Serviços Personalizados
Journal
Artigo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Acessos
Acessos
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares em
SciELO
Similares em
SciELO -
 Similares em Google
Similares em Google
Compartilhar
DYNA
versão impressa ISSN 0012-7353
Dyna rev.fac.nac.minas vol.81 no.185 Medellín maio/jun. 2014
https://doi.org/10.15446/dyna.v81n185.37234
http://dx.doi.org/10.15446/dyna.v81n185.37234
Flower wastes as a low-cost adsorbent for the removal of acid blue 9
Residuos de flores como adsorbentes de bajo costo para la remoción de azul ácido 9
Ana María Echavarria-Alvarez a & Angelina Hormaza-Anaguano b
a Facultad de Ciencias, Universidad Nacional de Colombia, Colombia. amechavarria@gmail.edu.co
b Facultad de Ciencias, Universidad Nacional de Colombia, Colombia. ahormaza@unal.edu.co
Received: February 22th, 2013. Received in revised form: December 2th, 2013. Accepted: April 10th, 2014.
Abstract
This paper describes the use of flower wastes (carnation, rose and daisy) as a potential, alternative and low-cost adsorbent for the removal of Acid Blue 9 (AB9). The best conditions to achieve an efficient adsorption were evaluated in a batch process. With an acidic pH of 2.0, a removal exceeding 90% was obtained using concentrations of AB9 of 15.0 mgL-1 and a dosage of adsorbent of 4.0 gL-1. The equilibrium of the process was modeled using the Langmuir and Freundlich isotherms, obtaining a better fit with the latter one. Kinetic studies indicated a better fit of the process to a pseudo-second order model and negligible effect of temperature. In addition, the bromatological characterization of the adsorbent is shown.
Keywords: adsorption; isotherms; kinetics, acid blue 9; flower wastes.
Resumen
El presente artículo describe el uso de residuos de flores (claveles, rosas y margaritas) como un adsorbente potencial, alternativo y de bajo costo para la remoción del colorante azul ácido 9 (AB9). Las mejores condiciones para lograr una adsorción eficiente fueron evaluadas en un proceso discontinuo. Un pH ácido de 2.0 permitió obtener una remoción superior al 90%, usando concentraciones de AB9 de 15.0 mgL-1 y una dosificación de adsorbente de 4.0 gL-1. El equilibrio del proceso fue modelado usando las isotermas de Langmuir y Freundlich, obteniendo un mejor ajuste con la última. Estudios cinéticos señalaron un proceso de pseudo-segundo orden y un efecto de la temperatura poco significativo. Adicionalmente, se presenta la caracterización bromatológica del adsorbente.
Palabras clave: adsorción; isotermas; cinética; azul ácido 9; residuos de flores.
1. Introduction
Dyes are widely used in textiles, paper, plastics, rubber, leather, cosmetic, pharmaceutical and food industries. The presence of these dyes in water, even at very low concentrations is highly visible and undesirable [1]. Water pollution due to discharge of colored wastewater negatively affects aquatic life and consequently, the overall ecosystem. Dyes reduce the light penetration required for the photosynthetic activity and therefore, the water self-purification process is reduced. More than 700.000 tonnes and around 1.000 different dyes and pigments are manufactured annually worldwide, 10% of them are discharged in wastewater [2,3].
Carcinogenic agents as benzydine and other aromatic amines compounds that can be transformed in more toxic derivatives as a result of microbial activity, are also used in dye manufacture [4,5].
Brilliant blue (acid blue 9) is a synthetic acid dye, with anionic nature belonging to triphenylmethanes, commonly used for flower dyeing, textile and in food industries. It is a highly stable compound and thus causes the color of effluents for extended periods of time. For the treatment of colored effluents, chemical, physical and biological conventional technologies have been tested; however, the physicochemical ones are often expensive and generate toxic sludge. Likewise, on a large scale it becomes a difficult issue in biological treatments [2].
The adsorption process has been proven to be one of the best water treatment technologies around the world and activated carbon is undoubtedly considered as universal adsorbent for the removal of diverse types of pollutants from water[6]. To diversify the abundantly available agricultural waste, it has been proposed to convert it into activated carbons [7,8]. Nevertheless it has considerable production costs [2]. Therefore, it is necessary to find alternative, efficient and inexpensive methods for the treatment of colored wastewater. Biosorption has several advantages among the methods analyzed for the removal of dyes in solution, and a large number of low-cost adsorbents derived from agricultural, poultry waste and even ashes have been successfully tested [9–20]. Structurally agricultural materials consist of lignin, cellulose, hemi-cellulose and some proteins, which make them effective biosorbents [21].
