Serviços Personalizados
Journal
Artigo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Acessos
Acessos
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares em
SciELO
Similares em
SciELO -
 Similares em Google
Similares em Google
Compartilhar
DYNA
versão impressa ISSN 0012-7353versão On-line ISSN 2346-2183
Dyna rev.fac.nac.minas v.78 n.170 Medellín dez. 2011
ELECTRICAL FURNACE FOR PRODUCING CARBIDE COATINGS USING THE THERMOREACTIVE DEPOSITION/DIFFUSION TECHNIQUE
DISEÑO DE UN HORNO ELÉCTRICO PARA PRODUCIR RECUBRIMIENTOS DE CARBUROS METÁLICOS UTILIZANDO LA TÉCNICA DE DEPOSICIÓN POR DIFUSIÓN TERMORREACTIVA
FABIO CASTILLEJO
PhD (c) en Ingeniería - Ciencia y tecnología de los materiales, Grupo de Ciencia e Ingeniería de Materiales (CIM), Universidad Santo Tomás de Aquino, Colombia, fabiocastillejo@usantotomas.edu.co
DIANA MARULANDA
PhD en Ingeniería - Ciencia y tecnología de los materiales, Grupo de Ciencia e Ingeniería de Materiales (CIM), Universidad Santo Tomás de Aquino, Colombia, dmaritzamc@gmail.com.co
OLIVO RODRIGUEZ
Ingeniero Mecánico, Grupo de Ciencia e Ingeniería de Materiales (CIM), Universidad Santo Tomás de Aquino, Bogotá, Colombia, olivarryyy@gmail.com
JHON OLAYA
PhD en Ciencia e Ingeniería de Materiales, Departamento de Ingeniería Mecánica y Mecatrónica, Universidad Nacional de Colombia, Sede Bogotá, jjolaya@unal.edu.co
Received for review March 28th, 2011, accepted July 18th, 2011, final version August, 16th, 2011
ABSTRACT: In this work, the design of an electrical furnace for producing transition metal-based hard coatings using the thermo-reactive deposition and diffusion (TRD) technique is described. Performance of the system was tested through production of vanadium carbide (VC) and niobium carbide (NbC) coatings on steel AISI D2. X-ray diffraction (XRD) and optical microscopy techniques were used to study phase formation and microstructure, respectively. Hardness was determined by using Knoop microhardness measurements. XRD results showed the presence of VC and NbC, and as MEB results clearly show, the formation of regular thickness coatings. The results obtained allow for assessing that the designed and built furnace fulfills the requirements of the TRD technique for obtaining different types of hard coatings.
KEYWORDS: Thermoreactive deposition and diffusion (TRD), coatings, VC, NbC
RESUMEN: En este trabajo se describe el diseño de un horno eléctrico que permite obtener recubrimientos duros basados en metales de transición, utilizando la técnica de deposición por difusión termorreactiva (TRD). El desempeño del sistema construido se evaluó a través de la producción de recubrimientos de carburo de vanadio (VC) y carburo de niobio (NbC) sobre aceros AISI D2. Se utilizaron las técnicas de Difracción de Rayos X (DRX) y Microscopía óptica para estudiar la formación de fases y la microestructura de los recubrimientos, respectivamente. La dureza se determinó utilizando medidas de microdureza Knoop. Los resultados de DRX confirmaron la presencia de carburo de vanadio y carburo de niobio mientras los resultados de MEB mostraron claramente la formación de recubrimientos con espesor homogéneo. Los resultados obtenidos permitieron verificar que el horno diseñado y construido cumple con los requerimientos de la técnica TRD para obtener recubrimientos duros de diferentes clases.
PALABRAS CLAVE: Difusión termorreactiva, DTR, VC, NbC, horno eléctrico
1. INTRODUCTION
Surface treatments are applied on different materials in order to improve mechanical properties such as microhardness, wear resistance and low friction coefficient, and chemical properties such as corrosion resistance and oxidation resistance [1,2]. The thermo reactive deposition/diffusion treatment is one of the processes used for this purpose that allows for one to obtain carbide, carbonitride, and nitride coatings on steels or substates whose carbon content is higher than 0.3 % atomic weight [1,2,3]. The TRD process may be applied through salt baths [4-7], fluidized bed [8], and pack method [9-10].
The TRD method uses a molten borax bath where carbide/nitride forming elements such as V, Nb, Ti, and Cr are combined with carbon from the substrate to produce a carbide layer with equiaxed or elongated grains [1,2]. As layer growth depends on carbon diffusion, the process requires high temperatures, from 800 °C to 1250 °C, in order to maintain an adequate deposition rate [5]. A carbide layer thickness of 4 to 28 mm may be obtained depending on bath temperature, test time, and substrate material [10]. The coated steels may be cooled and reheated for hardening, or the bath temperature may be selected to correspond to the steel austenitizing temperature, permitting the steel to be quenched directly after coating [1]. Figure 1 shows a schematic of a typical cycle for the TRD process.
