane and other light saturated hydrocarbons into aromatic hydrocarbons, (c) dehydrogenate propane to propene, and (d) perform the oxidative dehydrogenation of propane to propene:
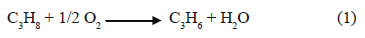
which leads to the formation of very stable products, making the reaction thermodynamically favorable.
Structural changes induced in Pd supported catalysts by the addition of a second metal or metal-support interaction have a profound effect on catalyst activity and selectivity for reactions spanning from selective hydrogenation to selective oxidation [7,10-14]. There have been reports of highly active and selective catalytic systems for the reaction of alkane dehydrogenation constituted by elements of group VIII (transition metals) and promoted by molybdenum [10,15-17]. An outstanding performance of a Pd/Al2O3 catalyst has been reported on aspects like propene yield and selectivity from propane [10]. The combination of Pd and g-Al2O3 were found to be necessary for the formation of superficial oxygenated species. The influence of g-Al2O3 may be due to the strong superficial acidity; alumina's acid centers are considered to be active places for the heterolytic dissociation of hydrocarbons [13,18,19].
In this study, the performances of Pd/g-Al2O3, Mo/g-Al2O3, and Pd-Mo/g-Al2O3 catalysts were investigated in the reaction of oxidative dehydrogenation of propane in order to determine the Pd-Mo interaction in terms of their structural properties and their catalytic activity. High-resolution TEM coupled with energy dispersive x-ray spectroscopy (EDS) was employed to study the nature of the Pd-Mo interaction.
2. MATERIALS AND METHODS
2.1. Catalysts synthesis
Wet impregnation was used in the preparation of coimpregnated catalysts. The support (alumina, Alpha-Aesar, g-phase > 99.97 %, surface area; 79 m2/g, pore volume: 0.31 cm3/g) was impregnated with an aqueous solutions of palladium chloride (PdCl2) and ammonium heptamolybdate ([NH4]Mo7O24.6H2O) of different concentrations to generate the proposed metallic loads on the support; the concentrations of the aqueous solution concentrations were theoretically calculated. During the addition of the impregnating solution, the support was kept under a moderate and constant vacuum, mixed in an ultrasound mixer (Branson B220) during 90 minutes; the remaining aqueous solution was evaporated at 120 oC during 3 hours; the resulting dried sample was heated in an inert atmosphere during 4 hours at 600 oC. A total of 13 catalysts were prepared, including monometallic catalysts that were prepared for comparison purposes. All bimetallic catalysts have a total metal loading of 5 % in weight.
An oxidative pretreatment procedure was applied to all the catalysts synthesized. During pretreatment, a catalyst sample was placed under dry air atmosphere with a total flow of 70 ml/min (STP) and heated at a constant increment of temperature (5 oC/min) from an ambient temperature up to 450 oC. These conditions were kept during 120 minutes and then the temperature was increased to 600 oC using the same increment. The condition of 600 oC was maintained during 240 minutes, natural cooling was allowed, and finally the catalyst was stored in an inert atmosphere (He) to avoid its degradation.
2.2. Catalyst Characterization
The total surface area was measured by the adsorption of nitrogen (N2) at 77 K, using a Coulter model Onmisorb100 CX adsorption system; the calculation of the total superficial area was done using the BET method; values of the total pore volume were obtained using Gurvitsch's method. The active surface areas of the reduced catalyst samples were measured through the irreversible H2 adsorption at atmospheric pressure and room temperature. Morphological analyses of the catalyst were carried out in a transmission electron microscope (TEM) using a high-resolution JEOL 2010 microscope coupled with an EDS) system; powdered samples were analyzed by XRD using Ni-filtered Cu-Ka radiation.
