Serviços Personalizados
Journal
Artigo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Acessos
Acessos
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares em
SciELO
Similares em
SciELO -
 Similares em Google
Similares em Google
Compartilhar
DYNA
versão impressa ISSN 0012-7353versão On-line ISSN 2346-2183
Dyna rev.fac.nac.minas v.78 n.170 Medellín dez. 2011
ETHANOL PRODUCTION FROM PAPER SLUDGE USING KLUYVEROMYCES MARXIANUS
PRODUCCIÓN DE ETANOL DE LODOS PAPELEROS USANDO KLUYVEROMYCES MARXIANUS
LINA MARÍA MADRID
Ingeniera Química. Chemical Engineering Department, Universidad de Antioquia, lunanita@gmail.com
JUAN CARLOS QUINTERO DÍAZ
Doctor en ingeniería. Química. Chemical Engineering Department, Associate Professor, Universidad de Antioquia, jcquinte@udea.edu.co
Received for review: September 9th, 2009; accepted: September 13rd, 2010; final version: October 14nd, 2010
ABSTRACT: Recycled paper sludge is a promising raw material for ethanol production. In this study, we first evaluated the effects of ethanol concentration, solids load, and cellulose crystallinity on the enzymatic hydrolysis of cellulose to produce reducing sugars. We then evaluated the production of ethanol by either saccharification and simultaneous fermentation (SSF) or separated hydrolysis and fermentation (SHF) using the yeast Kluyveromyces marxianus ATCC 36907. We found that cellulose hydrolysis decreased as ethanol concentrations increased; at 40 g/L ethanol, the reducing sugar production was decreased by 79 %. Hydrolysis also decreased as solids load increased; at 9 % of solids, the cellulose conversion was 76 % of the stoichiometric production. The ethanol yield and cellulose conversion rate were higher with SSF as opposed to SHF processes at 72 h of treatment.
KEYWORDS: Paper sludge, ethanol, Kluyveromyces marxianus, fermentation, hydrolysis
RESUMEN: El lodo de papel reciclado es una materia prima promisoria para la producción de etanol. En esta investigación, nosotros primero evaluamos el efecto de la concentración de etanol, la carga de sólidos y la cristalinidad de la celulosa sobre la hidrólisis enzimática de la celulosa. También se evaluó la producción de etanol con lodo de papel mediante el proceso de sacarificación y fermentación simultanea (SSF) e hidrólisis y fermentación separada (SHF) empleando la levadura Kluyveromyces marxianus ATCC 36907. El etanol presenta un efecto inhibitorio sobre la hidrólisis de la celulosa. Una concentración de etanol de 40 g/L genera una reducción del 79 % en la producción teórica de azúcares reductores. Al incrementar la concentración de sólidos se observa una reducción en la conversión de la celulosa a azúcares reductores obteniéndose un 76 % de conversión con 9 % de sólidos. Una mayor conversión y productividad en etanol fueron alcanzadas en el proceso SSF en relación al proceso SHF en 72 horas de tratamiento.
PALABRAS CLAVE: Lodo papelero, etanol, Kluyveromyces marxianus, fermentación, hidrólisis
1. INTRODUCTION
Ethanol is currently a predominant liquid fuel to be used as an alternative for gasoline partial substitution. Sugar cane is the principal raw material for carburant ethanol production in Colombia. Although ethanol from lignocellulosic wastes can be produced from a wide variety of agricultural or forest crops, the main disadvantages of these sources are finding an adequate pretreatment that improves the cellulose bioavailability with low-cost collection and storage systems. Recycled paper sludge is a cellulosic waste without these disadvantages.
Paper sludge is formed as a by-product of the production of paper from recycled paper and is composed of very short cellulose fibers which cannot be withheld by paper recycling machines. Fifteen to 20 % of recycled paper contains short fibers, and this percentage is lost in the production of paper, thus generating paper sludge [1]. As the use of recycled paper in Colombia is approximately 500 thousand tons per year, the sludge generation may be from 200 to 300 tons per day. In Colombia, Medellin is a city that generates a large portion of this paper sludge, approximately 100 tons per day [2]. Since recycled paper sludge is composed of approximately 50 % cellulose, has no economic value, and is deposited in landfills, this waste becomes a very attractive raw material for the production of different biotechnological products, such as ethanol [3]. For ethanol production, the cost of the use of cellulose from paper sludge is lower than that which is from native cellulosic materials, because paper sludge does not require triturating and milling operations, and its hydrolysis requires lower enzyme usage. This enzyme usage is considerably less because sludge fibers have a low polymerization degree and the low lignin content reduces enzyme inactivation through adsorption in the surface of sludge fibers [4].
