Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
DYNA
Print version ISSN 0012-7353
Dyna rev.fac.nac.minas vol.82 no.191 Medellín May/June 2015
https://doi.org/10.15446/dyna.v82n191.45296
DOI: http://dx.doi.org/10.15446/dyna.v82n191.45296
Effect of cellulose nanofibers concentration on mechanical, optical, and barrier properties of gelatin-based edible films
Efecto de la concentración de nanofibras de celulosa sobre las propiedades mecánicas, ópticas y de barrera en películas comestibles de gelatina
Ricardo David Andrade-Pizarro a, Olivier Skurtys b & Fernando Osorio-Lira c
a Facultad de Ingeniería, Universidad de Córdoba, Montería, Colombia. rdandrade@correo.unicordoba.edu.co
b Universidad Técnica Federico Santa María, Valparaíso, Chile. olivier.skurtys@usm.cl
c Universidad de Santiago de Chile, Santaigo, Chile. fernando.osorio@usach.cl
Received: Augusto 30th, de 2014. Received in revised form: November 4th, 2014. Accepted: November 14th, 2014
This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.

Abstract
The effect of gelatin, glycerol, and cellulose nanofiber (CNFs) concentrations on the mechanical properties, water vapor permeability, and color parameters of films was evaluated. The results indicate that the color is only affected by the gelatin concentration. Mechanical tests indicated that with increasing concentration of gelatin and CNFs, there is an increase in tensile strength, whereas an increase in glycerol concentration causes an increase in elongation, making the films more flexible. An increased concentration of gelatin and glycerol makes the film more permeable to water vapor, while an increase in the concentration of CNFs reduces this property. Finally, the addition of CNFs to gelatin-based films improves their mechanical and barrier properties (water vapor) without affecting the appearance (color) of the films.
Keywords: barrier properties; tensile strength; elongation; color.
Resumen
Se evaluó el efecto de la concentración de gelatina, glicerol y nanofibras de celulosa (NFC) sobre las propiedades mecánicas, permeabilidad al vapor de agua, y los parámetros de color de películas a base de gelatina. Los resultados indican que el color es influenciado sólo por la concentración de gelatina. Las pruebas mecánicas indican que al aumentar la concentración de gelatina y NFC hay un aumento en la resistencia a la tracción, mientras que un aumento en la concentración de glicerol provoca un aumento en el porcentaje de elongación, haciendo que las películas sean más flexibles. Un aumento en la concentración de gelatina y glicerol aumenta la permeabilidad al vapor de agua, mientras que un aumento en la concentración de NFC reduce esta propiedad. Finalmente, la adición de NFC en películas a base de gelatina mejora sus propiedades mecánicas y de barrera (vapor de agua) sin afectar a la apariencia (color) de las películas
Palabras clave: propiedades de barrera; resistencia a la tracción; elongación, color.
1. Introduction
Edible films have been considered for food preservation given their ability to improve food quality. Current interest in edible films is due to the need to develop packaging that is readily degradable and non-aggressive to the environment, as well as the need to improve the availability of new markets for the materials used in the fabrication of these films [1,2]. Generally, edible films are applied in combination with other technologies (cooling, controlled or modified atmosphere, heat treatment, etc.) in order to improve their quality, safety or to increase their shelf life, since they can act as a barrier, improve mechanical properties, avoid damaging different parts of a food, and even serve as carriers of additives and active components [3,4].
In the preparation of edible films a variety of biopolymers are used, such as polysaccharides, proteins, and lipids, alone or in combination, to enhance their individual properties. Films based on proteins are often brittle and inflexible and thus require the addition of plasticizers such as glycerol [5], which modify the organization of the polymeric three-dimensional protein network, reducing intermolecular attractive forces and increasing the volume, favoring free chain mobility [6]. Moreover, the form of protein is of great importance for the formation of the networks that make up the matrix. The high molecular weight proteins and fibrils, such as gelatin, may form larger networks that improve mechanical properties [5]. However, given their hydrophilic nature, gelatin films have a poor water vapor barrier. Different alternatives have been tested to improve this water vapor barrier property of gelatin-based films, including the addition of hydrophobic compounds such as lipids [7,8], modifying the polymer network via cross-linking protein chains [9,10] and addition of nanocomposites, including cellulose nanofibers [11].
Cellulosic nanofibers (CNF) have been gaining considerable interest as reinforcement as they are more effective than their microsized counterparts in reinforcing polymers as they form a percolated network connected by hydrogen bonds, provided there is a good dispersion of the nanofibers in the matrix [12,13]. Many researchers reported that the addition the CNF improved water vapor barrier properties of biopolymer films, such as chitosan films [14,15], Pullulan films [16], and kappa-carrageenan films [13].
To evaluate the efficacy and quality of edible coatings, different parameters of the coated food in storage can be determined (loss of water, respiration rate, texture, color, pH, etc). Measurements can also be taken directly from the film, including mechanical, thermal, optical, and barrier properties [17,18]. Barrier properties (H2O, O2, and CO2) can greatly influence the stability of foods sensitive to the oxidation of lipids, vitamins, and pigments or substantial loss of water. However, an edible coating with very good barrier properties can become ineffective if its mechanical properties do not allow the integrity of the coating to be maintained during handling, packaging, and transport. The coatings must be resistant to breakage and abrasion in order to reinforce the structure of the food and facilitate handling and/or be sufficiently flexible to accommodate any deformation of the product without tearing. Another important aspect to consider is consumer acceptance, as coating materials can produce sensory changes in the product.
