Serviços Personalizados
Journal
Artigo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Acessos
Acessos
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares em
SciELO
Similares em
SciELO -
 Similares em Google
Similares em Google
Compartilhar
DYNA
versão impressa ISSN 0012-7353
Dyna rev.fac.nac.minas vol.79 no.176 Medellín nov./dez. 2012
KINETIC AND THERMODYNAMIC EQUILIBRIUM OF ASPHALTENES SORPTION ONTO FORMATION ROCK: EVALUATION OF THE WASH IN THE ADSORPTIVE PROPERTIES
CINETICA Y EQUILIBRIO TERMODINAMICO DE LA SORCION DE ASFALTENOS EN ROCA: EVALUACIÓN DEL LAVADO EN LAS PROPIEDADES ADSORTIVAS
CAMILO A. FRANCO
Ing. de Petróleos Universidad Nacional de Colombia - Sede Medellin. caafrancoar@unal.edu.co
JULIANA GIRALDO
Ing. Química Universidad Nacional de Colombia - Sede Medellin. jgiraldocas@unal.edu.co
MARCO A. RUIZ
MSc, Profesor asociado Universidad Nacional de Colombia - Sede Medellín. maruiz@unal.edu.co
BENJAMIN A. ROJANO
Ph.D. Profesor asociado Universidad Nacional de Colombia - Sede Medellín. brojano@unal.edu.co
FARID B.CORTÉS
Ph.D. Profesor asociado Universidad Nacional de Colombia - Sede Medellín. fbcortes@unal.edu.co
Received for review March 15th, 2012, accepted June 19th, 2012, final version July, 15th, 2012
ABSTRACT: The main objective was to obtain kinetic and thermodynamic equilibrium of asphaltene sorption onto powdered rock obtained from the Guadalupe formation (washed and unwashed) at different times, temperatures and concentrations. However, the effect of temperature in the equilibrium sorption was not significant. Rock powders were characterized by N2 sorption at -196°C. Equilibrium sorption of asphaltenes in rock powders, within a range of concentrations from 250 to 1500 ppm, was determined using a static method. Sorption curves show Type I behavior, according to IUPAC. The Langmuir model was used for computing the monolayer asphaltene content values for the sorption at different temperatures, showing a good fit with respect to the experimental data. Sorption kinetics of asphaltenes in rock samples is a function of the concentration and for concentrations of 1500 ppm equilibrium is reached at about 90 min. Pseudo first order and pseudo second order kinetic models were applied to the experimental data for the different concentrations for the two rock samples, with a better result for the pseudo-first order kinetic model. The washing effect on the equilibrium was not significant; however the adsorptive capacity of the rock without washing was slightly higher than the rock washed with n-heptane and toluene.
KEYWORDS: Asphaltene, formation rock, sorption equilibrium, kinetic sorption.
RESUMEN: El principal objetivo del estudio fue obtener la cinética y el equilibrio termodinámico de la sorcion de asfaltenos en polvo de roca obtenida de la formación Guadalupe (lavada y sin lavar), a diferentes tiempos, temperaturas y concentraciones. Sin embargo, el efecto de temperatura en el equilibrio de sorción no era significativo. Los polvos de roca fueron caracterizados por sorción de N2 a -196°C. El equilibrio de sorcion de los asfaltenos en los polvos de roca, dentro de un rango de concentraciones desde 250 hasta 1500 ppm, fue determinado usando un método estático. Las curvas de sorcion muestran un comportamiento Tipo I, según la IUPAC. El modelo de Langmuir se usó para calcular los valores del contenido de asfaltenos monocapa para la sorcion a diferentes temperaturas, mostrando un buen ajuste con respecto a los datos experimentales. La cinética de sorcion de los asfaltenos en muestras de rocas es función de la concentración y para concentraciones de 1500 ppm el equilibrio es alcanzado alrededor de 90 min. Se aplicaron los modelos cinéticos de pseudo primer orden y pseudo segundo orden a los datos experimentales para las diferentes concentraciones para las dos muestras de rocas con un mejor resultado para el modelo cinético de pseudo-primer orden. El efecto de lavado en el equilibrio no fue significativo, sin embargo la capacidad adsortiva de la roca sin lavar fue ligeramente mayor que la roca lavada con n-heptano y tolueno.