In this study, the use of three locally available, renewable and previously untested adsorbents, carnation, rose and daisy stalks for the removal of acid blue 9 were evaluated. The aim of this investigation was to use low-cost adsorbents to develop a low-priced dye-removal technology. The effect of various parameters on the process such as pH, adsorbent dosage and initial concentration of dye were analyzed. Also, in order to find information on the physico-chemical characteristics of the adsorption, the equilibrium of the process is modeled using the Freundlich and Langmuir isotherms and the kinetics through the equations of the first and second order.
2. Materials and Methods
2.1. Collection and preparation of the flower wastes
The stems of flowers (carnation, rose and daisy) were obtained from a local market. They were washed and dried at 80 °C for 48 hours. Subsequently, they were milled to produce particles of the desired mesh size (500-700 µm) (Physis).
2.2. Flowers waste characterization
2.2.1. Bromatological analysis
Determination of the main components of the studied adsorbent was carried out according to the Van Soest method [22] including the evaluation of acid detergent fiber (ADF), neutral detergent fiber (NDF) and lignin. With this data the content of cellulose and hemicellulose was estimated. Starch content, ash and nitrogen were determined by using the same method. The tests were conducted at the Laboratory of Chemical and Bromatological Analysis of the Universidad Nacional de Colombia - Sede Medellín.
2.3. Chemicals
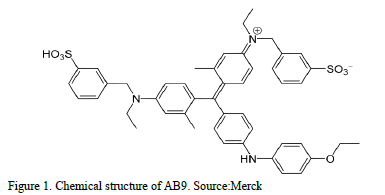
The textile dye Acid Blue 9 (AB9, CI 42090; industrial grade; molecular weight 792.84, molecular formula C37H34N2OS3Na2) was obtained from Merck, Colombia and used without further purification, its chemical structure is shown in Fig. 1.
2.4. Stock solution
A stock solution (500 mgL-1) was prepared by dissolving a determined amount of AB9 in distilled water. This solution was diluted to obtain the desired concentrations.
2.5. Dye concentration analysis
Dye Concentration was determined spectrophotometrically using UV-Vis spectrophotometer (Perkin-Elmer, Lambda 35). The absorbance was recorded at a wavelength of (l = 629 nm), which correspond to the maximum adsorption peek of AB9 and the concentration was determined in the calibration curve.
2.6. Biosorption studies
In order to determine the best conditions for dye removal, adsorption tests were carried out in batch and kept in a shaker Heidolp Unimax 2010 at 25±2 °C and 120 rpm. After stirring, samples were centrifuged in a Fisher Scientific and the supernatant was measured for the determination of remnant dye concentration.
2.6.1. Effect of pH on dye adsorption
The pH of the dye solution was adjusted to a range of 2.0-10.0. This modification was carried out using 1.0 M HCl / NaOH (Merck). Each of the adsorbents (carnation, rose and daisy) was added in 30 mg to 10 mL solution containing 15 mg of dye / L.
2.6.1. Effect of flower waste dose on removal efficiency
Adsorbent dosage was varied in the range of 30-800 mg sorbent / mg of dye. A 10 mL solution with 15 mg of dye / L at pH 2.0 was used. The adsorbent used in this experiment and in the following was an equimolar mixture (carnation, rose and daisy) to get results that could be extrapolated to real situations.
2.6.2. Effect of initial dye concentration on dye sorption
The effect of initial dye concentration was analyzed in a range of 0-15 mg / L. The adsorbent dose (40 mg) was added to 10 mL of solution with variable concentration of dye at pH 2.0.
