Serviços Personalizados
Journal
Artigo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Acessos
Acessos
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares em
SciELO
Similares em
SciELO -
 Similares em Google
Similares em Google
Compartilhar
DYNA
versão impressa ISSN 0012-7353
Dyna rev.fac.nac.minas vol.81 no.187 Medellín set-/out. 2014
https://doi.org/10.15446/dyna.v81n186.40806
http://dx.doi.org/10.15446/dyna.v81n187.40806
Adaptation and validation of a methdology for determing ammonia flux generated by litter in naturally ventilated poultry houses
Adaptación y validación de una metodología para determinar emisiones de amoniaco generado en camas de galpones avícolas con ventilación natural
Jairo Alexander Osorio-Saraz a, Ilda de Fatima Ferreira-Tinoco b, Richard S. Gates c, Keller Sullivan Oliveira-Rocha d, Enrique Miguel Combatt-Caballero e & Fernanda Campos-de Sousa f
a Facultad de Ciencias Agrarias, Universidad Nacional de Colombia, Colombia. aosorio@unal.edu.co
b Facultad de Ciencias Agrarias, Universidad Federal Vicosa, Brasil. iftinoco@ufv.br
c Departamento de Ingeniería Agrícola y Biosistemas Universidad de Illinois, USA. rsgates@illinois.edu
d Facultad de Ingeniería Agrícola, Universidad Federal Vicosa, Brasil. kellersullivan@yahoo.com.br
e Departamento de Suelos, Universidad de Córdoba, Colombia. ecombatt@hotmail.com
f Facultad de Ciencias Agrarias, Universidad Federal Vicosa, Brasil. Fernanda.sousa@ufv.br
Received: November 11th, de 2013. Received in revised form: June 9th, 2014. Accepted: July 22th, 2014
Abstract
The present work aimed to adapt and validate a precise and simple application method defined as the "Saraz method for the determination of ammonia emissions" (SMDAE), based on the method of mass diffusion, to determine ammonia flux due to mass convection from broiler litter. It was found that ammonia flux (N"A) can be obtained by the SMDAE diffusion method. The SMDAE method presents a recovery efficiency for volatilized ammonia of 77 ± 4% and can be used for ammonia concentrations as high as 0.5 ppm.
Keywords: Ammonia flux; poultry houses; broiler litter, air quality.
Resumen
El objetivo del presente trabajo fue avalar y validar un simple y preciso método definido "método Saraz para determinar emisiones de amoniaco" (SMDAE), basado en un método de difusión de masa, para determinar el flujo de amoniaco debido a la convección de la masa (NA") generada en la cama aviaria. Se encontró que el flujo de amoniaco puede ser obtenido por el método difusivo SMDAE. El método SMDAE presentó una eficiencia en la recuperación del amoniaco volatilizado de 77 ± 4% y puede ser usado con concentraciones de amoniaco mayores 0.5 ppm.
Palabras Clave: Flujo de amoniaco; instalaciones aviarias; calidad del aire.
1. Introduction
Knowledge on ammonia emission rates generated from manure in animal confinements is very important, due to its direct relation to negative health effects and productivity of animals and people [1]. Many studies were performed based on the reduction of ammonia emissions from manure by the minimization of nitrogen excretions in the feces due to dietary changes. This procedure is the first step for reducing NH3 emissions from agricultural installations [2, 3]. However, despite the efficiencies obtained by the technique for reducing ammonia by diet manipulation, ammonia emissions cannot be reduced until 100%.
Some methodologies have been developed and validated to determine ammonia gas emissions generated by animal manure, and have been applied in both open and closed animal production installations. However, these obtain different efficiencies in the recovery of the total ammonia nitrogen (TAN), volatilized.
The methodologies most commonly used are those, which involve mass balances, external, and internal tracer gas and the passive methods [4-9].
Most methodologies for ammonia quantification present good performance in closed installations. However, these methods require adaptations for open installations. When using tracer gases, external tracer gases are less efficient compared with internal tracer gases [10,11]. The passive flux method requires predominant air flow in the direction of the flux collector, while the greatest difficulty of the mass balance method is to find the convective mass coefficient [12,13].