PALABRAS CLAVE: Asfalteno, roca, equilibrio de sorcion, cinética de sorcion.
1. INTRODUCTION
Asphaltenes are defined as the fraction of oil, bitumen or vacuum residue that is insoluble in low molecular weight paraffins such as n-heptane or n-pentane, while being soluble in light aromatic hydrocarbons such as toluene, pyridine or benzene [1,2]. The structure of asphaltenes is formed by polyaromatic cores attached to aliphatic chains containing heteroatoms such as nitrogen, oxygen, sulfur, and metals such as vanadium, iron and nickel [1-8].Asphaltenes contain polar and non-polar groups (“amphiphilic” behavior) and tend to form colloidal aggregates. It has widely been documented that viscosity of heavy oils can dramatically increase by the asphaltene aggregation phenomenon [9,10].
Content of asphaltenes in a crude oil does not entail asphaltene related production problems. Most of the heavy oils with the greatest asphaltenes concentration are usually very stable during production, and asphaltenes do not promote well plugging on those wells. Quite the contrary asphaltenes precipitation problems are more common in lighter crude oils that contain minor amounts of asphaltenes in sub-saturated reservoirs that are at pressures above bubble point. The potential for asphaltenes to plug systems as constituents of heavy oil is more visible downstream, forming rigid oil/water emulsions and creating organic fouling problems in refineries.
Several researchers have studied the adsorption of asphaltenes onto solid surfaces focused on understanding process phenomenology [11-21]. Gonzalez et al.[22] and Acevedo et al. [23]reported that the isotherms of asphaltene sorption onto mineral surfaces followed a Langmuir-type behavior, with a maximum adsorption capacity of 1-2 mg/m2. Pernyeszi et al.[24] studied the effect of different types of materials (quartz, bentonite, cliptonite and two cores taken at different well depths) on the asphaltene adsorption process. Authors found that isotherms showed a type I behavior according to the IUPAC criteria. Other works studying the adsorption of asphaltenes on adsorptive clays also concluded that isotherms exhibited a type I behavior, indicating the formation of a monolayer onto the porous matrix surfaces [25,26].
R. Z. Syunyaev, R. M. Balabin, I. S. Akhatov and J. O. Safieva [27], computed asphaltene adsorption parameters (maximum adsorption capacity, adsorption equilibrium constant and adsorption kinetic constants) for quartz, dolomite and mica sandstones, using Near Infrared Spectroscopy (NIRS). Adsorption data was a good fit to the Langmuir model. Mica sandstone presented greater adsorption capacity compared to other minerals.
Alkafeef, S. F. et al. [28]studied asphaltene adsorption onto porous rock surfaces. Adsorption data were obtained by electrokinetic potential measurements on several sandstone samples with asphaltenic oil. Langmuir's isotherm was used as a theoretical approach to adsorption phenomenon.
Adsorption phenomena occur due to interactions between asphaltene aggregates and chemical compounds in minerals on rock surfaces. The governing forces in this behavior are electrostatic, charge transfer interaction, Van der Waals, steric repulsion and hydrogen bridge bonding [29-31].
Previous studies report that asphaltene and resin adsorption in aqueous and organic media onto clay minerals affect rock wettability, and decrease clay expansion, swelling and cationic exchange capacity. Adsorption phenomenon is deeply affected when an aqueous film is present in the medium[32].
José L. Mendoza de la Cruz et al.[20] studied asphaltene fraction adsorption from a Mexican oil dissolved in toluene on sandstone, limestone and dolomite rocks. For asphaltene concentrations lower than 6000 ppm, monolayer adsorption was observed. For higher concentrations, a multilayer adsorption was observed.
