Serviços Personalizados
Journal
Artigo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Acessos
Acessos
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares em
SciELO
Similares em
SciELO -
 Similares em Google
Similares em Google
Compartilhar
DYNA
versão impressa ISSN 0012-7353
Dyna rev.fac.nac.minas vol.80 no.180 Medellín jul./ago. 2013
CONVERSION OF D-XYLOSE INTO FURFURAL WITH ALUMINUM AND HAFNIUM PILLARED CLAYS AS CATALYST
CONVERSION DE D-XILOSA A FURFURAL CON ARCILLAS PILARIZADAS CON ALUMINIO Y HAFNIO COMO CATALIZADORES
WILLIAM CORTÉS
M.Sc. Departamento de Ciencias Básicas, Universidad Jorge Tadeo Lozano, Bogotá, Colombia, williamg.cortes@utadeo.edu.co
YINETH PIÑEROS-CASTRO
Ph.D. Facultad de Ciencias Naturales e Ingeniería, Universidad Jorge Tadeo Lozano, Bogotá, Colombia, yineth.pineros@utadeo.edu.co
ANA MARÍA CAMPOS ROSARIO
Ph.D. Departamento de Ciencias Básicas, Universidad Jorge Tadeo Lozano, Bogotá, Colombia, ana.campos@utadeo.edu.co
Received for review December 9th, 2011, accepted March 15th, 2013, final version March, 21th, 2013
RESUMEN: El proceso industrial utilizado para la producción de furfural es la deshidratación de pentosas, la cual se lleva a cabo por medio de ácidos minerales, altamente corrosivos y contaminantes como catalizadores. Debido a los efectos ecológicos y toxicológicos asociados a este proceso, y a la dificultad en la separación del producto, el desarrollo de nuevos catalizadores sólidos ácidos ha tenido una significativa expansión en los últimos años. El presente trabajo de investigación se enfocó en la producción de furfural a partir de D-xilosa empleando como catalizadores sólidos ácidos, arcillas pilarizadas con aluminio y hafnio. Después de 4 h de reacción a temperaturas entre 140 y 170 °C, los resultados evidenciaron una conversión entre el 50 y el 80%, mientras que los niveles de selectividad alcanzados estuvieron entre el 40 y el 65%. Finalmente, la estabilidad de los catalizadores fue evaluada por medio de un tratamiento térmico, antes de usarlo de nuevo. En cuatro reacciones consecutivas no fue observada una disminución significativa en la selectividad. En conclusión, las arcillas pilarizadas evidencian ser catalizadores activos, selectivos y estables para la deshidratación de pentosas.
PALABRAS CLAVE: xilosa, deshidratación de pentosas, arcilla pilarizada, furfural.
ABSTRACT: The industrial practice used to produce furfural is the dehydration of pentoses, which is carried out using highly corrosive and contaminating mineral acid catalysts. Because of the ecological and toxicological threats of this process and the complexity of product separation associated with these substances, the development of new acid solid catalysts has expanded in recent years. The present work is focused on the production of furfural from D-xylose using aluminum and hafnium pillared clays as catalysts. After 4 h of reaction at temperatures ranging between 140 and 170°C, conversion rates of 50-80% were observed, and selectivity levels of 40-65% were reached. Finally, the stability of the catalyst was further investigated by applying a thermal treatment prior to reuse. A significant decrease in the selectivity was not observed in four consecutive reactions. Consequently the pillared clays appear to be active, selective and stable catalyst for pentose dehydration.
KEYWORDS: xylose, pentose dehydration, pillared clays, furfural.
1. INTRODUCTION
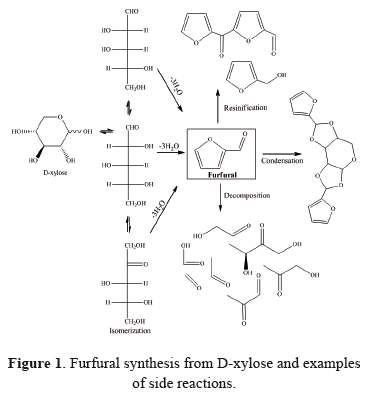
Biomass conversion through green chemical methods is an important process because biomass is one of the most widely available and renewable resources that can be used as a low cost raw material for the production of biofuel, bioenergy and value-added organic products. Among the methods used for biomass conversion, the acid-catalyzed dehydration of monosaccharides has been the target of many studies due to the development of renewable chemicals such as furfural and 5 hydroxymethylfurfural.
