Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
DYNA
Print version ISSN 0012-7353
Dyna rev.fac.nac.minas vol.83 no.197 Medellín May/June 2016
https://doi.org/10.15446/dyna.v83n197.42729
DOI: http://dx.doi.org/10.15446/dyna.v83n197.42729
Analysis and characterization of starchy and cellulosic materials after enzymatic modification
Análisis y caracterización de materiales amiláceos y celulósicos después de modificación enzimática
Julián A. Quintero a, Javier A. Dávila b, Jonathan Moncada c, Oscar H. Giraldo d & Carlos A. Cardona e
a Escuela de Ingeniería Bioquímica, Pontificia Universidad Católica del Valparaiso. Valparaiso, Chile. julian.quintero@ucv.cl
b Chemical Engineering Program, Universidad Jorge Tadeo Lozano, Bogotá, Colombia. javiera.davilar@utadeo.edu.co
c Universidad Nacional de Colombia, sede Manizales, Colombia. jmoncadab@unal.edu.co
d Department of Physics and Chemistry, Universidad Nacional de Colombia, sede Manizales, Colombia. ohgiraldoo@unal.edu.co
e Department of Chemical Engineering. Universidad Nacional de Colombia, sede Manizales, Colombia. ccardonaal@unal.edu.co
Received: March 21th, 2014. Received in revised form: November 5th, 2015. Accepted: December 20th, 2015.
This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.
Abstract
This study concerns the application of enzymatic hydrolysis to modifying starchy and cellulosic materials. Corn, cassava, upright elephant ear and sugarcane bagasse were used in order to evaluate the influence of the enzymatic hydrolysis over the structural, thermal stability and crystallinity of these materials. Differential Scanning Calorimetry (DSC) was used to evaluate the thermal properties while Scanning Electron Microscope (SEM), X-ray Diffraction (XRD) and Brunauer, Emmett and Teller (BET) methods were used for the structural and morphological analysis. Corn presented the highest starch yield (g of starch/g of raw material) with 40.4%. For all materials used, the crystallinity increased due to enzymatic hydrolysis suggesting that amorphous zones are attacked first. The gelatinization temperature of the starches increased as the crystallinity increased. The granules and fibers, except for upright elephant ear starch, did not change their size with enzymatic treatment and the superficial area did not increase significantly with the enzymatic treatment.
Keywords: Starch, Cellulose, Upright elephant ear, Cassava, Sugarcane bagasse, enzymatic hydrolysis, structural analysis, thermal analysis.
Resumen
Este estudio se enfoca en la modificación por hidrólisis enzimática de materiales amiláceos y lignocelulosicos. Maíz, yuca, bore y bagazo de caña de azúcar fueron usados para evaluar la influencia de la hidrólisis enzimática sobre las propiedades estructurales y térmicas de estos materiales. Calorimetría de barrido diferencial (DSC) fue usado para evaluar estabilidad térmica y cristalinidad, mientras métodos como Microscopia electrónico de barrido (SEM), difracción de rayos X (XRD) y Brunauer-Emmett-Teller (BET) fueron usados para análisis estructurales y morfológicos. El maíz presento el rendimiento (g de almidón/g de materia prima) más alto con 40.4%. Para todos los materiales usados, la cristalinidad aumento debido a la hidrolisis enzimática sugiriendo que las zonas amorfas son atacadas primero. La temperatura de gelatinización de los almidones incremento conforme aumento la cristalinidad. Los gránulos y fibras, excepto para el bore, no cambiaron su tamaño con el tratamiento enzimático y el área superficial no incremento significantemente con la hidrolisis enzimática.
Palabras clave: Almidón, celulosa, bore, yuca, bagazo de caña de azúcar, hidrolisis enzimática, análisis estructural, análisis térmico.
1. Introduction
Starchy and cellulosic materials are important feedstocks in the agroindustrial sector worldwide because they turn into a platform for sugar production and subsequently other important products. Crops such as corn, cassava, potato and tubers in general are good sources of starch, which can potentially be used in the commercial production of ethanol fuel [1, 2]. Sugarcane, on the other hand, is one of the principal sources of cellulosic material. Both, starchy and cellulosic materials should be modified to increase their yield in the enzymatic hydrolysis. Starch has traditionally been hydrolyzed by acids, but the specificity of the enzymes, and the absence of secondary reactions among other advantages mean that enzymatic hydrolysis an excellent technique for such processes [3,4].
Both, starchy and cellulosic materials can be hydrolyzed with different types of enzymes. Materials such as corn stover, poplar, eucalyptus, corn cob, rice straw, wheat straw among others have been hydrolyzed using enzymes such as endoglucanases, cellulases and xylanases obtaining good yields [4]. The enzyme a-amylase has been used to carry out the hydrolysis of materials such as cornstarch and rice starch reaching yields above of 90% [5,6]. The sugar yield of the enzymatic hydrolysis of starchy and cellulosic materials can be defined by some important characteristics such as particle size, crystallinity, and accessible surface area [7]. These key parameters are defined for the feedstock characteristics such as the nature of the enzyme, its temperature, pH, and the operating conditions used in the enzymatic hydrolysis.