2.3. Catalytic activity
The catalytic activity tests for the ODHP reaction were conducted on micro-reaction equipment (Autoclave Engineers model BTRS-jr). The reaction system consists of a stainless steel reactor tube (6.3 mm internal diameter) containing 100 mg + 2 of oxidized catalyst sample; the heating of the reactor was done externally through an electric resistance with temperature control. Reaction conditions were either 400 oC or 500 oC and 101.325 kPa of pressure. Preliminary experiments indicate the absence of mass transfer restrictions for the reaction conditions. The feeding consisted of propane and oxygen mixtures of high purity (99.99 %). A molecular sieve was used at the entrance of the reactor with the objective of retaining impurities coming from the feeding line; the gas flows were controlled with mass flow controllers (Bronkhorst High-Tech series F-200 and F201). Feed and product gases were analyzed by a gas chromatograph with of a programmable temperature controller (Tremetrics 9001) connected on-line to the reactor; chromatographic analyses were performed using a Porapak Q column (particle diameter mesh 80-100). The ratio of oxygen/propane in the feed was set according to the stoichiometric of reaction (1). Several tests were carried out without catalysts to confirm the absence of homogeneous reactions.
3. RESULTS
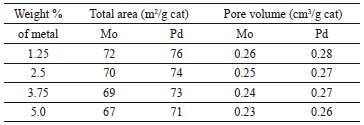
Tables 1 and 2 summarize the results obtained for the mono and bimetallic catalysts in terms of total surface area and pore volume; the TSA of monometallic catalysts are inversely proportional to metal loading; both for the Mo and Pd sets of impregnated catalysts. On the other hand, the bimetallic catalysts show values of TSA and TPV that are inversely proportional to the Pd weight content and directly proportional to the Mo weight content. Behaviors like those presented by the TSA and TPV are usually related to changes in the mechanisms of the active agent adsorption on the support [20,21].
Table 1. Total surface area and pore volume of bimetallic catalysts
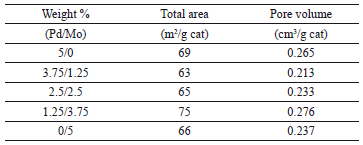
Table 2. Total surface area and pore volume of monometallic catalysts
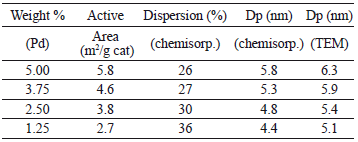
Results of the active surface area, dispersion, and particle diameter for the bimetallic and Pd loaded monometallic catalysts are presented in Tables 3 and 4. The ASA and diameter of particle values of monometallic Pd catalysts are directly proportional and almost lineal with the Pd load. The bimetallic catalysts present ASA values smaller than their respective monometallic Pd catalyst, for these catalysts there is no direct proportionality of the Pd loading with the dispersion or ASA values, they presented a minimum dispersion value at 2.5/2.5 loading of Pd/Mo with a corresponding maximum of particle diameter of 9.8 nm; the higher value of dispersion obtained for the bimetallic catalysts was at 1.2/375 Pd/Mo loading, with a corresponding minimum particle diameter of 6.6 nm.
Table 3. Metal surface area, dispersion and particle diameter of bimetallic catalysts
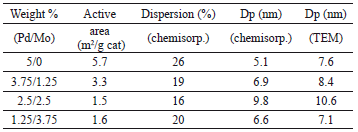
Table 4. Metal surface area, dispersion and particle diameter of monometallic Pd catalysts
The ASA results obtained by molecular chemical adsorption of H2 were verified by using high-resolution TEM pictures of the impregnated catalyst and the histogram of the particle size distribution obtained using Digital Micrograph 2.5 software. A minimum of 180 particles were employed to determine the average particle size by TEM pictures. The values of the diameter of the Pd particles obtained via chemisorption are in good agreement with those obtained by TEM, which is an indication of the reliability of H2 chemisorption as a tool to determine the Pd particle size in this type of supported catalysts.
ODHP reaction was used to test the catalytic activity of the catalysts. The reactants and products considered in the chromatographic analyses were: propane, oxygen, propene, carbon dioxide, water, and carbon monoxide. Figure 1 shows the propane conversion and propene selectivity for the bimetallic catalysts as a function of the Mo loading. For both reaction temperatures (400 and 500 oC), the trend of the propene selectivity is inversely proportional to the propane conversion. The neglectable contribution of the monometallic Mo catalyst in the conversion of propane at 400 oC and 500 oC is evident, implying that the catalytic active material is Pd for the ODH reactions, as reported in the literature [8-10,13,15]; the conversion for the reaction at 400 oC fluctuates as a function of the content of Mo with a maximum value of 11.1 % for the 1.25/3.75 Pd/Mo catalyst. In terms of selectivity, the results at 400 oC showed a maximum of 32 % for the 3.75/1.25 Pd/Mo catalysts.