The aim of this work is to evaluate adsorption equilibrium of Guadalupe formation rock powder after being washed with n-heptane and toluene. So far, no asphaltene adsorption studies have been reported for asphaltenes extracted from Colombian oils.
2. EXPERIMENTAL
2.1. Materials
Asphaltenes were isolated from “The HOCHA” crude oil, which is heavy oil (19.2°API) produced from a reservoir located in the south of Colombia. The rock powder was obtained from a sample extracted from Guadalupe Formation.
2.2 . Methods
2.2.1. Asphaltene isolation protocol
Asphaltene samples were isolated by following a standardized procedure. An excess of n-heptane (99% Sigma Aldrich) was added to the crude oil in a volume ratio of 40/L The mixture was sonicated for 2 h at 25 °C and further stirred at 300 rpm for 20 h. The precipitated fraction was filtered using a 8 mm Whatman filter paper and washed with n-heptane at a ratio of 4/1 (g/mL). Asphaltene samples were centrifuged at 5000 rpm for15 min and left to rest for 24 h. The cake was washed with n-heptane several times until the color of the asphaltenes became shiny black. Then, the sample was dried in a vacuum oven at 25 °C for 12 h. Finally, the obtained asphaltene was homogenized in a mortar [2].Precipitated asphaltenes were dissolved in toluene to prepare a stock solution at 2000 mg/L. Solutions with different concentrations, 150, 250, 400, 750, 1000, 1500 and 2000 mg/L, were prepared from the stock solution.
2.2.2. Rock powder obtention and washing
From the rock sample belonging to the Guadalupe formation, 9g of rock powder was milled. Half of the powder obtained was washed with 10ml toluene and n-heptane for each gram of powder, and then the sample was filtered until the discarded toluene was colorless. The other half was used without any treatment.
2.2.3. Samples characterization
The powder samples were characterized by N2 adsorption at -196°C.Nitrogen adsorption isotherms were obtained with an Autosorb-1 from Quantacrome after outgassing samples overnight at 140 °C under high vacuum (10-6mbar). BET surface area (SBET) values were calculated using the model of Brunauer, Emmet and Teller (BET) presented by Rouquerol et al. [33].
2.2.4. Adsorption experiments
A calibration curve of UV absorbance versus asphaltene concentration at 400 nm was constructed from the prepared solutions with known concentrations. Toluene was used as a solvent agent for dilution, as well as the blank in the calibration curve construction [4,34].
A constant mass of the rock powder sample (100 mg) was added to the asphaltene solutions of constant volume (10 mL). Each solution was stirred at 200rpm for 10 h at 25, 40, 55 and 70 °C in order to reach the sorption equilibrium. The time evolution of the measured asphaltene concentration indicated that a time of 1 h was enough to reach the sorption thermodynamic equilibrium. When asphaltene solutions with high concentrations (greater than 250 mg/L) were tested, an additional dilution with toluene was necessary in order to obtain the desired absorbance value within the calibration curve.
3. MODEL
3.1. Kinetic model
In order to predict the rate at which asphaltenes adsorb onto rock surfaces, two adsorption kinetic models, pseudo-first-order and pseudo-second-order, have been used. The pseudo-first-order kinetic model is expressed in [35] as:
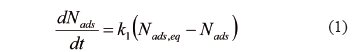
where 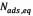 (mg/g) and
(mg/g) and 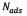 (mg/g) are the amounts of adsorbed asphaltene on rock powder at equilibrium and at time t, respectively, and
(mg/g) are the amounts of adsorbed asphaltene on rock powder at equilibrium and at time t, respectively, and 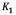 (h) is the kinetic constant of pseudo-first-order adsorption. The model is expressed in a linear form to plot the experimental data, in order to obtain the slope and intercept and determine
(h) is the kinetic constant of pseudo-first-order adsorption. The model is expressed in a linear form to plot the experimental data, in order to obtain the slope and intercept and determine 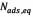 and.
and. 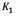 .