Furfural (2-furfuraldehyde) is currently obtained through the dehydration of pentoses, and an average of 250.000 tonnes per year is produced in China, where over 70% of the global production is located [1]. Furfural applications range from raw materials for furan chemical compounds to solvents (e.g., oil refining). Industrially, furfural is used as a building block for polymers such as polyester, polyimide, and polyurethane. Moreover, bulk chemicals such as tetrahydrofuran (THF), tetramethylene chloride pyrrole and pyridine are obtained from furfural [2].
Furfural is also widely used as a solvent for the refinement of lubricants and the separation of butadiene from mixtures of butene in synthetic rubber fabrication. Furfural is also used in the manufacturing of different types of thermosetting copolymer resins, such as furfural-phenol, furfural-acetone, furfural-urea and furfural-formol, as well as other high-molecular-weight plastics, such as polyvinylfurfural [2].
Furfural is only produced through the dehydration of pentoses (mainly from D-xylose). Pentose dehydration reactions are currently carried out at high temperatures using an acid catalyst, such as hydrochloric acid, sulfuric acid or acetic acid [3, 4]. Under these conditions, the selectivity of furfural does not exceed 70%, unless is continuously extracted with supercritical fluids [5].
Concentrated sulfuric acid is generally used as a catalyst. However, this process corrodes the reactor and causes environmental problems due to, the large quantity of wastewater needed to dilute the acid and complications arising from separation and recycling [4]. The process can also produce many secondary reactions that contribute to selectivity losses, including the condensation of furfural and intermediate products and the production of other compounds, such as glyceraldehyde and lactic acid [5].
Several efforts have been made to develop new low-cost catalytic processes and more selective and environmentally safe methods for the conversion of monosaccharides into furans [6].
Therefore, different solid acid catalysts have been investigated, including zeolites [7], heteropolyacids [6] and microporous supports functionalized with sulfonic groups [8] among others. These catalysts show high activity, selectivity, stability, and ease of separation from the reaction products and are categorized as super acid catalysts due to their acid strength, which may be similar to or greater than the acidity exhibited by strong mineral acids, such as concentrated sulfuric acid.
Nevertheless, other solid acid catalysts, such as pillared clays (PILCs), have not previously been investigated for reactions such as pentose dehydration. PILCs have been developed over the past few years as a promising alternative for acid-catalyzed reactions due to their high activity and selectivity. The pillarization process consists of expanding and supporting layers of a 2:1-type clay mineral with inorganic molecules known as pillars to obtain highly porous clays with increased accessibility to acid sites on their surfaces and high hydrothermal stability [9-13].
Moreover, the oxides or pillars may be converted into active sites with different levels of acidity, depending on the characteristics of the cations incorporated. Thus, cations such as Al, Zr, Hf and Ce have been evaluated for a wide range of reactions, especially those requiring sites with medium acidity and moderate temperatures [9, 10]. Consequently, the catalytic potential of pillared clays is suitable for the dehydration of saccharides.
Considering the previously discussed framework, it seems appropriate and necessary to: a) explore new technologies that exploit the large number of different types of materials that can be used as source of pentoses and similar sugars, such as lignocellulosic biomass; b) develop methods to extract these sugars; and c) investigate their transformation into commercially attractive chemical products.
Motivated by these ideas, the aim of the present investigation was to produce furfural from D-xylose using pillared clays as solid acid catalysts.
2. MATERIALS AND METHODS
2.1. Catalyst preparation
Natural vermiculite found in the region of Santa Marta, Colombia, was used as the starting clay. Modifications were performed on the fraction with particle sizes less than 150 mm. This fraction was separated from the bulk material by sieving and was used without any purification pre-treatment. The cation exchange capacity of the started material was 1.10 meq g-1 [12].