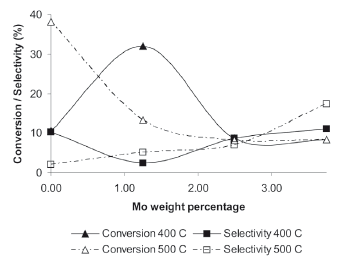
Figure 1. Propane conversion and propene selectivity as a function of Mo weight loading
The behavior of the conversion at 500 oC differs substantially from the tendency obtained at 400 oC. It starts with a maximum value of 38 % for the 5 % monometallic Pd catalyst, then decreases to values between 13 to 8 % as the Mo loading increases. On the contrary, the selectivity profile shows a constant increase with the increment in the percentage of Mo loading until its maximum value of 17 % for the 1.25/3.75 Pd/Mo.
As presented in Tables 3 and 4, the active surface area of the catalysts differ from one to another as a function of the catalyst loading; in order to compare the catalysts under the same conditions, the specific activity of propene was calculated and defined as milimoles of propene produced by square meter of active area per second, and plotted as a function of Mo weight percentage for the ODHP reaction at 400 and 500 oC (Fig. 2). All Pd/Mo catalysts showed an increment in the specific activity with the percentage of Mo (from 0 to 3.75) and their maximum specific activity value of 4.26 milimoles of propane/m2 active area per second was obtained with the 1.25/3.75 Pd/Mo catalysts.
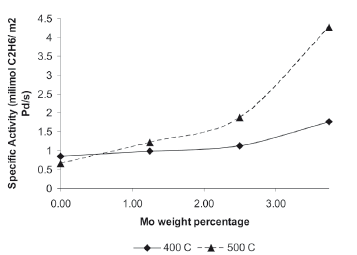
Figure 2. Specific activities of propane as a function of Mo weight loading
In order to compare the performance of bimetallic catalysts against monometallic catalysts with the same Pd loading, the specific activity of the monometallic catalyst was determinate and the ratio b (defined as the specific activity of the bimetallic catalyst divided by the specific activity of the monometallic catalyst with same Pd loading) was calculated; the results of these calculations are presented in Fig. 3. A b ratio equal to 1 implies that the bimetallic catalyst is as active to produce propene as its equivalent monometallic catalyst; a b ratio below 1 indicates that the monometallic catalyst is more active than the bimetallic catalyst; and, in the same way, a b ratio above 1 means that the bimetallic catalyst is more active than the monometallic catalyst.
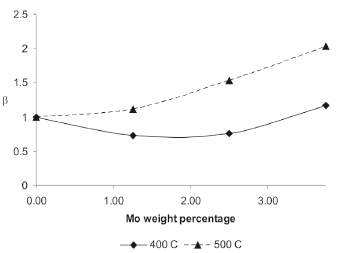
Figure 3. The b ratio as a function of Mo weight percentage
The results from Fig. 3 indicate that for the reaction test at 400 oC, the bimetallic catalysts are less active for producing propene than the monometallic catalyst with equivalent Pd loading, except for the 1.25/37.5 Pd/Mo catalyst with a b value slightly larger than 1 (b = 1.17). The 0 % Mo weight loading correspond to the 5 % Pd catalyst and therefore b = 1. On the contrary, for the reaction test performed at 500 oC, all the bimetallic catalysts presented values above 1, having their maximum in the 1.25/37.5 Pd/Mo catalyst with a b value of 2.03.