.
The pseudo-second-order kinetic model is expressed in [36,37] as:
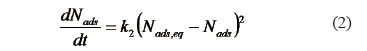
The second order kinetic constant 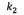 (h) and the amount of asphaltenes adsorbed at the equilibrium
(h) and the amount of asphaltenes adsorbed at the equilibrium 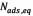 .
.
(mg/g) are obtained from the slope and intercept of the experimental data plot, fitted to the linear form of the model.
3.2. Langmuir Model
The Langmuir model has been widely used to correlate experimental data of equilibrium sorption [38].This model assumes that the process occurs on a homogeneous surface by monolayer adsorption. It was originally derived from kinetic data of adsorption and desorption, taking into account that equilibrium is obtained when the rates of adsorption and desorption are equal. The Langmuir equation can be expressed as follows:
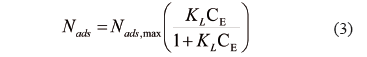
where 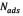 is the amount of asphaltenes adsorbed onto rock powder (mg/g),
is the amount of asphaltenes adsorbed onto rock powder (mg/g), 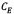 is the equilibrium concentration of asphaltenes in the solution phase (mg/L),
is the equilibrium concentration of asphaltenes in the solution phase (mg/L), 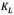 is the Langmuir equilibrium adsorption constant related to the affinity of binding sites (L/mg), and is defined as the monolayer saturation capacity,
is the Langmuir equilibrium adsorption constant related to the affinity of binding sites (L/mg), and is defined as the monolayer saturation capacity,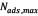 representing the maximum amount of asphaltenes per unit mass of nanoparticles for complete monolayer coverage (mg/g).
representing the maximum amount of asphaltenes per unit mass of nanoparticles for complete monolayer coverage (mg/g).
3.3. Thermodynamic properties
The thermodynamic properties were calculated in order to characterize the adsorption phenomenon and process spontaneity. They were estimated from the classical expression for the Gibbs free energy change 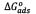 [KJ/mol])[4]:
[KJ/mol])[4]:
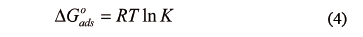
where R is the universal constant for ideal gases, T is the absolute temperature, and K is the adsorption equilibrium constant. K is related to the energy of adsorption and can be expressed as 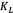 Cs, where Cs is the solvent molar concentration. The changes of sorption enthalpy (
Cs, where Cs is the solvent molar concentration. The changes of sorption enthalpy ( 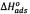 [KJ/mol]) and entropy (
[KJ/mol]) and entropy ( 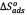 [KJ/K.mol]) were calculated from the plot of
[KJ/K.mol]) were calculated from the plot of 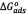 versus temperature [39].
versus temperature [39].
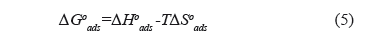
To determine the Langmuir constant it is necessary to know the molecular weight of the asphaltene. However, due to its dependence on the chemical nature of the crude oil from which asphaltenes were isolated, and the intrinsic complexity of the asphaltene structure, this property is still an unsolved issue [17].For this reason, thermodynamic properties were calculated taking into account the extremes of the typical range of molecular weight of asphaltenes reported in the literature (750-5000 g/mol) [2].
4. RESULTS AND DISCUSSION
4.1. Sorption isotherm of asphaltene
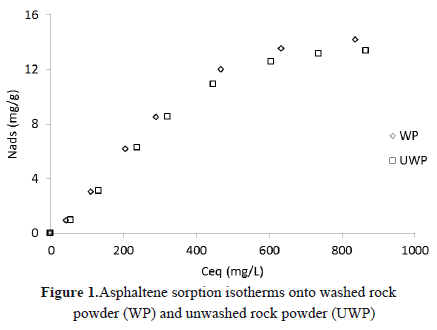
The rock powder has 15 m2/g of external surface area. Sorption isotherms of asphaltene onto washed sample and unwashed samples at 25°C are shown in Figure 1, where the unwashed sample shows a higher adsorptive capacity than the other sample.This phenomenon might have happened because during washing, some components present in the rock surface with affinity towards asphaltenes were removed. However, the difference between the adsorption capacities is not remarkable.
