Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
DYNA
Print version ISSN 0012-7353On-line version ISSN 2346-2183
Dyna rev.fac.nac.minas vol.79 no.172 Medellín Mar./Apr. 2012
BIOLEACHING OF COPPER SULPHIDE FLOTATION CONCENTRATE IN BATCH REACTION SYSTEM USING MESOPHILE AND THERMOPHILE MICROORGANISMS
BIO-LIXIVIACIÃN DE CONCENTRADO DE FLOTACIÃN DE SULFUROS DE COBRE EN SISTEMA DE REACCIÃN EN BARCADA UTILIZANDO MICROORGANISMOS MESÃFILOS Y TERMÃFILOS
RENATA DE BARROS LIMA
D.Sc. Chemistry, Federal University of Rio de Janeiro, Brazil, renatadbarros@gmail.com
SELMA GOMES FERREIRA LEITE
D.Sc. Chemical Engineering, Federal University of Rio de Janeiro, Brazil, selma@eq.ufrj.br
GABRIELLA SILVA FRANCISCO PEREIRA
Chemical Engineering, Federal University of Rio de Janeiro, Brazil, bibipereira@yahoo.com.br
ISABELLA CESARIO DE AMARAL
Chemistry, Technical Educational Foundation Souza Marques,Rio de Janeiro, Brazil, isacesario@gmail.com
LUIS GONZAGA SANTOS SOBRAL
Ph.D. Chemical Engineering, CETEM â Centre for Mineral Technology, Brazil, Lsobral@cetem.gov.br
Received for review April 7th, 2011, accepted August 10th, 2011, final version August, 28th, 2011
ABSTRACT: This work aims at optimizing the use of consortiaof mesophile and thermophile microorganisms (moderate and extreme ones) in the leaching process of copper sulphidesflotation concentrate, containing approximately 30% bornite (Cu5FeS4) and 70% chalcopyrite (CuFeS2), for extracting copper.The objective of the bioleaching tests was to evaluate the performance of those microorganisms, varying specific parameters such as: the composition of the leaching solution, the presence of an energy source (ferrous sulphate), and the inoculation of cultures . To ensure optimal conditions for the microorganisms to act, the pH, temperature, and stirring speed were controlled. Finally, from the microbial action and using agricultural fertiliser (N:P:K sources), a copper extraction of more than 85% was achieved.
KEYWORDS: Bioleaching, copper, sulphide, chalcopyrite, acidophilic microorganism.
RESUMEN: Este trabajo tiene como objetivo optimizar el uso de consorcios de microorganismos mesofÃlicos y termofÃlicos (moderados y extremos) en el proceso de lixiviación de concentrado de flotación de sulfuro de cobre, conteniendo aproximadamente 30% de bornita (Cu5FeS4) e 70% de calcopirita (CuFeS2), para la extracción de cobre. El objetivo de los experimentos de la biolixiviación fue evaluar el desempeño de esos microorganismos, variando parámetros especÃficos como: composición de la solución lixiviante, presencia de fuentes de energÃa (sulfato ferroso) y la inoculación de culturas. Para asegurar las condiciones óptimas para la actuación de los microorganismos fueron controlados valores de pH, temperatura, y velocidad de agitación. Finalmente, por acción microbiana y utilización de fertilizantes agrÃcolas (fuente de N:P:K), la extracción de cobre alcanzada fue superior a 85%.
PALABRAS CLAVE: Biolixiviación, cobre, sulfato, calcopirita, microorganismos acidófilos
1. INTRODUCTION
The copper extraction from copper sulphides flotation concentrates follows the conventional technological process due to their specific mineralogical characteristics. Thus, the copper sulphides present in the aforementionedconcentrates are directly converted into blister copper (impure metallic copper) by the flash smelting process, and further electro-refined.Although the pyrometallurgical process presents the advantage of transforming the distinct copper sulphides into metallic copper in a single step, it however hasthe disadvantages of generatingsulphur dioxide (SO2) along withheavy metal emissions such as cadmium, arsenic, mercury, bismuth, selenium, etc. Such a gas stream requires a specific treatment in order to avoid issuing these heavy metals into the environment.
The bioleaching treatment of sulphide concentratesis more attractive than the pyrometallurgical process as it eliminatesgas emissions.In the hydrometallurgical treatment, the copper, along with a large part of the metallic impurities, is released as soluble sulphate. The copper ions are recovered from leaching solution either by cementation or purification/concentration (solvent extraction), and followed by electrowinning.