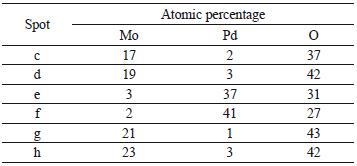
TEM coupled with EDS was employed to understand the nature of the Pd-Mo interaction. Figures 4(a) and 4(b) show the TEM images of the Pd/Mo 1.25/3.75 and 2.5/2.5 loading ratios, respectively; the corresponding results of the EDS patterns for some representative areas on the catalyst surface for Mo, Ti, and O are shown on Table 5. In order to obtain the distribution of the components on the surface of the support, EDS scan areas were selected in the vicinity of the Pd particle and in areas away from the Pd particles. The results clearly show that the coimpregnation procedure of Pd and Mo results in a catalyst with individual Pd particles and a relatively well-dispersed Mo on the surface of the alumina. The EDS scans showed that there is Mo in the vicinity of the Pd particles indicating close interaction between them. Energy dispersive X-ray specstroscopy (EDS) detection limits are approximately 1 % by weight [22,23]. Additional peaks showed up in the EDS analysis: Al from the support, and traces of Cu and C from the TEM grid sample holder, but these were not reported in Table 5.
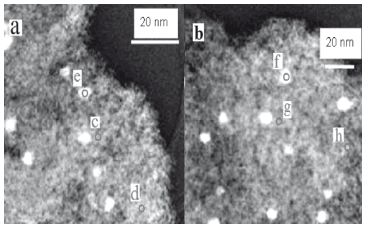
Figure 4. TEM image of (a) 1.25/3.75 Pd/Mo, and (b) 2.5/2.5 Pd/Mo catalysts
Table 5. EDS chemical analysis of samples 1.25/3.75 Pd/Mo and 2.5/2.5 Pd/Mo catalysts on spots labeled in Fig. 4
X-ray difracction results of the calcined catalyst are summarized in Fig. 5; all of the XRD profile showed the characteristic peaks of alumina (2q = 25.7, 35.2, 37.9, 43.4, and 52.8). The monometallic catalyst containing 5% Mo does not present any peak corresponding to any molybdenum oxide compound, which may indicated that Mo species are amorphous. A clear peak of PdO is observed at 2q = 34.1 for the monometallic 5% Pd catalysts, the bimetallic catalyst also showed this peak; however, the position of the peak is slightly shifted to the left, indicating a possible weak interaction between the Pd and Mo. No clear evidence of the formation of a Pd-Mo alloy or solid solution is observed.
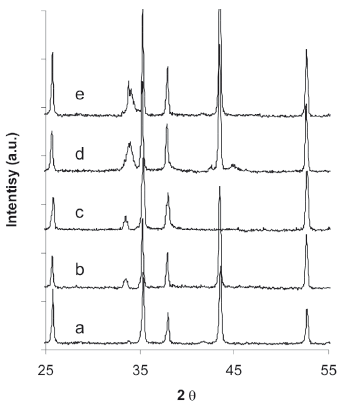
Figure 5. XRD profiles of calcined catalysts (a) 0/5 Pd/Mo, (b) 1.25/3.75 Pd/Mo, (c) 2.5/2.55 Pd/Mo, (d) 3.75/1.255 Pd/Mo, and (e) 5/0 Pd/Mo
The differences shown by the monometallic catalysts, when compared to the bimetallic catalysts in terms of TSA and dispersion values, are often related to the different conditions that the dissolved complex of Pd and Mo in the impregnating solution encountered during the impregnation process [24-26]. On one hand, the aqueous solution of PdCl2 behaves as a strong acid (with a pH around 2.7); but meanwhile, the aqueous solution (NH4)Mo7O24.6H2O shows a weak acid character (with a pH around 5.8). Due to the fact that the point of zero charge (PZC) of the support is 7.6, for both monometallic impregnations of Mo and Pd, the electrically charged density of the alumina surface is positive.
