Serviços Personalizados
Journal
Artigo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Acessos
Acessos
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares em
SciELO
Similares em
SciELO -
 Similares em Google
Similares em Google
Compartilhar
DYNA
versão impressa ISSN 0012-7353
Dyna rev.fac.nac.minas vol.83 no.197 Medellín maio/jun. 2016
https://doi.org/10.15446/dyna.v83n197.50318
DOI: http://dx.doi.org/10.15446/dyna.v83n197.50318
Differences between the use of ferric sulphate and ferric chloride on biodesulfurization of a large coal particle
Diferencias entre el uso de sulfato férrico y cloruro férrico en la biodesulfurización de un carbón con tamaño de partícula grueso
Gerardo Andrés Caicedo-Pineda a & Marco Antonio Márquez-Godoy b
a Facultad de Minas, Universidad Nacional de Colombia, Medellín, Colombia. gacaiced@unal.edu.co
b Facultad de Minas, Universidad Nacional de Colombia, Medellín, Colombia. mmarquez@unal.edu.co
Received: April 29th, 2015. Received in revised form: November 05th, 2015. Accepted: February 20th, 2016.
This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.
Abstract
Three iron sources (FeSO4, Fe2(SO4)3 and FeCl3) at different concentrations (150, 700 and 1250 mg Fe/L) were evaluated on large coal particle biodesulfurization processes at Erlenmeyer level. A consortium of Acidithiobacillus ferrooxidans (ATCC 23270) and Acidithiobacillus thiooxidans (ATCC 15494) was used in all the assays. By using 150 mg Fe2+/L (from FeSO4), pyrite biooxidation was 31.14% after 12 days. When Fe2(SO4)3 replaced FeSO4, oxidation improved by 21.16%. The assays using the highest concentrations of sulphate sources also obtained the same increase. However, Fe2(SO4)3 assays had a better sulphate removal from coal. This suggests that using the smallest concentration of Fe2(SO4)3 is a good alternative to boost the pyrite oxidation rate and avoid the formation of precipitates. Additionally, biooxidation in the FeCl3 assays decreased, indicating, a priori, that the microorganisms were not able to adapt properly to Cl- ions.
Keywords: coal biodesulfurization; pyrite oxidation; ferric chloride, ferric sulphate; Acidithiobacillus ferrooxidans; Acidithiobacillus thiooxidans.
Resumen
Se evaluaron tres fuentes de hierro (FeSO4, Fe2(SO4)3 y FeCl3) a diferentes concentraciones (150, 700 y 1250 mg Fe/L) en procesos de biodesulfurización de un carbón con tamaño de partícula grueso, utilizando un consorcio de Acidithiobacillus ferrooxidans (ATCC 23270) and Acidithiobacillus thiooxidans (ATCC 15494). Al usar 150 mg Fe2+/L (FeSO4), luego de 12 días se obtuvo 31.14% de pirita oxidada. Al reemplazar FeSO4 por Fe2(SO4)3, la oxidación mejoró en un 21.16%. Aunque los ensayos con las mayores concentraciones de sulfatos obtuvieron un resultado similar, al utilizar Fe2(SO4)3 permitió mejor remoción de sulfatos del carbón. Lo anterior sugiere que basta con utilizar la menor concentración de Fe2(SO4)3 para mejorar el índice de oxidación de pirita y evitar formación de precipitados. Por otra parte, los ensayos con FeCl3 tuvieron una disminución en la tasa de biooxidación, indicando a priori, que los microorganismos no fueron capaces de adaptarse adecuadamente a los iones Cl-.
Palabras clave: biodesulfurización de carbón; oxidación de pirita; cloruro férrico; sulfato férrico; Acidithiobacillus ferrooxidans; Acidithiobacillus thiooxidans.
1. Introduction
Among the elements contained in coal, sulphur plays an important role in almost all systems that use the material, especially combustion processes. The generation of sulphuric-volatile compounds (SO2, SO3) causes atmospheric pollution, contributing to acid rain generation [1]. In order to limit sulphur oxide emissions, some research proposes precombustion processes as good methods to reduce sulphur forms from coal. Sulphur on coal is present in two basic forms: organic, part of the coal structure, and inorganic, formed basically by sulphides (mainly pyrite) and sulphates [2].