Improved hydrolysis rates and ethanol yields have been achieved using the saccharification and simultaneous fermentation (SSF) process, compared with the two-step process of separated hydrolysis and fermentation (SHF) [5,6]. This improvement occurs mainly because the cellobiose and glucose inhibition over cellulolytic enzymes is reduced. Nevertheless, the ethanol production process using SSF is limited by factors such as: (a) high values of load of solids, potentially generate mixing problems; (b) ethanol concentration, since increasing amounts of ethanol inhibit cellulolytic enzymes; (c) cellulose crystallinity which makes cellulose recalcitrant to enzymatic hydrolysis; and (d) differences in optimal temperatures between enzyme activity and yeast growth. Despite the advantages of paper sludge as a source of cellulose, its biotechnological applications-including ethanol production-have not been evaluated in Colombia.
In the present study, we investigated the effects of ethanol concentration, cellulose crystallinity, and solids load on the enzymatic hydrolysis of cellulose to produce reducing sugars. We also evaluated the ethanol production from recycled paper sludge by either SSF or SHF using a thermotolerant yeast.
2. MATERIALS AND METHODS
2.1 Microorganism
Kluyveromyces marxianus var. marxianus ATCC 36907, a thermotolerant yeast, was kept in malt extract agar at 4 oC. For fermentation cultures, a colony from the agar plate was taken and sown at 38 oC in a culture medium containing 50 g/L glucose, 2.5 g/L yeast extract, 5 g/L peptone, 1 g/L KH2PO4, 0.62 g/L MgSO4.7H2O, and 2.5 g/L (NH4)2SO4.
2.2 Enzymes
Two enzymes were used for the hydrolysis assays. The enzyme Celluclast 1.5 L, produced by Trichoderma reesei, had an activity of 70 FPU/mL. This enzyme catalyzes the hydrolysis of cellulose to glucose, cellobiose, and other polymers of glucose. The other enzyme, Novozym 188, produced by Aspergillus niger, had a β-glucosidase (cellobiase) activity of 750 CBU/mL. Both enzymes were provided by Coldanzimas Ltda. (the representative of Novozymes in Colombia).
2.3 Substrates
Recycled paper sludge samples were composed of 70 % moisture content (wet base), 78.4 % cellulose (dry base), and 21.6 % inorganic material (dry base); samples were supplied by a local paper factory located in Medellin. Microcrystalline cellulose (Merck) and filter paper (Whatman No. 1) were used as models of crystalline and amorphous cellulose, respectively.
2.4 Enzymatic hydrolysis
Hydrolysis assays were carried out in triplicate to evaluate the effects of ethanol concentration, cellulose crystallinity, and solids load on the production of reducing sugars.
The effect of ethanol concentrations (0-40 g/L) on hydrolysis was tested using microcrystalline cellulose (9 % w/v solids load). These assays were performed in 250 mL Erlenmeyer flasks with a solution containing citrate buffer pH 4.5 and 24 PFU Celluclast 1.5L/g of cellulose, at 50 ºC in a shaker at 200 rpm for 5 h. The effect of cellulose crystallinity on hydrolysis was assessed using 9 % w/v of either microcrystalline cellulose or filter paper. The effect of solids load was evaluated using recycled paper sludge between 3 % and 9 % w/v. Crystallinity and solids load assays were carried out in 250 mL Erlenmeyer flasks with a solution containing citrate buffer pH 4.5 and an enzyme combination (Celluclast 1.5 L at 24 UPF/g of cellulose and Novozym 188, with these two enzymes at 10 % v/v Novozym/Celluclast), at 50 ºC in a shaker at 200 rpm for 72 h.
In all these assays, samples were taken periodically and after centrifugation, at 5000 rpm for 10 min, their supernatant liquid was used for the analysis of reducing sugars described in Section 2.7.