The structural formula, which has been determined in previous work, is [12]:
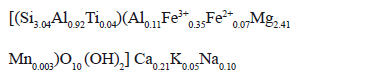
Polymeric solutions of Al and Hf were prepared using 12 mmol of (Al + Hf) per gram of clay. A 0.1 mol L-1 solution of AlCl3 was slowly added to a 0.1 mol L-1 HfCl4 solution at room temperature under constant stirring. Subsequently, the temperature was increased to 60°C, and a 0.2 mol L-1 NaOH solution was added dropwise under vigorous stirring until an OH/Al molar ratio of 2 was reached. After the addition, the solution was maintained at 60°C for 2 h.
After aging for 36 h at room temperature, the pillaring solution was slowly added to a vigorously stirred suspension of clay (2% wt/vol) at 80°C. When the addition was complete, the temperature was maintained for 4 h. The final clay suspension was aged for 12 h at room temperature. Subsequently, excess salt was eliminated via distilled water washings (until the conductivity was near the value for that of distilled water). Finally, the materials were dried at 60°C and calcined at 400°C (5 °C min-1) for 2 h.
The materials were labeled according to the name of the two metals followed by the quantity added. Thus, Al-Hf 11.5-0.5 corresponds to vermiculite modified with a solution of 11.5 mmol of Al and 0.5 mmol of Hf g-1 clay.
2.2. Characterization of pillared clays
X-ray diffraction (XRD) spectra of powder samples were obtained using a Phillips PW1710 spectrometer equipped with a copper anticathode. Elemental analysis of the samples was performed by inductively coupled plasma mass spectrometry. A detailed description of the synthesis and characterization techniques has been provided in previous studies [9-13].
The acidity was evaluated by temperature-programmed desorption of ammonia (TPD-NH3). The experiments were performed using pellets of each catalyst (30 mg), which possessed a particle size between 125 and 250 mm.
Catalyst pretreatment was carried out in-situ in helium at 400 °C for 1 h at 30 ml min-1. Subsequently, the temperature was reduced to 150 ºC, and the solid was exposed to NH3/N2 (5 wt% NH3) at 30 ml min-1 for 1 h. Excess of ammonia was removed with helium at the same flow for 30 min.
The desorption process was monitored by a Pfeiffer Omnistar Quadrupole Mass Spectrometer that was connected to the reactor on-line and possessed a fast response time (transit time ≤5 s). Desorption was performed at temperatures ranging between 150 and 500ºC at a rate of 15ºC min-1 in a flow of helium of 30 ml min-1. The identification of ammonia was achieved using the signal m/e= 15. Usually, the signals m/e 17 or 16 can be used for ammonia quantification.
The quantification of the total acidity was calculated by integrating the desorption area. To assess the acid strength distribution, the TPD-NH3 profiles were treated by mathematical analysis with Grams AI software.
2.3. Catalytic evaluation
Xylose dehydration tests were performed in a 50-mL temperature-controlled stainless-steel reactor stirred at 500 rpm. In a typical test, D-xylose (0.75 g), the catalyst (0.5 g) and deionized water (25 mL) were added to the reactor. The starting time was registered when the system reached the reaction temperature. Once the reaction time was complete, the sample was extracted and centrifuged. The product was analyzed by HPLC using an RPC-18 150 x 4.6 mm chromatographic column. The mobile phase was prepared by mixing 40 mL of methanol and 1 mL of glacial acetic acid and completing the volume to 1 L with distilled water. The column was maintained at 20 °C, and the product was detected by UV at 270 nm. The injection volume was 20 mL.
Once the reaction was finished, xylose was quantified by an analytical method using 3, 5-dinitrosalicylic acid (DNS) [14]. The analytical solution was prepared by adding 1.6 g of sodium hydroxide, 30 g of sodium and potassium tartrate, and 1 g of DNS to distilled water to obtain a total volume of 1 L. Quantification was carried out using a calibration curve obtained from an aqueous solution of D-xylose® and a Senway spectrophotometer at a wavelength of 540 nm. The conversion of D-xylose and the selectivity of furfural were estimated based on Equations 1 and 2, respectively [15]:
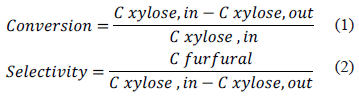
where C xylose, in is the initial concentration of xylose; C xylose, out is the concentration of xylose after the reaction; and C furfural is the concentration of furfural after the reaction.
