1. Introduction
Papaya (Carica papaya L.) is cropped mainly in tropical and subtropical countries, and is very desirable for its pleasant taste and aroma [1]. It is a source of high nutrient content (vitamin A and C) and minerals such as calcium, magnesium and potassium [2]. Papaya fruits are also transformed into various value-added products as follows: purees, juices, jams, jellies, candy, syrup, among others [3,4]. From a biological point of view, papaya is subjected to high post-harvest losses due to rapid senescence which shortens its shelf life [2]. Due to its high perishability, papaya losses during transportation and storage are estimated at least 25% [3]. To minimize these losses, it is necessary to find alternative methods to extend the shelf life while maintaining its quality properties close to those of fresh fruit. Freezing is an alternative in food preservation, to maintain color, flavor and nutritional characteristics[5,6]. However, after freezing-thawing, food present some drastic changes in quality, mainly shows in drip loss (which is due to cell damage) [7,8] and abrupt changes in mechanical properties associated with texture changes (loss of turgor during thawing, resulting in flaccidity and shrinking) [9], among other properties. Irreversible damages occurred during freezing, are caused because a portion of the aqueous fraction freezes forming large ice crystals that destroy the cellular structure of fresh fruits and vegetables [10], therefore cell membranes lose their osmotic state and semi permeability [11].
An alternative technique to reduce quality losses during freezing of fruits and vegetables is partially remove freezable water of the food in order to reduce the water content and limit ice crystal damage in foods [12]; In this sense, the osmodehydrofreezing technique is used as a complement to freezing, which consisting an osmotic dehydration(OD) application for the product followed by the freezing process [11]. OD can partially reduce a freezable water quantity of the product, which consists on the extraction of water from a food product that is immersed in a hypertonic osmotic solution (OS) for a specific time and temperature [13]. The OS must be highly concentrated in osmotic solutes and close to room temperature [8]. According to previous studies, osmodehydrofreezing has been used in various fruits and vegetables with positive results, reducing quality losses once defrosted the product, as in melon [14] apples [8], eggplant [6], kiwis and pears [8]. In papaya, no research evidence is available in the scientific literature where OD and freezing are combined, except the work of Ayala et al. 2014 [15], on that research a concentration of sucrose (65 ° Brix) was used different to those of this work.
The aim of this research was to evaluate the influence of osmotic treatments application on freezing rate, drip loss and changes in mechanical properties in terms of elastic modulus (EM) and the stress of yield limit (σL) in papaya samples under freezing storage for 10, 20, 30 and 60 days.
2. Materials and methods
2.1. Sample preparation
Papaya (variety cultivar Maradol) were selected with an uniform soluble solids content (11.30 ± 0.33°Brix).These were prime-quality samples, following Technical Standard NTC 1270 [16]. The fruits were washed, peeled (using a stainless steel knife), and cut into halves to remove seeds from the center of each fruit piece, samples of cut fruit were 20 mm height and 15 mm diameter cylinders using a stainless steel cylindrical hollow punch.
2.2. Osmotic treatments
Osmotic treatments were carried out in sucrose solution of 45 and 55°Brix. Sucrose was used as solute due to its wide use in the dehydration fruit processing [17]. Concentration levels were selected according to previous research on osmotic dehydration in papaya fruits [18,19]. The weight ratio of osmotic solution for papaya was 20:1 to maintain a constant concentration of the osmotic solution during OD [15,20]. The OS was kept at 26.3±0.3°C and constantly stirred at 800 rpm using a mechanical stirrer (Kika Labor Technik Pol Col, USA) to avoid the crusting phenomenon resulting from the presence of sugar on the samples surface.
To partially reduce the freezable water of fruits, papaya samples were removed in two-time periods of OD (30 and 90 min) in both OS to 44 and 55°Brix to get different moisture contents. The moisture content (MC) of treated and non-treated samples was determined by using the method 934.06 of AOAC (1990) and soluble solids were analyzed measuring the refractive index with a refractometer (Abbe Atago, Zeiss, USA), at 20 ° C.
2.3. Freezing-thawing storage
Non-treated samples (NTS) and osmotically treated samples (OTS) were stored at 8°C in resalable plastic bags before freezing for 12 h, to promote an internal equilibration of concentration [7]. Afterwards, they were frozen at -40°C in a freezer (Revco, USA) and stored in a commercial freezer at -18°C during 10, 20, 30 and 60 days. For each storage time, the samples were removed from the freezer and taken a commercial refrigerator at 8°C for 18 h to ensure complete thawing process [17,18]. Once papaya samples were thawed, physical properties were evaluated (freezing rate, DL and mechanical properties in terms of ME and σL).
2.4. Determination of physical properties
2.4.1. Freezing rate
The freezing rate of samples was determined following eq.1, for this, the time and freezing temperature were recorded using a Dattalogger Thermometer (Sper Scientific, USA.) coupled with a type thermopar K type, inserted into a geometric center of the fruit. With the data obtained from the product temperature and the freezing time, freezing curves were constructed.
Where FR is the freezing rate, T o is the initial temperature of the sample (°C), T f is the final temperature of the sample (ºC) and Δt is the freezing time interval (min).
