Serviços Personalizados
Journal
Artigo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Acessos
Acessos
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares em
SciELO
Similares em
SciELO -
 Similares em Google
Similares em Google
Compartilhar
DYNA
versão impressa ISSN 0012-7353
Dyna rev.fac.nac.minas vol.82 no.190 Medellín mar./abr. 2015
https://doi.org/10.15446/dyna.v82n190.42638
DOI: http://dx.doi.org/10.15446/dyna.v82n190.42638
The influence of the glycerin concentration on the porous structure of ferritic stainless steel obtained by anodization
Influencia de la concentración de glicerina en la estructura porosa obtenida por anodización de acero inoxidable ferrítico
Alexander Bervian a, Gustavo Alberto Ludwig b, Sandra Raquel Kunst c, Lílian Vanessa Rossa Beltrami d, Angela Beatrice Dewes Moura e, Célia de Fraga Malfatti f & Claudia Trindade Oliveira g
a Universidade Federal do Rio Grande do Sul, Porto Alegre, Brazil, alexbervian@yahoo.com.br
b Universidade Federal do Rio Grande do Sul, Porto Alegre, Brazil, gustavolludwig@gmail.com
c Universidade Federal do Rio Grande do Sul, Porto Alegre, Brazil, tessaro.sandra@gmail.com
d Universidade Federal do Rio Grande do Sul, Porto Alegre, Brazil, lvrossa@yahoo.com.br
e Universidade Feevale, Novo Hamburgo, Brazil, angelab@feevale.br
f Universidade Federal do Rio Grande do Sul, Porto Alegre, Brazil, materiaisxenergia@gmail.com
g Universidade Feevale, Novo Hamburgo, Brazil, ctofeevale@gmail.com
Received: September 6th, 2012. Received in revised form: November 1th, 2013. Accepted: November 25th, 2013.
This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.
Abstract
Anodizing of ferritic stainless steel has been used for decoration purposes to obtain a barrier effect. The most commonly used electrolyte for this process is INCO (5M H2SO4 + 2,5M CrO3). INCO electrolyte with glycerin addition induces the formation of ordered porous structures, because glycerin reduces the electrolyte conductivity. Ferritic stainless steel was anodized in electrolyte composed by 2M H2SO4 with glycerin addition in different concentrations, without chromium oxide addition. It was observed that the addition of 90 v/v% glycerin promoted a reduction in the electrolyte conductivity, which caused an increase in the anodizing potential. The glycerin addition to the electrolyte diminished the oxide dissolution in the electrolytic solution, promoting the formation of an oxide with an ordered porous structure.
Keywords: Anodizing; ferritic stainless steel; glycerin; porous structure.
Resumen
La anodización de acero inoxidable ferrítico ha sido utilizada con fines decorativos, para obtener una capa de barrera. El electrólito más comúnmente utilizado para este proceso es el INCO (5M H2SO4 + 2,5M CrO3). Con adición de glicerina induce la formación de estructuras porosas ordenadas, pues la glicerina reduce la conductividad del electrólito. Se anodizaron muestras de acero inoxidable ferrítico, en electrólito de 2M H2SO4 con la adición de glicerina en diferentes concentraciones, sin adición de óxido de cromo. Se observa que la adición de glicerina en un 90 v/v % promovió la reducción de la conductividad del electrólito, lo que ocasionó un aumento del potencial de anodización. Los resultados muestran que la adición de glicerina al electrólito redujo la disolución del óxido en la solución electrolítica y promovió la formación de un óxido con una estructura porosa ordenada.
Palabras clave: Anodización, acero inoxidable ferrítico, glicerina, estructura porosa.
1. Introduction
The resistance of stainless steel to corrosion is high for a wide range of conditions [1], mainly environmental ones. As chromium is the main alloy element, the addition of nickel and molybdenum improve resistance to corrosion [2]. These materials are less corrosion susceptible and more resistant to attack than common steel, but they are not completely immune [3].