3. RESULTS AND DISCUSSION
3.1. Catalyst characterization
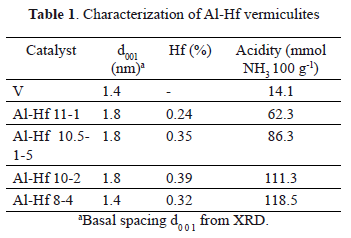
An increase in the interlayer spacing due to cationic exchange processes in clay is a characteristic that allows the success of a pillarization process to be determined and can be assessed by the d001 signal in the XRD pattern. This signal was located at 1.4 nm [12] for the starting mineral clay and 1.8 nm for the pillared clays [9, 10]. As shown in table 1, all of the clays were successfully pillared, except for Al-Hf 8-4.
The large amount of Hf added in the preparation of Al-Hf 8-4 increased the acidity in the pillaring solution, which resulted in the formation of aluminum oligomeric species instead of polymeric species and then reduction in the basal space (1.43 nm for Al-Hf 8-4 clay compared with 1.8 nm for the other clays) [11].
Regarding the quantity of hafnium used in the synthesis, Hf incorporation rates of 0.5 - 2 mmol g-1 of clay were achieved. When larger quantities were used (4 mmol), the efficiency decreased (Table 1).
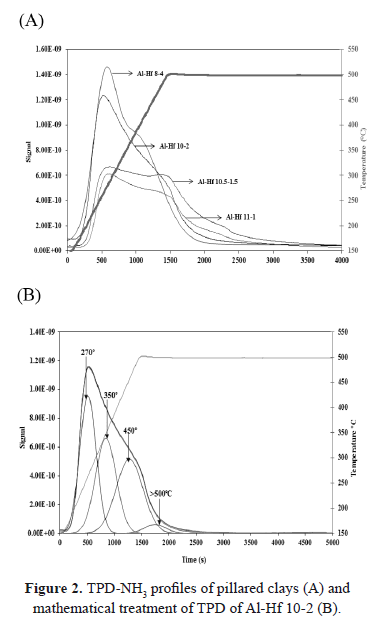
The heterogeneity of the acid surface of pillared clays was observed in the TPD-NH3 spectra. The components observed after mathematical treatment displayed maximum peaks between 180-250ºC, 280-330ºC and 380-500ºC. These ranges are related to ammonia absorbed at weak, medium, strong, and very strong acid sites, respectively [16].
Even though the acidity increased with an increase in the content of hafnium (Table 1), the TPD-NH3 profiles displayed significant differences in the amount and strength of the acid sites (Figure 2). Al-Hf 8-4 and Al-Hf 10-2 exhibited an increase in the formation of weak acid centers, whereas an increase in the number of medium and strong acid sites was detected in Al-Hf 10.5-1-5 and Al-Hf 11-2.
3.2. Catalytic evaluation
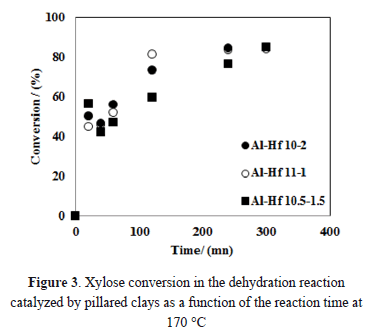
The reaction was carried out at 120 °C, 140 °C and 170 °C. According to the literature, the maximum conversion of D-xylose is achieved after heating for 3 to 4 h [7]. Therefore, an initial experiment was performed at 170 °C, and the conversion rate was determined as a function of time to select the reaction time.
The trends observed for several different catalysts were also exhibited by the pillared clays, as shown in Figure 3. The reaction time was established as 4 h because high conversion rates of D-xylose were observed at this point.