Starch is made up of two types of glucose polymers: amylose that is essentially a linear chain molecule, and amylopectin, which is branched. Amylose and amylopectin are packed into granules, representing the crystalline and amorphous parts of the structure. There is evidence that crystals consist of parallel double helices formed from the short chains of amylopectin. Two types of crystals or polymorph structures (A and B) have been identified in starch granules, which can be distinguished by the packing density of the double helices [8].
Sugarcane bagasse is a polydisperse particulate, which is present as fiber and rind particles. This material has high length-width ratios and corresponds mostly to stalk fibro-vascular bundles. Sugarcane bagasse matter is mostly made up of cells walls. Thus, bagasse particles have cavities that correspond to cell lumina. Besides, there are pith particles, which are finer and near unitary length-width ratio originated from stalk parenchyma [9].
Different techniques have been used to analyze some of the important characteristics of starch and cellulosic materials. Scanning Electron Microscope (SEM) has been used to relate granule morphology to starch genotype and sugarcane sizes [9, 10]. Shapes of starchy and cellulosic materials vary widely depending on the source. Generally, oval, elliptic, spherical and irregular shapes predominate among all the granule shapes. X-ray Diffraction (XRD) has been used to reveal the presence and characteristics of the crystalline structure of the starch granules and the patterns of sugarcane bagasse [11,12]. Differential Scanning Calorimetry (DSC) is used to measure changes of physicochemical properties in gelatinized starch and to measure the rate and extent of starch recrystallization [13]. The specific surface area of the starch granules is measured using Brunauer-Emmett-Teller (BET) technique [14].
Given that enzymatic hydrolysis is an interesting biorefining alternative to obtain a sugar-based platform from starchy and cellulosic materials, the analysis of the effect of enzyme treatment on specific material characteristics is of relevant importance to finding suitable raw materials and sustainable technologies. In this sense, this work shows a primary technical analysis of different starchy and cellulosic materials after enzyme treatment and their potential as future biorefinery feedstocks. Taking into account the importance of factors such as particle size, crystallinity and accessible surface area in starchy and cellulosic materials, this paper also presents a study of the characterization of native and enzyme-treated starchy and cellulosic materials after enzymatic modifications. Corn, cassava and upright elephant ear are studied for starch recovery while, sugarcane bagasse is considered a source of fiber with a high cellulose content. Structural and thermal properties were compared for each material in its native and enzyme-treated states.
2. Materials and methods
2.1. Raw materials
Cassava (Manihot esculenta) and yellow dent corn (Zea mays) were obtained from a local Colombian market. Sugarcane bagasse (Saccharum) was obtained from a small mill, which produces commercial sugarcane juice. Upright elephant ear roots (Alocasia macrorrhiza), were collected from a farm belonging to the National Service of Learning (SENA) in Manizales-Colombia (center-west of Colombia). These raw materials were thoroughly washed with enough water to remove impurities. Cassava and upright elephant ear were manually peeled, and then processed for starch recovery. Corn grains were separated from the corncobs and subsequently processed for starch recovery. While, sugarcane bagasse was milled in a mill blade and the obtained fibers were dried (for 48 h at 75 ºC) and sieved.
2.2. Starch recovery
Cassava and upright elephant ear starch were recovered by the conventional method used in tropical countries. This method consists of root washing, peeling, milling, and filtering [15]. The starch solution obtained from filtering was decanted for 24 h, after which, the residual liquid was extracted and the solid starch was dried at 60 ºC to constant weight.
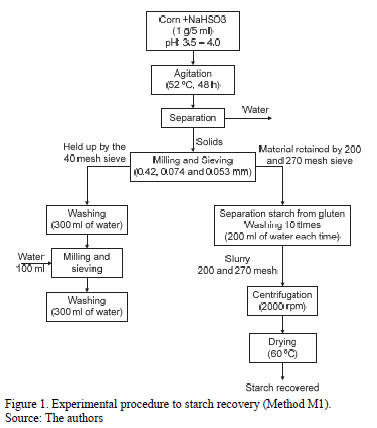
Starch from corn was recovered using two methods. In the first method (M1) exposed by [16], starch recovery was calculated as the ratio of the weight of starch recovered from wet milling to the total weight of the starch present in the corn. To give a better idea of the work undertaken, Fig. 1 shows the experimental procedure followed to recover starch. In the second method (M2), lactic acid and sodium bisulfite were used for the steeping step, and the starch content was measured according to the method reported by [17].