Bioleaching consists of a natural process of sulphide dissolution, resulting from the action of a group of microorganisms that oxidizes sulphide minerals, releasing the metals in their soluble ionic species. Examples of metals which may be extracted by bioleaching are:copper, through chalcopyrite (CuFeS2), bornite (Cu5FeS4) and covellite (CuS); uranium through uraninite (UO2) [1].
The majority of secondary copper sulphides, such as digenite, bornite, and covellite, may be successfully bioleached by mesophile microorganisms. However, chalcopyrite leaching, chalcopyrite being the main copper sulphide mineral, is still a challenge due to slow extraction kinetics [2].
In general, one of the most common solutions forthe slow kinetics of the bioleaching processes is raising the temperature. However, the use of extreme thermophile microorganisms may presenta series of difficulties such as: low oxygen solubility in water; less tolerance to high concentrations of metals and catalysts, such as silver; and presenting lower resistance to the shearing of their cell membranes, than themesophile microorganisms [3â4].However, the industry has preferred the use of moderate thermophile microorganisms, as they are more resistant to high pulp densities and high heavy metal concentrations, when compared with extreme thermophile microorganisms [5].The use of bioleaching at high temperatures and thermophile microorganisms increases the copper extraction efficiency in comparison with those obtained by using mesophile onesat room temperature. The typical copper extraction obtained by chalcopyrite bioleaching with mesophile microorganisms is nearly 30%, while copper extraction higher than 90% may be obtained in a shorter period of time using the thermophile ones [6].
The leaching of chalcopyrite is an acid consumption reaction. Thus, as the leaching reaction goes on, a consequent increase of pH takes place, which provokes the precipitation of ferric ionssuch as jarosite. Therefore, lower pH values may result in higher copper extractionand lowerjarosite precipitation [6]. Thus, a constant monitoring and maintenance of pH valuesis necessary.The optimum and minimum pH values for the growth of microorganisms used in the bioleaching process are directly linked to the species used, in fact that pH 2.5(optimun) and 1.3(minimum) values for A. ferrooxidans and pH 1.7/ 1.0values for L. ferrooxidans[7].
A wide range of sulphides may be oxidized by the microorganisms found in acid rock drainage from mined areas. These microorganisms may act in an expanded temperature range (from 30 to 70 oC), acid pH range (1.8 to 2.2) and high Fe3+/Fe2+ ions ratios [8].
The bioleaching of sulphide minerals is based on the ability of acidophilic microorganisms in oxidizing ferrous ions (reaction 1) and/ or reducing sulphur (reaction 2). The bioleaching processes may lead to significant heat generation produced by sulphide oxidation exothermal reactions. In the leaching solution, temperature values from 50 to 80 oC were observed [9, 10,11].
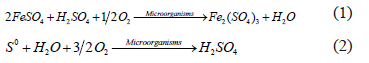
What makes the bioleaching process a very interesting alternative for replacing the conventional processes is the ability of certain iron or sulphur oxidant microorganisms, such as the genders Acidithiobacillus and Leptospirillum, which grow in highly acid environments and in the presence of heavy metals. In addition, if we compare the costs of the bioleaching process with the costs of the operation of a conventional plant, it is possible a reduction of up to 50% [12].
The microorganisms used in bioleaching acquire energy through the oxidation of sulphur reduced compounds and/or ferrous ions, speeding upthe copper extraction by generating ferric ions and protons, which together form the oxidant environment for the sulphide mineral. The dissolution of sulphide can occur through the indirect mechanism (contactless), in which the ferric ion and protons in solution, attack the mineral; and the mechanism by direct contact, where the dissolution of the mineral occurs by the contact of those microorganisms with the mineral surface [13], as shown in Figure 1.
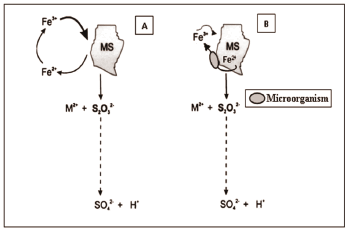
Figure 1. Bioleaching mechanisms: [A] Indirect and [B] direct contact. [apud14].