The purpose of this work was to investigate the influence of the concentration of gelatin, glycerol, and cellulose nanofibers on the mechanical, optical, and water vapor permeability of edible films.
2. Materials and methods
2.1. Materials
Type B gelatin from bovine skin (180 Bloom) was purchased from Rousselot (Brazil), and glycerol was purchased from Sigma (Sigma-Aldrich, Chile). CNFs were provided by the New Materials Research Group (Pontificia Bolivariana University, Colombia), and these were obtained from agro-industrial waste (banana peel) as reported by Zuluaga et al. [19].
2.2. Preparation of film-forming solution (FFS)
FFS were prepared with distilled water. Gelatin was hydrated at room temperature (20 ± 2 ºC) for 30 min and then heated at 50 ºC for 30 min with continuous stirring until it was completely dissolved. Glycerol and cellulose nanofibers were added at different concentrations (based on dry gelatin weight). CNFs were dispersed uniformly with the aid of an ultrasound equipment operating at 40 kHz (Branson Model 2210, USA) for 30 min. FFSs were prepared at concentrations of 0.8 and 2.2%w/v, glycerol concentration varied between 10 and 30% w/w based on gelatin, and CNF concentration varied between 1 and 5% w/w based on gelatin.
2.3. Preparation of films
Gelatin-based edible films were obtained by the casting technique, which consists in dehydrating an FFS, which is conveniently applied on a support. 40 mL of the FFS were poured into teflon plates with a diameter of 15.5 cm. The plates were maintained at 22 ºC in a laboratory oven (LDO-150F model, LabTech, Korea) for 24 h. After drying, the films were peeled off from the plate surface.
2.4. Edible film thickness
The film thickness was measured with a digital micrometer (Mitutoyo Co., Japan) with a sensitivity of 1 mm. The thickness was expressed as the average of 10 random measurements of the films cut and adapted for mechanical and water vapor permeability tests.
2.5. Color parameter of edible films
The color of the films was determined using a Miniscan XE Plus colorimeter (HunterLab, USA), and the D65 (daylight) CIELab scale was used to determine the parameters L*, a*, and b*, where L* indicates the degree of brightness from 0 for black to 100 for white, a* indicates the position between red (+a*) and green (-a*), and b* indicates the position between yellow (+b*) and blue (-b*). The color of the film was expressed as the color difference (dE*) according to eq. (1).
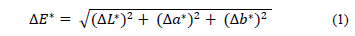
where dL*, da*, and db* are the differences between the sample color parameter and the white board color standard (L* = 94.8, a* = -0.78, b* = 1.43), which is used as the background for determining the color film [20].
2.6. Mechanical properties of edible films
The mechanical properties of edible films were determined by stress and puncture tests using a universal testing machine (Zwick/Roell, Germany). The tensile strength (TS) and elongation (E%) of the films were determined according to the ASTM D882-95 method. Film specimen strips (100 mm × 25 mm) were cut and conditioned in a desiccator containing saturated potassium carbonate (purity ≥ 99%, Sigma-Aldrich) solution with 50% (RH) at 22 ºC for 4 days prior to testing. The initial distance of separation between the tensile grips and velocity were adjusted to 50 mm and 1 mm/s, respectively. TS was calculated by dividing the maximum load at break by the initial specimen's cross-sectional area (thickness for width (25 mm)). E (%) was calculated by dividing the extension at breaking of the specimen by the initial gage length of the specimen (50 mm) and expressed as a percentage (%). Each test trial per film consisted of seven replicate measurements.
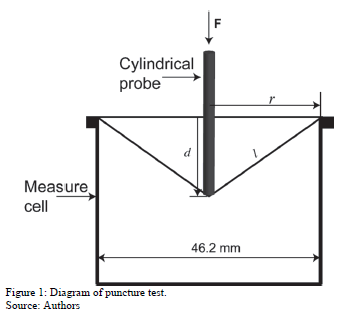
The puncture test was run according to Gontard et al. [21]. Each film was mounted on a 46.2-mm-diameter puncture cell and perforated by a smooth-edged cylindrical probe (5 mm in diameter) moving at 1 mm/s (see Fig. 1).
The puncture strength (PS) and the percentage deformation (Ep) were calculated by eq. (2) and (3).
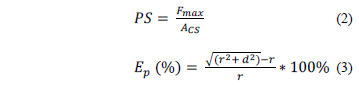
where Fmax is the maximum applied force (N), ACS is the cross-section of the film situated in the cell (ACS = 2rd), r is the initial radius of the film, d is the film thickness, and d is the movement of the probe from the point of contact with the film to the breaking point [22].
2.7. Water vapor permeability of films
The water vapor transmission rate (WVTR) and water vapor permeability (WVP) were determined gravimetrically at 22 ºC, according to the method proposed by Gontard et al. [23]. Four edible films without visual defects were selected (no bubbles or fractures), and these were cut with a diameter of 21.6 mm and placed on a glass cell containing distilled water. A high-vacuum silicone grease (Merck, Germany) was used to seal the film with the cell. The cells were placed in a desiccator containing silica gel (~0% RH) and maintained at 22 ºC in a laboratory oven (Labtech LDO-150F, Korea). The cells were weighed on an analytical balance (Precisa ES - DR 225SM, Germany) every 2 hours during the firsts 8 hours and then at 24 hours.