The presence of determined levels of chromium in a stainless steel promotes resistance to corrosion [2]. Chromium dissolved in the iron lattice forms a thin oxide film on the steel surface, referred to "passive layer," protecting the metal against corrosion [4].
Metal corrosion protection has been widely studied [5-7]. Anodizing is an electrochemical process that promotes the obtaining of a protective oxide on the metal surface. This process is carried out by the immersion of the metal in an electrolytic bath, in which the metal is the anode; cathode can be an inert metal, for instance, platinum. The Anodizing process enhances the obtaining of barrier and porous oxide layers [8].
Barrier oxides obtained by the anodizing process are able to hinder electron conduction, which enhances a resistive character and a relatively uniform thickness, in addition to being compact, dense and thin [9]. In order to bring about the formation of a barrier layer, the electrolyte must not attack the formed oxides [10].
On the other hand, the formation of porous oxides is favored with electrolytes that chemically attack the oxide. It takes place due to the dissolution of the barrier layer in the electrolyte, which generates attacked spots on the surface. That generation takes place inside, specifically in the smallest pores, in the center of the hexagonal cells. A single potential is developed in each attack point, which then becomes a source of current. In the oxide-electrolyte interface there are ions that supply oxygen to transform the attacked point to oxide. At the same time, the reduction of the thickness of the barrier layer which occurs due to the action of dissolving the electrolyte in the pore base tends to decrease. Then, there is a pore lengthening that facilitates the ions' pass and the heat outflow, aiding the oxide film dissolution [10,11].
Currently, the anodizing process is widely used in stainless steel for aesthetic purposes [12], so the works developed on anodizing such as surface treatment for stainless steel are very few [13].
The electrolyte INCO (5M H2SO4 + 2,5M CrO3) is widely used because it is extremely conductive [14]. In any case, given its high toxicity, chromium has to be replaced, and this has provided the motivation for the research of alternative electrolytes. Studies showed that valve metal anodizing in electrolyte containing glycerin induces the formation of ordered porous structures, due to the fact that glycerin reduces the electrolyte conductivity [15].
An increasing interest in applications for glycerin has been motivated by the increase of biodiesel production, since glycerin is one of its co-products. Glycerin ensures high viscosity and low conductivity to the electrolyte. It promotes the formation of porous oxides on the metals during the anodizing process [16].
The aim of this study is to evaluate the influence of different glycerin concentrations on the 2M H2SO4 electrolyte, in the properties of the oxides formed on ferritic stainless steel during the anodizing process.
2. Experimental
2.1. Elaboration of the samples
AISI 430 ferritic stainless steel plates of 20 mm x 20 mm x 0.5 mm were supplied by Aços Riograndense, with the composition that is shown in Table 1.
Samples were sanded down with silicon carbide paper (200 - 4000) and polished using 1 mm diamond paste and neutral alcohol in the Pentec polishing machine Polipan-U.
After the surface preparation, electric contact was mounted with a copper wire and two layers of acetone-based lacquer on the unpolished side and about 2 mm on each border on the polished side. After this process, the samples were anodized in the solutions specified in Table 2. Each sample remained with an exposed area of approximately 2.5 cm2 during the anodizing process.
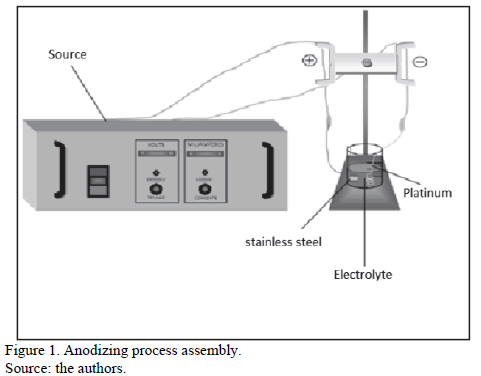
Tests were carried out at room temperature. A current density of 4 mA.cm-2 during three minutes [17]. The sample of AISI 430 stainless steel was the anode and a platinum wire was the cathode. The equipment used possesses a 0 - 300 V potential source and a 0 - 500 mA current source. Figure 1 shows the assembly of the anodizing process.