2.3. Enzymatic hydrolysis
The hydrolysis of starch from cassava, upright elephant ear and corn was conducted with a-amylase (HT-340L, Bacterial source: Bacillus licheniformis; supplied by Proenzimas, Cali, Colombia), which allows a partial hydrolysis under special conditions due to its action on starch polymers. On the other hand, Cellulase (Celulase CE 2, Fungal source: Trichoderma longbrachiatum; supplied by Proenzimas, Cali, Colombia) was used for sugarcane bagasse modification. Before hydrolysis, all materials were dried in an oven for 48 h at 60 ºC [18]. Dried starches were milled in a ball mill (KM1, Germany) obtaining a dry powder. Enzymatic hydrolysis was carried out in a shaker (UNITRONIC OR, J.P. Selecta, S.A., Barcelona).
Enzymatic modifications were based on procedures exposed by [19]. Starch modification using the a-amylase occurred by preparing a 1/50 dilution of enzyme in a buffer solution, which gave an activity level of 340.000 Modified Wohlhemuth Units (1 MWU = quantity of enzyme that will dextrinize one milligram of starch in 30 minutes at pH 6 and 50 ºC). The buffer was prepared by combining deionized water with 2.4 g/L of sodium phosphate (monobasic; Sigma) and 0.39 g/L of sodium chloride (Sigma). The pH was adjusted to 6.9 using an aqueous solution of sodium hydroxide (Carlo ERBA Reagent). The starches where added at 2:3 v/v ratio of starch to enzyme solution. The modification took place at pH 6.9 at 25 ºC for 7 days. The starch was then washed repeatedly and then dried in a convection oven for 24 h at 40 ºC.
Cellulose hydrolysis was carried out with a 1/2 and 1/6 cellulase solution in citrate buffer at a ratio of 10 ml of the enzyme solution to 1 g of sugarcane bagasse. The mixture was kept at 50 ºC in a shaker for 6 days. After enzymatic hydrolysis of the starchy materials and sugarcane bagasse, the reducing sugar concentrations were measured according to the method proposed by Nelson and Somogyi [20].
2.4. SEM, DSC, XRD and BET analysis
After hydrolysis, several analyses were carried out using Scanning Electron Microscope (SEM), Differential Scanning Calorimetric (DSC), X-ray Diffraction (XRD) and Brunauer-Emmett-Teller (BET). All samples were dried in a vacuum oven (3 mm Hg) at 60 ºC for 3 days. SEM micrographs were obtained in a JEOL JSM-5910LV microscope. Calorimetric plots were measured in a DSC Q 100 (Module DSC Standard Cell FC, TA Instruments) unit in a temperature range of 25-200 ºC for starchy materials and 25-355 ºC for sugarcane bagasse, using a heating rate of 10 ºC/min, under nitrogen atmosphere with a flow of 50 mL/min. The X-ray diffraction plots were obtained in a Rigaku (MiniFlex II) unit with CuKa at 30 kV and 15 mA. The diffraction angle ranged from 35º to 2º with a scan rate of 5 º/min. Finally, the superficial area was determined in a Micromeritics ASAP 2020 unit (Micromeritics Instrument Corporation).
3. Results and discussion
3.1. Starch recovery
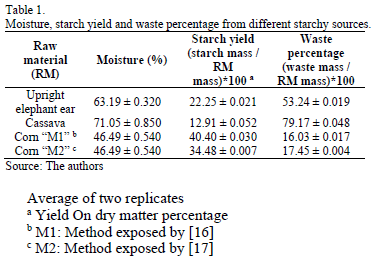
Table 1 shows the moisture, starch yield and the waste percentage determined for the starchy materials. From these results, we can see that the lowest yield corresponds to cassava due to its high moisture content therefore, for this raw material, removing water becomes in an important step that requires an efficient technique. [21]. Thus, the starch recovered from corn using the method M1 had the highest yield in contrast to method M2. The highest yield was obtained for corn also with method M2. On the other hand, cassava showed the highest waste production, while treated corn using method M1 showed the lowest. This fact suggests that the sugar yield and waste production in the starch recovery processes are strongly inversely related.
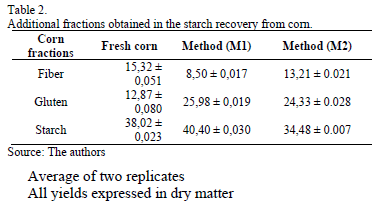
Additionally, the cornstarch recovery process generated other products, which can be considered commercial byproducts in a wet milling or dry milling concepts. In this sense, Table 2 shows additional fractions of fiber and gluten as byproducts from corn hydrolysis. Thus, starches obtained from the three selected raw materials were not completely pure given the remaining protein. The starch purities obtained were 74.03 ± 0.520 %, 73.92 ± 0.410% and 74.03 ± 0.658 for upright elephant ear, corn and cassava, respectively.
3.2. Sugarcane bagasse treatment
Sugarcane moisture was 65.02 ± 0.080 %. The crust corresponded to 92.85 ± 0.002% of the dry matter while marrow was only 7.15 ± 0.002 % of the dry matter. Consequently, the subsequent analyses were performed based on sugarcane crust due to its greater contribution.