The water vapor transmission rate was determined according to Eq. (4).
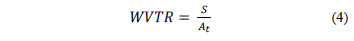
where S is the slope of the mass loss of the cells over time and At is the area (m2) of the water vapor transfer rate.
Existence of a stagnant air layer inside and above the cup generates significant resistance to water vapor transport; therefore, it was necessary to correct the WVTR value according to the methodology proposed by Gennadios et al. [5].
2.8. Statistical design and analysis
The Box-Behnken statistical screening design was used to statistically develop the model and to study and evaluate the main effects, interaction effects, and quadratic effects of the independent variables (gelatin, glycerol, and CNF concentrations) on the mechanical, optical, and water vapor barrier properties of gelatin-based edible films. The response surface methodology was applied to analyze the effect of independent variables on response variables. A second-order polynomial model (eq. 5) was used to predict the experimental behavior.
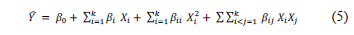
where Y is the predicted value of the response; b0, bi, bii, and bij are the regression coefficients for interception, linear, quadratic, and interaction effects, respectively; k is the number of independent parameters (k = 3 in this study); and Xi and Xj are the coded levels of the experimental conditions. Analysis of variance (ANOVA) was applied to determine significant effects of gelatin, glycerol, and CNF concentrations on the properties of gelatin-based edible films. The quality of the developed model was determined by the coefficients of determination (R2) and root mean square error (RMSE). This study design was analyzed and three-dimensional response surface plots were drawn using JMP 9.0.1 software (SAS Institute).
3. Results and discussion
3.1. Color of edible films
Color evaluation is an important quality parameter for potential industrial applications of edible films, because consumers often associate aspects such as brightness and color of food-which can be affected by the coatings and edible films-with food quality. All edible formulations were assessed visually and the films obtained were transparent, with the absence of insoluble particles and generally good appearance.
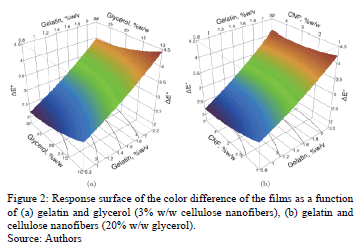
The ANOVA shows that gelatin concentration (p = 0.004) significantly affects the color difference (dE*) of edible films, with a significance level at 95%. Fig. 2 shows the response surface of the behavior of the color difference of edible films in terms of concentrations of gelatin-glycerol (Fig. 2a) and gelatin-CNF (Fig. 2b). We can see that in the presence of glycerol and CNFs, dE* increases linearly with the concentration of gelatin, irrespective of the other constituents. This increase was approximately 71.6% when the concentration of gelatin rose by 0.8% w/v (dE* = 2.23) to 2.2% w/v (dE* = 3.83). This increase is due to the greater amount of solids contained in the film, which in turn increases the concentration of gelatin. Vanin et al. [24] reported that the concentration of gelatin-based films (2% w/v) and type of plasticizer (glycerol, diethylene glycol and propylene glycol) do not affect dE*.
The behavior of the color difference (dE*) of edible films may be represented as a function of the concentrations of gelatin (G), glycerol (g), and CNFs (C) according to eq. (6). This equation has values of R2 and RMSE of 0.94 and 0.302, respectively. This indicates the suitability of the proposed model to represent the experimental data.
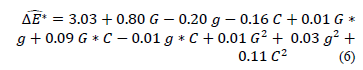
Considering only factors that significantly influence the color difference of edible films, eq. 6 can be expressed as eq. (7).
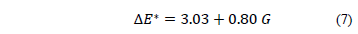
3.2. Mechanical properties of edible films
3.2.1. Stress test of edible films
• Tensile strength (TS) of edible films
The ANOVA showed that the linear effects of gelatin (p = 0.0017), NFC concentration (p = 0.0074) and the quadratic effect of glycerol concentration (p = 0.0312) significantly affect the tensile strength (TS) of edible films, with a significance level of 95%. The values for the fracture of edible films agree with those reported by Chambi and Grosso [25] for gelatin-based films (TS ≈ 60 MPa). Gelatin, however, has a loosely organized structure that may be renatured during the film formation process [26], as it is able to reacquire part of the triple helix structure of collagen. According to Siew et al. [27], it increases the chain organization optimized molecular packing, resulting in an increase in mechanical properties.
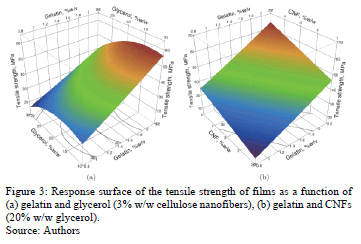
Fig. 3 shows the response surface of the tensile strength of the edible films. An increased gelatin concentration (see Fig. 3a) produces an increase in the value of the tensile strength of the edible films. High gelatin concentrations can cause a greater number of physical connections during the formation of the network, so that greater force is required to rupture the films [28]. Moreover, the addition of CNFs increases the tensile strength (Fig. 3b), which has been observed in different polymeric matrices: chitosan [12] and starch [29]. George and Siddaramaiah [30] reported that for gelatin films (10%) the addition of 4% bacterial cellulose nanocrystals produces an increase of 30% in the tensile strength. The increased tensile strength due to the addition of CNFs suggests a uniform dispersion of the fibers within the gelatin matrix, and good CNF-gelatin adhesion interactions, as suggested by Gardner et al. [31] and Xu et al. [32] for other matrices. The addition of glycerol had a quadratic effect on TS. This was an unexpected result given that although it is a well known fact that increasing glycerol concentration decreases fracture stress, Sobral et al. [33] observed a decrease, shaped like a parabolic segment, in the TS of films made of meat myofibrillar proteins (between 25 and 100g glycerin/100g protein), acidified with acetic acid or lactic acid.