High-sulphur coals (S > 3% w) generally present pyrite at a high rate (60-80% of total sulphur) [3,4]. For this reason, a lot research has focused on coal depyritization processes. Biological processes have many economic and environmental advantages in comparison to chemical and physical options [5,6]. Nowadays, the research evaluates physical, chemical and biological parameters in order to seek alternatives with possible industrial applications [6].
Basically, biodepyritization consists of the oxidation of sulphides catalysed by acidophilic microorganisms, in an aqueous medium, generating soluble sulphates [1,7,8]. A typical culture medium contains Fe2+ from ferrous sulphate. Bacteria like A. ferrooxidans, oxidize Fe2+ producing Fe3+ by eq. (1). Then, Fe3+ attacks the pyrite, releasing more Fe2+ ions by eq. (2) which are taken by bacteria, creating a cycle of Fe3+ regeneration [8,9].
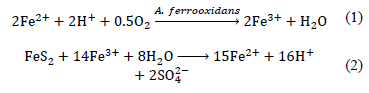
It is known that the use of fine-particle coal increases the exposed surface area of pyrite, improving its leaching. However, pyrite oxidation is not the only variable to consider in the design of the process. Precipitate generation is also important to define the best parameters for operation. Using fine particles increases the effect of coal compounds such as alkalinizing agents and carbonates. This induces the formation of undesirable compounds (i.e. jarosite) which could reprecipitate over the coal, reducing the efficiency of global desulfurization [4,6]. Recent studies showed low or null precipitate generation when large-particle coal is used, although pyrite oxidation decreased [10].
The use of large-particle coal also presents important advantages to the process. This minimizes risks to health, because volatile particles of fine coal could cause damage to the airways of humans and animals who come into contact with the material. Additionally, large-particle coal does not require heavy grinding and/or milling processes and it is easy to transport to regions far from the source. All of these are important factors when using a low-cost material. Besides, a wide range of cheap systems can treat coal at this size, from processes such as packed-bed bioreactors (heaps) [6] and even to stirred tank bioreactors. Previous research reports on a pilot plant level reactor using -1" Tyler mesh coal [11]. The low-specific gravity of the material (around 1.30) allowed it to be maintained in suspension without requiring a high agitation.
Multistage processes and/or long residence times would help to improve pyrite oxidation [5,12]. However, these also increase the costs of treatment. Previous research found that replacing ferrous sulphate by ferric salts also helps to accelerate pyrite oxidation, because it minimizes the lag phase, where the bacteria begin to oxidize ferrous ions [8,13]. This might be an alternative to boost the pyrite oxidation rate in processes using large-particle coal. Therefore, the research presented here evaluated how ferric ions from two different iron sources (sulphate and chloride) interacted with desulfurization processes of pyrite-rich coal mediated by a consortium of acidophilic bacteria. Chemical and mineralogical techniques allowed the behaviour of the process to be evaluated.
2. Experimental Procedure
2.1. Coal
High-volatile bituminous coal samples were collected from "La Angostura" mine (Morales, Cauca, Colombia). The sample was ground to a particle size between -8 +30 Tyler mesh (2.38 mm < particle size < 0.50 mm). Table 1 shows proximate analyses, sulphur forms and iron composition in coal samples.
2.2. Microorganism
A consortium of A. thiooxidans (ATCC 15494) and A. ferrooxidans (ATCC 23270) were selected from the collection of Laboratorio de Biomineralogía y Biohidrometalurgia of the Universidad Nacional de Colombia - Sede Medellín. The microorganisms were previously adapted to coal biodesulfurization, according to an established protocol [3]. Inoculums preparation was carried out in 350 mL flasks, with a working volume of 150 mL, containing 1g of coal per 10 mL of solution. The solution was composed of 10% v of inoculum (5x108 cells/mL), 150 mg Fe2+/L (from FeSO4.7H2O) and modified T&K medium [14], defined as: 0.50 g/L of (NH4)2SO4, 0.50 g/L of MgSO4.7H2O, 0.50 g/L of KH2PO4 and 1.5 mL/L of H2SO4 98% . The microorganism was adapted to ferric sulphate (92% of total iron as Fe3+ ions) and ferric chloride, replacing ferrous sulphate. The cultures were incubated in a shaker for 12 days, at 30 ± 1 ºC, using a mixing rate of 180 ± 2 rpm. The incubation was repeated several times and no variations of the biodesulfurization ratio among the three last processes were observed.