For the same concentration, in this case, 860 mg/L, the adsorptive capacity for the unwashed sample was 14.17 mg/g, while for the washed sample was 13.31 mg/g, 9% less than the unwashed sample. According to this result, the adsorption response in both samples is very similar.
Type I behavior was observed in the sorption isotherms of asphaltenes onto rock powder. These results are in agreement with Jose L. Mendoza de la Cruz [20], who studied the asphaltene adsorption onto sandstone, limestone and dolomite.
The Langmuir model was used for analyzing the asphaltene adsorption isotherms. The model parameters calculated for the isotherms of asphaltene adsorption on rock powder samples are presented in Table 1. As indicated by the values of the regression coefficients, the experimental data were adequately described by the model, presenting a Type I behavior [2].For each rock powder sample, the asphaltene content of the monolayer (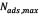 ), calculated by linear regression, was equal for all adsorption temperatures. Figure 2 shows isotherms behavior for the asphaltene sorption onto the washed sample (a) and the unwashed sample (b) at 25, 40, 55 and 70°C.
), calculated by linear regression, was equal for all adsorption temperatures. Figure 2 shows isotherms behavior for the asphaltene sorption onto the washed sample (a) and the unwashed sample (b) at 25, 40, 55 and 70°C.
The Langmuir model had correlation coefficients of 0.92 for de washed sample and 0.96 for the unwashed sample. There was a slight difference between the experimental data and the values obtained with the model mainly for high concentrations.
4.2. Thermodynamics properties of asphaltene sorption
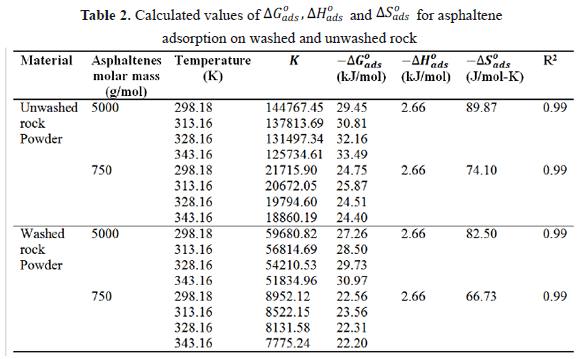
The calculated thermodynamic parameters for the sorption of asphaltenes on the washed and unwashed samples are given in Table 2. The change in the free Gibbs energy (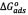 ) was inversely proportional to temperature. A negative value of
) was inversely proportional to temperature. A negative value of 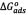 indicates the status of feasibility and spontaneity of thermodynamic processes, i.e., it characterizes the natural tendency of a system to undergo spontaneous changes [33,38].The
indicates the status of feasibility and spontaneity of thermodynamic processes, i.e., it characterizes the natural tendency of a system to undergo spontaneous changes [33,38].The 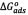 values obtained were in agreement with those reported by Nassar [2]. The negative value of
values obtained were in agreement with those reported by Nassar [2]. The negative value of 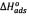 suggests the exothermic nature of the sorption process. This is in agreement with the finding that the adsorption is rapid and decreases with the temperature [2].
suggests the exothermic nature of the sorption process. This is in agreement with the finding that the adsorption is rapid and decreases with the temperature [2].