2.5 Hydrolysis and separated fermentation
For the first step, hydrolysis, paper sludge samples were prepared in citrate buffer pH 4.5 to achieve a concentration of 6 % w/v, sterilized at 121 ºC for 15 min, and then an enzyme combination (Celluclast 1.5 L at 24 UPF/g of cellulose and Novozym 188, with these two enzymes at 10 % v/v Novozym/Celluclast) was added. The hydrolysis process was carried out in triplicate using 500 mL Erlenmeyer flasks with 190 mL working volume, at 50 ºC in a shaker at 200 rpm for 24 h.
For the second step, fermentation, a yeast inoculum containing the Kluyveromyces marxianus biomass in 10 mL citrate buffer pH 4.5 (this biomass was obtained from 10 mL of yeasts cultured for 17 h, as described in Section 2.1.) was added and supplemented with the reagents indicated in the culture medium in Section 2.1., except glucose, at 38 ºC and 200 rpm for 48 h.
2.6 Saccharification and simultaneous fermentation
Paper sludge samples were mixed with the culture medium (as described in Section 2.1., but without glucose) and citrate buffer pH 4.5 to achieve a concentration of 6 % w/v. This mixture was sterilized at 121 ºC for 15 min and then an enzyme combination (Celluclast 1.5L at 24 UPF/g of cellulose and Novozym 188, with these two enzymes at 10 % v/v Novozym/Celluclast) was added. Subsequently, a yeast inoculum containing the Kluyveromyces marxianus biomass in 10 mL citrate buffer pH 4.5 (this biomass was obtained from 10 mL of yeast cultured for 17 h, as described in Section 2.1.) was added.
Along the SHF and SSF processes, samples were taken periodically for 72 h. These samples were centrifuged at 5000 rpm for 10 min, filtered using 0.45 mm membranes, and we determined their content of ethanol and reducing sugars.
2.7 Analytical methods
Reducing sugars were determined using the dinitrosalicylic acid method and spectrophotometer readings of the developed color at 540 nm, as previously described [7]. Ethanol concentrations were determined by gas chromatography in a Sigma 300 Perkil Elmer chromatograph using a DB-WAX column (30 m x 0.53 mm, 1.0 mm).
3. RESULTS AND DISCUSSION
3.1 Enzymatic hydrolysis
Hydrolysis of crystalline cellulose to reducing sugars was decreased significantly as the initial concentration of ethanol increased (Fig. 1). This finding suggests an inhibitory effect of ethanol on the enzymatic activity of cellulases. The inhibition associated with ethanol was linear between 0 and 20 g/L ethanol, reached 68 % at 20 g/L, and decreased productivity by 79 % at 40 g/L. The same inhibitory effect of ethanol on cellulose hydrolysis had been observed using cellulases produced by P. decumbens and thrichoderma. Since previous studies on cellulase activity had shown that the cellulase enzyme complex consists of endoglucanases, exoglucanases, and b-glucosidases, and that ethanol has no effect on either endoglucanase or b-glucosidase activities [8,9], it can be inferred that ethanol affects the exoglucanase activity of the complex.
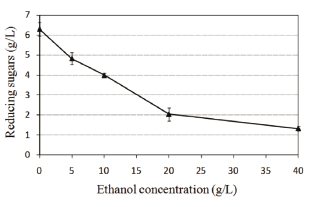
Figure 1. The effect of ethanol concentration on the enzymatic hydrolysis of crystalline cellulose employing the enzyme Celluclast 1.5 L
It has been observed that ethanol modifies the adsorption of exoglucanases on cellulose, reducing its catalytic action, and that ethanol extraction from the enzymatic system restores the activity of exoglucanases [8]. Thus, it can be concluded that ethanol is a reversible inhibitor. To increase the hydrolysis rate of cellulose and to reduce ethanol inhibition, simultaneous ethanol extraction and SSF have been recommended.
The effect of two cellulose structures (amorphous vs. crystalline) on their hydrolysis is shown in Fig. 2. The yield of reducing sugars at 72 h of incubation was 71 % for filter paper (amorphous) and 19 % for microcrystalline cellulose (crystalline). Moreover, the initial rate of hydrolysis was 3.3 times higher with filter paper compared with microcrystalline cellulose. Similar results had been reported previously, i.e., the hydrolysis rates mediated by fungi cellulases had been 3 to 30 times faster using amorphous cellulose compared with crystalline cellulose [10]. Additionally, the degree of cellulose crystallinity inversely correlates with the degree of accessibility to cellulolytic enzymes [11], so strategies such as the addition of surfactants have been used to increase the hydrolysis of crystalline substrates [12].