Remarkably, of the three components tested, the concentration of gelatin had the most influence on TS, showing an increase of 124% when the gelatin concentration increased from 0.8% w/v (TS = 23.50 MPa) to 2.2% w/v (TS = 52.72 MPa), while an increase in the concentration of CNFs from 1% w/w (TS = 29.05 MPa) to 5% w/w (TF = 47.17 MPa) causes an increase of 62.4% in the TS.
Eq. (8) shows the relationship representing the TS based on the concentration of gelatin (G), glycerol (g), and CNFs (C). In this equation, all factors are encoded independently if they are significant.
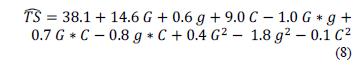
The values of R2 (0.96) and RMSE (4.54) indicate the goodness of the quadratic polynomial in the representation of the experimental data.
• Elongation of edible films
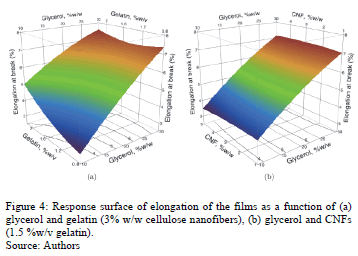
ANOVA shows that the elongation (%) of the films is significantly influenced (95%) by the glycerol concentration (p = 0.0123). Fig. 4 shows the response surface of elongation of edible films in function of the concentrations of glycerol-gelatin (Fig. 4a) and glycerol-CNF (Fig. 4b). An increase in glycerol concentration from 10% w/w (E = 3.99%) to 30% w/w (E = 6.91%) causes an increase of 73.2% in the percentage elongation. This effect of glycerol on elongation has been widely reported by several authors [22,24,34]. The addition of plasticizer in the preparation of edible films reduces the interactions between the chains of biopolymers [28]. According to Jongjareonrak et al. [35] and Ayala et al. [36], the glycerol molecule is a small chain and is hygroscopic; thus, it is easily inserted between the protein chains, attracting more water into the structure of the film and making it more flexible. Remarkably, Carvalho [37] reported that gelatin concentration does not affect the elongation of films made of gelatin and sorbitol. Besides, in contrast to this study, Azeredo et al. [12] observed that CNF significantly affect the elongation of films. This is possibly explained by a better adhesion of CNF to the matrix used (gelatin) in this study.
The elongation percentage (E, %) of the edible films can be represented according to eq. (9) with the factors encoded: gelatin concentration (G), glycerol (g), and CNFs (C). The accuracy of the model (eq. 9) in the representation of the experimental data is shown by the obtained values of R2 = 0.89 and RMSE = 0.84.
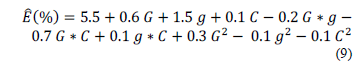
3.2.2. Puncture test
• Puncture resistance test
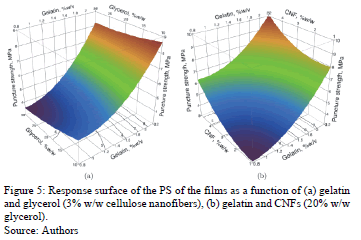
The ANOVA shows that the puncture strength (PS) is influenced significantly by the gelatin concentration (p = 0.0498) and CNFs (p = 0.0189). Fig. 5 shows the response surface of the behavior of the PS of edible films in terms of concentrations of gelatin-glycerol (Fig. 5a) and gelatin-CNF (Fig. 5b). An increase in the gelatin concentration from 0.8 to 2.2% w/v causes an increase in PS of 90.2%, whereas when the concentration of CNFs increases from 1 to 5%, the PS value increases by 121.1%. The contribution of these compounds to the mechanical properties was described in Section 3.2.1.
• Puncture deformation
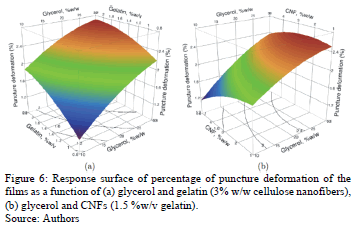
The ANOVA shows that the puncture deformation was significantly affected only by the linear factor: glycerol concentration (p = 0.0305). In Figs. 6a and 6b, we can see that increasing the glycerol concentration increases the percentage of puncture deformation by approximately 40%.
The addition of plasticizers (glycerol) reduces the intermolecular forces between the protein molecules, thereby increasing the flexibility and extensibility of the films [38,39].
These data corroborate those obtained in the stress test. According to Vanin et al. [24], in gelatin matrix, glycerol has a greater effect on the puncture deformation than ethylene glycol and propylene glycol.