2.6. Equilibrium studies
The obtained data from equilibrium tests were analyzed using the Langmuir and Freundlich isotherms, in order to obtain information on the surface of the adsorbent. There are several theoretical models; however, the Langmuir and Freundlich isotherms are the most common. Quantification of the amount of dye attached to the biomass was performed using the eq. (1):
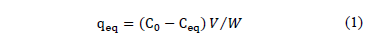
Where:
qeq: Amount of adhered dye to biomass [mg/g]
C0, Ceq: Initial and equilibrium concentration of the contaminant [mgL-1]
V: Volume of dye solution used [L]
W: Mass of added sorbent [g]
2.7.1. Langmuir isotherm
The empirical model of Langmuir sets up the existence of a uniform layer in which there is a finite number of equivalent active sites distributed homogeneously. This model states through eq. (2) that:
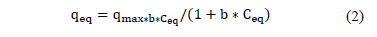
With:
qmax: Langmuir constant denoting the maximum adsorption capacity of biomass [mg/g]
b: Langmuir constant that indicates the affinity for the active sites
Specific constants can be obtained from the intercept and slope of linearized eq.(2):
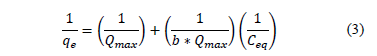
2.7.2. Freundlich isoterm
In this model a mixed monolayer is considered in which the active sites are not independent or equivalent. The specific adsorption capacity is given by eq. (4):
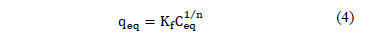
With:
Kf: Freundlich constant related to biomass adsorption capacity
n: Freundlich constant that indicates the intensity of adsorption
From the slope and intercept of the linearized equation, the value of the constants can be determined through eq. (5):
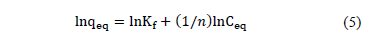
2.7. Kinetic studies
Kinetic studies were performed in order to determine whether the controlling steps are mass transfer or chemical reaction processes. Pseudo - first order and pseudo - second order equations were used, ignoring the movement of the dye ion from the liquid bulk to the liquid film or boundary layer surrounding the adsorbent.
The first order kinetic is based on the adsorbent capacity and is generally expressed by eq. (6).
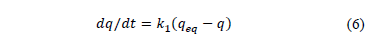
Where qeq and q are the amounts of adsorbed dye on the biosorbent at equilibrium and at time t, respectively (mg/g) and k1 is the rate constant of first-order biosorption (1/min). After integration and applying boundary conditions, t = 0 to t = t and q = 0 to q = q; the integrated form of eq. (6) becomes:
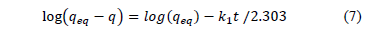
A fit of the experimental data through the straight line of log (qeq – q) vs. t would imply the applicability of this kinetic model. In this case, the parameter qeq must be known. It might happen that qeq is unknown and the adsorption process becomes extremely slow, the amount adsorbed being significantly smaller than the equilibrium amount. For this reason, it is necessary to extrapolate the experimental data to t = ∞ or use trial and error. Moreover, the pseudo - first order model usually fit just over the first period of sorption (20 - 30 min) and does not describe the entire process well [23].
On the other hand, the pseudo - second order kinetic describes all stages of adsorption: external film diffusion, adsorption and internal particle diffusion. The model is based on the adsorbent capacity and assumes the adsorption processes involves chemisorption mechanism. Furthermore, it considers adsorption to be the rate controlling step and is expressed as follows:
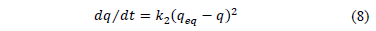
Where k2 is the rate constant of second-order biosorption (g/mg/min). For the boundary conditions t = 0 to t = t and q = 0 to q = q; the integrated form of eq. (8) becomes:
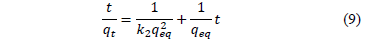
If the second order kinetic is applicable, the plot of t/q against t should give a linear relationship. There is no need of knowing any parameter beforehand. The rate constant is also expressed as a function of temperature by the following Arrhenius type relation:
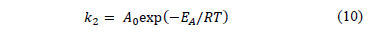
Where A0 is the frequency factor of sorption and EA is the activation energy of sorption. The magnitude of activation energy may give an idea about the type of sorption.
3. Results and Discussion
3.1. Bromatological analysis
It is important to highlight the lack of reports about bromatological composition of these three types of flowers and their mixture. This fact reflects the flower stalks have been little explored as adsorbent material. Table 1 shows the chemical analysis for its major components. The percentages of cellulose, hemicellulose and lignin polymers for the foliage mixture satisfy the required conditions of a potential adsorbent, as has been reported for other agricultural residues with considerable adsorption capacity [24,25]. The ash content is lower when compared to the average value reported for other adsorbents [24–26].