To determine the electrolyte conductivity a microprocessed conductivimeter pH determination was made at 25 ºC. These equipments are found at the Central Analítica at Universidade Feevale.
Morphological analysis of the obtained oxides was undertaken by scanning electronic microscopy (SEM). With the SEM micrographs and the Image Tool 3.0 software, pore diameter was measured. Through the energy dispersive spectrometer (EDS) coupled to the SEM equipment, a chemical elements mapping of the samples was undertaken.
Surface structural characterization was carried out by X-ray diffraction (XRD) in a Bruker AXS D5000. The Ka beam was generated by a copper tube with 40 kV and 25 mA.
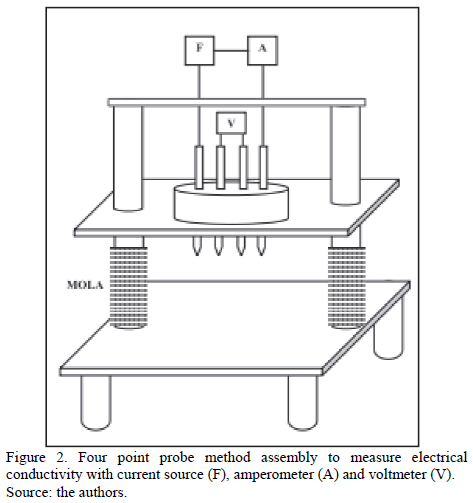
The four point probe method was used to determine the oxides electrical conductivity. A variable potential/current source 20 V/20 A, fabricated by Universidade Feevale was used, applying a voltage to close the circuit. Current was measured with a Minipa ET 2042C tester and for voltage an Agilent 34401 tester was used. The assembly can be seen in
Atomic force microscopy (AFM) was carried out in a SHIMADZU SPM 9500J3 in contact mode at LAPEC laboratory in the Universidade Federal do Rio Grande do Sul (UFRGS).
3. Results and discussion
3.1. Electrolyte pH and conductivity
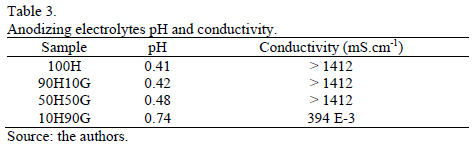
Table 3 shows the pH and conductivity values corresponding to the different anodizing electrolytes. All the samples showed an acidic character; however, the increase in the glycerin percentage promoted a slight increase in the pH value. It was not possible to measure conductivity for the samples 100H, 90H10G and 50H50G due to the limitations of the equipment used. Sample 10H90G showed less conductivity compared to the other samples.
3.2. Anodizing curves
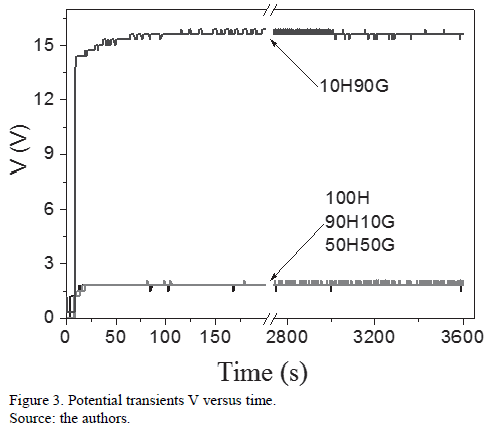
Figure 3 shows the potential transients measured during anodizing. 10H90G had an anodizing potential of 1.5 V; those points out were about the formation of an oxide on the sample surface. Samples 100H, 90H10G and 50H50G had anodizing potentials close to 1.5 V for 1 hour and maintained the same behavior. As formerly stated, the conductivity of these samples are too high. This can be related to the intense oxide dissolution, hindering their accrual on the sample surface. It keeps the anodizing potential reduced for those samples.