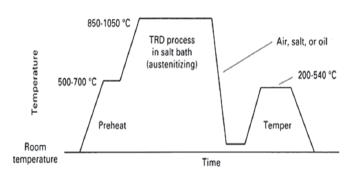
Figure 1. Schematic of typical TRD processing [6]
According to that which was exposited in the previous paragraph, process temperature considerably influences coating properties and therefore furnace characteristics will also influence deposition conditions and coating properties. This is why this work presents the design to build an electric furnace to produce coatings using the TRD technique fulfilling process requirements. In addition, in order to assess furnace performance, vanadium carbide (VC) and niobium carbide (NbC) coatings were obtained on AISI D2 steel and they were characterized to study phase formation, thickness, and microhardness.
2. EXPERIMENTAL SETUP
2.1 Furnace design
The furnace geometry selected for the design was a cylindrical shape with 1200 °C maximum working temperature. The furnace dimensions were 0.06 m in radius and 0.176 m in height, and using these dimensions a furnace working volume of 1922 cm3 was obtained. This volume requires 1500 W power at 1200 ºC [11], and it was used to calculate the electrical resistance needed at room temperature, obtaining 8.06 W. As the furnace must operate at high temperatures, kanthal (an iron-chromium-aluminum alloy) was selected as a resistance material because it improves high temperature performance where common metallic elements may cause problems, and also because its operation temperature and lifetime are higher than nikel-chromium alloys [11].
The following expression was used in order to calculate kanthal resistance at the maximum working temperature (Rt) [11]:

where R is the room temperature resistance and Ct is the kanthal resistivity coefficient at 1200 ºC [11]. A resistance value of 8.32 W at 1200 ºC was obtained. After this step, the wire length was determined in such way that it fulfilled the electric requirements: When using kanthal, wire diameter in function of power and voltage requirements (110 VAC) is recommended. Taking this into account, a wire diameter in the range from 1.290 to 2.052 mm should be used [11]. In this work, 1.5 mm was used as middle value, because lower diameters showed a short lifetime while greater diameters delayed the furnace heating process due to the decrease in kanthal current.
The wire length was calculated using the wire resistivity (ρ = 1.45 x 10-6 W m [11]) through the following expression:

obtaining 10.21 m wire length. This resistive wire length (kanthal) was coiled with 16 mm internal diameter and it was distributed in ten high alumina ceramic cores with a 15 mm diameter (Fig. 2).
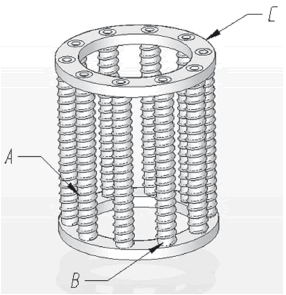
Figure 2. Furnace's internal ceramic structure. A: Kanthal, B: cores, C: core support
The furnace's insulating ceramic fiber thickness (Fig. 3) was determined taking the Fourier equation for heat conduction in steady state into account [12]:

Where 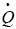 is the heat per time unit, K is the thermal conductivity coefficient, A is the area where heat flows, and the differential corresponds to temperature gradient.
is the heat per time unit, K is the thermal conductivity coefficient, A is the area where heat flows, and the differential corresponds to temperature gradient.
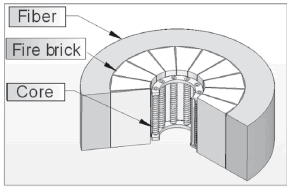
Figure 3. Furnace isometric view
The cylindrical geometry and radius distribution shown in Fig. 4 were used in order to solve (3) and the result is shown in expression (4).
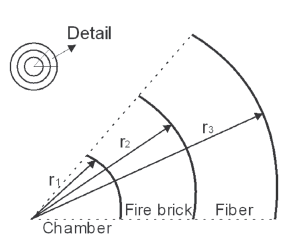
Figure 4. Cylindrical geometry of the designed furnace
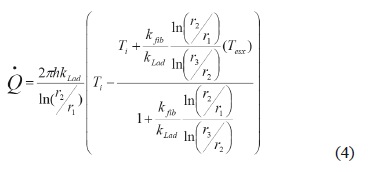
In expression (4), h is furnace height; kLad and kfib are thermal conductivity coefficients of fire brick and ceramic fiber, respectively; r1, r2, and r3 are the internal radius, the radius from the center to the fire brick, and the radius from the center to the furnace exterior surface, respectively (Fig. 3); Ti is the internal temperature and Text is the temperature on the furnace external surface. Considering an internal temperature of 1200 ºC and a 50 ºC external temperature, and taking the manufacturer constants (kLad = 0.36 W/m°C, kfib = 0.29 W/m°C) into account, heat loss through the furnace external walls was calculated, and this value was used in order to calculate the kW/h cost when using the furnace during 16 consecutive hours. The ceramic fiber's thickness was determined by taking into account the cost for furnace manufacturing and the cost related to the calculated kW/h for heat losses. Iterations were performed for the two costs and the result is shown in Fig. 5. The intersection point was taken as a reference point, selecting 3 cm of fiber thickness. Using this thickness, approximately 300 W of heat losses is expected.