The adsorption of molybdate anions (Mo7O246-) is facilitated by the positive electrically-charged surface of the alumina, leading to a well-distributed adsorption of molybdate ions all over the support surface, as observed in the EDS analysis of the selected catalysts. The chemistry of PdCl2 is more complex; it has been reported that in the presence of Cl- ions, Pd in aqueous solutions could form several types of complexes, [Pd(H2O)4']+2, [Pd(H2O)3']+1, among others [27-29]. The adsorption of these positively-charged palladium complexes will be hampered by the positive electrically-charged surface. This is reflected by the better dispersion of the low-loaded palladium monometallic catalysts. In the case of the bimetallic coimpregnated catalysts, the mixing of the aqueous solutions of palladium chloride (PdCl2) and ammonium heptamolybdate ([NH4]Mo7O24.6H2O) produces a mixed solution with an average pH of 4.5. It has been established that at pH values between 4.0 to 5.0, a hydrolytic reaction occurs in the PdCl2 solution, and that a colloidal Pd(OH)2 species is formed [19,30]. At this pH value, the molybdate anions (Mo7O246-) continue being the dominant species during the impregnation process [31]. This new condition of simultaneous Pd and Mo species in a solution interacting with the positively electrically-charged surface of the alumina propitiates the selective adsorption of the (Mo7O246-) anions on the surface, followed by the precipitation of the Pd particles during the evaporation of the remaining aqueous solution. This sequential-type of adsorption mechanism explains the behavior of the ASA with regard to the percentage of Mo; MoOx selective species deposited on the pore walls available for Pd(OH)2 colloidal species, reducing the ASA via the formation of larger Pd crystallites. These larger Pd crystallites could reside either on top or in the surroundings of the MoOx species impregnated on the support surface [20], in concordance with the EDS results; also observed in other transition metal supported catalysts [32].
In summary, the results obtained by the characterization of the catalysts showed that: (a) the addition of Mo to Pd catalyst generate a loss in terms of active surface area, (b) there is no formation of a Pd-Mo alloy or solid solution, (c) Pd particles are in close vicinity with MoOx species, and (d) the Mo species are amorphous. The results from the activity test of the ODHP reaction showed an increment in the specific activity towards the formation of propene when Mo was added. The absence of the Pd-Mo alloy and solid solution formation implies no alteration of the chemical structure of the active material; this fact, along with the virtual inactivity of the Mo monometallic catalyst lead to the conclusion that the improved specific activity must be the result of the interaction of the MoOx species and the Pd crystallites. This interaction could be related to a modification in the mechanisms of reaction in which the MoOx might facilitate the insertion of oxygen in the propane molecule, avoiding its oxidation to CO, CO2, or other oxygenated hydrocarbon compounds. The modification on the mechanisms of reaction explains the strong dependency of the specific activity and b values with temperature; a kinetic and reaction mechanism study is out of the scope of this article but is the subject of ongoing studies in our group.
4. CONCLUSIONS
The performance of Pd-Mo supported in alumina catalysts for the reaction of the ODHP has been evaluated in comparison to single-component Pd and Mo supported catalysts. The highly dispersed Pd/Al2O3 catalyst prepared by impregnation showed a high conversion of propane but a poor selectivity to propene; the supported monometallic Mo catalyst presented no catalytic activity for the ODHP reaction. The Pd-promoted Mo catalyst showed lower dispersion values but a significantly higher specific activity to the formation of propene when compared to the monometallic Pd catalyst under similar reaction conditions. Characterization studies revealed that a close vicinity of Mo and Pd was obtained by the coimpregnation method used during the synthesis of the catalyst, although there is no formation of a Pd-Mo alloy or solid solution.
ACKNOWLEDGMENT
Financial support from the European Commission within the framework of the project ALR/B7-3011/94.04-6.0108.9, CATALISE-ALFA is gratefully acknowledged.