3.3. Morphological analysis
In the micrographs, sample 100H (Figure 4a-b) shows an important presence of pores formed due to the intense oxide dissolution in the electrolyte.
Sample 90H10G (Figure 4c-d) has an intense porous surface, but with less oxide dissolution compared to the 100H sample. Micro-arc oxidation or dielectric breakdown studies carried out in anodizing of magnesium, in an electrolyte that contained silicate with glycerin addition, showed that there was an improvement in the coating, with less pores and microcracks. Glycerin addition to the electrolyte increases the micro-arc oxidation stability; it reduces the spark size. Consequently, smaller and more homogeneous pores are obtained [18].
In sample 50H50G (Figure 4e-f), a smaller number of pores with less depth compared to the former ones are observed. In sample 10H90G (Figure 4g-h), there was less pore formation. These results prove that with an increase in the amount of glycerin in the electrolyte reduces oxide dissolution, making the surface homogeneous with a reduced number of pores. According to the study [19], glycerin addition increases the electrolyte viscosity, that reduces the oxide dissolution and, with that, pore formation.
Authors [17] have reported behaviors like those observed in this work. Another work [20] observed that the polarization curve pointed out that the dissolution is accompanied by hydrogen evolution; meanwhile, oxygen reduction can accompany passive film growing. The film seems to be finely porous and reveals cracks along the stainless steel grain boundaries, due to the improved dissolution in these areas. Film growth can be obtained by dissolving cycles of stainless steel, capable of activating or transpassivation, and subsequent deposition of the material on the film.
The proposed iron passivation mechanism [21-23] indicates that iron dissolves as ferrous ion to form ferric hydroxide with water; finally, film formation takes place by ferric hydroxide dehydration. These processes can be simplified according to equations [1-3], although it is known that the anions in solution, specially the oxygenated species, participate in the passivation reaction, where M represents the metal, M(OH)3 is the metallic hydroxide and M2O3(H2O)3, the passive film [22].
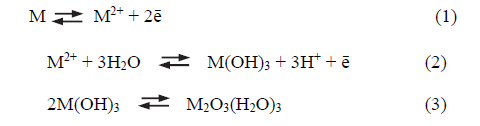
As stated above, glycerin addition to the electrolyte provoked a reduction in the number of pores on the samples surface. However, samples 90H10G and 90H10G showed the highest pore diameter (Table 4), promoted by the glycerin addition, compared to 100H. Sample 10H90G had the least number of pores as well as the smallest pore diameter, when compared to the other samples.
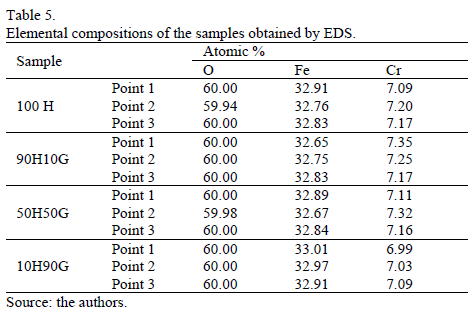
Figure 5 shows the EDS images and the point at which elemental determination was undertaken. Table 5 shows the EDS results for the anodized samples. It is observed that all the samples, the oxygen, iron and chromium remains unaltered with the anodizing electrolyte. So, it is possible to foresee the oxide formation according to the amount of oxygen.
It was expected that for sample 100H the amount of oxygen would be the smallest. But, namely, the presence of chromium in stainless steel promotes the formation of a thin oxide on the surface of the metal. That is why it is thought that the high amounts of oxygen present in the samples come from the alloy natural oxide, instead of evidencing the formation of a specific oxide after the anodizing.