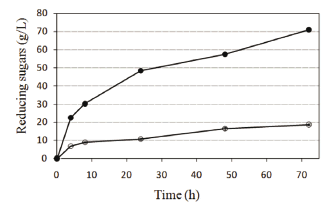
Figure 2. The effect of cellulose structure (amorphous vs. crystalline) on its enzymatic hydrolysis using a combination of the enzymes Celluclast 1.5L and Novozym 188 with 9 % w/v solids load. Symbols: microcrystalline cellulose (○) and filter paper Watman No. 1 (●)
Since recycled paper sludge has both a low polymerization degree and a high degree of machination, suggesting a low crystallinity index, sludge becomes an attractive raw material for fermentable sugar production.
A high solids load concentration is desirable in hydrolytic processes, because it can produce high sugar concentrations and generate high ethanol yields in SSF; however, a high solids load can also generate agitation problems and the inhibition of enzyme by a product.
To test whether the solids load had an effect on the enzymatic hydrolysis of recycled paper sludge, we evaluated solids load concentrations (ranging from 3 % to 9 % w/v) subjected to enzymatic hydrolysis using recycled paper sludge samples (Fig. 3). Reducing sugars concentrations exhibited a typical pattern of batch process, with a quick release of sugars during the first hours of treatment and a subsequent reduction of the generation rate. Sixty-nine percent of the total production of reducing sugars was released within the first 24 h at 3 % substrate concentration; this percentage was 77 % at 9 % concentration. With respect to the stoichiometric production of sugars, the cellulose final conversion of the process decreased from 86 % to 76 % as solids load was increased from 3 % to 9 %, respectively. These findings suggest that the substrate concentration is a limiting factor in the hydrolysis process and are in agreement with what other investigators have reported previously, a decrease in cellulose hydrolysis efficiency as solids load is increased [13]. Also, a 33 % reduction in cellulose conversion when solids concentrations increased from 3 % to 40 % has been reported [14]. This effect can be explained by product inhibition during hydrolysis.
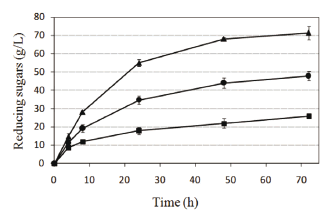
Figure 3. The effect of solids load of recycled paper sludge on its enzymatic hydrolysis at three solids concentrations 3 % (■), 6 % (●), and 9 % (▲)
When assays at 9 % solids load were performed, an inadequate mixing behavior was observed; therefore, an adequate homogenization during our assays cannot be assured. Mixing problems increased as solids load increases, and a considerable increase of mixing power is required above 10 % solids concentration [15,16].
Comparing filter paper conversions (Fig. 2) with recycled paper sludge conversions at 9 % solids (Fig. 3), similar values were observed, 76 % and 71 %, respectively. This similarity suggests that amorphous cellulose is present in paper sludge, a characteristic that makes paper sludge a convenient and promising substrate for processes such as hydrolysis and fermentation.
3.2 Fermentation processes
Recycled paper sludge was subjected to either the SHF or SSF processes. The profiles of reducing sugar concentration and ethanol concentrations over time using either process are shown in Figs. 4 and 5. The reducing sugars concentration reached 33 g/L during the hydrolysis step in the SHF process. After hydrolysis, within the first 6 hours of the fermentation step, sugar was consumed simultaneously for ethanol formation in a nearly stoichiometric relationship. Between 42 and 72 h, the ethanol concentration slowly increased to 12.2 g/L. The sugar concentration also increased because hydrolysis continued in this step and sugar consumption in fermentation was less than sugar generation. The oxygen transfer to the system could cause the reduction in fermentative capacity of yeasts before the first 6 h. Moreover, an 8.2 g/L ethanol concentration was obtained during the first 6 h of treatment using the SSF process (Fig. 5). This ethanol production was above 50 % of the yield at the end of treatment (14 g/L ethanol). An ethanol concentration of 10.5 g/L was reached at 24 h of treatment, equivalent to 85 % of the concentration at 72 h. These results show high ethanol yield in the SSF process beginning in the first hours of treatment. At the end of the process time, the SSF method yielded 59 % ethanol compared to 51 % by SHF. Overall, it can be concluded that the SSF process showed better productivity and conversion of cellulose into ethanol compared with the SHF process, as the findings widely reported. Low capital costs and high ethanol yields are main advantages of the SSF process [17].