2.3. Coal biodesulfurization processes
The assays were prepared in 350 mL flasks, with a working volume of 150 mL, using a coal/culture medium ratio of 1:10 (g of coal: mL solution). The variables evaluated were: (i) proportion of iron added (mg/L): 150 (C1), 700 (C2) and 1250 (C3) and (ii) iron source: ferrous sulphate (SF(II)), ferric sulphate - with 8% Fe2+ ions - (SF(III)) and ferric chloride (CF(III)). All processes were incubated for 12 days, under similar conditions to the inoculum preparation and with the respective replica and abiotic control.
All the experiments were monitored with measures of pH and redox potential (ORP), using a pH/ORP-meter SCHOTT Handylab. Total and ferrous iron in solution was determined in a spectrophotometer Thermo GENESYS UV 10, employing the method E 394-09, according to ASTM standard. At the end of the experiments, sulphur forms in coal samples were measured by the ASTM D 2492-02 method.
Additionally, the mineralogical composition of the original and treated samples was established by XRD (X-ray diffraction). XRD analysis were made to -200 Tyler mesh coal in a Rigaku Miniflex II X-ray Diffractometer, using a step by step method, with 5° start angle, 70° stop angle, 0.01°step angle, and counting time of 1 s. The minerals present on the coal were quantified by XRD using a Rietveld refinement. All simulations and calculations were carried out in the program X'Pert HighScore Plus©, using the data base PDF2.
3. Results
3.1. Biodesulfurization process
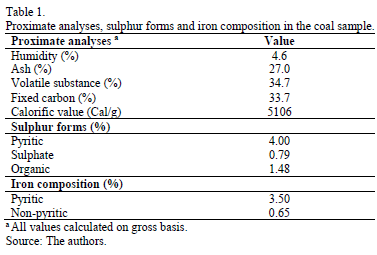
Fig. 1 shows pH behaviour along the process. The initial value was the highest in the ferrous sulphate assays. In contrast, ferric chloride assays CF(III) C2 and CF(III) C3 started at the lowest pH. In the first days of the process, ferrous sulphate assays increased in value, where SF(II) C2 and SF(II) C3 assays reached the maximum (1.71 at day 3). Subsequently, SF(II) C1 assay did not have significant changes; however, SF(II) C2 and SF(II) C3 assays had a continuous pH decreasing up to day 12, especially by using 1250 mg Fe2+/L (1.51).
The assays using ferric salts also raised the pH value on the first day, although it was not as pronounced as the values observed in the ferrous sulphate assays. Later, all the assays had a pH decreasing, which was dependant on the iron source and concentration. CF(III) C3 assay showed the lowest value (1.37).
As regards the abiotic controls, they presented a pH increase on the first day (Fig. 1b) and then did not evidence significant changes during the process, except for SF(III) C3 and CF(III) C3 abiotic controls, whose values decreased after day 1, although they did not reach the same values as their corresponding bioassays.
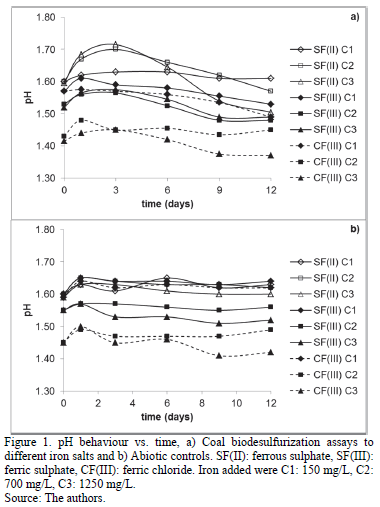
Fig. 2 presents redox potential (Eh). In the same way as the pH values, initial Eh depended on iron source and the initial concentration. Ferrous sulphate assays had the lowest values (below 450 mV). In contrast, the values were over 530 mV in the assays using ferric salts, especially by using ferric chloride. Nonetheless, all the assays had a similar behaviour after day 1. Eh of the assays tended to stabilize between 550-560 mV after day 6 (day 9 for CF(III) C1 assay), except for CF(III) C2 and CF(III) C3 assays, where the value did not surpass 500 mV. On the other hand, all the abiotic controls tended to reach 413 mV after day three.