3.4. XRD analysis
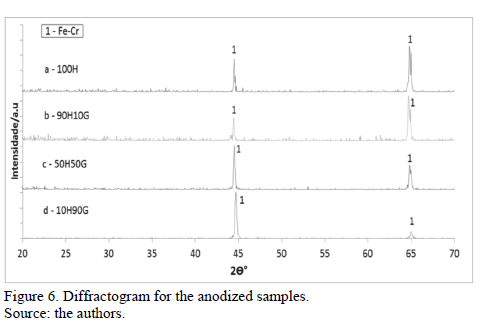
Figure 6 shows the X-ray diffractograms for the samples anodized in different electrolytes.
It is possible to observe that the systems 100H, 90H10G, 50H50G and 10H90G were amorphous. Meanwhile, anodized samples with different electrolyte composition did not affect the substrate chemical composition. Only iron and chrome were detected because they are substrate components.
3.5. AFM
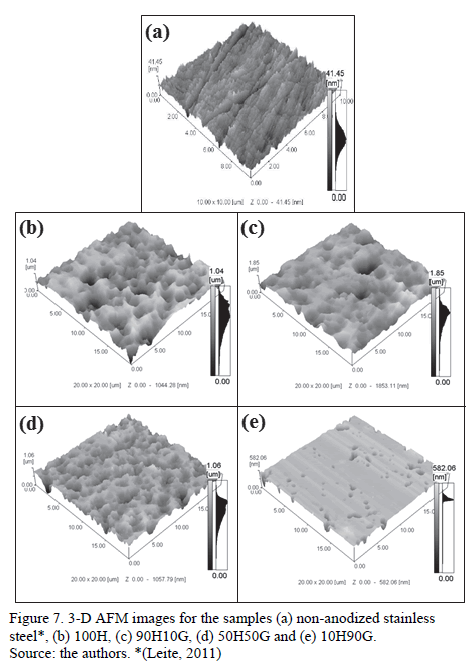
Figure 7 shows the 3-D atomic force microscopy images for the samples.
It can be observed that the samples had a rough surface, confirming what was observed in the SEM results.
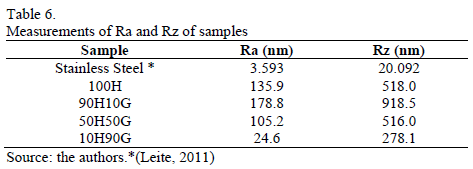
Table 6 shows the average roughness (Ra) and the depth roughness (Rz) of the samples. Anodized samples present higher Ra and Rz values than the original stainless steel. The increase in the glycerin concentration in the electrolyte (10H90G) diminished the Ra and Rz values, which indicates a reduction of the attack to the oxide. On the other hand, Rz value is too high for all the electrolytes, indicating the presence of deeper pores on the anodized samples when compared to the non-anodized steel.
3.6. Electrical conductivity
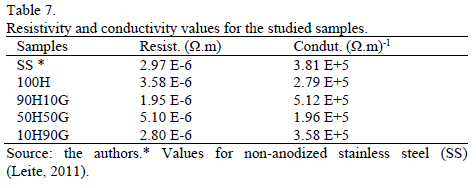
Table 7 shows the resistivity and conductivity values for the studied samples. It can be observed that all the conductivity values are found in an order of 105 (W m)-1. This is related to the decrease in the oxide dissolution process; however, it cannot be stated that there was the formation of an oxide layer, based only on these results.
4. Conclusions
The study conducted showed that the addition of glycerin to the 2M H2SO4 electrolyte diminished its electrical conductivity, which increases the potential transient during the anodizing process.
The decreased electrolyte conductivity promoted a decrease in the formed oxide dissolution, allowing its deposition on the metallic substrate, forming a porous oxide layer. Regarding the SEM images the formation of a porous oxide on the sample surface promoted by anodizing was verified.
The sample anodized in electrolyte with the highest glycerin concentration (10H90G) had the highest number of pores, but with a reduced diameter compared to the other samples.
In spite of the modification of the electrolyte composition with the addition of glycerin that made its conductivity vary, XRD evidenced that the chemical composition of the substrate remained the same.