3.3. Enzymatic hydrolysis
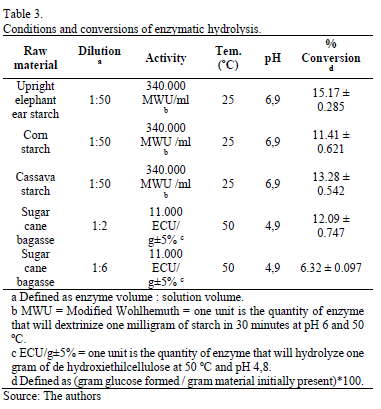
Conditions and conversion of enzymatic hydrolysis are shown in Table 3. The highest starch conversion was reached for upright elephant ear starch followed by cassava starch [22]. This fact suggests that starch granules from roots are more exposed to the enzymatic attack. Sugarcane bagasse with a dilution of 1:2 v/v presented grater conversion than that obtained from a dilution of 1:6 v/v. This behavior was expected due to the low enzyme concentration.
In the hydrolysis process, a-amylase breaks only the a-1.4 linkages presented in amorphous zones producing glucose units and partial hydrolysis is accomplished by correct hydrolysis conditions. This procedure was necessary for holding the starch granule structure while exposing the hydroxyl groups of the amorphous starch chain. A similar process occurs with the cellulose from sugarcane bagasse. In the partial acid hydrolysis, cellulose was broken down into cellobiose (glucose dimer), cellotriose (glucose trimer), and cellotetrose (glucose tetramer), whereas upon complete acid hydrolysis, it is broken down into glucose. The b-1,4 glycoside linkages cause cellulose to be in a low surface area crystalline form.
3.4. SEM, DSC, XRD and BET analysis
3.4.1. Analysis of SEM micrographs
Scanning electron microscope (SEM) images of the starch granules and bagasse are shown in Fig. 2.
Native upright elephant ear starch granules (Fig. 2a) are particles with spherical and ovoidal shapes with diameters of between 5 and 15 µm and a smooth surface. Enzyme-treated upright elephant ear starch tends to get agglomerated while native granules were scattered. Modified starch granules changed their diameters to 1-5 µm, and their shape was altered obtaining irregular arrangements with a rough surface (see Fig. 2b). Native starch granules from corn are shown in Fig. 1c, the particles presented a spherical shape with some irregularities, diameters of between 5 and 10 µm and a semi-porous surface. Enzyme-treated cornstarch granules kept their size but presented some fractures due enzymatic attack (see Fig. 2d).
Micrograph from Fig. 2e shows native cassava starch granules, which present irregular shapes and sizes (between 5 and 10 mm) with a smooth surface. Enzyme-treated cassava starch granules, as well as upright elephant ear starch, tend to agglomerate but to a lesser extent. Fig. 2f shows the deterioration caused by enzymatic action, but with no change in the size. Enzymatic treatment generated deformations and breaking of the granules but no pores on the particle surface [22].
In general for all starches, enzyme treatment did not create new visible pores, but did cause some fractures and shape alteration, the most appreciable enzymatic effect occurred with upright elephant ear starch granules. Here, a-amylases did not have the ability to create pores on particle surfaces as observed by other authors using glucoamylases as a modifying agent [23,24].
Native bagasse fibers are shown in Fig. 1g. These are elongated with lengths of over 200 µm and are formed with several smooth surface layers. Hydrolyzed bagasse fibers with an enzyme to buffer ratio of 1:2 v/v are shown in Fig. 1h. There is evidence of layers breaking by enzymatic action exposing the inner channels of the fiber, but fiber length was kept after hydrolysis. Bagasse fibers hydrolyzed with an enzyme to buffer ratio 1:6 v/v, presented minor signs of enzymatic degradation.
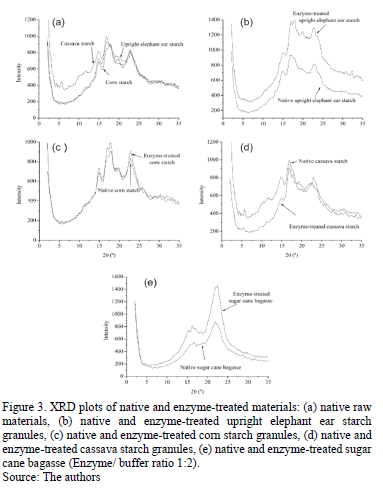
3.4.2. Analysis of XRD plots
XRD patterns for starchy materials are shown in Fig. 3a. The patterns and characteristic of the starches are similar; however, the difference depends on the natural source of each starch. According to starch classification from X-ray diffraction, starch form Corn is classified as type A, because it presents the highest peaks at around 2q (angle of diffraction) values of 14.92º, 17.18º, 17.8º and 22.7º, which are characteristic angles for this sort of starch [25]. Thus, upright elephant ear and cassava starch presented the maximum peak at 16.82º and 16.98º, respectively. Moreover, it has peaks of less intensity at 2q values of 20.80º, 22.86º and 24.20º for upright elephant ear starch and 19.70º, 22.8º and 24.2º for cassava starch. As a result, upright elephant ear and cassava starches were classified as type B.