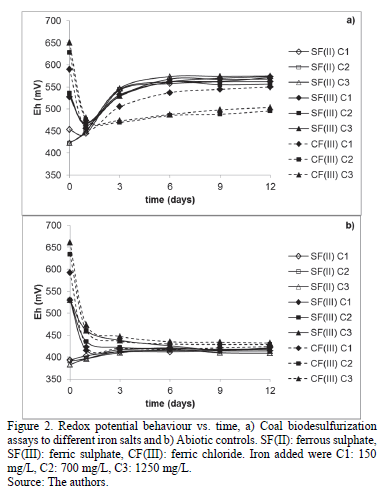
All assays and abiotic controls showed iron dissolution on day 1 (Fig. 3). The quantity removed was around 12% in SF(III) C3, CF(III) C3 assays and abiotic controls and 10% in the other assays. After day 2, the abiotic controls of SF(III) C3 and CF(III) C3 assays reached a maximum removal of around 19%, while the abiotic controls of the other assays were between 12-14% (Fig. 3b). Moreover, SF(II) C1, SF(III) C1 and CF(III) C1 assays reached a removal between 33-35% at the end of the process (Fig. 3a) and SF(III) C2 and SF(III) C3 assays a maximum around 40%. In contrast, CF(III) C2 and CF(III) C3 assays had a removal below 25%. This correlated with the low redox potential observed in Fig. 2.
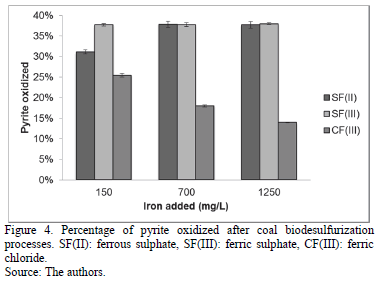
Fig. 4 presents pyrite oxidized at the end of the process. All the assays using ferric sulphate and SF(II) C2 and SF(II) C3 assays reached the maximum of pyrite oxidation (37.80%). In contrast, CF(III) C2 and CF(III) C3 assays obtained the minimum values (18.00% and 14.00%), showing a decreased pattern when the iron concentration increased (similar to observed in the Fig. 2, 3). SF(III) C3 and CF(III) C3 abiotic controls reached a chemical pyrite oxidation of around 9.00%, while the others did not have a significant value (below 3%).
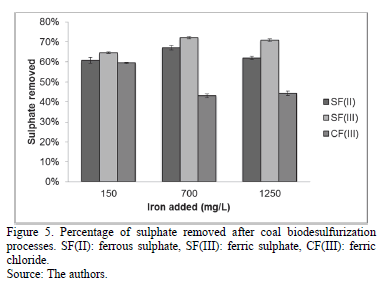
Although the assays using the two highest iron concentrations of ferrous and ferric sulphate reached the maximum of pyrite oxidized, SF(III) C2 and SF(III) C3 assays had the best sulphate removal of around 70.00% (Fig. 5). On the other hand, sulphate removed was low in CF(III) C2 and CF(III) C3 assays, similar to that observed in the parameters shown in Fig. 2, 3.
3.2. X-ray diffractogram analyses
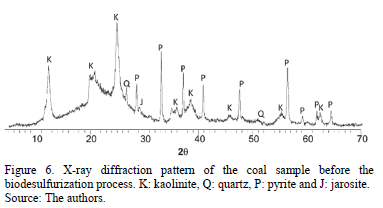
The X-ray diffractogram of non-treated coal (Fig. 6) showed the presence of pyrite, kaolinite, quartz and jarosite. The curvature of baseline between 2q=13°-23° corresponds to the amorphous phase of coal [15].
The Rietveld refinement for the semi-quantification of XRD patterns, showed 7.5% w of pyrite, 28.86% w of kaolinite, 1.69% w of quartz and not significant amounts (below 0.5%) of jarosite. A goodness-of-fit below 1.1% and weighted residual profile below 20% were observed, considered good to accept this method [16,17].
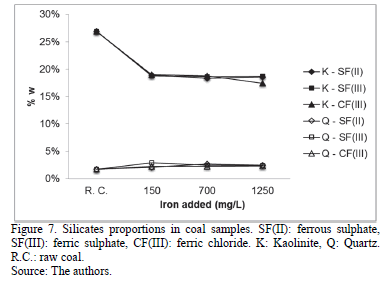
Treated coal samples presented a decrease of kaolinite content (Fig. 7), reporting an average percentage of 18.57% (± 0.45%) between all the assays without a specific correlation with respect to the evaluated variables. Quartz did not show significant changes. As additional data, jarosite content was not significant (less than 0.4% w).