Thus, each of the mentioned methodologies has advantages and disadvantages, and a common disadvantage to all is the high cost of operation. Other volatilization models have also been used to predict ammonia emissions based on different circumstances and types of poultry installation [14,15]. The acquisition of mass transfer coefficient (hm), which is an important parameter in the volatilization model of ammonia present in manure, is found in literature with wide variation, and it is a disadvantage of the model.
A methodology used in the study of soils for determining nitrogen (N) loss from the soil by volatilization of TAN makes use of a collector chamber for ammonia fixation by diffusion, where quantification is performed by acid - base titration, using the Kjeldhal method [16]. In this methodology, nitrogen recovery efficiencies of roughly 70% are encountered [17-20].
Based on these facts, the present study aimed to obtain the ammonia flux value due to mass convection (N"A) of broiler litter, using the ammonia mass flux (SMDAE) which is based on the mass diffusion method (J"A).
2. Material and methods
The present study was developed in the Laboratories of the Department of Agricultural Engineering, at the Federal University of Viçosa, Brazil, and in a conventional commercial broiler house working jointly with the Pif-Paf Alimentos S/A company, located in the municipality of Viçosa, MG, Brazil. According to the Köppen classification, the climate of the region is type Cwb - high altitude tropical with wet summers and pleasant temperatures. This study was performed during the summer, with an average temperature of 22°C and relative humidity ranging between 50 and 70%.
2.1. Characteristics of the facilities
The commercial poultry house utilized in this investigation housed 14000 Cobb chickens, with housing density of 12 birds m-2 and the following dimensions: 100 m x 13 m (Length x Width), 3 m high ceilings, 0.50 m overhang and 20° roof inclination angle.
The poultry house, with little thermal insulation, which is commonly observed in Brazil and South America, was open with natural ventilation during the experimental phase and the litter was composed of fresh coffee hulls.
2.2. Mass diffusion method proposed for determining ammonia mass flux called SMDAE
A passive flux method used by [19] and [21] was adapted and validated for determining ammonia flux from the litter of poultry buildings. This adapted method, called "Saraz method for determination of ammonia emissions" (SMDAE), is based on the mass diffusion method for the determination of ammonia flux from broiler bedding, based on the total volatilized ammonia content that is volatilized and captured.
2.2.1. Measuring equipment
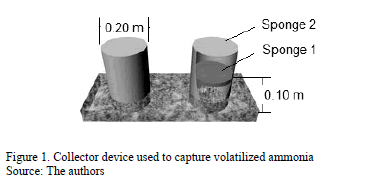
A common PVC pipe measuring 20 cm in diameter and 30 cm in height was used to construct the NH3 capturing device. Two polyurethane sponges measuring 20 cm in diameter each and thickness of 2 cm were placed in the tube so that they were 10 cm (Sponge 1) and 30 cm (Sponge 2) from the base of the PVC collector. The function of sponge 1 was to directly capture the ammonia flux produced by the poultry litter bedding, while sponge 2 was used to prevent contamination by exterior gases that might affect the values of the ammonia captured by sponge 1 (Fig. 1).
2.2.2. Appropriate time for ammonia capture
The objective of this experiment was to encounter the ammonia flux from the bed and simulate the natural conditions of this emission in real time, in order to determine the appropriate ammonia adsorption period for the collector device. Thus, the tests performed lasted 1, 2, 3, 4, 12, 16, 22 and 24 hours, with three repetitions for each time.
2.2.3. Location of the collector devices and collection of experimental data
Data collection was performed on three consecutive days in each week of the birds lives, between 22-28, 29-35, and 36-48 days of the productive cycle. In compliance with the studies performed by [6] it was considered that, on the first 14 days, ammonia emissions are minimal and, after this time, emissions increase linearly.