Crust and marrow bagasse have different structures and crystallinity because crust bagasse presented two diffraction peaks at 2q values, (18.04º and 21.9º). However, marrow bagasse presented only a peak at 2q values (21,86º), which is characteristic of the cellulose structures [26,27].
Fig. 3b shows the diffraction patterns for native and enzyme-treated upright elephant ear starch. Enzyme-treated upright elephant ear starch presents peaks with more intensity and definition than native starch suggesting that crystallinity was increased. Enzymatic attack promoted the formation of two peaks at 2q values of 14.98º and 17.84º, the XRD pattern being more similar to a type A starch. Fig. 3c shows native and enzyme-treated cornstarch which did not present differences in 2q values for their diffraction peaks. Nonetheless, these peaks were more defined and with greater intensity for enzyme-treated starch, indicating that hydrolysis generated a more crystalline structure. The latter also happened to upright elephant ear starch. Fig. 3d shows the enzyme-treated cassava starch which presents diffraction peaks at 2q (14.92º and 17.76º), indicating that the XRD pattern was more similar to a type A starch. Diffraction peaks for this starch presented less intensity. Fig. 3e shows the hydrolyzed crust bagasse, which exhibited defined diffraction peaks and high intensity for both evaluated ratios (1:2 v/v and 1:6 v/v). This Figure also shows the diffraction pattern for enzyme to buffer ratio of 1:2 v/v. Bagasse became more crystalline with the enzyme treatment. As a consequence, enzymatic hydrolysis promoted the crystallinity by attacking the amorphous zones first.
3.4.3. Analysis of BET surface area
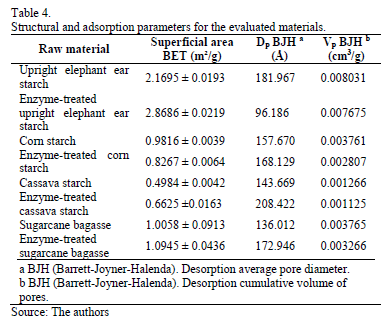
Nitrogen adsorption isotherms were obtained using the BET method. According to Brunauer's classification [28], all starchy materials exhibited type II isotherms (Isotherm with large deviation from Langmuir model absortion) with hysteresis from the middle zone of the curve indicating the presence of meso and macro pores. Sugarcane bagasse also showed a type II isotherm with a more pronounced hysteresis. This indicates that at high pressures, condensation in meso and macro pores is higher, which makes it more difficult to evaporate the nitrogen. Considering the latter, Table 4 shows the structural parameters obtained from the isotherms by the BET method.
Upright elephant ear starch was the material with the highest superficial area for both native and enzyme-treated cases. Enzymatic hydrolysis did not significantly change the superficial area for all tested materials. The specific surface areas obtained are low compared to other commercial materials. As a result, there is concern about the viability of using these materials as adsorbents. However, it is important to evaluate the performance of these materials as adsorbents in dehydration processes. This is relevant since enzymatic treatment may have exposed more functional groups (hydroxyl groups) that can absorb water. However, such groups could not be found using the BET method. Thus, pore diameters obtained by the BJH method [29] confirmed the existence of macro pores in the studied materials.
3.4.4. Analysis of DSC plots
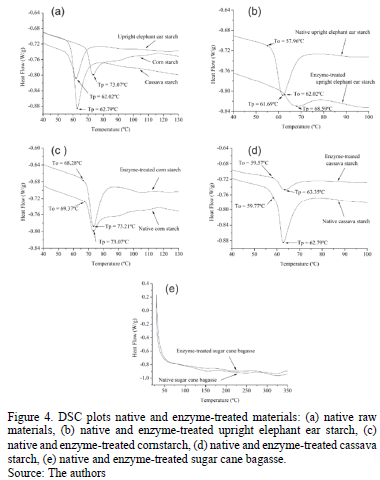
Fig. 4 shows the DSC patterns for native and enzyme-treated materials. DSC patterns for starches show the endothermic transition known as gelatinization, which occurs when the starch is heated in presence of water. Excess water was used to ensure the existence of a single peak. The onset, Ti, and the peak, Tp, temperatures were determined from the intercepts with the baseline. Fig. 4a shows the DSC pattern for native upright elephant ear, cassava, and corn, for which gelatinization peak temperatures were 62.02 ºC, 62.79 ºC and 73.07 ºC respectively. This is in alignment with literature values for corn and cassava starch [30]. There is no open literature reporting on the thermal properties of upright elephant ear starch.
Peak temperatures for cassava and upright elephant ear starches were similar, while cornstarch had a peak temperature 10 ºC higher than the roots. Other authors have reported that at higher temperatures, the granular structure has more ordered areas [31]. It has also been suggested that gelatinization of large starch particles occurs at a lower temperature than that of small particles [32].