3.2. Effect of pH value on the adsorption process
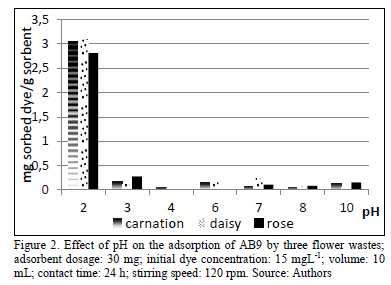
Experimental results show that the adsorption of AB9 is a process highly dependent on the pH. Maximum removal was observed at pH 2.0 with the three kinds of adsorbents (carnation, rose and daisy). By contrast, the adsorption is minimal at higher pH values (3.0-10.0), (Fig.2).
The pH range used did not include extreme values due to degradation of the dye at these points. The dye adsorption capacity at pH 2.0 was 2.08, 3.04 and 3.09 mg/g sorbent, for rose, carnation and daisy, respectively. Similar phenomenon was reported with anionic dyes, with a maximum adsorption at pH 2.0 [15], [23]. This result could be explained by electrostatic interactions between active sites and dissolved molecules. At low pH values, active sites on the surface of
the adsorbent are protonated and then positively charged, which enhances electrostatic forces of attraction with anionic molecules, such as AB9. When the pH is increased, the adsorbent is negatively charged and therefore raises repulsion with the dye molecule.
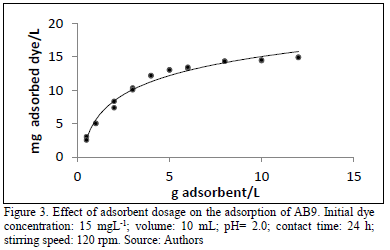
3.3. Effect of sorbent dose on adsorption process
The experiments showed as expected that the adsorption of AB9 increased when a greater amount of adsorbent is used (Fig. 3). At low doses of adsorbent (1.0 gL-1) the quantity of dye removed was 5.04 mgL-1. Nevertheless, when using doses of adsorbent higher than 5.0 gL-1, the amount of removed dye increased to 14 mgL-1 approximately. Akar and coworkers [28], also found similar results for removal of acid blue 40 on Thuja orientalis biomass. Is clear that with higher doses of adsorbent for a determined dye concentration, the available sites over the surface increase, and hence, more dye molecules are retained by the sorbent. In Fig. 3 the influence of the sorbent mixture dosage on dye removal is plotted.
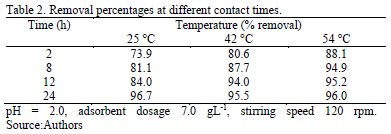
Preliminary studies were performed at different contact times (Table 2). Satisfactory removal of the dye at 24 hours was achieved suggesting this interval as adequate to perform further studies.
3.4. Effect of initial dye concentration
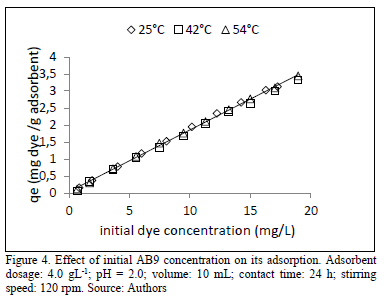
It was determined that initial dye concentration is highly influential on the removal percentage. As the initial concentration of AB9 increases, more molecules are adsorbed per unit mass of adsorbent until a constant value, later all active sorption sites are saturated, and then any transfer from the liquid phase is not possible. The increase of the initial dye concentration from 1.0-18.0 mgL-1, improved the uptake capacity from 0.1-3.0 mg of dye adsorbed / g adsorbent in the three assessed temperatures 25, 42 and 54 °C, (Fig. 4).
The temperatures were selected in order to be consistent with a process in which there is a minimal energy demand. It is important to remark that we seek to design an economical and efficient process for its possible scaling. As can be observed, temperature had a minimal effect on removal effiency. Aksu and Dönmez [23] also found similar trends for the adsorption of acid blue 161 by Trametes versicolor and remazol reactive blue for various types of yeast, respectively.
This initial increase in adsorption process with dye concentration is due to the more availability of dye molecules in solution to be adsorbed. The major quantity of dye influences the increase in driving force and decreases the resistance to mass transfer from the liquid phase to solid phase. Higher dye concentrations were not used in order to avoid exceeding the limit for the absorbance measurements.