REFERENCES
[1] Dahl, I., Grande K., Jens K., Rytter E. and Slagtern A., Oxidative hydrogenation of propane in lithium hydroxide-lithium iodide melts. Applied Catalysis. 77, pp. 163-174. 1991 [ Links ]
[2] Kumar, B., Effect of silica support on V-Mg-O catalysts during ODH of propane: Experimental and modeling using genetic algorithms. International Journal of Chemical Reactor Engineering. 4, A28. 2006 [ Links ]
[3] Leveles, L., Seshan, K., Lercher, J. and Lefferts, L., Oxidative conversion of propane over lithium-promoted magnesia catalyst - II. Active site characterization and hydrocarbon activation. Journal of Catalysis. 218, pp. 307-314. 2003 [ Links ]
[4] Liu, W., Lai, S., Dai, H., Wang, S., Sun, H. and Au C., Oxidative dehydrogenation of n-butane over mesoporous VOx/SBA-15 catalysts. Catalysis Letters. 113, pp. 147-154. 2007 [ Links ]
[5] Madeira, L. and Portela, M., Catalytic oxidative dehydrogenation of n-butane. Catalysis Reviews-Science and Engineering.44, pp. 247-286. 2002 [ Links ]
[6] Perez-Reina, F., Rodriguez-Castellon, E., Jimenez-Lopez, A. Dehydrogenation of propane over chromia-pillared zirconium phosphate catalysts. Langmuir;15, pp. 8421-8428. 1999 [ Links ]
[7] Savary, L., Costentin, G., Mauge. F., Lavalley, J., Elfallah, J., Studer, F., Guesdon, A. and Ponceblanc´, H., Characterization of AgMo3P2O14 catalyst active in propane mild oxidation. Journal of Catalysis;169, pp. 287-300. 1997 [ Links ]
[8] Vitry, D., Dubois, J. and Ueda, W., Strategy in achieving propane selective oxidation over multi-functional Mo-based oxide catalysts. Journal of Molecular Catalysis A-Chemical, 220, pp. 67-76. 2004 [ Links ]
[9] Feng, Z., Liu. L. and Anthony, R., Reactions of propane on modified metal-oxides. Journal of Catalysis. 136, pp. 423-431. 1992 [ Links ]
[10] Hamed, O. and Henry, P., Oxidation of olefins by palladium(II). 16. A new palladium(II)-catalyzed asymmetric chlorohydrin synthesis. Organometallics l. 17, pp. 5184-5189. 1998 [ Links ]
[11] Aksoylu, A., Misirli, Z. and Onsan, Z., Interaction between nickel and molybdenum in Ni-Mo/Al2O3 catalysts: I - CO2 methanation and SEM-TEM studies. Applied Catalysis A-General,168, pp. 385-397. 1998 [ Links ]
[12] Zea, H., Lester, K., Datye, A., Rightor, E., Gulotty, R., Waterman W. and Smith M., The influence of Pd-Ag catalyst for ethylene hydrogenation restructuring on the activation energy in ethylene-acetylene mixtures. Applied Catalysis A-General, 282, pp. 237-245. 2005 [ Links ]
[13] Yazawa, Y., Yoshida, H., Takagi, N., Komai S., Satsuma, A. and Hattori, T., Oxidation state of palladium as a factor controlling catalytic activity of Pd/SiO2-Al2O3 in propane combustion. Applied Catalysis B-Environmental, 19, pp. 261-266. 1998 [ Links ]
[14] Zea, H., Chen, CK., Lester, K., Phillips, A., Datye, A., Fonseca, I and Phillips J., Plasma torch generation of carbon supported metal catalysts. Catalysis Today l, 89, pp.237-244. 2004 [ Links ]
[15] Shao, H., Zhong, S. and Guo, J., Pd-Cu/V2O5-SiO2 catalyst for propane oxidative dehydrogenation with CO2 to propylene. Chinese Journal of Catalysis, 25, pp. 143-148. 2004 [ Links ]
[16] Lezla, O., Bordes, E., Courtine, P. and Hecquet, G., Synergetic effects in the Ni-Mo-O system - Influence of preparation on catalytic performance in the oxidative dehydrogenation of propane. Journal of Catalysis,170,pp. 346-356. 1997 [ Links ]
[17] Liu, Y., Wang, J., Zhou, G., Xian, M., Bi Y. and Zhen, K., Oxidative dehydrogenation of propane to propene over barium promoted Ni-Mo-O catalyst. Reaction Kinetics and Catalysis Letters, 73, pp. 199-208. 2001. [ Links ]