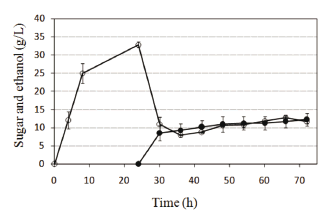
Figure 4. Separate hydrolysis and fermentation (SHF) using recycled paper sludge, a combination of the enzymes Celluclast 1.5L and Novozym 188, and the yeast Kluyveromyces marxianus. Symbols: ethanol (●) and reducing sugars (○)
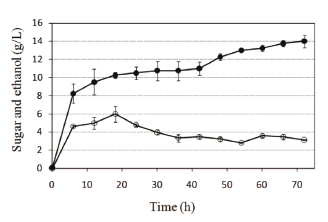
Figure 5. Simultaneous saccharification and fermentation (SSF) using recycled paper sludge, a combination of the enzymes Celluclast 1.5L and Novozym 188, and the yeast Kluyveromyces marxianus. Symbols: ethanol (●) and reducing sugars (○)
In the first hours of the SSF process (Fig. 5), higher ethanol yields were obtained compared with the SHF process, because the enzymatic inhibition was absent (low ethanol and low sugar concentrations). After 6 h, a significant decrease in ethanol production rate was observed from 1.35 to 0.21 g/L per h, probably because of inhibitory ethanol concentrations and a consequent reduction in sugar production. Since the ethanol production rate is proportional to the fermentable sugar concentration, a reduction in the sugar production leads to a reduction in the ethanol production rate. We found that ethanol concentrations above 5 g/L drastically affect cellulose hydrolysis (Fig. 1); thus, in the SSF and SHF processes, this phenomenon could also be present. Alternatively, it had been reported that a high competitive inhibition is exhibited in the presence of glucose by the A. niger enzyme β-glucosidase and its constant inhibition is between 0.3 and 1.2 g/L [18]. High values of glucose concentration, present in both the SSF and SHF processes (according to measured reducing sugars), are inhibitory to cellulose hydrolysis. This inhibition is less in the SSF process because reducing sugar concentrations are between 3 and 4 g/L after 30 h, while values above 10 g/L were obtained in the SHF process in the same time frame.
In summary, we believe that in both the SSF and SHF processes, the relatively high amount of sugar production observed can be reduced by increasing sugar consumption. This reduction could be achieved using yeast inoculums higher than 5 % of what we used in the present study. Also, the simultaneous ethanol production and removal should ameliorate the observed inhibitory effect of ethanol on cellulose hydrolysis.
3. CONCLUSIONS
In our study, cellulose hydrolysis is significantly inhibited at 5 g/L or higher ethanol concentrations; and based on the literature, this is probably through the inhibition of exoglucanases, more precisely, by the reduction of the cellulose adsorption on cellulose fibers in the presence of ethanol. This inhibition seems reversible as it disappears if ethanol is extracted. Therefore, the SSF process could generate higher yields if ethanol is simultaneously removed during fermentation. Furthermore, since amorphous cellulose is more susceptible to enzymatic hydrolysis than crystalline cellulose, it seems that our recycled paper sludge samples had amorphous cellulose and could be easily used to produce reducing sugars. We also observed that the initial rate of paper sludge hydrolysis increases as solids load increases; however, there is less reduction of the sugar yield as solids increase, because of the product inhibition. The SSF process for ethanol production from paper sludge resulted in higher productivity and ethanol concentrations than the SHF process. Since in our studies, using the SSF and SHF processes sugar concentrations were not close to zero, yeast inoculums above 5 % could allow for us to reach sugar concentrations close to zero, thus, eliminating its inhibitory effect on cellulose hydrolysis.
ACKNOWLEDGMENTS
We thank Colciencias for supporting our research through the Young Investigator Scholarship Program. We are grateful to Coldanzimas Ltda. for generously providing the enzymes.