4.3. Sorption Kinetics of asphaltene
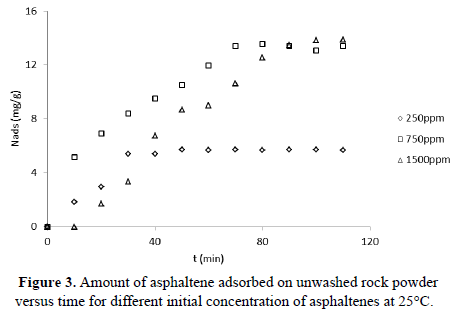
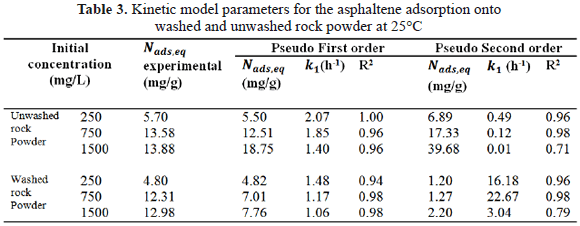
To determine kinetic parameters, the amount of asphaltenes adsorbed was calculated from concentration measurements at different times, for 250,750 and 1500 mg/L, as shown in Figure 3 and Figure 4 for unwashed and washed samples respectively. The results indicate that equilibrium is reached faster for lower initial concentration (30 min for 250 mg/L; 70 min for 750 and 1500 mg/L). Kinetic constants (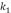 and
and 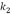 ) and amounts of adsorbed asphaltene at the equilibrium (
) and amounts of adsorbed asphaltene at the equilibrium (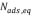 ) obtained are summarized in Table 3. Furthermore, experimental data fitted better to pseudo-first-order model, (Table 3.) which suggests that adsorption of asphaltene onto rock powder follows a first order kinetic. Finally, values obtained with kinetic models of pseudo-first order are in agreement with experimental data obtained in this study as shown in Table 3.
) obtained are summarized in Table 3. Furthermore, experimental data fitted better to pseudo-first-order model, (Table 3.) which suggests that adsorption of asphaltene onto rock powder follows a first order kinetic. Finally, values obtained with kinetic models of pseudo-first order are in agreement with experimental data obtained in this study as shown in Table 3.
CONCLUSIONS
The asphaltene sorption isotherms of rocks (washed and unwashed) at 4 temperatures (25, 40, 55 and 70°C) and different concentration levels were determined using the static method. The equilibrium asphaltene content slightly increased with decreases in temperature at a constant asphaltene concentration. Additionally, the temperature dependence of sorption isotherms could be predicted with reasonable accuracy with Langmuir model. However, the temperature effect is not significant.
The monolayer moisture content values for desorption at different temperatures were determined using a Langmuir model. However, the temperature effect is not significant. Superior results were obtained when the washed powder rock.
The thermodynamics properties of asphaltene adsorption on rocks reaffirm the spontaneity and exothermic nature of the adsorption process. Equilibrium constants, adsorption capacities, and thermodynamic parameters for asphaltene adsorption onto rocks were computed and compared with the literature, which is agreement.
ACKNOWLEDGEMENTS
The authors are grateful to COLCIENCIAS and Universidad Nacional de Colombia, for logistical and financial support
REFERENCES
[1] Bouhadda, Y., Bormann, D., Sheu, E., Bendedouch, D., Krallafa, A. and Daaou, M., Characterization of Algerian Hassi-Messaouda asphaltene structure using Raman spectrometry and X-ray diffraction, Fuel, 86, pp.1855-1864, 2007. [ Links ]
[2] Nassar, N., Asphaltene adsorption onto alumina nanoparticles: kinetics andthermodynamic studies. Energy & Fuels,24, 4116-22,2010. [ Links ]
[3] Energy Information Administration. World consumption of primary energy by energy type and selected country groups, 1980-2006.Available: www.eia.doe.gov/pub/international/iealf/table18.xls. [cited on september 23th of 2010. [ Links ]
[4] Babadagli, T., Development of mature fields, Journal of Petroleum Science and Engineering, 57, pp. 221-246, 2007 [ Links ]
[5] Ruiz, R. and Rojas, J., THAI Toe-to-Heel air injection-numerical simulation of THAI heavy oil recovery process. White Papers-Schlumberger, 2007. [ Links ]
[6] Navarrete, N., Major new oil developments. Technological & operational, challenges & solutions. Colombia potential. WPC Regional Meeting - IV Oil & Gas Investment Conference, 2010. [ Links ]
[7] Heavy Oil Info, Available www.heavyoilinfo.com/recovery-methods.[Cited on october 12th of 2011] [ Links ].