The puncture deformation percentage (Ep, %) of the edible films can be represented according to Eq. (10) with the factors encoded: gelatin concentration (G), glycerol (g), and CNFs (C). The accuracy of the model (eq. 10) in the representation of the experimental data is shown by the obtained values of R2 = 0.86 and RMSE = 0.32.
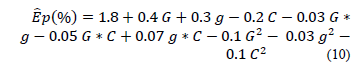
3.3. Water vapor permeability of edible films
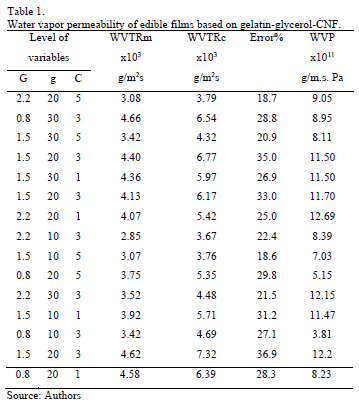
Table 1 shows the water vapor transfer rate (WVTR) and water vapor permeability (WVP) of edible films of gelatin, glycerol, and CNFs. The measured values of water vapor transfer rate (WVTRm) underestimate the corrected values of water vapor transfer rate (WVTRc), introducing errors of 18 to 37%.
The ANOVA shows that with a confidence level of 95%, the water vapor permeability of edible films is influenced significantly by the linear effects of gelatin (p = 0.0038), glycerol (p = 0.0211), and CNFs (p = 0.0214) and the quadratic effect of gelatin concentration (p = 0.0240). Fig. 7 shows the response surface of the behavior of water vapor permeability of edible films in terms of concentrations of gelatin-glycerol (Fig. 7a) and gelatin-CNF (Fig. 7b).
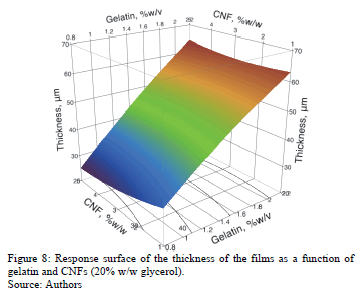
An increase in the gelatin concentration from 0.8 to 2.2% w/v causes an increase in WVP of 150%, which may be caused by swelling of the film due to water, which creates various structures in the film. In addition, increasing the concentration of gelatin produces a linear increase of the thickness (see Fig. 8), which agrees with that reported by Carvalho [37]. Benbettaïeb et al. [40] observed that when the thickness increased from 52 to 159 mm, WVP linearly increased. In ideal polymeric structures, the water vapor permeability is independent of the thickness of the film [38], and does not particularly apply to hydrophilic films. An increase in the film thickness provides a greater resistance to mass transfer through it and consequently the partial pressure of water vapor in equilibrium at the surface of the inner film increases. Significantly, the permeability to water vapor in gelatin-based films is not an inherent property of the films, because the rate of water vapor transmission through hydrophilic films varies nonlinearly with the gradient of partial pressure of water vapor [41].
Moreover, increasing the concentration of glycerol (10 to 30% w/w) increases the WVP by 31%. Several authors have observed this behavior [24,38,39,42]. The addition of glycerol results in a reorganization of the network formed by the protein, reducing the intermolecular attractive forces and increasing the free volume and chain mobility; thus, increasing the diffusion coefficient of water [43,44]. On this point, Nemen [45] reported that an increase in the content of glycerol (hydrophilic component) increases the amount of polar groups present in the gelatin film.
Finally, of the three components of the formulations, only the CNF concentration decreased the WVP values. The addition of 1 to 5% w/w of CNF causes a 32% decrease in WVP. Several authors have reported that the addition of CNFs decreases the permeability of films prepared from alginate [14], chitosan [12], and gelatin [30]. This could be due to interaction between CNFs and hydrophilic sites of the gelatin and the fact that CNFs have low hygroscopicity. According to Lagaron et al. [46], the presence of crystalline fibers increases tortuosity, which causes slower diffusion processes and hence lower permeability.
Eq. (11) shows the relationship representing the water vapor permeability according to the concentration of gelatin (G), glycerol (g), and CNFs (C). In this equation, all factors are encoded independently if they are significant. The values of R2 (0.92) and RMSE (1.27) indicate the goodness of the quadratic polynomial in the representation of the experimental data.
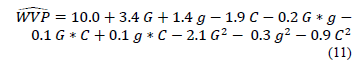
WVP (x 1011) is the water vapor permeability at g/m s Pa.
4. Conclusion
Films made of gelatin, glycerol, and cellulose nanofibers (CNFs) have a good visual appearance, confirming the good dispersion of CNFs in the gelatin matrix. Gelatin concentration increases the color of the films linearly.
Determination of the mechanical properties (stress and puncture tests) of edible films shows that the concentration of gelatin and CNFs increases the fracture stress and puncture resistance, suggesting a uniform dispersion in the matrix and CNF and good CNF-gelatin adhesion interactions. Moreover, an increase in glycerol concentration increases the flexibility and elongation of gelatin-based films.
Water vapor permeability is also influenced by the concentration of gelatin, glycerol, and CNF. An increase in the concentration of gelatin and glycerol increases the WVP by 150% and 31%, respectively. However, the addition of CNF in the gelatin matrix causes a reduction of WVP of 31%, because the nanofibers provide a more tortuous path to water vapor passing through the film.