2. OBJECTIVE
This work aims at evaluating the use in separate of the agricultural fertiliser N:P:K (10:10:10)and P. A. chemical reagents (MKM culturemedium) in presence and absence of ferrous sulphate, extracting copper from a flotation concentrate containing chalcopyrite (CuFeS2) and bornite (Cu5FeS4) during the bioleaching process.
3. EXPERIMENTAL
3.1. Flotation Concentrate
The flotation process is known as one of the methods that can separate minerals based on differences of surface properties of minerals. In this process, the minerals particles, with hydrophobic surfaces, join the air bubbles generated on the lower part of the flotation column and fluctuate to form foam on the top of the column. On the other hand, the particles with hydrophilic surfaces do not join the bubbles and sink, forming a tail [15].
The copper sulphides flotation concentrate used contains approximately 30% of bornite (Cu5FeS4) and 70% of chalcopyrite (CuFeS2). Thus, it is perfectly possible that microorganisms obtain their sources of energy from that flotation concentrate, where, with the oxidation of chalcopyrite by oxygen in the air, the formation of ferrous sulphate (3) takes place â source of Fe2+; however, this ferrous sulphate is rapidly oxidized by oxygen in the presence of microorganisms, producing ferric sulphate (4), which attacks the chalcopyrite forming, still, more ferrous sulphate, repeating, thus, the oxidation cycle (5) [16]:
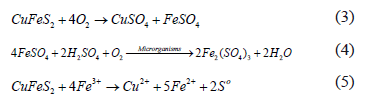
On the other hand, the source of sulphur can be obtained through generating elemental sulphur from the indirect dissolution of chalcopyrite, where it is oxidized by microorganisms to sulphuric acid, which maintains the iron in solution (6) [16], as shown in the following:
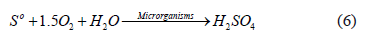
3.2. Microorganisms and culture medium. For accomplishing this study three microbial consortia were used:
- Mesophiles:Leptospirilum sp., Acidithiobacillus ferrooxidans e Acidithiobacillus thiooxidans;
- Moderate Thermophiles: Species not informed;
- Extreme Thermophiles: Acidianus brierleyi DSM 1651 and 6334, Acidianus infernus DSM 3191, Metallosphaera sedula ATCC 33909, Sulfolobulus metallicus DSM 6482, Sulfolobulus acidocaldarius ATCC 49426, Sulfolobulus shibatae DSM 5389.
3.2.1. Mesophile Microorganisms
The mesophile microorganisms of Acidithiobacillus genus were represented by species Acidithiobacillus ferrooxidans and Acidithiobacillus thiooxidans. This group of microorganisms operates in temperature ⤠40 oC and are the microorganisms more commonly used in bioleaching of sulphide minerals [17].
The Acidithiobacillus ferrooxidans strain oxidizes, besides ferrous ions, sulphur (compounds) and grows in the pH range of 1.0 to 6.0. The optimum pH, to achieve the maximum growth speed, is between 2.0 and 2.5.In relation to temperature, the microorganisms survive in a temperature interval from 2 to 40 oC, being the optimum range from 28 to 35 oC.[18].
Microorganisms of the species Acidithiobacillus thiooxidans oxidize sulphur and act in an optimum temperature range from 25 to 30 oC and optimum pH of 2.0 for its maximum growth.
The source of energy for such microorganisms can be derived from the oxidation of one or more reduced sulphur compounds, being sulphate obtained as a result of that oxidation [19].
3.2.1.1. Composition of the culture medium for mesophile microorganisms
In general, the species A. ferrooxidans may be grown in T& K culture medium [20] and 9K [21]. As the tests carried out use the two species together (Acidithiobacillus ferrooxidans and Acidithiobacillus thiooxidans), it is necessary that only one type of culture medium be used. Therefore, the microorganisms were grown in 9K culture medium, where only the source of energy is different.
Composition of 9K medium [21]: (NH4)2SO4: 4.28 g/L, K2HPO4: 0.71 g/L, MgSO4.7H2O: 0.71 g/L, KCl: 0.14 g/L and Ca(NO3)2: 0.014 g/L; source of energy: FeSO4.7H2O: 147.4 g/L, for cultivating A. ferrooxidans and So: 1.0 g/L, for cultivating A. thiooxidans. Such medium is acidified with 10N H2SO4 up to pH 1.8 before using it.