[8] Groenzin, H. and Mullins, O., Molecular Size and Structure of. Asphaltenes from Various Sources, Energy & Fuels, 14, pp. 677-684, 2000 [ Links ]
[9] Acevedo, S., Castillo, J., Fernandez, A., Goncalves, S. and Ranaudo, M., Study of Multilayer Adsorption of Asphaltenes on Glass Surface by Photothermal Surface Deformation. Relation of this Adsorption to Aggregate Formation in Solution, Energy & Fuels, 12, pp. 386-390, 1998 [ Links ]
[10] Mousavi-Dehghani, S. and Riazi, M., Vafaie-Sefti, M., Mansoori, G.J., An analysis of methods for determination of on sets of asphaltene phase separations, Pet. Sci. Eng., 42, pp.145-156, 2004 [ Links ]
[11] Xing, C. and Hilts, R., Shaw, J., Sorption of Athabasca Vacuum Residue Constituents on Synthetic Mineral and Process Equipment Surfaces from Mixtures with Pentane Energy & Fuels, 24, pp.2500-2513, 2010. [ Links ]
[12] Turgman-Cohen, S. and Fischer, D., Kilpatrick, P., Asphaltene adsorption onto self-assembled monolayers of alkyltrichlorosilanes of varying chain length, ACS Applied Materials & Interfaces, 6, pp.1347-1357, 2009 [ Links ]
[13] Turgman-Cohen, S., Fischer, D., Kilpatrick, P. and Genzer, J., Asphaltene adsorption onto Self-Assembled monolayer of mixed Aromatic and Aliphatic Trichlorosilanes, Langmuir, 25, pp. 6260-6269, 2009 [ Links ]
[14] Yan, N. and Masliyah, J., Demulsification of solids-stabilized oil-in-water emulsions, Colloids and Surfaces A: Physicochem. Eng. Aspects, 117, pp.15-25, 1996. [ Links ]
[15] Yan, N. and Masliyah, J., Characterization and demulsification of solids-stabilized oil-in-water emulsions Part 2. Demulsification by the addition of fresh oil, Colloids and Surfaces A: Physicochem. Eng. Aspects, 96, pp. 243-252, 1996. [ Links ]
[16] Ese, M., Galet, L., Clausse, D., SJer emuJ., Properties of Langmuir Surface and Interfacial Films Built up by Asphaltenes and Resins: Influence of Chemical Demulsifiers, J. Colloid Interface Sci, 220, pp. 293-301, 1999. [ Links ]
[17] Rudrake, A., Karan, K. and Horton, J.A., combined QCM and XPS investigation of asphaltene adsorption on metal surfaces, J. Colloid Interface Sci., pp. 332,22-31, 2009. [ Links ]
[18] Abdallah, W. and Taylor, S., Surface characterization of adsorbed asphaltene on a stainless steel surface, Nuclear Instruments and Methods in Physics Research B, pp. 258,213-217, 2007. [ Links ]
[19] Xie, K. and Karan, K., Kinetics and Thermodynamics of Asphaltene Adsorption on Metal Surfaces: A Preliminary Study, Energy & Fuels, 19, pp. 1252-1260, 2005. [ Links ]
[20] Mendoza, J., Castellanos, I. and Ortiz A., BUENROSTRO E., DURMetal Surfaces: Study of monolayer to multilayer adsorption of asphaltenes on reservoir rock minerals, Colloids and Surfaces A: Physicochem. Eng. Aspects, pp. 340,149-154, 2009. [ Links ]
[21] Kumar, K., Dao, E. and Mohanty, K., AFM study of mineral wettability with reservoir oils, J. Colloid Interface Sci. pp. 289:206-217, 2005. [ Links ]
[22] Gonzalez, G. and Moreira, M. B. C., The wettability of mineral surfaces containing adsorbed asphaltene. Colloids Surf., 58,293- 302,1991 [ Links ]