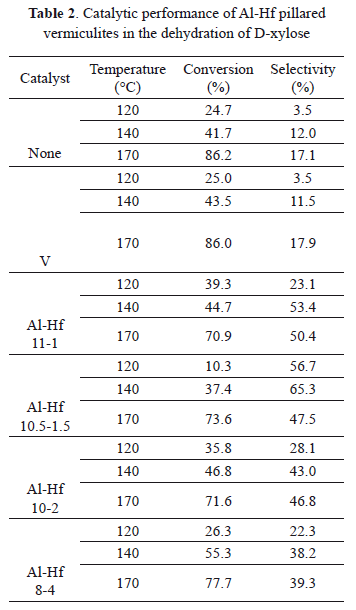
Once the reaction time was established as 4 h, the catalytic activity of the pillared clays as a function of the temperature was determined using water as a solvent. The results are shown in Table 2.
As shown in Table 2, when the reaction was carried out without catalyst or with vermiculite clay without pillaring treatment, the rate of conversion ranged from 20% to 80% as the temperature increased from 120 °C to 170 °C. The maximum selectivity was 17%, when the reaction was carried out at 170 °C.
The positive influence of the pillaring process in vermiculite clay was evidenced by the fact that natural clay did not have an effect on the furfural conversion or selectivity.
In contrast, when the reaction was catalyzed by Al and Hf pillared clays at 120 °C, the conversion of D-xylose was 39.3% (Al-Hf 11-1). At 140°C and 170°C, the conversion rate increased to 55.3% (Al-Hf 8-4) and 77.7%, respectively.
The conversion rates achieved with Al-Hf vermiculites using water as a solvent were comparable to those obtained with other catalysts, such as zeolites, which yield conversions ranging from 45% to 80% under similar reaction conditions [17-18]. Nevertheless, these reactions were carried out in water/organic solvent mixtures, such as water/toluene [17, 19] and water/dimethyl sulfoxide [8].
According to most reports, dehydration reactions of pentoses are performed in biphasic systems (water/organic solvent) to reduce the formation of degradable side products through consecutive polymerization reactions [15]. However, in the present study, water was employed as an environmentally friendly reaction medium.
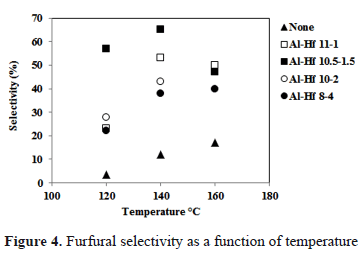
The selectivity of the pillared clays in the production of furfural was dependent on the temperature (Figure 4). The selectivity reached 53.4% at 120°C (Al, Hf 11-1) and increased to 65.3% (Al-Hf 10.5-1.5) at 140°C. However, a loss of selectivity was observed at 170°C for the catalyst.
These results are related to the acidity of the catalyst. For Al-Hf 10.5-1-5 and Al-Hf 11-2, an increase in the number of sites of medium and strong acidity was observed, despite the low total acidity compared with that of other pillared clays.
A decrease in the selectivity of the reaction has been observed with other acid catalysts [20] due to the complex mechanism, which involves a series of secondary reactions. However, the effects of competitive adsorption and changes in the surface properties of the catalyst during the reaction cannot be ruled out [20]. In fact, a brown color is observed on the catalyst after the reaction, which may be related to the presence of nonvolatile organic compounds on the surface of the material [20].
Even though the selectivity decreased at 170°C, the values were comparable and even superior compared with those obtained using other solid acid catalysts, such as zeolites (55% at 170 °C) [17], microporous silico-alumino-phosphates [20], and nanolayers exfoliated with titanium and niobium, which show selectivity values of 35% and 60%, respectively [21]. Consequently, the activity and selectivity values of the pillared clays are significant because they were achieved at 140°C.
The application of the Al-Hf 11-1 catalyst at 170 °C resulted in a conversion of 70.9% of D-xylose and a furfural selectivity rate of 50.4%. Moreover, at 140°C, Al-Hf 10.5-1.5 led to 37.4% conversion and 65.3% selectivity. Such pillared clays appear promising for the dehydration of pentoses to furfural.