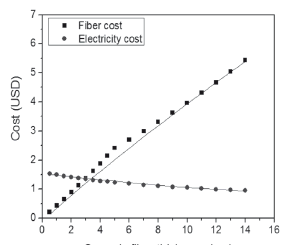
Figure 5. Costs in the function of ceramic fiber thickness. Costs are given in United States dollars (USDs).
2.2. Sample preparation
The designed and built furnace was used to produce NbC and VC coatings on AISI D2 steels (1.48 % C, 11.92 % Cr, 0.96 % Si, 0.45 % Mn, 0.75 % V, Fe balance). Before the treatments, cylindrical samples measuring 12 mm in diameter and 4 mm of thickness were ground up to 1200 mesh emery paper until they achieved a mirror-like finish. Subsequently, the samples were cleaned in acetone and dried in sequence using compressed air before starting thermal treatment.
The treatments for producing VC were carried out in a molten mixture constituted by 16 % ferro-vanadium (Fe-V), 3 % aluminum, and 81 % borax (Na2B4O7). The treatments for producing NbC coatings were performed in a molten mixture constituted by 16 % ferro-niobium (Fe-Nb), 3 % aluminum, and 81 % borax. The samples were treated at 1050 ºC for 4 h and were quenched in a salt bath directly from the molten mixture. Eight treatments were carried out in order to obtain average values.
2.3. Characterization of the layers
The crystallographic phases of the coatings and preferential orientations were identified by x-ray diffraction using an X-PertPro Panalytical system (q-2q configuration, 45 kV, 40 mA, and monochromatized CuKa radiation with a wavelength of 1.56 amstrongs). The step size was 0.02° with a 40 s step time.
Cross-sectional samples were polished and etched with Vilella's reagent at 3 % for observation through optical microscopy and hardness measurements. Thickness measurements were performed using an optical microscopy Leco while microhardness measurements were carried out by applying a 0.24 N load. Both values were determined as the average of 9 measurements.
3. RESULTS AND DISCUSSION
Figures 6 and 7 show diffraction patterns for VC and NbC coatings, respectively. In the first case, it was verified that the layer consisted of vanadium carbide (VC) with high intensity peaks in orientations (111) and (200), and low intensity peaks in orientations (200), (311), and (222) according to database JCPDS 01-073-0476 with crystalline structure fcc. In the second case, the layer consisted of niobium carbide (NbC) with high intensity peaks in orientations (111) and (200), and low intensity peaks in orientations (110), (220), (311), and (222) according to database JCPDS 00-038-1364 with crystalline structure fcc. The formation of iron boride (FeB) was also observed in low proportion with orientations (020), (111), and (021) (JCPDS 00-032-0463) in the latter case. Orientation (021) is visible through deconvolution (not shown) of the peak, located around 44.8º. FeB formation could be due to the thermochemical reduction in the molten mixture which freed boron atoms to diffuse into the substrate and to react with the iron of the base metal [13]
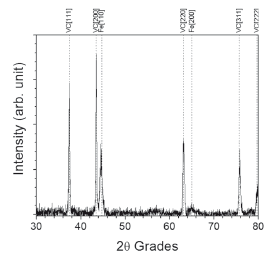
Figure 6. XRD pattern for VC coating
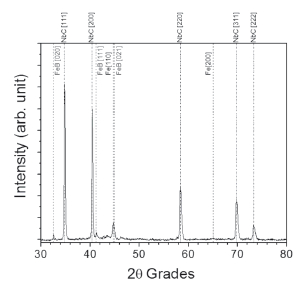
Figure 7. XRD pattern for NbC coating
Figure 8 (a) and (b) show cross-sectional micrographs for the coatings, and in both cases it is observed that the layers were homogeneous with thickness regularity. Vanadium carbide layer thickness was 11.93 ± 0.32 mm while the niobium carbide layer thickness was 10.75 ± 0.3 mm.