4. Discussion
Ferric salts influenced pH at the beginning of the process. They ionise to Fe3+ and the respective anion (Xn-) in water solution by eq. (3). Then, Fe3+ hydrolyses by eq. (4)-(6) and releases H+ to the medium [18]. This explains why the assays using ferric salts started at lower values than assays using ferric sulphate. Besides, the equilibrium reaction of Cl- and H+ generates a stronger acid than SO42- and H+, influencing low pH.
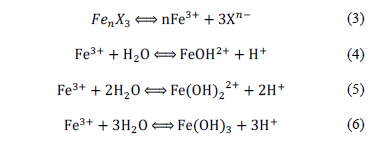
Additionally, the initial redox potential values depended on the concentration of Fe3+ ions, as this parameter is directly related to the Fe3+/Fe2+ ion exchange ratio [4,19-21]. Ferric sulphate assays had a lower Eh than ferric chloride assays, because the reagent contained 8% of Fe2+ ions.
The following sections provide information on how both initial pH and Eh variations affect assays and abiotic controls during the processes for each iron source.
4.1. Sulphate salts effect
The increment of pH on day 1 (Fig. 1) indicated H+ consumption by the A. ferrooxidans to biooxidize Fe2+, as shown in eq. (1). The results obtained agreed with the reaction mechanism, because the assays using the highest concentrations of Fe2+ consumed more H+, which explains why the highest pH values were similar to those reported by other authors [22]. Moreover, Fig. 7 indicates that kaolinite also might have consumed acid in its dissolution by eq. (7) [23,24].
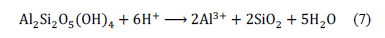
The subsequent pH decrease indicated that the acid production came from pyrite oxidation by eq. (2) and Fe3+ hydroxylation by eq. (4)-(6) [18], which produced more H+ after the lag phase. Therefore, assays using the two highest concentrations have a better pH decreasing, because they had a higher iron concentration in the culture medium. Since the coal did not present common alkalinizing compounds such as carbonates [3,4,10], it did not have a significant participation on pH changes around the process.
The replacement of ferrous sulphate by ferric sulphate allowed the onset of sulphide oxidation to be faster by eq. (2). The extra acidity helped to avoid pH increase and improved iron and sulphate removal in relation to assays using ferrous sulphate (Fig. 3, 5), proving pH value affects solubility. Additionally, iron concentration also affected the process. Early research showed that values above 1200 mg/L induce the generation of precipitates as hydroxisulphates by eq. (8), compounds poorly insoluble at the conditions used [4,18,25,11]. This explains why SF(II) C3 and SF(III) C3 assays decreased iron removal after day 9 (Fig. 3).
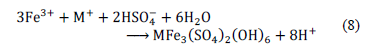
Moreover, the similar pyrite oxidation reached by SF(II) C2, SF(II) C3, SF(III) C2 and SF(III) C3 (around 38%) indicate that Fe2+ concentrations above 800 mg/L produced sufficient Fe3+ to obtain a good Fe3+/Fe2+ ion exchange, regardless of the iron sulphate source. The only advantage of using high ferric sulphate concentrations was to reduce precipitates formation, because it did not affect redox potential behaviour after day 3 (Fig. 2). Ferric sulphate abiotic controls demonstrated that Fe3+ added oxidized pyrite on the first days, especially by using the highest concentration. However, Fig. 2b, 3b showed Fe3+ ions did not regenerate again due to the absence of the microorganism (stabilization of Eh to 413 mV and non-significant iron removal after day 3). This suggests that bacterial activity is the main controller of Fe3+/Fe2+ ratio.
Nevertheless, the assay SF(III) C1 also obtained the best pyrite oxidation, 20% higher than the assay SF(II) C1. It indicated that the biodepyritization process (under the evaluated conditions) might obtain an increase of pyrite oxidized and a sulphate removal (64.51%) by adding 150 mg Fe3+/L.