Fig. 4b shows the change in onset and peak temperatures by enzymatic hydrolysis of upright elephant ear starch. Onset and peak temperatures were increased by 4 ºC and 7 ºC, respectively. The latter suggests that upright elephant ear starch became more crystalline. Degradation temperature was affected too, changing from 193.5 ºC to 162 ºC. This fact represents that thermal stability is decreased with partial hydrolysis. On the other hand, Fig. 4c shows that gelatinization temperatures for cornstarch were not significantly altered. However, the degradation temperature decreased with de partial hydrolysis, implying the existence of some structural change. Degradation temperatures for native and enzyme-treated cornstarch were 174 ºC and 152 ºC, respectively.
Fig. 4d shows that gelatinization temperatures for cassava starch were not significantly altered. However, the degradation temperature increased with the partial hydrolysis, suggesting that some structural changes occurred. Degradation temperatures for native and enzyme-treated cassava starch were 168 ºC and 179 ºC, respectively. In general, gelatinization temperatures had the tendency increase with partial hydrolysis. This increment was related to the increase of crystallinity and decreasing particle size.
Finally, Fig. 4e shows that sugarcane bagasse did not present melting curves because of the high cristallinity of cellulose. Some small curves were obtained but these correspond to water elimination. In the evaluated range of temperatures, the degradation temperature was not reached.
4. Conclusions
Both, starchy and cellulosic materials presented changes after enzymatic modification. Size reduction, shape alterations, fractures, changes in the surface area, and pore formation the most important changes caused by the enzymatic modification.
From the raw materials used, corn and upright elephant ear presented the highest starch yield and reducing sugar production respectively. Sugarcane bagasse did not require a further recovery process, and only a structural components separation (such as crust and marrow) was necessary. In general, enzymatic treatment increased crystallinity as shown in XRD patterns. Enzymatic hydrolysis generated alterations over starch granules as was observed in SEM images.
Upright elephant ear starch granules were more affected by hydrolysis than other materials, suffering a greater size reduction. The enzyme altered the surfaces of both, corn and cassava, but particle size was not affected. Enzymatic hydrolysis of sugarcane bagasse showed an appreciable fiber alteration exposing its inner channels. This work reveals that analyses of these types of materials are necessary to understand and evaluate the performance of the enzymatic hydrolysis as they offer valuable information about the influence of the structural and thermal properties on their adsorption performance.
Acknowledgment
The authors would like to thank the Nano-Structured and Functional Materials Laboratory (National University of Colombia at Manizales) for technical assistance in XRD and BET analyses, Dr. Andrés Rosales R., Magnetism and Advanced Materials Laboratory (National University of Colombia at Manizales) for technical assistance in DSC analyses, Dr. Medardo Pérez, Advanced Microscopy Laboratory (National University of Colombia at Medellin) for technical assistance in SEM analyses.
References
[1] Cardona, C.A. and, Sánchez, O.J., Fuel ethanol production: Process design trends and integration opportunities. Bioresource Technology 98, pp. 2415-2457, 2007. DOI:10.1016/j.biortech.2007.01.002. [ Links ]
[2] Quintero, J.A., Montoya, M.I., Sánchez, O.J., Giraldo, O.H. and Cardona, C.A., Fuel ethanol production from sugarcane and corn: Comparative analysis for a Colombian case. Energy 33 pp.385-399. 2008. DOI:10.1016/j.energy.2007.10.001 [ Links ]
[3] Sánchez, O.J. and Cardona, C.A., Trends in biotechnological production of fuel ethanol from different feedstocks. Bioresource Technology 99, pp. 5270-5295. 2008. DOI: 10.1016/j.biortech.2007.11.013 [ Links ]
[4] Van Dyk, J.S. and Pletschke, B.I., A review of lignocellulose bioconversion using enzymatic hydrolysis and synergistic cooperation between enzymes-Factors affecting enzymes, conversion and synergy. Biotechnology Advances 30, pp. 1458-1480. 2012. DOI: 10.1016/j.biotechadv.2012.03.002 [ Links ]
[5] Apar, D.K. and Özbek, B., s-Amylase inactivation during corn starch hydrolysis process. Process Biochemistry 39, pp. 1877-1892. 2004, DOI: 10.1016/j.procbio.2003.09.014 [ Links ]