[18] Lebars, J., Vedrine, J., Auroux, A., Trautmann, S. and Baerns, M. Role of surface-acidity on vanadia silica catalysts used in the oxidative dehydrogentaion of ethane. Applied Catalysis A-General, 88, pp. 179-195. 1992. [ Links ]
[19] Hubbard, C., Otto, K. and Gandhi, H., Ng K. The influence of sulfur-dioxide on propane oxidation activity on supported platinum and palladium. Catalysis Letters, 30 pp. 41-51. 1995. [ Links ]
[20] Aksoylu, A., Isli, A. and Onsan, Z., Interaction between nickel and molybdenum in Ni-Mo/Al2O3 catalysts: III. Effect of impregnation strategy. Applied Catalysis A-General, 183, pp. 357-364. 1999. [ Links ]
[21] Bourikas, K., Kordulis, C. and Lycourghiotis, A., The role of the liquid-solid interface in the preparation of supported catalysts. Catalysis Reviews-Science and Engineering, 48, pp. 363-444. 2006 [ Links ]
[22] Slezakova, K, Pires, J., Pereira, M., Martins, F. and Alvim-Ferraz, M., Influence of traffic emissions on the composition of atmospheric particles of different sizes-Part 2: SEM-EDS characterization. Journal of Atmospheric Chemistry, 60, pp. 221-236. 2008 [ Links ]
[23] Khwaja, H., Parekh, P., Khan, A., Hershey, D., Naqvi, R., Malik, A. and Khan, K., An In-Depth Characterization of Urban Aerosols Using Electron Microscopy and Energy Dispersive X-Ray Analysis. Clean-Soil Air Water, 37, pp.544-554. 2009 [ Links ]
[24] Lekhal, A., Glasser, B. and Khinast, J., Influence of pH and ionic strength on the metal profile of impregnation catalysts. Chemical Engineering Science, 59, pp. 1063-1077. 2004 [ Links ]
[25] Hao, X., Quach, L., Korah, J., Spieker, W. and Regalbuto, J., The control of platinum impregnation by PZC alteration of oxides and carbon. Journal of Molecular Catalysis a-Chemical, 219, pp.97-107. 2004 [ Links ]
[26] D'souza, L., Regalbuto, J. and Miller, J., Preparation of carbon supported cobalt by electrostatic adsorption of [Co(NH3)(6)]Cl-3. Journal of Catalysis, 254, pp. 157-169. 2008 [ Links ]
[27] Gammons, C., Experimental investigations of the hydrothermal geochemistry of platinum and palladium .5. Equilibria between platinum metal, Pt(II), and Pt(IV) chloride complexes at 25 to 300 degrees C. Geochimica Et Cosmochimica Acta, 60, pp. 1683-1694. 1996 [ Links ]
[28] Zheng, W, and Wu, L., Preparation and pore size shrinkage of palladium-ceramic composite membrane by electroless plating under hydrothermal conditions. Materials Science and Engineering a-Structural Materials Properties Microstructure and Processing, 283, pp. 122-125. 2000. [ Links ]
[29] Hsu, L., Lechler, P. and Nelson J., Hydrothermal Solubility of Palladium in Chloride Solutions from 30-Degrees-C to 700-Degrees-C - Preliminary Experimental Results. Economic Geology and the Bulletin of the Society of Economic Geologists, 86, pp. 422-427. 1991 [ Links ]
[30] Sun, R., Tryk, D., Hashimoto, K. and Fujishima, A., Formation of catalytic Pd on ZnO thin films for electroless metal deposition. Journal of the Electrochemical Society, 145, pp. 3378-3382. 1998 [ Links ]
[31] Kohler, S., Ekerdt, J., Kim, D. and Wachs, I., Relationship between structure and point of zero charge for molybdenum and tungsten supported on alumina. Catalysis Letters,16, pp. 231-239. 1992 [ Links ]
[32] Rojas, H., Martinez, J. and Reyes, P., Kinetics behavior in the hydrogenation of furfural over Ir catalysts supported on TiO2. Dyna, 77(163): p. 151-159. 2010. [ Links ]