Seeking to observe the influence of waterers and feeders on ammonia flux, compared to other regions of the poultry house, four collector devices were installed in the vicinity of the feeders and four, in the vicinity of the waterers. Ammonia flux measurements were taken during 9 days between 8:00 to 10:00 AM and 3:00 to 5:00 PM.
2.2.4. Determination of the quantity of ammonia captured
To capture volatilized ammonia, each sponge was impregnated with 80 ml of a solution composed of sulfuric acid (1 mol L-1) and glycerine (3 %), corresponding to an adaptation of the ammonia fixation method by diffusion, whose quantification is performed by acid-base titration by the Kjeldhal method [16].
Eighty mL of a potassium chloride (KCl) solution at a concentration of 0.5 mol L-1 added to 40 mL of water were used to extract the ammonia captured in the sponge. This solution mixed with the sponge was prepared in a Tecnal model TE-0363 nitrogen distillation column. After distillation, the condensed sample was titrated with hydrochloric acid (HCl) at a concentration of 0.5 mol L-1. The NH3 concentration (g NH3) captured by the sponge was obtained by the volume of the tilter solution (mL), the solution concentration (mol L-1), and number of moles of NH3 The SMDAE mass flux was obtained by using the eq. (1).
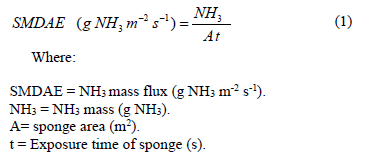
2.2.5. Determination of the SMDAE efficiency
To determine the efficiency of the proposed SMDAE method for ammonia recovery, it was determined the difference between the quantity of NH3 volatilized from the litter and the quantity of NH3 recovered by the sponge. Ten replications were performed to achieve this value.
2.3. The theoretical proposed SMDAE diffusion method and the mass convection method
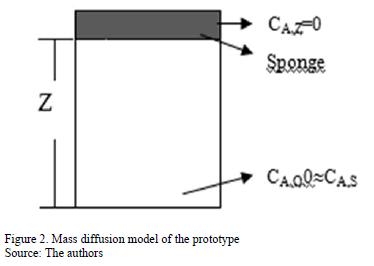
The proposed SMDAE diffusion method is derived from Fick's Second Law. A schematic of the prototype is presented in Fig. 2, where CA,s (g m-3) corresponds to concentration of specie A at the litter bedding surface, CA, Z (g m-3) concentration at height Z of the sponge; J"A that is equal to the ammonia emission flux SMDAE captured by the sponge (g m-2 s-1); and DAB is the diffusion coefficient of ammonia in the air (0.28 x 10- 4 m2 s-1) according to [22].
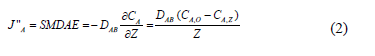

For the mass convection model, a boundary limit model was used for concentration of a chemical species on a flat surface, where N"A is the ammonia flux (g m-2 s-1) and hm the mass diffusion coefficient. This coefficient is a function of the Reynold's number (Re) and the Schmidt number (Sc); V is the average wind speed at the height of the birds; L is the length of the installation; and n is the viscosity of the air. Mass flux by convection is determined as [22]:

For the case in which it is considered outside the boundary limit, mass flux is determined as:

Because flow in the building is turbulent, the mass convection coefficient is calculated as:
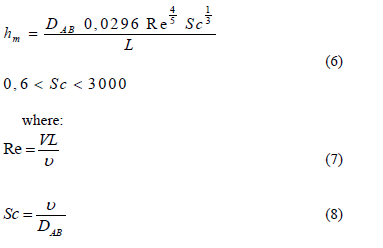
2.4. Statistical analyses
After the experiments, the data obtained from both measurement methods (SMDAE diffusion and convection models (N"A) were treated and analyzed statistically, and a non parametric one-way analysis of variance (Kruskal-Wallis test), with method as treatment, was conducted.
To determine the incidence of variables such as location (waterer and feeder) and the time of the day a non parametric one-way analysis of variance (Kruskal-Wallis test), with period as treatment was conducted.