Based on the aforementioned results, it can be concluded that ferritic stainless steel anodizing in 2M H2SO4 and glycerin promoted the formation of an oxide on the sample surface, but the latter one maintained its chemical structure. Due to the addition of glycerin, oxide dissolution in the electrolyte was minimized. This promotes the formation of pores on the oxide, which harms its protective performance.
Acknowledgments
The present work was carried out with the support of CAPES, A Brazilian Government entity focused on human resources formation. The authors also thank the financial support of Brazilian agencies CNPq and FAPERGS.
References
[1] Arevalo, J.L.M., Millan, A.R. y Barón, J.A., Oxidación en vapor de agua del acero inoxidable AISI 317 a 700 y 750 °C. DYNA, 2013. [Online]. 80 (179), pp. 151-156, 2013. Available at: http://www.redalyc.org/resumen.oa?id=49627363018. [ Links ]
[2] Callister Júnior, W.D.J., Fundamentos da ciência e da engenharia de materiais: uma abordagem integrada. 2. ed. Rio de Janeiro, RJ: LTC, 2006. [ Links ]
[3] Huntz, A.M., Reckmann, A., Haut, C., Severac. C., Herbst, M., Resende, F.C.T. and Sabioni. A.C.S., Oxidation of AISI 304 and 439 stainless steels. Material Science and Engineering: A, 447, pp. 266-276, 2007. http://dx.doi.org/10.1016/j.msea.2006.10.022 [ Links ]
[4] Giosa, J.A., Influência de diferentes meios oxidantes na repassivação de um aço inoxidável ferrítico Tipo AISI 430. Universidade de Minas Gerais. Ciência e Engenharia dos Materiais, 2003. [ Links ]
[5] Barbalat, M., Caron, D., Lanarde, L., Meyer, M., Fontaine, S., Castillon, F., Vittonato, J. and Refait, PH., Estimation of residual corrosion rates of steel under cathodic protection in soils via voltammetry. Corrosion Science, 73, pp. 222-229, 2013. http://dx.doi.org/10.1016/j.corsci.2013.03.038 [ Links ]
[6] Behzadnasab, M., Mirabedini, S.M. and Esfandeh, M., Corrosion protection of steel by epoxy nanocomposite coatings containing various combinations of clay and nanoparticulate zirconia. Corrosion Science, 75, pp. 134-141, 2013. http://dx.doi.org/10.1016/j.corsci.2013.05.024 [ Links ]
[7] Kreislova, K. and Geiplova, H., Evaluation of corrosion protection of steel bridges. Procedia Engineering, 40, pp. 229-234, 2010. http://dx.doi.org/10.1016/j.proeng.2012.07.085 [ Links ]
[8] Robert, S.A., Anodizing. Electrochemistry Encyclopedia. Northbrook, IL 60065-0622, USA 2002. [ Links ]
[9] Abal - Associação brasileira do alumínio. Guia técnico do alumínio, tratamento de superfície. 3. ed. São Paulo, 3, pp. 39-55, 1996. [ Links ]
[10] Parkhutik, V.P. and Shershulsky, V.I., Theoretical modeling of porous oxide growth on aluminium. Journal of Physics D: Applied Physics, 25, pp. 1258-1263, 1992. http://dx.doi.org/10.1088/0022-3727/25/8/017 [ Links ]
[11] Cohen, S.M., Replacements for chromium pretreatments as aluminum. Corrosion, 51, pp. 71-78, 1995. http://dx.doi.org/10.5006/1.3293580 [ Links ]
[12] Wang, J.H., Duh, J.G. and Shih, H.C., Corrosion characteristics of coloured films on stainless steel formed by chemical, INCO and AC processes. Surface and Coatings Technology, 78, pp. 248-254, 1996. http://dx.doi.org/10.1016/0257-8972(94)02414-6 [ Links ]