3.5. Equilibrium studies
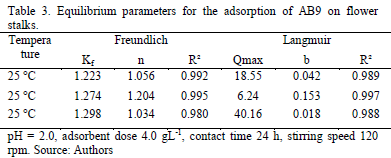
The obtained data from the equilibrium study of adsorption process of AB9 were modeled according to Langmuir and Freundlich isotherms at different temperatures (25, 42 and 54 °C). Taking into account the correlation coefficient, the Freundlich isotherm showed a better fit with an average R2 = 0.992 in the range of concentrations and temperatures evaluated, even though both models fitted very well (Table 3).
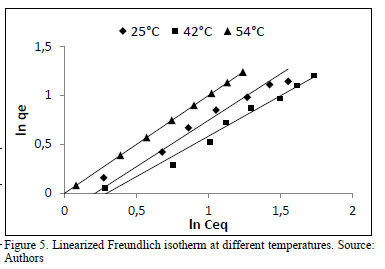
The linearized plots of Freundlich isotherms at different temperatures are presented in Fig. 5
The Freundlich constant Kf increased from 1.22-1.29 with a rise in temperature of 25-54 °C, indicating its slight influence on the process. The higher the temperature, the better the uptake capacity. This might be due to a rise in interactions and collisions frequency among active surface and dye molecules. The value of n, however, shows an irregular pattern, reaching its highest value at 42 °C (n = 1.204). The intensity of biosorption seems to increase until 42 °C and then registers a lower value at 54 °C (n = 1.034).
This behavior could be explained by repulsive interactions at the surface, which reduce binding force between sorbent and solute. This result suggest, first that the increasing temperature has a negligible effect on the performance of the process and secondly, the surface of the adsorbent presents heterogeneous nature with different binding sites and nonequivalent adsorptive energies including electric interferences rather than a homogenous and equivalent monolayer. Osma and coworkers [15] reported a similar adjustment for the adsorption of reactive black 5 on sunflower waste.
According to the Langmuir model, it was not possible to determine a clear relation between temperature and adsorption affinity, nor with adsorption capacity.
3.6. Kinetic studies
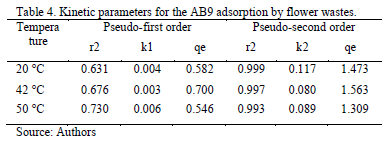
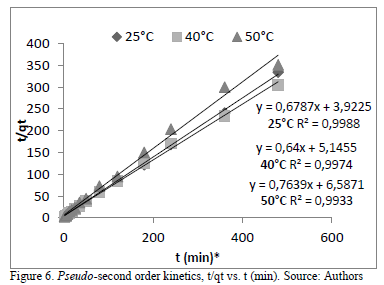
The adsorption process showed a better fit for the pseudo - second order model kinetics, Table 4 and Fig. 6. Therefore, the reaction takes place in heterogeneous conditions because it depends on the amount of solute adsorbed in a time t and on the equilibrium. It indicates that the rate limiting step might be chemical biosorption involving the exchange of electrons between the dye ions and the adsorbent. Results show a negligible effect on temperature. Similar results were obtained for the adsorption of remazol reactive dye and yeasts and AB40 by cone biomass of T.orientalis [23], [28].
In physical adsorption the energy requirements are usually low (no more than 4.2 kJmol-1) since the forces involved are weak. Chemical adsorption is specific and involves forces much stronger than in physical adsorption [27]. Therefore, the kinetic parameters found such as activation energy (EA = 8838.8 Jmol-1) and Arrhenius constant (Ao = 0.003) indicate a chemical nature for the adsorption process of AB9 by flower stems (Table 4).
4. Conclusions
The adsorption experiments demonstrated the great potential of flower stalks as a low-cost and easily available adsorbent for the removal of AB9. It was observed that the extent of adsorption increased by lowering of initial pH up to 2.0, the uptake capacity being a maximum at this value. It was also noted that the specific adsorption capacity decreases with increasing the ratio of adsorbent-dye. The process followed Freundlich isotherm model, showing a slight increase in the uptake capacity with temperature. The adsorption process was better described by pseudo-second order kinetics.
Although the potential of flower wastes as adsorbent for the removal of synthetic dyes was proved, further research is required in order to improve its adsorptive capacity, for example, through chemical modification of the surface of this material. We also suggest the evaluation of isotherms different to Freundlich and Langmuir in order to support the observations of this study. The structural characterization of the material through SEM and IR analysis is being developed in order to get a deeper understanding of the process.