3.2.2. Thermophile Microorganisms
A consortium acidophilic moderate thermophiles microorganisms was isolated directly from the flotation concentrate. Currently, the group of moderate thermophile microorganism includes mainly the following microorganisms: Acidithiobacillus caldus, Leptospirillum ferriphilum, Acidimicrobium ferrooxidans Sulfobacillusthermosulfidooxidans and Sulfobacillus acidophilus. They can grow by oxidizing ferrous ions, sulphur and/or sulphide minerals. The optimum growth temperature and pH ranges are from 45 to 60 oC and from 1.3 to2.5, respectively [22].
The majority of extreme thermophile microorganisms is classified as Archaea and survive in temperatures higher than 60oC. Almost always, have a fragile cell wall, which prevents them to survive in stirred systems, in the presence of solid, due to the consequent shear effect that occurs on their cell wall, limiting their industrial application [23]. In addition to these aggravating factors, other factors contribute to make things even worth for using such microorganisms, such as reducing the solubility of oxygen in the leaching solution and the low resistance to high concentrations of metals (high ionic strength) and catalysts as silver [5].
In the growth of extreme thermophile microorganisms, the following strains were used: Acidianus brierleyi, Acidianus infermus, Metallosphaera sedula, Sulfolobulus metallicus, Sulfolobulus acidocaldarius, Sulfolobulus shibatae. The optimum temperature for growing those microorganisms is 68 oC.
3.2.2.1. Composition of culture medium for thermophile microorganisms
The MKM culture medium, Modified Kelly Medium, [24], which contains mineral salts and iron (from the flotation concentrate, which contains around 70% of chalcopyrite (CuFeS2) and 30% of bornite (Cu5FeS4)) as a source of energy, it is commonly used for the cultivating moderate and extreme thermophiles microorganisms.
The composition of MKM culture medium: (NH4)2SO4: 0.4g/L, MgSO4.7H2O: 0.4g/L, K2HPO4: 0.04g/L, energy source: FeSO4.7H2O: 10g/L, So: 5g/L, Chalcopyrite (CuFeS2): 10g/L, Pyrite (FeS2):5g/L and yeast extract: 0.2 g/L (only in thermophile microorganisms cultivation). The pH of the culture medium was adjusted to 1.7 using concentrated sulphuric acid. The inoculation of microorganisms in the culture medium was accomplished in a 10% v/v ratio [25].
3.3. Bioleaching tests using mesophiles, moderate and extreme thermophile microorganisms
The mesophile microorganisms begin the bioleaching process. Subsequently, the moderate thermophiles are used, and, finally, followed by the extreme thermophile, according to the experimental conditions listed in the Table 1 as follows:
Table 1 âExperimental conditions used in the stirred flask bioleachingwith 1% of pulp density (w/v).

The tests were carried out in Erlenmeyer flask of 250 mL capacity. In each flask 1.5 g of copper concentrate and 150 mL of leaching solution were added.
In each new stage, the flotation concentrate was filtered and reused, maintaining the solid/liquid ratio in 1%.
The tests for evaluating the performance of the microorganisms were accomplished varying the following parameters:
- Salty solution (commercial agricultural fertilizer N:P:K â10:10:10 â in tap water/ chemicals in deionised water);
- Culture medium composition in relation to energy source (with/ without Fe2+);
- Presence of microbial inoculum.
Being the pH adjusted, periodically, in the range of 1.5 to 1.8 and the stirring speed maintained at 150 rpm. The temperature, in each test, was kept constant in accordance with the microorganisms used. The tests were carried out in duplicate.
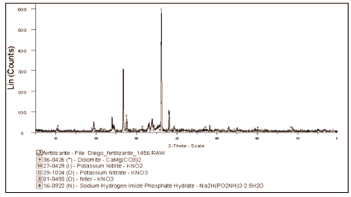
Figure 2. X-ray difratograms of fertilizer sample
4. RESULTS AND DISCUSSIONS
4.1. Analysis of the microorganisms activity
4.1.1. Considering the redox potential (Eh) and pH:
The redox potential is related with the ferric/ferrous ions ratio by the Nernst equation:
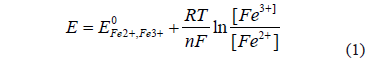
Ferrous ions, from the chalcopyrite dissolution and the culture medium-bearing ferrous sulphate, are oxidized by microorganisms to ferric ions through the following equations:
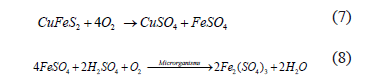
Thus, it is realized that the ferric/ferrous ions concentrations ratio is increasing, which canbe observed by monitoring the redox potential.that increases during the leaching process.