[13] Oliveira, C.T., Falcade, T., Menezes, T.L. e Malfatti, C.F., Anodização de aços inoxidáveis como tratamento superficial para aplicação em temperaturas elevadas, in 30º Congresso Brasileiro de Corrosão e 3rd International Corrosion Meeting, 2010. [ Links ]
[14] Evans, T.E., Hart, A.C. and Skedgell, A.N., The nature of the film on colored stainless steel. Transactions of the Institute of Metal Finishing, 5 (3), pp. 108-112, 1973. [ Links ]
[15] Lee, B.-G., Choi, J.-W., Lee, S.-E., Jeong, Y.-S., Oh, H.-J. and Chi, C.-S., Formation behavior of anodic TiO2 nanotubes in fluoride containing electrolytes, Trans. Nonferrous Met. Soc. China, 19 (4), pp. 842-845, 2009. http://dx.doi.org/10.1016/S1003-6326(08)60361-1 [ Links ]
[16] Muratore, F., Baron-Wiecheć, A., Gholinia, A., Hashimoto, T., Skeldon, P. and Thompson, G.E., Comparison of nanotube formation on zirconium in fluoride/glycerol electrolytes at different anodizing potentials, Electrochimica Acta, 58, pp. 389-398, 2011. http://dx.doi.org/10.1016/j.electacta.2011.09.062 [ Links ]
[17] Leite, P., Obtenção de estrutura porosa e condutora em aço inoxidável ferrítico. Trabalho de Conclusão de Curso (Monografia) - Curso de Engenharia Industrial Química, Universidade Feevale, Novo Hamburgo, Brasil, 2011. [ Links ]
[18] Wu, D., Liu, X., Lu, K., Zhang, Y. and Wang, H., Influence of C3H8O3 in the electrolyte on characteristics and corrosion resistance of the microarc oxidation coatings formed on AZ91D magnesium alloy surface. Applied Surface Science, 255, pp. 7115-7120, 2009. http://dx.doi.org/10.1016/j.apsusc.2009.02.087 [ Links ]
[19] Martin, F., Frari, D.D., Cousty, J. and Bataillon, C., Self-organisation of nanoscaled pores in anodic oxide overlayer on stainless steels. Electrochimica Acta, 54, pp. 3086-3091, 2009. http://dx.doi.org/10.1016/j.electacta.2008.11.062 [ Links ]
[20] Doff, J., Archibong, P.E., Jones, G., Koroleva, E.V., Skeldon, P. and Thompson, G.E. Formation and composition of nanoporous films on 316L stainless steel by pulsed polarization. Electrochimica Acta, 56, pp. 3225-3237, 2011. http://dx.doi.org/10.1016/j.electacta.2011.01.038 [ Links ]
[21] Ogura, K., Iron passivation in various buffer solutions. Journal of Electroanalytical Chemistry and Interfacial Electrochemistry, 79, pp. 149-157, 1977. http://dx.doi.org/10.1016/S0022-0728(77)80407-5 [ Links ]
[22] Ogura, K. and Majima, T., Formation and reduction of the passive film on iron in phosphate-borate buffer solution. Electrochimica Acta, 23, pp. 1361-1365, 1978. http://dx.doi.org/10.1016/0013-4686(78)80017-6 [ Links ]
[23] Ogura, K. and Sate, K., Passivity of metals. Eletrochemistry Communications, 443, 1978. [ Links ]
A. Bervian, possui curso técnico profissionalizante em Química pela Fundação Liberato Salzano Vieira da Cunha. Possui graduação em Engenharia Industrial Química pela Universidade Feevale, em 2012. Mestre em Engenharia pela Universidade Federal do Rio Grande do Sul, em 2014 (PPGEM - Capes 7). Doutorando acadêmico com dedicação exclusiva pela Universidade Federal do Rio Grande do Sul (PPGE3M - Capes 7) Principais linhas de pesquisa: Elaboração e caracterização de revestimentos metálicos protetores para substratos metálicos utilizados em alta temperatura (SOFC).