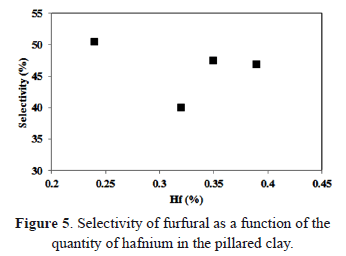
Regarding the amount of hafnium added to the pillaring aluminum solution in the vermiculites, Figure 5 shows the relationship between the quantity and the catalytic behavior of the catalyst.
The effect of the second metal in the pillarizing solution is evident. Namely, an increase in the hafnium content reduced the selectivity for furfural on Al-Hf vermiculites. This behavior is related to a reduction in the structural properties of the solid, which may be attributed to a decrease in the basal space in solid Al-Hf 8-4 and reduced laminar order or reduced pillarization.
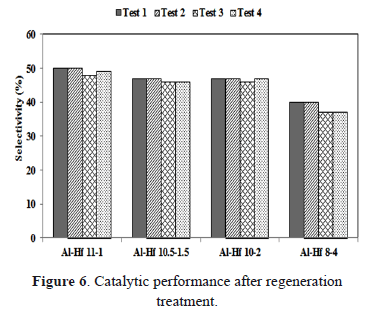
Finally, the stability of the catalyst was further investigated by applying a thermal treatment (400°C, 3 h, in air) to remove carbonaceous matter prior to reuse. A significant decrease in the selectivity was not observed in four consecutive reactions (Figure 6). Hence, the pillared clays appear to be stable after regeneration treatment prior to reuse.
4. CONCLUSIONS
Al-Hf pillared vermiculites with a maximum Hf content of 2 mmol Hf g-1 clay are promising catalysts for the dehydration of pentose in water as a solvent, producing furfural with an average conversion rate of 78% and a selectivity rate of 50% after 4 h at 170 °C.
The conversion of D-xylose to furfural represents a new pathway for the development of different clean technologies focused on the use of saccharides as raw materials to synthesize other chemical products. The reaction conditions may be extrapolated to agricultural residues with high pentose contents, such as lignocellulosic materials.
ACKNOWLEDGMENTS
The authors thank Ministerio de Agricultura y Desarrollo Rural, Cámara Induarroz of ANDI and Universidad de Bogotá Jorge Tadeo Lozano for supporting the Project “Aplicación de tecnologías para el aprovechamiento de las fracciones celulósica y hemicelulósica de la cascarilla de arroz” based on this research.
The corresponding author wishes to express their gratitude to Dr. Rafael Molina and Dr. Sonia Moreno from Universidad Nacional de Colombia for allowing the evaluation of the catalysts synthesized during her doctoral research.
REFERENCES
[1] Tong, X., Ma, Y. and LI, Y., Biomass into chemicals: Conversion of sugars to furan derivatives by catalytic processes. Applied Catal. A: Gen, 385, pp. 1-13, 2010. [ Links ]
[2] Moreau, C., Belgacem, M. and Gandini, A., Recent catalytic advances in the chemistry of substituted furans from carbohydrates and in the ensuing polymers. Topics in Catalysis, 27, pp. 11-30, 2004. [ Links ]
[3] Vásquez, M., Oliva, M., Téllez, L. and Ramírez, J., Hydrolysis of sorghum Straw using phosphoric acid: Evaluation of furfural production, Bioresour. Technol., 98, pp. 3053-3060, 2007. [ Links ]
[4] Antal, M., Lessomboon, T., Mok, W. and Richards, G., Mechanism of formation of 2-furaldehyde from D-xylose, Carbohydr. Res., 217, pp. 71-85, 1991. [ Links ]
[5] Shi, X., Wu, Y., Li, P., Yi, H., Yang, M. and Wang, G., Catalytic conversion of xylose to furfural over the solid acid SO42-/ ZrO2-Al2O3/SBA-15 catalyst, Carbohydr. Res., 346, pp. 480-487, 2011. [ Links ]