Figure 3, shows that the lowest values of redox potential are observed in the control tests curves, without ferrous sulphate and without microbial inoculum, for both fertiliser and for chemicals. However, the peak values correspond to the tests where inoculation was done.
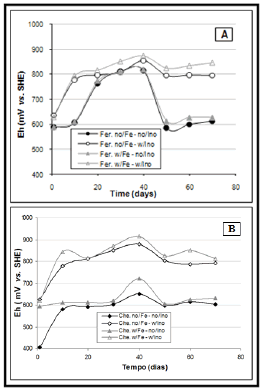
Figure 3. Redox potential readings (mean values), in mV vs. SHE, in the bioleaching tests using agriculture fertilizer (A) and chemicals (B).
Among the tests with inoculum, the effect of adding ferrous sulphate to the culture medium could be analyzed. As the Fe2+ concentration increases due to the sulphide oxidation, more ferric ions are produced and, therefore, the Eh values are higher. When the curve goes down, as observed on Figure 3, is due to the onset of another test, when a new culture is used.
Therefore, through the redox potential analysis, it is possible to infer the importance of the microorganisms regarding to ferrous ions oxidation.
In the bioleaching tests, all microorganisms used are acidophilus. Therefore, to keep the pH in the right range was of utmost importance for thatextractive process to take place.
The pH values in the plots of Figure 4, below, correspond to the measurement of it before adjusting it in the optimum range of 1.5 to 1.8 for evaluating the microorganismsâ activity. This is due to the oxidation of elemental sulphur, formed during the chalcopyrite dissolution, to sulphuric acid by the microorganisms, increasing the acid content of the leaching solution, according to the following reactions:
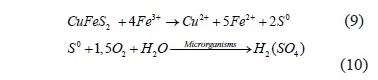
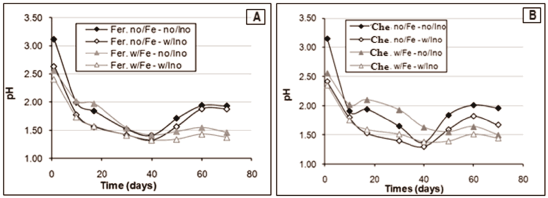
Figure 4. Initial pH values (mean values) of the bioleaching tests using agriculture fertilizer as nutrient source (A) and chemicals (B)
In the tests containing microbial inoculum and ferrous sulphate lower pH values were observed, due to a higher Fe3+ concentration (oxidation product), by shifting the reaction (9) toward generating elemental sulphur, producing consequently sulphuric acid (10) as the oxidative process goes on [26].
4.1.2. Considering the Ferrous and ferric concentrations:
Figure 5, below, highlights the leaching process, using agriculture fertiliser in tap water, ferrous sulphate and microbial inoculum.
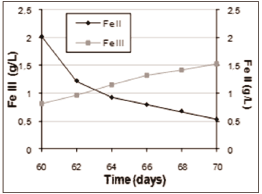
Figure 5. Measurement of Fe2+ e Fe3+ ions concentrations, in g/L, in the leachate produced during the bioleaching process using extreme thermophile microorganisms.
following the ferrous and ferric ions concentrations during the use of extreme thermophile microorganisms, it is possible to conclude that the Fe2+ concentration decreases, as the Fe3+ concentration increases. This is due to the oxidation of ferrous ions by the microorganisms present in the bulk, according toequation (8). Such oxidation is shown in Figure 3, where it is possible to observe an increase in redox potential (819 mV vs. SHE to 840 mV vs. SHE) during the aforementioned test, using agriculture fertilizer, with ferrous sulphate and microbial inoculums. This increase in the ferric ions concentration is important for extracting copper, according to reaction (9), with consequent shifting of equilibrium toward Cu2+ generation.
4.1.3. Considering the copper extraction:
According to the results of redox potential, pH and ferric and ferrous ions concentrations, the test where agricultural fertiliser was used as NPK source, presented better copper extraction â 86%.