G.A. Ludwig, possui graduação em Engenharia Industrial Mecânica pela Universidade Feevale, em 2011. Mestre em Engenharia pela Universidade Federal do Rio Grande do Sul, em 2013 (PPGEM - Capes 7). Doutorando acadêmico com dedicação exclusiva pela Universidade Federal do Rio Grande do Sul (PPGE3M - Capes 7) Principais linhas de pesquisa: Elaboração e caracterização de revestimentos metálicos protetores para substratos metálicos utilizados em alta temperatura (SOFC).
S.R. Kunst, possui curso técnico profissionalizante em Química pela Fundação Liberato Salzano Vieira da Cunha. Possui bacharelado em Engenharia Industrial Química. Mestre em engenharia com dedicação exclusiva na Universidade Federal do Rio Grande do Sul (PPGEM - Capes 7). Doutoranda acadêmica com dedicação exclusiva na Universidade Federal do Rio Grande do Sul (PPGE3M - Capes 7), sendo as principais linhas de pesquisas: Elaboração e Caracterização de precursores silanos na proteção do aço galvanizado, flandres e alumínio e elaboração e caracterização de camada de difusão gasosa para células a combustíveis de hidrogênio.
L.V.R. Beltrami, graduada em Engenharia Química e Mestre pelo Programa de Pós-Graduação em Engenharia de Processos e Tecnologias (PGEPROTEC) da Universidade de Caxias do Sul (UCS), na área de processamento de polímeros e desenvolvimento de compósitos biodegradáveis. Atualmente é doutoranda pelo Programa de Pós-Graduação em Engenharia de Minas, Metalúrgica e de Materiais (PPGE3M) da Universidade Federal do Rio Grande do Sul (UFRGS), com orientação da Profª Drª Célia de Fraga Malfatti.
A.B.D. Moura, possui graduação em Engenharia Mecânica pela Universidade do Vale do Rio dos Sinos, em 1985, mestrado em Engenharia Mecânica pela Universidade Federal do Rio Grande do Sul, em 1996 e doutorado em Engenharia Mecânica pela Universidade Federal do Rio Grande do Sul, em 1999. Atualmente é professora titular e coordenadora do curso de Engenharia Mecânica e do curso de Industrial Mecânica, da Universidade Feevale. Professora e orientadora do Mestrado de Tecnologia dos Materiais e Processos Industriais. Parecerista da Revista Árvore. Tem experiência na área de Engenharia Mecânica (projeto, manutenção e operação). Pesquisadora na área de Fenômenos de Transportes, atuando principalmente nos seguintes temas: poluição atmosférica, modelo de dispersão, dispersão atmosférica, transferência de calor, fenômenos de transporte, carvão vegetal e gerenciamento ambiental.
C. de F. Malfatti, possui graduação em Engenharia Metalúrgica pela Universidade Federal do Rio Grande do Sul com mestrado e doutorado em Engenharia - área de concentração Ciência e Tecnologia dos Materiais, pela Universidade Federal do Rio Grande do Sul e pela Université Paul Sabatier. Atualmente é professora e pesquisadora na Universidade Federal do Rio Grande do Sul. Tem experiência na área de Engenharia de Materiais e Metalúrgica, com ênfase em eletroquímica, revestimentos inorgânicos e revestimentos compósitos, revestimentos metálicos e corrosão. Atua principalmente no desenvolvimento relacionado aos seguintes temas: nanotecnologia aplicada ao tratamento de superfícies metálicas, tratamento de superfície metálicas para aplicações na área de biomateriais, tecnologias e materiais para conversão e estocagem de energia, revestimentos protetores e caracterização eletroquímica.
C.T. Oliveira, professora e pesquisadora, doutora em engenharia na área de ciênica dos materiais e engenheira metalúrgica, possui experiência na área de tratamento de superfície, principalmente em revestimentos protetores, porosos e não-porosos, para finalidade de proteção contra corrosão, aderência de tintas, uso como dielétricos em capacitores eletrolíticos, e obtenção de nano-óxidos para aplicação diversificada.