Figure 8. Optical micrographs of TRD treated AISI D2 in the built furnace, (a) vanadium carbide, (b) niobium carbide
The hardnesses of the layers were 2270.9 ± 46.88 HK for VC, and 2180.5 ± 25.5 HK for NbC. Some indentations performed for these measurements can be observed on the micrographs (Fig. 8). These levels of hardness are in the range expected for VC layers [1] and NbC layers [14]. Uncoated AISI D2 was tempered after the TRD treatment, presenting a hardness of 686, 13 ± 37 HK which is within the hardness values reported in the literature [15].
4. CONCLUSIONS
In this study, the design of an electrical furnace to produce coatings through the use of the thermo reactive deposition/diffusion technique was described. Vanadium carbide and niobium carbide coatings were successfully produced in order to assess furnace performance.
In the baths containing ferroniobium or ferrovanadium, the layers presented regular thickness. In the first bath, the layer is made up of NbC, with a hardness of 21809 ± 25 HV and a thickness of 10.75 ± 0.4 mm; while in the second case, the layer is made up of a VC measuring 2270.9 ± 46 HK of hardness and 11.93 ± 0.3 mm thickness.
Through the use of this equipment and based on the obtained results, future research will be conducted to study the properties of the coatings, such as corrosion and wear resistance.
ACKNOWLEDGMENTS
This research has been financially supported by The Fundación para la promoción de la ciencia y la tecnología of the Banco de la República through Project #2608.
REFERENCES
[1] Olaya, J.J., Rodil, S.E, Marulanda, D.M., Recubrimientos de nitruros metálicos depositados con ubm: tecnología eficiente y ambientalmente limpia, DYNA, Vol. 77, pp. 60-69, 2010 [ Links ]
[2] Fan, X.S., Yang, Z.G., Xia, Z.X., Zhang, C. and Che, H.Q., The microstructure evolution of VC coatings on AISI H13 and 9Cr18 steel by thermo-reactive deposition process. Journal of Alloys and Compounds, 505, L15-L18, 2010. [ Links ]
[3] Nakanishi, K., Takeda, H, Tachikawa, H. and arai, t., Fluidized bed carbide coating process-development and its application, The 8th International Congress on Heat Treatment of Materials, Heat and Surface 92, Kyoto Nagoya, Japan, 1992. [ Links ]
[4] Arai, T. and Komatsu, N., Carbide Coating Process by Use of Salt Bath and its Application to Metal Forming Dies, in Proceedings of the 18th International Machine Tool Design and Research Conference, 14-16, pp. 225-231, Sept 1977. [ Links ]
[5] Arai, T., Carbide Coating Process by Use of Molten Borax Bath in Japan, J. Heat Treat., Vol. 18 (2), pp. 15-22, 1979. [ Links ]
[6] Arai, T., Fujita, H., Sugimoto, Y. and Ohta, Y., Vanadium Carbonitride Coating by Immersing into Low Temperature Salt Bath, in Heat Treatment and Surface Engineering, George Krauss, Ed., ASM International, pp. 49-53, 1988. [ Links ]
[7] Campbell, I.E., Barth, V.D., Hoeckelman, R.F. and Gonser, B.W. Salt Bath Chromizing, J. Electrochem. Soc, Vol 96 (4), pp. 262-273, 1949. [ Links ]
[8] Arai, T., Endo, J. and Takeda, H., Chromizing and Boriding by Use of a Fluidized Bed, in Proceedings of the 5th International Congress: Heat Treatment of Materials Conference, pp. 1335-1341, 20-24 Oct 1986. [ Links ]
[9] Glowachi., Z. and Jastrzebowski, K., Karbidbildungen und-umwandlungen beim Vanadieren, Neue Hütte, Vol 29 (6), pp. 220-222, 1984. [ Links ]
[10] Xiujuan L., Huachang, W., Dongwei L., Yanxi W., School of Material Science and Engineering, Wuhan University of Technology, Wuhan 430070, China. Surface and coatings Technology 201, pp. 2414-241, 2006. [ Links ]
[11] Kanthal Handbook, Heating alloys for electronic household appliances, Sweden, 2003. [ Links ]
[12] Cengel, Y. Heat transfer, a practical approach. Second edition. McGraw-Hill, 2003. [ Links ]
[13] Matiasovský, K., Chrenková-Paucírová M., Fellner, P. and Makyta, M., Surf. Coat. Technol. 35 pp. 133-149, 1988. [ Links ]
[14] Oliveira, C.K.N., Muñoz Riofano c, R.M. and Casteletti, L.C., Surface & Coatings Technology 200, pp. 5140 - 5144, 2006. [ Links ]
[15] Oliveira, C.K.N., Benassi C.L. and Casteletti, L.C., Surf. Coat. Technol. 201, pp. 1880 -1885, 2006. [ Links ]