4.2. Chloride salts effect
Cl- ions had a negative impact on bacterial activity. These findings are similar to previous research which reported that concentrations above 1000 mg/L of Cl- inhibited bacterial growth, reducing the regeneration of Fe3+ ions, and therefore pyrite biooxidation rate [26,27]. 800 mg/L and 1350 mg/L of Fe3+ provided 1520 mg/L and 2565 mg/L of Cl- respectively. Although this work evidenced microorganism activity, results showed the adaptation stages were not sufficient to counterattack all chloride toxicity for these concentrations.
Furthermore, although ferric chloride assays had the lowest pH values (Fig. 1), the extra acidification did not help to increase sulphate removed (actually, it diminished), contrasting with the pH-sulphate solubility relation observed in ferric sulphate assays. The commercial use of ferric chloride as a coagulant in water treatment, especially to remove phosphate [28], is well known,. Ferric chloride probably attacked KH2PO4 from the culture medium and K+ inducted to the formation of precipitates as hydroxisulphates, reacting with the iron and sulphate product of oxidation of pyrite. The possible precipitation of ferric ions and some nutrients in the bacteria (K, P) might have affected sulphate-removed ratio, as observed in Fig. 5.
5. Conclusions
The main contribution to replace ferrous sulphate by ferric sulphate was to support acidity to the media, which helps to improve sulphate removal. Nonetheless, the present work found that replacing the common iron source of the culture medium by ferric sulphate in small quantities (150 mg/L of Fe3+) was a good alternative to increase the pyrite oxidation rate within the same configuration process. However, this efficiency has to be tested in other scenarios, for example using high pulp concentration, because initial Fe3+ ions (from ferric sulphate) only acted as an accelerant and not as a catalyser.
Furthermore, ferric chloride was not efficient as an iron source of Fe3+ ions. Although Fe3+ promoted chemical oxidation of pyrite during the first days, Cl- was toxic to the microorganism and furthermore acted as an inductor of the sulphate precipitates. Both factors considerably affected pyrite biooxidation and sulphate removal ratios.
Moreover, besides pyrite and sulphate removal, all the assays presented kaolinite dissolution by acid leachate. This improved raw coal, because as inorganic matter decreases, calorific capacity increases, obtaining a better quality product. However, this phenomenon only depended on the acidity of the medium and not on the variables assessed. However, although ferric chloride did not work, future studies might evaluate other kinds of ferric salts, in order to determine a positive influence of the anions on pyrite oxidation and sulphate removal ratios.
Acknowledges
To the Biomineralogy and Biohydrometallurgy Laboratory (National University of Colombia) where the investigation was carried out.
References
[1] Juszczak, A., Domka, F., Kozlowski, M. and Wachowska, H., Microbial desulfurization of coal with Thiobacillus ferrooxidans bacteria, Fuel, 74, pp. 725-728, 1995. DOI: 10.1016/0016-2361(94)00019-N [ Links ]
[2] Calkins, W.H., The chemical forms of sulfur in coal: A review, Fuel, 73, pp. 475-84, 1994. DOI: 10.1016/0016-2361(94)90028-0 [ Links ]
[3] Caicedo, G.A. and Márquez, M.A., Selection procedures of consortia bacterial with A. ferrooxidans like bacteria and A. thiooxidans like bacteria in coal biodesulfurization process. Revista Facultad de Ingeniería Universidad de Antioquia [online], 52, 2010. [date of reference April 28th of 2015]. Available at: http://www.scielo.org.co/pdf/rfiua/n52/n52a08.pdf [ Links ]
[4] Cardona, I.C. and Márquez, M.A., Biodesulfurization of two Colombian coals with native microorganisms, Fuel Process Technology, 90, pp. 1099-1106, 2009. DOI: 10.1016/j.fuproc.2009.04.022 [ Links ]