[6] Apar, D.K. and Özbek, B., a-Amylase inactivation during rice starch hydrolysis. Process Biochemistry 40, pp. 1367-1379. 2005. DOI: 10.1016/j.procbio.2004.06.006 [ Links ]
[7] Zhang, Q., Zhang, P., Pei, Z.J. and Wang, D., Relationships between cellulosic biomass particle size and enzymatic hydrolysis sugar yield: Analysis of inconsistent reports in the literature. Renewable Energy 60, pp. 127-136. 2013. DOI: 0.1016/j.renene.2013.04.012 [ Links ]
[8] Shujun, W., Jinglin, Y., Jiugao, Y., Jiping, P. and Hongyan, L., Structure characterization of C-type starch granule by acid hydrolysis. Food Hydrocolloids 22, pp. 1283-1290. 2008. DOI: 10.1016/j.foodhyd.2007.06.012 [ Links ]
[9] Driemeier, C., Oliveira, M.M., Mendes, F.M. and Gómez, E.O., Characterization of sugarcane bagasse powders. Powder Technology 214, pp. 111-116, 2011. DOI: 10.1016/j.powtec.2011.07.043 [ Links ]
[10] Fannon, J.E., Hauber, R.J. and Bemiller, J.N., Surface pores of starch granules. Cereal Chemistry, 69, pp. 284-288, 1992. [ Links ]
[11] Hoover, R., Composition, molecular structure and physicochemical properties of tuber and root starches: A review. Carbohydrate Polymers, 45, pp. 253-267, 2001. DOI: 10.1016/S0144-8617(00)00260-5 [ Links ]
[12] Loh, Y.R., Sujan, D., Rahman, M.E. and Das, C.A. Sugarcane bagasse-The future composite material: A literature review. Resources, Conservation and Recycling. 75. pp. 14-22, 2013. DOI: 10.1016/j.resconrec.2013.03.002 [ Links ]
[13] Hyang-Aee, L., Nam-Hie, K. and Nishinari, K., DSC and rheological studies of the effects of sucrose on the gelatinization and retrogradation of acorn starch. Thermochimica Acta, 322, pp. 39-46, 1998. DOI: 10.1016/S0040-6031(98)00469-9 [ Links ]
[14] Chen, Y., Huang, S., Tang, Z., Chen, X. and Zhang, Z., Structural changes of cassava starch granules hydrolyzed by a mixture of a-amylase and glucoamylase. Carbohydrate Polymers, 85, pp. 272-275, 2011. DOI: 10.1016/j.carbpol.2011.01.047 [ Links ]
[15] Alarcon, M.F. y Dufour, D., Almidon agrio de yuca en Colombia. CIAT Centro International de Agricultura Tropical 1998. ISBN: 958-9439-67-5 [ Links ]
[16] Haros, M. and Suarez, C., Effect of drying, initial moisture and variety in corn wet milling. Journal of Food Engineering, 34, pp. 473-481, 1997. DOI: 10.1016/S0260-8774(97)00099-X [ Links ]
[17] Serna, S.S.O., Refinación de almidón y producción de jarabes glucosados a partir de sorgo y maíz. Transferencia de Programas de Graduados e Investigación. [Online]. 1998. Available at: http://www.mty.itesm.mx/die/ddre/transferencia/Transferencia42/eli-01.htm. [ Links ]
[18] Giraldo, G., Orrego, C., Grajales, L., Telis, V., Gabas, A. and Telis J., Effect of drying methods on the thermodynamic properties of blackberry pulp powder. DYNA, 78(168), pp 139-148, 2011. [ Links ]
[19] Beery, K.E., Gulati, M., Kvam, E. and Ladisch, M., Effect of enzyme modification of corn grits on their properties as an adsorbent in a skarstrom pressure swing cycle dryer. Adsorption, 4, pp. 321-335, 1998. DOI: 10.1023/A:1008846003116 [ Links ]
[20] Nelson, N., A Photometric adaptation of the Somogyi method for the determination of glucose. Journal of Biological Chemistry, 153, pp. 375-380, 1944. [ Links ]
[21] Breuninger, W.F., Piyachomkwan, K. and Sriroth, K., Chapter 12 - Tapioca/Cassava starch: Production and use. In: James,B. and Roy, W., Eds. Starch (Third Edition). San Diego: Academic Press, 2009, pp. 541-568. DOI: 10.1016/B978-0-12-746275-2.00012-4 [ Links ]
[22] Quintero, J.A. and Cardona, C.A., Ethanol dehydration by adsorption with starchy and cellulosic materials. Industrial Engineering Chemistry Research, 48, pp. 6783-6788, 2009. DOI: 10.1021/ie8015736 [ Links ]
[23] Aggarwal, P. and Dollimore, D., A thermal analysis investigation of partially hydrolyzed starch. Thermochimica Acta, 319, pp. 17-25, 1998. [ Links ]
[24] Aggarwal, P. and Dollimore, D., Degradation of starchy food material by thermal analysis. Thermochimica Acta, 357-358, pp. 57-63, 2000. DOI: 10.1016/S0040-6031(00)00368-3 [ Links ]
[25] Sajilata, M.G., Singhal, R.S. and Kulkarni, P.R., Resistant starch. A review. Comprehensive Reviews in Food Science and Food Safety, 5, pp. 1-17, 2006. DOI: 10.1111/j.1541-4337.2006.tb00076.x [ Links ]
[26] Filho, G.R., de Assunção, R.M.N., Vieira, J.G., Meireles, C.dS., Cerqueira, D.A., da Silva-Barud, H., Ribeiro, S.J.L. and Messaddeq, Y., Characterization of methylcellulose produced from sugar cane bagasse cellulose: Crystallinity and thermal properties. Polymer Degradation and Stability, 92, mpp. 205-210, 2007. DOI: 10.1016/j.polymdegradstab.2006.11.008 [ Links ]