Acknowledgements
The authors thank Universidad Nacional de Colombia – Sede Medellín, Dirección de Investigation, DIME, for the financial support through the Project code 20101007696 as well as to Universidad Nacional de Colombia - Vicerrectoría de Investigación for financing the Project GTI code 40000001102.
References
[1] Robinson, T., Chandran, B. and Nigam, P. Removal of dyes from a synthetic textile dye effluent by biosorption on apple pomace and wheat straw. Water research, vol. 36 (11), pp. 2824–2830, 2002. [ Links ]
[2] Robinson, T., McMullan, G., Marchant, R. and Nigam, P. Remediation of dyes in textile effluent: a critical review on current treatment technologies with a proposed alternative. Bioresource technology, vol. 77 (3), pp. 247–555, 2001. [ Links ]
[3] Zollinger, H. Color Chemistry Colour Chemistry: Synthesis, Properties of Organic Dyes and Pigments. Third. Zurich: Wiley-VCH, 2003, pp. 637. [ Links ]
[4] Anliker, R. and Clarke, E. A. Organic dyes and pigments, in Part A. Anthropogenic compounds, the handbook of environmental chemistry, vol. 3, O. Hutzinger, Ed. Springer, 1980, pp. 181 – 215. [ Links ]
[5] Kariminiaae-Hamedaani, H. R., Sakurai, A. and M. Sakakibara. Decolorization of synthetic dyes by a new manganese peroxidase-producing white rot fungus. Dyes and Pigments, 72 (2), pp. 157–162, 2007. [ Links ]
[6] Bhatnagar A. and Sillanpää M. Utilization of agro-industrial and municipal waste materials as potential adsorbents for water treatment: a review. Chemical Engineering Journal, vol. 157 (2–3), pp. 277–296, 2010. [ Links ]
[7] Rangabhashiyam, S., Anu, N. and Selvaraju, N. Sequestration of dye from textile industry wastewater using agricultural waste products as adsorbents. Journal of Environmental Chemical Engineering, vol. 1 (4), pp. 629–641, 2013. [ Links ]
[8] Nethajia, S., Sivasamya, A., Thennarasua G. and Saravananb, S. Adsorption of Malachite Green dye onto activated carbon derived from Borassus aethiopum flower biomass. Journal of Hazardous Materials, vol. 181 (1–3), pp. 271–280, 2010. [ Links ]
[9] Rafatullah, M., Sulaiman, O., Hashim R. and Ahmad, A. Adsorption of methylene blue on low-cost adsorbents: a review. Journal of Hazardous Materials, vol. 177 (1–3), pp. 70–80, 2010. [ Links ]
[10] Gupta V. K. and Suhas. Application of low-cost adsorbents for dye removal a review, Journal of environmental management, vol. 90 (8), pp. 2313–2342, 2009. [ Links ]
[11] G. Crini, Non-conventional low-cost adsorbents for dye removal: a review. Bioresource technology, vol. 97 (9), pp. 1061–1085, 2006. [ Links ]
[12] Srinivasan A. and Viraraghavan, T. Decolorization of dye wastewaters by biosorbents: a review. Journal of environmental management, vol. 91 (10), pp. 1915–1929, 2010. [ Links ]
[13] Hormaza A. and Suárez García E. Estudio del proceso de Biosorción de dos colorantes estructuralmente diferentes sobre residuos avícolas. Revista de la Sociedad Química del Perú, vol. 75 (3), pp. 329–338, 2009. [ Links ]
[14] Hirata, M., Kawasaki, N., Nakamura, T., Matsumoto, K., Kabayama, M., Tamura, T. and Tanada, S. Adsorption of Dyes onto Carbonaceous Materials Produced from Coffee Grounds by Microwave Treatment. Journal of Colloid and Interface Science, vol. 254 (1), pp. 17–22, 2002. [ Links ]
[15] Osma, J. F., Saravia, V., J. L. Toca-Herrera, J. L. and Couto, S. R. Sunflower seed shells: a novel and effective low-cost adsorbent for the removal of the diazo dye Reactive Black 5 from aqueous solution. Journal of Hazardous Materials, vol. 147 (3), pp. 900–905, 2007. [ Links ]
[16] Hameed, B. H. and Ahmad, A. Batch adsorption of methylene blue from aqueous solution by garlic peel, an agricultural waste biomass. Journal of Hazardous Materials, vol. 164 (2–3), pp. 870–587, 2009. [ Links ]
[17] Aksu, Z. Application of biosorption for the removal of organic pollutants: a review. Process Biochemistry, vol. 40 (3–4), pp. 997–1026, 2005. [ Links ]