2.5. Acquisition of experimental data
Background ammonia concentration data in the environment were obtained from an electrochemical detector "Gas alert Extreme Ammonia (NH3) Detector" from BW Technologies with a measuring range from 0-100 ppm, temperature between -4 to + 40°C, relative humidity from 15% to 90% and presenting an accuracy of ± 2%. Data collection was performed in twenty minutes interval.
Air temperature at sample height was measured (DS1820, Dallas Semiconductor, address). Energy was provided to the 1-wireTM system by a parasitic feed derived from the data transmission conductor, where only two conductors are necessary. Temperature measurements were made every five minutes. Air speed (m s-1) was measured with a digital wind gage (Testo 425), with a range between 0- 20 m s-1, precision of ± 0.5 (°C), accuracy of 1% (pressure) and 2.5% (m s-1) and 0.1°C, positioned five centimeters in front of each sponge on the upwind side. Air velocity data collection was performed in five minutes interval. Relative humidity of the air inside and outside of the poultry house was obtained at diverse points representing the entire poultry house, using independent systems (Hobo H8-032) with accuracy of ±0.7 at 21°C. Data collection was performed at one second intervals.
The pH of the poultry litter was determined in the laboratory using a digital pH meter, for which each sample of the bed collected in the installation was diluted in water at a 1:4 ratio (bed sample:water). Moisture content of the litter was determined in the laboratory as the mass difference between the dry and moist mass using an oven at 105°C.
3. Results and discussion
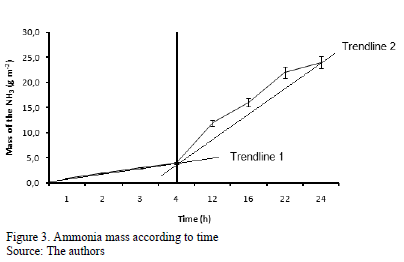
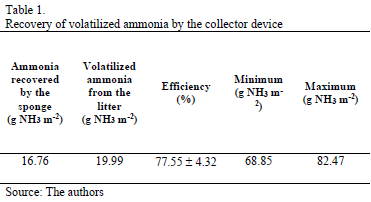
Fig. 3 presents the behavior of the ammonia mass captured by the collector device encountered by the mass diffusion method in function of the time,. It was observed that the behavior of the curve of ammonia for all replicates was linear in function of time, with a greater increase in emissions after the prototype was exposed for four hours. Hence, the prototypes were exposed for no more than two hours to facilitate sampling in the field and allows for a larger numbers of experimental replicates. Table 1 shows the ammonia mass recovery data and the ammonia recovery curve, according to its volatilization. The collector device used had a recovery efficiency of 77.55± 4.32 g NH3 m-2 and was efficient, compared to the experiments performed by [19, 23] and [21], who found 70% efficiency when using the chamber collector method. Moreover, the proposed method can capture ammonia concentrations exceeding 0.5 ppm.
After determining the efficiency of the collector device, the mass diffusion flux SMDAE was calculated by equation 3. The value of CA,s was obtained from the SMDAE. The ammonia fluxes were achieved by equation 4, by the mass convection model (N"A).
The mass convection coefficient (hm) was calculated from equation 6 for turbulent flow, temperatures between 25 and 30°C, and velocities at the concentration boundary limit ranging from 0.10 to 0.35 m s-1, where the values found in this experiment are in agreement with other experiments, including [24] and [25]. The values of hm achieved ranged from 5.15 x 10-4 to 1.34 x 10- 3 m s-1. These hm values did not differ from those reported by [26] and [27], who worked with velocities at the same range.
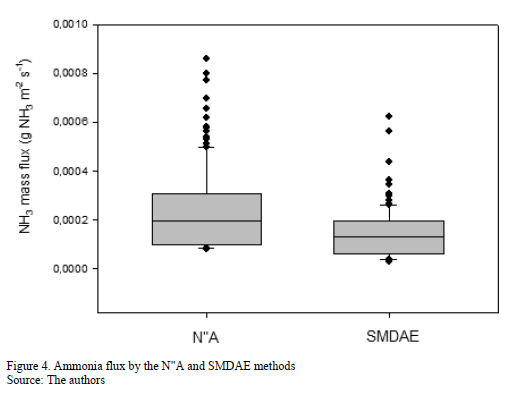
The analysis of variance between the N"A and SMDAE method achieved is shown in Fig. 4. The test reveals There was a significant difference between NH3 flux values determined by the SMDAE method with the emissions for mass convection obtained by the N"A (p<0.001), as expected, due to the incidence of wind in the N"A method.