The addition of CNF to the gelatin matrix can improve the mechanical properties (62.4%) and the water vapor permeability (32%) without affecting the color of the films, which are desirable characteristics in most applications of edible films and coatings.
Acknowledgement
The authors would like to thank Fondecyt (project 1130587). O.S is grateful to the Fondecyt (project 1120661) for financial support.
References
[1] Chillo, S., Flores, S., Mastromatteo, M., Conte, A. and Gerschenson, L., Del Nobile, M.A. Influence of glycerol and chitosan on tapioca starch-based edible film properties. J Food Eng, 88 (2), pp. 159-168, 2008. DOI: 10.1016/j.jfoodeng.2008.02.002 [ Links ]
[2] Koelsch, C., Edible water vapor barriers: properties and promise. Trends Food Sci Tech, 5(3), pp. 76-81, 1994. DOI: 10.1016/0924-2244(94)90241-0 [ Links ]
[3] Debeaufort, F., Quezada-Gallo, J. and Voilley, A., Edible films and coatings: Tomorrow's packagings. A review. Crit Rev Food Sci Nutr, 38 (4), pp. 299- 313, 1998. DOI: 10.1080/10408699891274219 [ Links ]
[4] Pavlath, A. and Orts, W., Edible films and coatings: Why, what, and how?. In Embuscado, M. and Huber, K, Eds, Edible films and coatings for food applications, Springer, 2th edition, pp. 1- 24, 2009. [ Links ]
[5] Gennadios, A., McHugh, T., Weller, C. and Krochta, J.M., Edible coatings and films based on proteins. In Krochta, J., Baldwin, E. and Nisperos-Carriedo, M., Eds., Edible coatings and films to improve food quality, Lancaster, PA: Technomic Publishing Company, pp. 201-277, 1994. [ Links ]
[6] Chang, C. and Nickerson, M.T., Effect of plasticizer-type and genipin on the mechanical, optical, and water vapor barrier properties of canola protein isolate-based edible films. Eur Food Res Technol, 238 (1), pp. 35-46, 2014. DOI: 10.1007/s00217-013-2075-x [ Links ]
[7] Ma, W., Tang, C., Yin, S., Yang, X., Wang, Q., Liu, F. and Wei. Z., Characterization of gelatin-based edible films incorporated with olive oil. Food Res Int, 49 (1), pp. 572-579, 2012. DOI: 10.1016/j.foodres.2012.07.037 [ Links ]
[8] McHugh, T. and Senesi, E., Apple wraps: A novel method to improve the quality and extend the shelf life of fresh-cut apples. J. Food Sci., 65 (3), pp. 480-485, 2000. DOI: 10.1111/j.1365-2621.2000.tb16032.x [ Links ]
[9] Chambi, H. and Grosso, C., Edible films produced with gelatin and casein cross-linked with transglutaminase. Food Res Int, 39 (4), pp. 458-466, 2006. DOI: 10.1016/j.foodres.2005.09.009 [ Links ]
[10] Mu, C., Guo, J., Li, X., Lin, W. and Li D., Preparation and properties of dialdehyde carboxymethyl cellulose crosslinked gelatin edible films. Food Hydrocolloid, 27 (1), pp. 22-29, 2012. DOI: 10.1016/j.foodhyd.2011.09.005 [ Links ]
[11] Chang, S, Chen, L., Lin, S. and Chen, H. Nano-biomaterials application: Morphology and physical properties of bacterial cellulose/gelatin composites via crosslinking. Food Hydrocolloid, 27 (1), pp. 137-144, 2012. DOI: 10.1016/j.foodhyd.2011.08.004 [ Links ]
[12] Azeredo, H., Mattoso, L., Avena-Bustillos, R, Filho, G., Munford, M., Wood, D. and McHugh, T., Nanocellulose reinforced chitosan composite films as affected by nanofiller loading and plasticizer content. J Food Sci, 75 (1), pp. N1-N7, 2010. DOI: 10.1111/j.1750-3841.2009.01386.x. [ Links ]
[13] Savadekar, N.R., Karande, V.S., Vigneshwaran, N., Bharimalla, A.K. and Mhaske, S.T., Preparation of nano cellulose fibers and its application in kappa-carrageenan based film. Int. J. Biol. Macromol, 51 (5), pp. 1008-1013, 2012. DOI: 10.1016/j.ijbiomac.2012.08.014 [ Links ]
[14] Azeredo, H., Miranda, K., Ribeiro, H., Rosa, M. and Nascimento, D., Nanoreinforced alginate-acerola puree coatings on acerola fruits. J Food Eng, 113 (4), pp. 505-510, 2012. DOI: 10.1016/j.jfoodeng.2012.08.006 [ Links ]
[15] Wu, T., Farnood, R., O'Kelly, K. and Chen, B., Mechanical behavior of transparent nanofibrillar cellulose-chitosan nanocomposite films in dry and wet conditions. J Mech Behav Biomed, 32, pp. 279-286, 2012. DOI: 10.1016/j.jmbbm.2014.01.014 [ Links ]
[16] Trovatti, E., Fernandes, S.C.M., Rubatat, L., Perez, D., Freire, C.S.R., Silvestre, A.J.D. and Neto, C.P., Pullulan-nanofibrillated cellulose composite films with improved thermal and mechanical properties. Compos Sci Technol, 72 (13), pp. 1556-1561, 2012. DOI: 10.1016/j.compscitech.2012.06.003 [ Links ]