[5] Klein, J., Technological and economic aspects of coal biodesulfurisation. Biodegradation [online], 9, 1998. [date of reference April 28th of 2015]. Available at: http://www.ncbi.nlm.nih.gov/pubmed/10022072 [ Links ]
[6] Cara, J., Vargas, M., Morán, A., Gómez, E., Martínez, O. and García Frutos, F.J., Biodesulphurization of a coal by packed-column leaching. Simultaneous thermogravimetric and mass spectrometric analyses, Fuel, 85, pp. 1756-1762, 2006. DOI: 10.1016/j.fuel.2006.02.014 [ Links ]
[7] Kodali, B., Rao, M.B., Narasu, M.L. and Pogaku, R., Effect of biochemical reactions in enhancement of rate of leaching, Chemical Engineering Science, 59, pp. 5069-5073, 2004. DOI: 10.1016/j.ces.2004.09.004 [ Links ]
[8] Sand, W., Gehrke, T., Jozsa, P.G. and Schippers, A., (Bio) chemistry of bacterial leaching-direct vs. indirect bioleaching, Hydrometallurgy, 59, pp. 159-75, 2001. DOI: 10.1016/S0304-386X(00)00180-8 [ Links ]
[9] Sand, W. and Gehrke, T., Extracellular polymeric substances mediate bioleaching/biocorrosion via interfacial processes involving iron(III) ions and acidophilic bacteria, Research in Microbiology, 157, pp. 49-56, 2006. DOI: 10.1016/j.resmic.2005.07.012 [ Links ]
[10] Caicedo, G.A., Márquez, M.A. and Moreno, C.X., Particle size, iron concentration and pH influence in biodesulfurization process of coal - laboratory tests. Revista Colombiana de Biotecnología [online], 13, 2011. [date of reference April 28th of 2015]. Available at: http://www.scielo.org.co/pdf/biote/v13n2/v13n2a20.pdf [ Links ]
[11] Caicedo, G.A., Prada, M., Peláez, H., Moreno, C.X. and Márquez, M.A., Evaluation of a coal biodesulfurization process (semi-continuous mode) on the pilot plant level. DYNA [online], 79(174), 2012. [date of reference April 28th of 2015]. Available at: http://www.scielo.org.co/pdf/dyna/v79n174/v79n174a15.pdf [ Links ]
[12] Loi, G., Mura, A., Trois, P. and Rossi, G., Bioreactor performance versus solids concentration in coal biodepyritization, Fuel Processing Technology, 40, pp. 251-260, 1994. DOI: 10.1016/0378-3820(94)90147-3 [ Links ]
[13] Nemati, M., Harrison, S.T.L., Hansford, G.S. and Webb, C., Biological oxidation of ferrous sulphate by Thiobacillus ferrooxidans: A review on the kinetic aspects, Biochemical Engineering Journal, 1, pp. 171-190, 1998. DOI: 10.1016/S1369-703X(98)00006-0 [ Links ]
[14] Tuovinen, O.H. and Kelly D.P., Studies on growth of thiobacillus ferrooxidans. I. Use of menbrane filters and ferrous iron agar to determine viable numbers, and comparison with 14CO2-fixation and iron oxidation as measures of growth, Archiv für Mikrobiologie, 88, pp. 285-298, 1973. DOI: 10.1007/BF00409941 [ Links ]
[15] Lu, L., Sahajwalla, V., Kong, C. and Harris D., Quantitative X-ray diffraction analysis and its application to various coals, Carbon, 39, pp. 1821-1833, 2001. DOI: 10.1016/S0008-6223(00)00318-3 [ Links ]
[16] McCusker, L.B., Von Dreele, R.B., Cox, D.E., Loue, D. and Scardi, P., Rietveld refinement guidelines, Journal of Applied Crystalography, 32, pp. 36-50, 1999. DOI: 10.1107/S0021889898009856 [ Links ]
[17] Kniess, C.T., Cardoso de Lima, J. and Prates, P.B., The quantification of crystalline phases in materials: Applications of rietveld method. in Shatokha, V., Ed. Sintering - Methods and Products [online], Intech, 2012, pp. 293-316. [date of reference April 28th of 2015]. Available at: http://goo.gl/NWoIs4 [ Links ]
[18] Daoud, J. and Karamanev, D., Formation of jarosite during Fe2+ oxidation by Acidithiobacillus ferrooxidans, Minerals Engineering, 19, pp. 960-967, 2006. DOI: 10.1016/j.mineng.2005.10.024 [ Links ]
[19] Bhatnagar, A., Dastidar, M.G. and Roychoudhury, P.K., Integrated technique for pyritic sulphur reduction in Indian (ASSAM) coal, Energy Conversion Management, 39, pp. 1121-1126, 1998. DOI: 10.1016/S0196-8904(98)00005-3 [ Links ]