REFERENCES
[1] Lark, N., Xia, Y., Quin, C., Gong, C.S., Tsao G.T., Production of ethanol from recycled paper sludge using cellulase and yeast Kluveromyces marxianus. Biomass Bioenergy, 12, pp. 134-43, 1997 [ Links ]
[2] Quinchía, A., Valencia, M., Giraldo, J., Uso de lodos provenientes de la industria papelera en la elaboración de paneles prefabricados para la construcción. Revista EIA, 8, pp. 9-19, 2007. [ Links ]
[3] Fan, Z., Lynd, L., Conversion of paper sludge to ethanol, II: process design and economic analysis. Bioprocess Biosyst Eng., 30, pp.35-45, 2007. [ Links ]
[4] Tu, M., P, X., Saddler, J., Adsorption of Cellulase on Cellulolytic Enzyme Lignin from Lodgepole Pine. J Agric Food Chem., 57, pp. 7771-8, 2009. [ Links ]
[5] Kádár, Z., Szengyel, Z., Réczey, K., Simultaneous saccharification and fermentation (SSF) of industrial wastes for the production of ethanol. Ind. Crops Prod., 20, pp. 103-10, 2004. [ Links ]
[6] Olofsson, K., Bertilsson, M., Lidén, G., A short review on SSF - an interesting process option for ethanol production from lignocellulosic feedstocks. Biotechnol Biofuels, 1, pp. 1-14, 2008. [ Links ]
[7] Miller, G.L., Use of DNS for determination of reducing sugars. Anal Chem., 31, pp. 426-8, 1959. [ Links ]
[8] Chen, H., Jina, S., Effect of ethanol and yeast on cellulase activity and hydrolysis of crystalline cellulose. Enzyme Microb Technol., 39, pp. 1430-1432, 2006. [ Links ]
[9] Ooshima, H., Ishitani, Y., Harano, Y., Simultaneous saccharification and fermentation of cellulose: Effect of ethanol on enzymatic saccharification of cellulose. Biotechnol. Bioeng., 27, pp. 389-97, 1985. [ Links ]
[10] Zhang, Y.P., Lynd, L.R., Toward an Aggregated Understanding of Enzymatic Hydrolysis of Cellulose: Noncomplexed Cellulase Systems. Biotechnol. Bioeng., 88, pp. 798-824, 2004. [ Links ]
[11] Fierobe, H.P., Bayer, E.A., Tardif, C., Czjzek, M., Mechaly, A., Belaich A. Degradation of Cellulose Substrates by Cellulosome Chimeras. substrate targeting versus proximity of enzyme components. J Biol Chem., 277, pp. 49621-30, 2002. [ Links ]
[12] Mizutani, C., Sethumadhavan, K., Howley, P., Bertoniere, N., Effect of a nonionic surfactant on Trichoderma cellulase treatments of regenerated cellulose and cotton yarns. Cellulose, 9, pp.83-9, 2002. [ Links ]
[13] Zhong, C., Lau, M.W., Balan, V. Optimization of enzymatic hydrolysis and ethanol fermentation from AFEX-treated rice straw. Appl Microbiol Biotechnol., 84, pp. 667-676, 2009. [ Links ]
[14] Jørgensen, H., Vibe-Pedersen, J., Larsen, J., Felby C., Liquefaction of lignocellulose at high-solids concentrations. Biotechnol Bioeng., 96, pp. 862-70, 2007. [ Links ]
[15] Fan, Z., Colin, S., Kimberly, L., Jeffery, M., Peter, Van W., Lee, R.L. Conversion of paper sludge to ethanol in a semicontinuous solids-fed reactor. Bioprocess Biosyst Eng., 26, pp.1615-7591, 2003. [ Links ]
[16] Cara, C., Moya, M., Ballesteros, I., Negro, M.J., Influence of solid loading on enzymatic hydrolysis of steam exploded or liquid hot water pretreated olive tree biomass. Process Biochem., 42, pp. 1003-9, 2007. [ Links ]
[17] Wingren, A., Galbe, M., Zacchi, G., Techno-Economic Evaluation of Producing Ethanol from Softwood: Comparison of SSF and SHF and Identification of Bottlenecks. Biotechnol Progr., 19, 1109-17, 2008. [ Links ]
[18] Yeoh, H.H., Tan, T.K., Chua, S.L., Lim G., Properties of ß-glucosidase purified from Aspergillus niger. MIRCEN Journal, 42, pp. 425-30, 1988. [ Links ]