[6] Karinen, R., Vilonen, K. and Niemelä, M., Biorefining: Heterogeneous catalyzed reactions of carbohydrates for the production of furfural and hydroxymethylfurfural, ChemSusChem, 4, pp. 1002-1016, 2011. [ Links ]
[7] Moreau, C., Durand, R., Pourcheron, C. and Razigade, S., Selective preparation of furfural from xylose over microporous solid acid catalyst. Ind. Crops Prod., 7, pp. 95-99, 1998. [ Links ]
[8] Dias, A., Pillinger, M. and Valente, A., Dehydration of xylose into furfural over micro-mesoporous sulfonic acid catalyst. Journal Catal. 229, pp. 414-423, 2005. [ Links ]
[9] Campos, A., Moreno, S. and Molina, R., Relationship between hydrothermal parameters as a strategy to reduce layer charge in vermiculite, and its catalytic behavior. Catal. Today. 133, pp. 351-356, 2008. [ Links ]
[10] Alvarez, A., Moreno, S., and Molina, R., Efecto del tamaño de agregado de arcilla en la oxidación de fenol en medio acuoso diluido. DYNA. 77 No 162, pp. 57-66, 2010. [ Links ]
[11] Campos, A., Gagea, B., Moreno, S., Jacobs, P. and Molina, R., Decane hydroconversion with Al-Zr, Al-Hf, Al-Ce pillared vermiculites. Appl. Catal., A. 345, pp. 112-118, 2008. [ Links ]
[12] Campos, A., Moreno, S. and Molina, R., Characterization of vermiculite by XRD and spectroscopic techniques. Earth Sci. Res. J., 13(2), pp. 108-118, 2009. [ Links ]
[13] Campos, A., Moreno, S. and Molina, R., Approach to a descriptive model of a vermiculite charge reduction by hydrothermal treatment. Clays Clay Miner. 58 (1), pp. 97-109, 2010. [ Links ]
[14] Silverstein, R., Bassler, G. and Morrill, T., Spectrometric identification of organic compounds, Jhon Wiley & Sons, New York, 1974. [ Links ]
[15] Jeong, G., Kim, E., Kim, S., Park, E. and Kim, S., Fabrication of sulfonic acid modified silica shells and their catalytic performance within dehydration of D-xylose into furfural. Microporous Mesoporous Mater. 144, pp. 134-139, 2011. [ Links ]
[16] Arena, F., Dario, R. and Parmaliana, A., A characterization study of the surface acidity of solid catalysts by temperature programmed methods, Appl. Catal. A: Gen, 170, pp. 127-137, 1998. [ Links ]
[17] Lima, S., Pillinger, M. and Valente, A., Dehydration of D-xylose into furfural catalyzed by solid acids derived from the layered zeolite Nu-6 (1). Catal. Commun.9, pp. 2144-2148, 2008. [ Links ]
[18] Moreau, C, Durand, R., Razigade, S., Duhamet, J., Faugeras, P., Rivalier, P., Ros, P. and Avignon, G., Dehydration of fructose to 5-hydroxymethylfurfural over H-mordenites. Appl. Catal., A. 145, pp. 211-224, 1996. [ Links ]
[19] Lima, S., Antunes, M., Fernandes, A., Pillinger, M., Ribeiro, M. and Valente, A., Catalytic cyclodehydration of xylose to furfural in the precense of zeolite H-Beta and a micro/mesoporous Beta/TUD-1 composite material. Appl. Catal. A. 388, pp. 141-148, 2010] [ Links ]
[20] Lima, S., Fernandes, A., Antunes, M., Pillinger, M., Ribeiro, F. and Valente, A., Dehydration of xylose into furfural in the presence of crystalline microporous silicoaluminophosphates. Catal. Lett. 135, pp. 41-47, 2010. [ Links ]
[21] Dias, A., Lima, S., Carriazo, D., Rives, V., Pillinger, M. and Valente, A., Exfoliated titanate, niobate and titanoniobate nanosheets as solid acid catalysts for the liquid-phase dehydration of D-xylose into furfural, J. Catal., 244, pp. 230-237, 2006. [ Links ]