[18] Garg, V. Basic dye (methylene blue) removal from simulated wastewater by adsorption using Indian Rosewood sawdust: a timber industry waste. Dyes and Pigments, vol. 63 (3), pp. 243–250, 2004. [ Links ]
[19] Forgacs, E., Cserháti, T. and Oros, G. Removal of synthetic dyes from wastewaters: a review. Environment international, vol. 30 (7), pp. 953–971, 2004. [ Links ]
[20] Fernández, B. and Ayala, J. Evaluation of fly ashes for the removal of Cu, Ni and Cd from acidic waters. Dyna, vol. 77 (161), pp. 141–147, 2010. [ Links ]
[21] Bansal, M., Garg, U., Singh, D. and Garg, V. K. Removal of Cr(VI) from aqueous solutions using pre-consumer processing agricultural waste: a case study of rice husk. Journal of Hazardous Materials, vol. 162 (1), pp. 312–320, 2009. [ Links ]
[22] Van Soest, P. J. and Wine, R. H. Use of detergents in the analysis of fibrous feeds IV. Determination of plant cell-wall constituents. Journal of the Association of Official Analytical Chemists, 50, pp. 50–55, 1967. [ Links ]
[23] Aksu, Z. and Dönmez, G. A comparative study on the biosorption characteristics of some yeasts for Remazol Blue reactive dye. Chemosphere, vol. 50 (8), pp. 1075–1083, 2003. [ Links ]
[24] Chuah, T. G., Jumasiah, A., Azni, I., Katayon, S. and Thomas Choong, S. Y. Rice husk as a potentially low-cost biosorbent for heavy metal and dye removal: an overview. Desalination, vol. 175, pp. 305–316, 2005. [ Links ]
[25] Doria, G. M., Hormaza, A. and Gallego Suarez, D. Cascarilla de arroz: Material alternativo y de bajo costo para el tratameinto de aguas contaminadas con Cromo (VI). Revista Gestión y Ambiente, vol. 14 (1), pp. 73–84, 2011. [ Links ]
[26] Valverde, A., Sarria, B. and Monteagudo J. Análisis comparativo de las características fisicoquímicas de la cascarilla de arroz. Scientia et Technica, vol. 5 (37), pp. 255-260, 2007. [ Links ]
[27] Aksu, Z., Tatli, A. I. and Tunç, Ö. A comparative adsorption/biosorption study of Acid Blue 161: Effect of temperature on equilibrium and kinetic parameters. Chemical Engineering Journal, vol. 142 (1), pp. 23–39, 2008. [ Links ]
[28] Akar, T., Ozcan, A. S., Tunali, S. and Ozcan, A. Biosorption of a textile dye (Acid Blue 40) by cone biomass of Thuja orientalis: estimation of equilibrium, thermodynamic and kinetic parameters. Bioresource technology, vol. 99 (8), pp. 3057–3065, 2008. [ Links ]
A. M. Echavarria-Alvarez, received the Bs. Eng in Biological Engineering at the Universida Nacional de Colombia, Sede Medellin in 2010, the MS degree in in Bioprocess Engineering Design at the Delft University of Technology, Netherlands in 2012. She was a very active member of the Research Group "Synthesis, Reactivity and Transformation of Organic Compounds", SIRYTCOR and her researching experience in the environmental topic allowed her to participate in several projects. Currently she works as a researcher in the Department of Environmental Processes at the Delft University of Technology.
A. Hormaza-Anaguano, received the Chemistry degree at the Universidad de Nariño in 1994, the MS degree in Chemical Science at the Universidad del Valle in 1997, and the PhD degree in Natural Sciences at the Johannes Gutenberg-University Mainz, Germany in 2003. She began working at the a la Universidad Nacional de Colombia Sede Medellín in 1997 as assistant professor and currently she is a full-time Associate Professor in the School of Chemistry, Facultad de Ciencias, Universidad Nacional de Colombia Sede Medellín. Since 2003 she is the Director of Research Group "Synthesis, Reactivity and Transformation of Organic Compounds", SIRYTCOR, whose research lines are focused on the treatment of industrial effluents, exploration and evaluation of alternative adsorbents, adsorption and desorption, and biological processes by solid state fermentation.