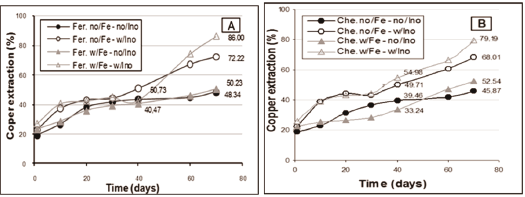
Figure 6. Copper extraction (mean values) during the bioleaching process using agriculture fertiliser as source of nutrients (A) and chemicals (B).
This result is corroborated by the difratogram shown in Figure 7 B, where it is observed a marked reduction in the chalcopyrite peak, when compared with the difratogram of Figure 7 A.
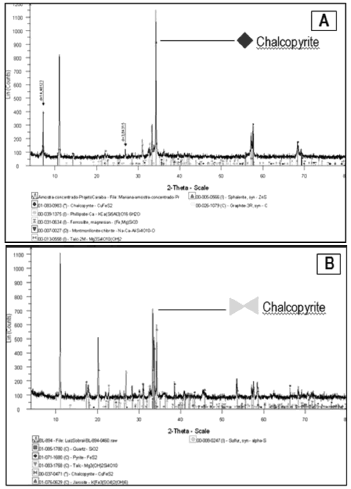
Figure 7. Difratograms referring to semi-quantitative analysis of the mineral species comprising the flotation concentrate before the bioleaching test (A) and after that process (B), using agriculture fertiliser as source of nutrients.
5. CONCLUSIONS
The performance of the reaction system using both chemical reagent and agriculture fertilizer as sources of nutrientsfor the microbial inoculum, in the bioleaching process, increased the copper extraction to 11% and 14%, respectively.
However, the commercial agriculture fertiliser presented better performance in copper extraction, which was of 86%,in comparison to 79% forchemical reagent . Thisis an indication of the possibility of using such fertilizer in a culture medium, waving for being usedin an industrial scale bioleaching process.
The addition of the ferrous sulphate, either using agriculture fertiliser or chemicals, in theexperiments, increased the ratio of copper extraction. In this context, the result suggested that the ferrous ions were available for microbial oxidation as an alternative source of energy in the process.
REFERENCES
[1] Rojas, J. J. G.,Biotecnologia en la Disolucion y Recuperacion de Metales. In: Congresso Peruano de Biotecnologia y Bioingenieria, Trujillo, Peru, 1998. [ Links ]
[2] Zhou, H., Zeng, W., Yang, Z., Xie, Y. and Qiu, G., Bioleaching of chalcopyrite concentrate by a moderately thermophilic culture in a stirred tank reactor. Bioresource Technology 100, pp. 515â520 (2009) [ Links ]
[3] Okibe, N., Gericke, M., Hallberg, K.B. and Johnson, D.B., Enumeration and characterization of acidophilic microorganisms isolated from a pilot plant stirred-tank bioleaching operation. Applied and Environmental Microbiology 69, pp. 1936â1943, 2003. [ Links ]
[4] Olson, G.J. and Clark, T.R., Fundamentals of metal sulfide biooxidation., Mining Engineering 56, pp. 40â46, 2004. [ Links ]
[5] Cancho, L., Blázquez, M.L., Ballester, A., González, F. and Muñoz ,J.A., Bioleaching of a chalcopyrite concentrate with moderate thermophilic microorganisms in a continuous reactor system, Hydrometallurgy 87, pp. 100â111, 2007. [ Links ]
[6] Vilcáez, J., Suto, K. and Inoue, C., Bioleaching of chalcopyrite with thermophiles: TemperatureâpHâORP dependence. Int. J. Miner. Process. 88, pp. 37â44, 2008. [ Links ]
[7] Yahya, A. and Johnson, D. B., Bioleaching of pyrite at low pH and low redox potentials by novel mesophilic Gram-positive bacteria. Hydrometallurgy 63, pp. 181â 188, 2002. [ Links ]
[8] Suzuki, I., Microbial leaching of metals from sulfide minerals. Biotechnology Advances, Department of Microbiology, University of Manitoba, Winnipeg, Canada, V. 19, pp. 119-132, 2001. [ Links ]
[9] Konishi, Y., Nishimura, H. and Asai, S., Bioleaching of sphalerite by the acidophilic thermophile Acidianus brierleyi Hydrometallurgy 47, pp. 339-352, 1998. [ Links ]
[10] Beck, J.V., The role of bacteria in copper mining operations, Biotechnology. Bioengineering. 9, pp. 487- 497, 1967. [ Links ]