[20] Malik, A., Dastidar, M.G. and Roychoudhury, P., Biodesulphurization of coal: Effect of pulse feeding and leachate recycle, Enzyme Microbial Technolology, 28, pp. 49-56, 2001. DOI: 10.1016/S0141-0229(00)00283-0 [ Links ]
[21] Meruane, G., Oxidación bacteriana de sulfato ferroso, PhD dissertation Thesis, Universidad de Chile, Santiago, Chile, 2002. [ Links ]
[22] Acharya, C., Kar, R.N. and Sukla, L.B., Bacterial removal of sulphur from three different coals. Fuel [online], 80, 2001. [date of reference April 28th of 2015]. Available at: http://goo.gl/QmA9TG [ Links ]
[23] Ford, K.J.R., Leaching of fine and pelletised Natal kaolin using sulphuric acid. Hydrometallurgy [online], 29, 1992. [date of reference April 28th of 2015]. Available at: http://www.sciencedirect.com/science/article/pii/0304386X9290008N [ Links ]
[24] Panda, A.K., Mishra, B.G., Mishra, D.K. and Singh, R.K., Effect of sulphuric acid treatment on the physico-chemical characteristics of kaolin clay, Colloids Surfaces A Physicochemical Engineering Aspects, 363, pp. 98-104, 2010. DOI: 10.1016/j.colsurfa.2010.04.022 [ Links ]
[25] Peláez, H., Prada, M., Caicedo, G.A., Moreno, C.X. y Márquez, M.A., Influencia de la relación inicial de Fe3+/Fe2+, en un proceso de biodesulfurización de carbones en suspensión. Revista Internacional de Contaminación Ambiental [online], 29, 2013. [date of reference April 28th of 2015]. Available at: http://www.journals.unam.mx/index.php/rica/article/view/25160 [ Links ]
[26] Deveci, H., Jordan, M.A., Powell, N. and Alp, I., Effect of salinity and acidity on bioleaching activity of mesophilic and extremely thermophilic bacteria, Transactions of Nonferrous Metals Society of China, 18, pp. 714-721, 2007. DOI: 10.1016/S1003-6326(08)60123-5 [ Links ]
[27] Gahan, C.S., Sundkvist, J.E. and Sandström A., A study on the toxic effects of chloride on the biooxidation efficiency of pyrite, Journal of hazardous materials, 172, pp. 1273-1281, 2009. DOI: 10.1016/j.jhazmat.2009.07.133 [ Links ]
[28] Caravelli, A.H., de Gregorio, C. and Zaritzky, N.E., Effect of operating conditions on the chemical phosphorus removal using ferric chloride by evaluating orthophosphate precipitation and sedimentation of formed precipitates in batch and continuous systems, Chemical Engineering Journal, 209, pp. 469-477, 2012. DOI: 10.1016/j.cej.2012.08.039 [ Links ]
G.A. Caicedo-Pineda, received his BSc. Eng. in Chemical Engineering in 2005, his MSc. degree in Biotechnology in 2008, and his PhD. degree in Engineering - Science and Technology of the Materials in 2015, all of them from the Universidad Nacional de Colombia, Manizales (Bs. Eng.) and Medellin (MSc. and PhD. degrees). From 2006 to 2010, he worked as a researcher in the Universidad Nacional de Colombia, Medellín. From 2011 to the present, he has worked as a Process Engineering Researcher in Biofenix S.A.S. and Inverdesarrollo S.A.S. His research interests include biomining, bioleaching, nanobiotechnology, bioremediation, biological wastewater treatment, green technology and raw materials bioimprovement. ORCID: 0000-0003-2320-0980
M.A. Márquez-Godoy, received his BSc. Eng. in Geological Engineering in 1989, his MSc. degree in Geology in 1995, and his PhD. degree in Geology in 1999. The BSc. Eng. degree was from the Universidad Nacional de Colombia, Medellín and both the MSc. and PhD. degrees from Fundação Universidade de Brasília, Brazil. From 1990 to 1995, he worked as a geologist administrator in Ministerio de Minas y Energía. From 1996 to 1997, he worked as a geologist in Ingeominas. Currently, he works as Full Professor in the Universidad Nacional de Colombia, Medellin. Additionally, he has worked as a professor in EAFIT (1984 to 1986) and the Universidad de Antioquia (2000-2001). His research interests include engineering, microbial biotechnology, mining engineering, biogeochemistry, geomicrobiology, mineral processing, ore deposits, and minerals. ORCID: 0000-0002-7462-2430