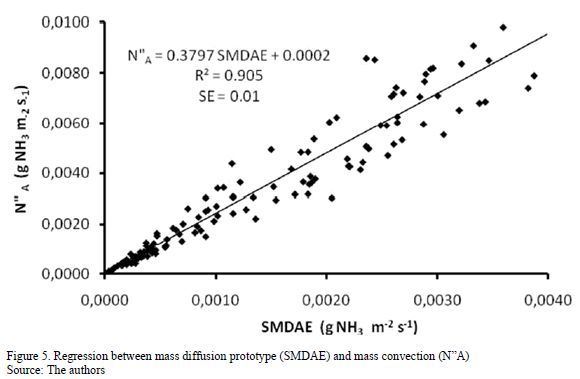
Fig. 5 shows the regression of the SMDAE model of mass diffusion and mass convection N"A, at the significance level of p< 0.05. The values of N"A underestimate the SMDAE in all cases, as demonstrated in Fig. 4. However, it was observed that the R2 coefficient was 0.91, which reveals a high correlation between the models and that the SMDAE method can be used to determine N"A from poultry manure in terms of natural ventilation. The values of N"A within the range 10-5 and 10-3 gNH3 m-2s-1 did not differ from those reported by [13, 27, 28, 29], who worked with mass transfer methods.
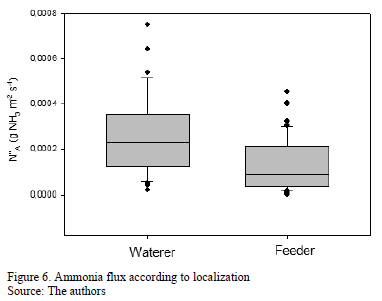
The boxplot of the Fig. 6 show the statistical analysis for the correlation between the convective mass flux (N"A) in function of the location of the samples (waterer and feeder). Results of the analysis of variance at the confidence level showed that There were significant differences (p<0.001). The difference in ammonia flux (N"A) from the litter in the areas of the feeders and waterers may be due to the lower moisture content near the feeders in comparison with the waterers. This was expected, since, according to [28, 30], the total volatilized ammonia (TAN) increases when the moisture of the litter bedding increases.
4. Conclusions
The SMDAE method has a good relationship with the N"A mass convection method, which is the method most commonly used for works with mass balances from ammonia sources, and presented a recovery efficiency of approximately 78% of total volatilized ammonia, and can capture ammonia at concentrations as high as 0.5 ppm. Therefore, this method could be considered efficient and used as an alternative to determine ammonia flux (N"A) from the poultry litter in installations with natural ventilation.
Acknowledgements
The authors are thankful to the National University of Colombia for this great opportunity; Colciencias-Colombia; the Brazilian Government Agency FAPEMIG; the National Counsel of Technological and Scientific Development (CNPq - Brazil) and Federal agency CAPES, for their financial support; and the Federal University of Viçosa (UFV-Brazil).