[11] Murr, L.E. and Brierley, J.A., The use of large-scale test facilities in studies of the role of microorganisms in commercial leaching operations, In: L.E. Murr, A.E. Torma, J.A. Brierley (Eds.), Metallurgical Applications of Bacterial and Related Microbiological Phenomena, Academic Press, New York, pp.491-520, 1978. [ Links ]
[12] Gibbs, H.E., Errington, M. and Pooley, F.D., Economics of bacterial leaching. Can. Metall. Q., V. 24(2), pp. 121-125, 1985. [ Links ]
[13] Rohwerder, T., Gehrke, T., Kinzler, K. and Sand, W., Bioleaching review part A. Appl. Microbiol. Biotechnol. 63, pp. 239â248, 2003. [ Links ]
[14] Schippers, A. and Sand, W., Bacterial Leaching of Metal Sulfides Proceeds by Two Indirect Mechanisms via Thiosulfate or via Polysulfides and Sulfur, Applied and Environmental Microbiology, pp. 319â321, 1999. [ Links ]
[15] Toru Nagaoka, T., Ohmura, N. and Saiki, H., A Novel Mineral Flotation Process Using Thiobacillus ferrooxidans, Applied And Environmental Microbiology, pp. 3588â3593 - American Society for Microbiology, 1999. [ Links ]
[16] Lima, R., Biolixiviação de Concentrado de Flotação de Sulfetos de Cobre. 2006. 90f. Dissertação (Mestrado em Tecnologia de Processos QuÃmicos e BioquÃmicos), Escola de QuÃmica, Universidade Federal de Rio de Janeiro, Rio de Janeiro, 2006. [ Links ]
[17] Bosecker, K., Bioleaching: Metal solubilization by micro-organisms. FEMS Microbiology Reviews, 20, pp. 591- 604, 1997. [ Links ]
[18] Brewis, T., Extración de metales por oxidación bacteriana. Mining, Abril, 1996. [ Links ]
[19] Holt, J.G. and Krieg, N.R. et al. Bergeyâs Manual of Determinative Bacteriology â Ninth Edition - Ed. Willians & Wilkins â USA, 1994. [ Links ]
[20] Tuovinen, O. H., Kelly, D. P., Studies on the growth of Thiobacillus ferrooxidans. Use of membrane filters and ferrous iron agar to determine viable number and comparison 14CO2 â fixation and iron oxadation as measures of growth. Arch. Microbiol. V.1, pp. 205-210, 1979. [ Links ]
[21] Garcia, O., Isolation and Purifications of Thiobacillus ferrooxidans and Thiobacillus thiooxidans from some Coal and Uranium Mines of Brazil. Rev. Microbiol., São Paulo, 22 (1), pp. 1-6, 1991. [ Links ]
[22] Zeng, W.M., Wu, C.B., Zhang, R.B., Hu, P.L., Qiu, G.Z., Gu, G.H. and Zhou, H.B., Isolation and identification of moderately thermophilic acidophilic iron-oxidizing bacterium and its bioleaching characterization, Trans. Nonferrous Met. Soc. China 19, pp. 222 - 227, 2009. [ Links ]
[23] Olson, G. J. and Clark, T. R., Bioleaching of molybdenite. Hydrometallurgy, 93, pp. 10â15, 2008. [ Links ]
[24] Olson, G. J., Brierley, J. A. and Brierley, C. L., Bioleaching review B: Processing in bioleaching: applications of microbial processes by the minerals industries. Applied Microbiology and Biotechnology, V. 63, pp. 249 â 257, 2003. [ Links ]
[25] Zapata, D., Henao, D. y Godoy, M., Caracterización Mineralógica de los Productos de Oxidación del Sistema Pirita-Esfalerita por Bacterias Nativas Oxidantes de Fe, DYNA, Año 75, No. 154, pp. 59-64, 2008. [ Links ]
[26] Sandstorm, A. and Petersson, S., Bioleaching of a complex sulphide ore with moderate thermophilic and extreme thermophilic microorganism. Hydrometallurgy 46 (1â2), pp. 181â190, 1997. [ Links ]