[17] Guilbert, S. and Gontard, N., Edible and biodegradable food packaging. In: Ackermann, J. and Ohlsson, T., eds., Foods and Packaging Materials-Chemical Interactions, England: The Royal Society of Chemistry, pp. 159-168, 1995. [ Links ]
[18] Wan, Y., Luo, H., He, F., Liang, H., Huang, Y. and Li, X., Mechanical, moisture absorption, and biodegradation behaviours of bacterial cellulose fibre-reinforced starch biocomposites. Compos Sci Technol, 69 (7-8), pp. 1212-1217, 2009. DOI: 10.1016/j.compscitech.2009.02.024 [ Links ]
[19] Zuluaga, R., Putaux, J., Cruz, J., Vélez, J., Mondragon, I. and Gañán, P., Cellulose microfibrils from banana rachis: Effect of alkaline treatments on structural and morphological features. Carbohyd Polym, 76(1), pp. 51-59, 2009. DOI: 10.1016/j.carbpol.2008.09.024 [ Links ]
[20] Sobral, P., Santos, J. and García, F., Effect of protein and plasticizer concentrations in film forming solutions on physical properties of edible films based on muscle proteins of a Thai Tilapia. J Food Eng, 70 (1), pp. 93-100, 2005. DOI: 10.1016/j.jfoodeng.2004.09.015 [ Links ]
[21] Gontard, N., Guilbert, S. and Cuq, J., Edible wheat gluten films: Influence of the main process variables on film properties using response surface methodology. J. Food Sci., 57 (1), pp. 190-195, 1992. DOI: 10.1111/j.1365-2621.1992.tb05453.x [ Links ]
[22] Sobral, P., Menegalli, F., Hubinger, M. and Roques, M., Mechanical, water vapor barrier and thermal properties of gelatin based edible films. Food Hydrocolloid, 15 (4-6), pp. 423-432, 2001. DOI: 10.1016/S0268-005X(01)00061-3 [ Links ]
[23] Gontard, N., Guilbert, S. and Cuq, J., Water and glycerol as plasticizers affect mechanical and water vapor barrier properties of an edible wheat gluten film. J. Food Sci., 58 (1), pp. 206-211, 1993. DOI: 10.1111/j.1365-2621.1993.tb03246.x [ Links ]
[24] Vanin, F., Sobral, P., Menegalli, F., Carvalho, R. and Habitante, A., Effects of plasticizers and their concentrations on thermal and functional properties of gelatin-based films. Food Hydrocolloid, 19 (5), pp. 899-907, 2005. DOI: 10.1016/j.foodhyd.2004.12.003 [ Links ]
[25] Chambi, H. and Grosso, C., Mechanical and water vapor permeability properties of biodegradables films based on methylcellulose, glucomannan, pectin and gelatin. Food Science and Technology (Campinas), 31 (3), pp. 739-746, 2011. DOI: 10.1590/S0101-20612011000300029 [ Links ]
[26] Achet, D. and He, X., Determination of the renaturation level in gelatin films. Polymer, 36 (4), pp. 787-791, 1995. DOI: 10.1016/0032-3861(95)93109-Y [ Links ]
[27] Siew, D., Heilmann, C., Easteal, A. and Cooney, R., Solution and film properties of sodium caseinate/glycerol and sodium caseinate/polyethylene glycol edible coating systems. J Agr Food Chem, 47 (8), pp. 3432-3440, 1999. DOI: 10.1021/jf9806311 [ Links ]
[28] Arvanitoyannis, I., Formation and properties of collagen and gelatin films and coatings. In Gennadios, A., Ed., Protein-based Films and Coatings, Boca Raton: CRC Press, pp. 730-739, 2002. [ Links ]
[29] Wan, Y.Z., Luo, H., He, F., Liang, H., Huang, Y. and Li, X.L., Mechanical, moisture absorption, and biodegradation behaviours of bacterial cellulose fibre-reinforced starch biocomposites. Composites Science and Technology, 69 (7- 8), pp. 1212-1217, 2009. DOI: 10.1016/j.compscitech.2009.02.024 [ Links ]
[30] George, J., Siddaramaiah., High performance edible nanocomposite films containing bacterial cellulose nanocrystals. Carbohydr Polym, 87 (3), pp. 2031- 2037, 2012. DOI: 10.1016/j.carbpol.2011.10.019 [ Links ]
[31] Gardner, D., Oporto, G., Mills, R. and Samir, M., Adhesion and surface issues in cellulose and nanocellulose. J Adhes Sci Technol, 22 (5-6), pp. 545-567, 2008. DOI: 10.1163/156856108X295509 [ Links ]
[32] Xu, Y., Ren, X. and Hanna, M., Chitosan/clay nanocomposite film preparation and characterization. J Appl Polym Sci, 99 (4), pp. 1684-1691, 2006. DOI: 10.1002/app.22664 [ Links ]
[33] Sobral, P., Ocuno, D. and Savastano, H., Preparo de proteínas miofibrilares de carne e elaboracao de biofilmes com dois tipos de acidos: propriedades mecanicas. Brazilian Journal of Food Technology, 1, 44-52, 1998. [ Links ]
[34] Maria, T., de Carvalho, R., Sobral, P., Habitante, A. and Solorza-Feria, J., The effect of the degree of hydrolysis of the PVA and the plasticizer concentration on the color, opacity, and thermal and mechanical properties of films based on PVA and gelatin blends. J Food Eng, 87 (2), 191-199, 2008. DOI: 10.1016/j.jfoodeng.2007.11.026 [ Links ]