References
[1] Tinôco, I.F.F., A Granja de Frangos de Corte. In: Mendes, A.A.; de Alencar-Naas, I.; Macari, M., (Eds.), Produção de Frangos de Corte. 1 ed. Campinas - SP, 2004. pp. 1-345. [ Links ]
[2] Panetta, D.M., Powers, W.J., Xin, H., Kerr, B.J. and Stalder, K.J., Nitrogen excretion and ammonia emissions from pigs fed modified diets. Journal of Environmental Quality, 35 (4), pp. 1297-1308, 2006. [ Links ]
[3] Ndegwa, P.M., Hristov, J., Arogo E.R. and Sheffield, E., A review of ammonia emission mitigation techniques for concentrated animal feeding operations, Biosystems Engineering, 100 (4), pp. 453-469, 2008. [ Links ]
[4] Welford, E.L., Stéphane, B.E., Lemay, P., Barber, M. and Godbout, S., Simulation of ammonia emissions from swine manure for various diet formulations. In: CSAE/SCGR Meeting Montréal, pp. 6 - 9, 2006. [ Links ]
[5] Nicholson, F.A., Chambers B.J. and Walker, E.A.W., Ammonia Emissions from broiler litter and laying hen manure management systems, Biosystems Engineering, 89 (2), pp. 175-185, 2004. [ Links ]
[6] Gates, R.S., Xin, H., Casey, K.D., Liang, Y. and Wheeler, E.F., Method for measuring ammonia emissions from poultry houses, J. Applied Poultry, 14 (1), pp. 622-634, 2005. [ Links ]
[7] Reidy, B., Mmgen, D.A., Hler, D.H., Eurichmenden, B., Van Evert, F.K., Hutchings, N.J., Luesink, H.H., Menzi, H., Misselbrook, T.H., Monteny, G.J. and Webb, J., Comparison of models used for national agricultural ammonia emission inventories in Europe: Liquid manure syst., Atmospheric Environment, 42 (14), pp. 3452-3464, 2008. [ Links ]
[8] Osorio J.A., Tinoco, F.F. and Ciro H.J., Ammonia: a review about concentration and emission models in livestock structures, DYNA, 76 (158), pp. 89-99, 2009. [ Links ]
[9] Osorio, J.A., Tinoco, I.F.F., Gates, R.S., Oliveira, M.P. and Mendes, L.B., Evaluation of different methods for determining ammonia emissions in poultry buildings and their applicability to open facilities, DYNA, 80 (178), pp. 51-60, 2013. [ Links ]
[10] Dore, C.J., Jones, B.M.R., Scholtens, R., Veld, J.W.H.H.I.T., Burgess, L.R. and Phillips, V.R., Measuring ammonia emission rates from livestock buildings and manure stores, Part 2: Comparative demonstrations of three methods on the farm, Atmospheric Environment, 38 (19), pp. 3017-3024, 2004. [ Links ]
[11] Phillips, V.R.; Scholtens, R., Lee, D.S., Garland, J.A. and Sneath, R.W., SE-Structures and Environment: A review of methods for measuring emission rates of ammonia from livestock buildings and slurry or manure stores, Part 1: Assessment of basic approaches. Journal of Agricultural Engineering Research, 7 (4), pp. 355-364, 2000. [ Links ]
[12] Keener, H.M. and Zhao, L., A modified mass balance method for predicting NH3 emissions from manure N for livestock and storage facilities, Biosystems Engineering, 99 (1), pp. 81-87, 2008. [ Links ]
[13] Teye, F.K and Hautala, M., Adaptation of an ammonia volatilization model for a naturally ventilated dairy building, Atmospheric Environment, 42 (18), pp. 4345-4354, 2008. [ Links ]
[14] Arogo, J., Westerman, P.W. and Liang, Z.S., Comparing ammonium ion dissociation constant in swine anaerobic lagoon liquid and deionized water, Transactions of the ASAE, 46 (5), pp. 1415-1419, 2003. [ Links ]
[15] Pinder, R.W., Pekney, N.J., Davidson, C.I. and Adams, P.J., A process-based model of ammonia emissions from dairy cows: Improved temporal and spatial resolution, Atmospheric Environment, 38 (9), pp. 1357-1365, 2004. [ Links ]
[16] Association of Official Analitical Chemists - AOAC, Official methods of analysis. 11. Ed., Washington D.C, 1015 P, 1970. [ Links ]
[17] Yang, P., Lorimor, J.C. and Xin, H., Nitrogen losses from laying hen manure in commercial high-rise layer facilities, Transactions of the ASAE, 43 (6), pp. 1771-1780, 2000. [ Links ]