[23] Acevedo, S., Ranaudo, M., Escobar, G. and Gutierrez, L., Adsorption of asphaltenes and resins on organic and inorganic substrates and their correlation with precipitation problems in production well tubing, Fuel, 74 (4), pp. 595-598, 1995 [ Links ]
[24] Pernyeszi, T., Patzko, A., Berkesi, O. and Dekani, I., Asphaltene adsorption on clays and crude oil reservoir rocks, Journal of colloids and surfaces A, 137, pp. 373-384. 1998 [ Links ]
[25] Acevedo, S., Ranaudo, M., Garcia, C., Casillo, J. and Caetano, M., GONCALVEZ S. Importance of asphaltene aggregation in solution in determining the adsorption of this sample on mineral surfaces, Colloids and Surfaces A: Physicochem. Eng. Aspects,166, pp.145-152, 2000. [ Links ]
[26] Sakanishi, K., Saito, I., Watanabe, I. and Mochida, I., Dissolution and demetallation treatment of asphaltene in resid using adsorbent and oil-soluble Mo complex, Fuel, 83, pp.1889-1893, 2004.R. [ Links ]
[27] SYUNYAEV Z., BALABIN, R. M. and Akhatov, I. S., Adsorption of Petroleum Asphaltenes onto Reservoir Rock Sands Studied by Near-Infrared (NIR) Spectroscopy, Energy Fuels, 23 (3), pp.1230-1236, 2009. [ Links ]
[28] Alkafeef, S. F., Gochin, R. J. and Smith, A. L., Surface Potential and Permeability of Rock Cores Under Asphaltenic Oil Flow Conditions, Paper SPE 30539-MS, 1995. [ Links ]
[29] Dubey, S.T. and Waxman, M.H., Asphaltene adsorption and desorption from mineral surfaces, in: SPE (Soc. Pet. Eng.) 18462 Oilfield Chemistry, 1989. [ Links ]
[30] Murgich, J., Intermolecular forces in aggregates of asphaltenes and resins, Petroleum Science and Technology, 20 (9-10), pp. 983-997, 2002 [ Links ]
[31] Moschopedis, S.E., Fryer,s J.F. and Speight, J.G., Investigation of asphaltene molecular weights, Fuel, 55 - 227, 1976 [ Links ]
[32] Kokal, S., Tang, T., Schramm, L. and Sayegh, S., Colloid Surf. A, 94,253, 2005 [ Links ]
[33] Rouquerol, F., Rouquerol, J. and Sing, K.S.W., Adsorption by powders and porous solids.London (UK): Academic Press; 1999. [ Links ]
[34] Dudásová, D., Simon, S., Hemmingsen, P. and Sjöblom, J., Colloids and Surfaces A:Physicochem. Eng. Aspects, 317, pp. 1-9, 2009 [ Links ]
[35] Alboudwarej, H, Pole, D., Svrcek, W.Y. and Yarranton, H.W, Adsorption of Asphaltenes on Metals. Ind Eng Chem Res,44,5585-92, 2005 [ Links ]
[36] Cortés, F.B. and Chejne, F., A rapid and novel approach for predicting water sorption isotherms and isosteric heats of different meat types, Meat Sci. 2010 doi:10.1016/j.meatsci.2010.07.017, 2010. [ Links ]
[37] Pinzón-Bedoya, M.L. and Vera, L.E., Kinetc Modeling Biosorption of Cr(iii) Using Orange Shell, Dyna, 160, pp. 95-106, 2009. [ Links ]
[38] Langmuir, I., Journal of the American Chemical Society, 38,2221-221e, 1916 [ Links ]
[39] Cortés, F. B., Chejne, F., Carrasco, F., Moreno, M., Castilla, C. and Pérez-Cadenas, A. F., Water adsorption on zeolite 13X: comparison of the two methods based on mass spectrometry and thermogravimetry Adsorption,16, pp. 141-146, 2010 [ Links ]