[27] Zhao, H., Kwak, J.H., Conrad, Z.Z., Brown, H.M., Arey, B.W. and Holladay, J.E., Studying cellulose fiber structure by SEM, XRD, NMR and acid hydrolysis. Carbohydrate Polymers, 68, pp. 235-241, 2007. DOI: 10.1016/j.carbpol.2006.12.013 [ Links ]
[28] Andrade, P.R.D., Lemus, M.R. and Perez, C.C.E., Models of sorption isotherms for food: Uses and limitations. VITAE Revista de la Facultad de Química Farmaceutica, 18, pp. 325-334, 2011. [ Links ]
[29] Webb, P.A. and Orr, C., Analytical methods in fine particle technology. norcross, First Edition ed, Micromeritics Instrument Corporation, USA, 1997. ISBN-13: 978-0965678308 [ Links ]
[30] Sandoval, M.L., Camerucci, M.A., Cavalieri, A.L. y Sian, A.N., Conformado por consolidación directa con almidón de precursores de materiales porosos de cordierita. [En línea]. 2007. Congresso Brasileiro de Cerâmica, No. 51, Salvador Brasil, [Consultado: Febrero de 2013] Disponible en: http://wwwconicetgovar/new_scp/detallephp?keywords=&id=20192&congresos=yes&detalles=yes&congr_id=1186567 [ Links ]
[31] Qi, X., Tester, R.F., Snape, C.E., Yuryev, V., Wasserman, L.A. and Ansell, R., Molecular basis of the gelatinisation and swelling characteristics of waxy barley starches grown in the same location during the same season. Part II. Crystallinity and gelatinisation characteristics. Journal of Cereal Science, 39, pp. 57-66. 2004. DOI: 10.1016/S0733-5210(03)00065-1 [ Links ]
[32] Bar, W.H., Park, Y.K. e Papini, R.S., Relacao entre intumescimento, gelatinizacao suscetibilidade dos amilos de mandioca e de milho . alfa-amilase bacteriana. Revista Brasileira de Tecnologia, 2, pp. 95-100, 1971. [ Links ]
J. Quintero, completed his BSc in Chemical Engineering in 2005, his MSc degree in Chemical Engineering in 2008, and his PhD degree in Automatic Engineering in 2011, all of them at Universidad Nacional de Colombia, Manizales, Colombia. Since 2011, he has been a postdoctoral researcher at the Universidad Católica de Valparíso, Valparaíso, Chile and since 2014 he has been an assistant professor at the School of Biochemical Engineering at the same university. His main research interests are: first and second generation biofuels production, process modelling and simulation, life cycle analysis, fermentation, enzymatic conversion and non-conventional separation operations. ORCID: 0000-0003-1224-5649
J. Davila, completed his BSc. Eng in Chemical Engineering in 2005 at Universidad Nacional de Colombia, Manizales; his MSc degree in Chemical Engineering in 2008, at Universidad del Valle, Cali, Colombia and his PhD degree in Automation in 2016, at Universidad Nacional de Colombia, Manizales, Colombia. From 2009 to 2012, he worked for Chemical Engineering Department at the Universidad de los Andes, Bogota, Colombia. Currently, he is a full professor in the Engineering Department, Universidad Jorge Tadeo Lozano, Bogotá Colombia. His research interests include: simulation, modeling and design of processes, alternative energy, supercritical fluids, and biorefineries. ORCID: orcid.org/0000-0002-4583-7512
J. Moncada, works as a junior researcher at the Copernicus Institute of Sustainable Development, Utrecht University, in the Netherlands. He holds BSc. and Msc. degrees in Chemical Engineering. He has worked as researcher and consultant in many projects related to the techno-economic and environmental assessment of biofuels and biorefineries. His research interests include the design and sustainability assessment of biorefinery systems, bio-based economy, sustainability assessment of novel bio-based routes for chemicals production, and integrated multiproduct biorefineries. ORCID: orcid.org/0000-0003-3620-8594
O.H. Giraldo, completed his BSc. in Chemical Engineering in 1992, at Universidad del Valle, Cali, Colombia, and his Dr. degree in 2000, at the University of Connecticut, USA. He is a full professor in the Physics and Chemistry Department, Universidad Nacional de Colombia, Manizales Colombia. His research interests include: Inorganic and nuclear chemistry, chromatographic, nanotechnology. ORCID: orcid.org/0000-0002-9213-6348
C.A. Cardona, completed his BSc. En in Chemical Engineering in 1993, his MSc. degree in Chemical Engineering in 1995 and his Dr. degree in 2001, all of them at the Estatal Academy Lomonosov, Russia. He is a full professor in the Chemical Engineering Department, Universidad Nacional de Colombia, Manizales, Colombia. His research interests include: industrial biotechnology, biofuels, agroindustrial waste usage, biorefineries and reactive distillation. ORCID: orcid.org/0000-0002-0237-2313