[18] Sangoi, L., Ernani, P., Lech, V. and Rampazzo, C. ,Volatilization of N-NH3 influenced by urea application forms, residue management and soil type in lab conditions, Ciência Rural, 33 (4), pp. 687-692, 2003. [ Links ]
[19] Renata, H., Cazetta J. and Moraes V.M., Frações nitrogenadas, glicidas e amonia liberada pela cama de frangos de corte em diferentes densidades e tempos de confinamento, Rev.bras. zootec, 3 (4), pp. 1795-1802, 2002. [ Links ]
[20] Leal, V.L.A., Salamanca, J.A. and Sadeghian, S., Pérdidas de nitrógeno por volatilización en cafetales en etapa productiva, Cenicafé, 58 (3), pp. 216-226, 2007. [ Links ]
[21] Araujo, E.S., Marsola, T., Miyazawa, M., Boddey, R.M., Urquiaga, S. and Rodrigues, B.J., Câmara coletora para quantificação do N-NH3 volatilizado do solo. In: XXXI Congresso Brasileiro de Ciência do Solo., Gramado RS - Brasil, pp. 1-6, 2007. [ Links ]
[22] Incropera, F.P. and Dewitt, D.P., Fundamentals of heat and mass transfer. Wiley, New York. pp. 344-369, 1999. [ Links ]
[23] Renata, H. and Cazetta, J., Método simples para determinar amônia liberada pela cama aviaria, Rev.bras. zootec, 30 (3) pp. 824-829, 2001. [ Links ]
[24] Brewer, S.K. and. Costello, T.A., In situ measurement of ammonia volatilization from broiler litter using an enclosed air chamber, Transaction of ASAE, 42 (5), pp 1415-1422, 1999. [ Links ]
[25] Menegali, I., Tinôco, I.F.F., Baêta, F.C., Cecon, P.R, Guimarães, M.C.C. and Cordeiro, M.B., Ambiente térmico e concentração de gases em instalações para frangos de corte no período de aquecimento, Revista Brasileira de Engenharia Agrícola e Ambiental, 13 (10), pp. 984-990, 2009. [ Links ]
[26] Ni, J., Mechanistic models of ammonia release from liquid manure: a review, J. Agric. Eng Res, 72 (1), pp. 1-17, 1999. [ Links ]
[27] Liu Z.L., Wang, D.B. and Beasley, S.S.B., Modelling ammonia emissions from broiler litter at laboratory scale, Transactions of the ASABE, 52 (5), pp. 1683-1694, 2009. [ Links ]
[28] Miragliotta, M.Y., Avaliação dos níveis de amônia em dois sistemas de produção de frangos de corte com ventilação e densidade diferenciados. Teses. Faculdade de Engenharia Agrícola, Universidade Estadual de Campinas, Campinas, Brasil, 2001. [ Links ]
[29] Redwine, J.S., Lacey, R.E., Mukhtar, S. and Carey, J.B., Concentration and emissions of ammonia and particulate matter in tunnel-ventilated broiler houses under summer conditions in Texas, Transactions of ASAE, 45 (4), pp. 1101-1109, 2002. [ Links ]
[30] Jones, T.A., Donnelly, C.A. and Dawkins, M.S., Environmental and management factors affecting the welfare of chickens on commercial farms in the United Kingdom and Denmark stocked at five densities, Poultry Science, 84 (8), pp. 1155-1165, 2005. [ Links ]
J. A. Osorio-Saraz, received the Bs. Eng. in Agricultural Engineering (1998), the Sp in Environmental Legislation (2001) and the MSc. degree in Materials Engineering (2006), all of them from the Universidad Nacional de Colombia, Medellín, Colombia. In 2011 he received a DSc. degree in Rural Constructions from the Federal University of Viçosa, Minas Gerais, Brazil. Since 2003 is Professor at the Universidad Nacional de Colombia, teaching and advising in the areas of Design of Livestock Housing, Materials Technology for Livestock Housing, air quality and animal welfare, thermal confort for animals, use of the CFD tool to predict air motion patterns in agro industry facilities. He is member of AMBIAGRO (Center for Research in Agro-Industrial Ambience and Engineering Systems) at the Department of Agricultural Engineering, Federal University of Viçosa and of the research group in agricultural engineering at the Universidad Nacional de Colombia. He is currently the Dean of the Faculty of Agricultural Sciences at Universidad Nacional de Colombia Medellin since 2012.