[35] Jongjareonrak, A., Benjakul, S., Visessanguan, W., Prodpran, T. and Tanaka, M., Characterization of edible films from skin gelatin of brownstripe red snapper and bigeye snapper. Food Hydrocolloid, 20 (4), pp. 492-501, 2006. DOI: 10.1016/j.foodhyd.2005.04.007 [ Links ]
[36] Ayala, G., Agudelo, A. and Vargas, R., Effect of glycerol on the electrical properties and phase behavior of cassava starch biopolymers. DYNA, 79 (171), pp. 138-147, 2012. [ Links ]
[37] Carvalho, R., Desenvolvimento e caracterizaçao de biofilmes a base de gelatina. Master's Thesis, Universidade Estadual de Campinas, Campinas, Brasil, 1997. [ Links ]
[38] Bertuzzi, M., Vidaurre, E.C., Armada, M. and Gottifredi, J., Water vapor permeability of edible starch based films. J Food Eng, 80 (3), pp. 972-978, 2007. DOI: 10.1016/j.jfoodeng.2006.07.016 [ Links ]
[39] Hanani, Z., McNamara, J., Roos, Y. and Kerry, J., Effect of plasticizer content on the functional properties of extruded gelatin-based composite films. Food Hydrocolloid, 31 (2), pp. 264-269, 2013. DOI: 10.1016/j.foodhyd.2012.10.009 [ Links ]
[40] Benbettaïeb, N., Kurek, M., Bornaz, S. and Debeaufort, F., Barrier, structural and mechanical properties of bovine gelatin-chitosan blend films related to biopolymer interactions. J Sci Food Agric, 94 (12), pp. 2409-2419, 2014. DOI: 10.1002/jsfa.6570, 2014. [ Links ]
[41] McHugh, T., Avena-Bustillos, R. and Krochta, J., Hydrophilic edible films: Modified procedure for water vapor permeability and explanation of thickness effects. J. Food Sci., 58 (4), pp. 899-903, 1993. DOI: 10.1111/j.1365-2621.1993.tb09387.x [ Links ]
[42] Cerqueira, M., Souza, B., Teixeira, J. and Vicente, A., Effect of glycerol and corn oil on physicochemical properties of polysaccharide films - a comparative study. Food Hydrocolloid, 27 (1), pp. 175-184, 2012. DOI: 10.1016/j.foodhyd.2011.07.007 [ Links ]
[43] Cuq, B., Gontard, N., Cuq, J. and Guilbert, S., Selected functional properties of fish myofibrillar protein-based films as affected by hydrophilic plasticizers. J. Agric. Food Chem, 45 (3), pp. 622-626, 1997. DOI: 10.1021/jf960352i [ Links ]
[44] Guilbert, S., Gontard, N. and Cuq, B., Technology and applications of edible protective films. Packag Technol Sci, 8 (6), pp. 339-346, 1995. DOI: 10.1002/pts.2770080607 [ Links ]
[45] Nemet, N., Soso, V. and Lazic, V., Effect of glycerol content and pH value of film-forming solution on the functional properties of protein-based edible films. Acta Periodica Technologica, 41, pp. 57-67, 2010. DOI: 10.2298/APT1041057N [ Links ]
[46] Lagaron, J., Catalá, R. and Gavara, R., Structural characteristics defining high barrier properties in polymeric materials. Mater Sci Tech Ser, 20 (1), pp. 1-7, 2004. DOI: 10.1179/026708304225010442 [ Links ]
R.D. Andrade-Pizarro, is Chemical Engineering in 1994, at the University of Atlántico, Colombia; completed a Specialization in Food Science and Technology in 2004, from the University of Magdalena, Colombia, and a PhD in Food Science and Technology at the University of Santiago Chile, Chile. Currently, he is an associate professor of the Food Engineering Department at the University of Cordoba, Colombia. Ricardo is member of the Society of Rheology (2009-present) and the International Society of Food Engineering (2008-preent). Also, he is a member of the scientific board of the Journal VITAE. His research interests include: Rheology and texture of foods, food processing and edible coatings by spraying.
O. Skurtys, completed a MSc in Aerodynamics, Fluid Mechanics, combustion and thermal sciences in 1999, and a PhD. in Engineering Sciences in 2004; all of them from the Université de Poitiers, École Nationale Supérieure de Mécanique et d'Aérotechnique in the Thermal Studies Laboratory. His research interests include: Fluid mechanics, Granular matter, Soft matter, Surface science, and edible coatings by spraying.
F. Osorio-Lira, received a BSc. Eng in Chemical Engineering in 1977, from Universidad de Chile, Chile, a MSc in Food Science in 1985, from Michigan State University, USA, and PhD. dual in Food Engineering and in Food Science in 1989, from Michigan State University, USA. His research interests include: Rheology and texture of foods, flow of non-Newtonian fluids, and edible coatings by spraying.