I. de F. Ferreira-Tinôco, received the Bs. Eng. in Agricultural Engineer (1980), the MSc. degree in Animal Structures (1988), the DSc. degree in Animal Sciencesm (1996), all of them from the Federal University of Lavras, Minas Gerais state, Brazil. She is currently associate professor at the Department of Agricultural Engineering of Federal University of Viçosa, Brasil, and coordinates the UFV branch of the following scientific exchange programs: (1) CAPES/FIPSE Agreement involving the University of Illinois and University of Purdue, in the U.S.A.; (2) Umbrella Agreement amongst UFV and Iowa State University, University of Kentucky (both in the U.S.A.); (3) Scientific and Technical Agreement amongst UFV and University of Évora (Portugal), Universidad Nacional de Colombia (Colombia), Iowa State University and University of Kentucky (U.S.A.). Also coordinates the Center for Research in Animal Microenvironment and Agri-Industrial Systems Engineering (AMBIAGRO) and is the President of the DEA-UFV Internation Relations Committee. Her research areas are design of rural structures and evaporative cooling systems, thermal confort for animals, air quality and animal welfare.
R.S Gates, received the Bs. Eng. in Agricultural Engineering at the University of Minnesota (1978), the MSc. degree in Agricultural Engineering (1980) and PhD. in Biological Engineering (1984), all of them from the Cornell University, USA. He is currently Professor at the Agricultural and Biological Engineering Department, University of Illinois. Among his honors and awards has several honorable mentions and awards ASABE since 1987 in the areas of livestock, environment, and biology; He is a member of the American Society of Agricultural and Biological Engineering (ASABE, 2010) His research interest onclude: Controlled environment systems analysis, control and simulation, Dietary manipulation in poultry and livestock for reduced aerial gases and building emissions; poinsettia propagation and hydroponic lettuce, Control systems development including fuzzy logic, heuristics and vapor pressure deficit, Livestock production models for real-time economic optimization.
E. M. Combatt-Caballero, received the Bs. Eng. in Agronomic Engineer in 1992, from the Universidad de Córdoba, Colombia, MSc. in Soils in 2004, from the Universidad Nacional de Colombia, Colombia, and Dr in 2010 from the Federal University of Viçosa, Brazil. He is currently a professor at the Universidad de Cordoba, Colombia and he is a head director of the soils Lab. His research interests include: crop physiology, chemistry and physics soil, soil analytical methods.
K. S. Oliveira-Rocha, received the Bs. in Information Systems with experience in Computer Science, in 2005 from University Center UNA, the MSc. degree in Agricultural Engineering in 2008, and Dr. degree in with emphasis in Energy in Agriculture in 2012, all of them from at the Department of Agricultural Engineering at the Federal University of Viçosa, Brazil. He has experience in the areas of instrumentation, process control, data acquisition using electronic devices addressable systems, numerical methods applied to engineering, simulation, grain drying and aeration, ventilation systems, machine vision, image processing. He is a member of AMBIAGRO (Center for Research in Agro-Industrial Ambience and Engineering Systems), Department of Agricultural Engineering, Federal University of Viçosa, Brazil.
F. Campos-de Sousa, received the Bs. Eng. in Agricultural and Environmental Engineering in 2012, the MSc. degree in Agricultural Engineering in the area of Rural Ambience Constructions in 2014, all of them from the Federal University of Viçosa, Brasil. She is currently a doctoral student in the field of Agricultural Engineering and Rural Constructions. She is currently member of AMBIAGRO (Center for Research in Agro-Industrial Ambience and Engineering Systems), Department of Agricultural Engineering, Federal University of Viçosa Brazil.