2.4.2. Drip Loss (DL)
The DL was calculated by the weight differences of the samples before and after the freeze-thawing process [17,19], using an analytical balance (Mettler Toledo AE200) with 0.001g accuracy. The eq. 2 was used to calculated DL.
Where m o is the weight of the sample before freezing (g) and m f is the weight of the sample after thawing (g).
2.4.3. Mechanical properties
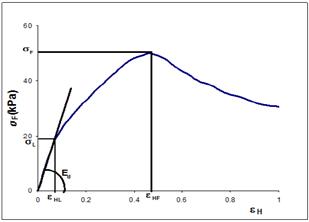
Mechanical properties of OTS and CS were analyzed through uniaxial compression test using a texturometer (model EZ-Test, Shimadzu, Japan), adapted with a 40mm diameter cylindrical plate. The measurements were performed at 27.1 ± 0.2°C, at a speed of 30 mm/min, until the sample was compressed to 90% of its initial height. The force-displacement curves obtained in the compression analysis were used to calculate the failure stress σ(t) and Hencky strain ε H from the eq.3 and eq. 4 respectively [20,21]. The typical curve of uniaxial compression is shown in Fig. 1.
Where σ(t) is the failure stress (true stress) (Pa), F is the compression force (N), A is the cross sectional contact area (m2), εH is the Hencky strain at failure (mm/mm),  is the initial sample length (mm) and d is the sample length of the sample at a time t compression (mm).
is the initial sample length (mm) and d is the sample length of the sample at a time t compression (mm).
The parameters of stress of yield limit ((L) and elastic modulus (EM) were analyzed in Fig. 1. The (L was determined at the end point of the linear relationship between stress and strain, the EM was obtained from the slope of the linear region before the yield point [21]. Therefore, the samples showed no macroscopic point of fracture, the failure stress ((F), wich is defined as the ultimate stress before the material fracture [12], could not be determined.
3. Experimental design
A factorial design 2x2x4 was applied with three factors: Concentration of OS (45 and 55°Brix), immersion time in OS (30 and 90 min) and freezing storage time (10, 20, 40 and 60 days). All of the measurements were performed three times. The results were analyzed using a 95% confidence level variance analysis using the Minitab 15.1 Version (Minitab, Inc., State College, Pennsylvania, 2009).
4. Results
4.1. Moisture content and soluble solids
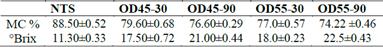
Initial moisture content (% wet basis) and °Brix of treated and non-treated samples are shown in Table 1. The osmodehydrated
Table 1 Average values of moisture content for papaya in fresh and osmotically dehydrated at 45 and 55ºBrix.
Source: The authors
samples reached moisture levels from 79.6 to 74.2% (wet basis) with a gain of solids of 17.5 to 22.5 °Brix. Treatments with higher concentration of osmotic solution (55°Brix) showed lower moisture content than those treated at 45°Brix for the same dehydration time. This is because the OS with higher concentration has greater driving force or greater osmotic pressure gradient between the solution of the environment and food, which had achieved an acceleration of the dehydration kinetics [26].
4.2. Freezing curve
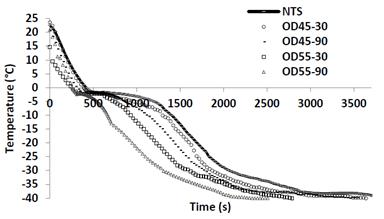
The freezing curves of the OTS and the NTS samples are shown in Fig. 2. In all treatments the phenomenon of sub cooling typical in food was present (water remains liquid below its freezing point). Ice formation (initial freezing temperature) in the freezing of fresh fruit began at about -1.5°C, while in the osmofrozen samples were lower ranging from -1.71 to 2.43°C (DO45-30 = -1.71 ° C; DO45-90 = -1.92; DO55-30 =-2.0 and DO55-90 = -2.43°C). Similar behavior was presented in yellow pitahaya [27]. The beginning of ice formation is associated with the removal of the latent heat of freezing. The lowest freezing temperatures in OTS are attributed to the solutes concentration (freezing-point depression). Freezing temperatures are reported in some fruits like apple (-2.20 ± 0.38°C to 12.28°Brix) in pear (-1.96 ± 0.24ºC for 9.55ºBrix) and in grape (-3.37 ± 0.35°C to 16.7°Brix) [28]. The freezing rate on fresh samples were slower (0.92°C/min) as compared to the osmotically treated (OD45-30=1.02°C/min;OD45-90=1.05°C/min; OD55-30=1, 21°C/min and OD55-90=1.45°C/min). These freezing rate are considered fast [29], which prevents the formation of large ice crystals within the structure of the food and aid in reducing mechanical damages to the samples.
4.3. Drip loss
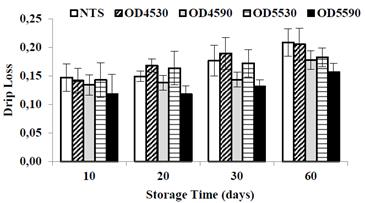
Dripp loss (DL), also called liquid phase loss. The DL during the freeze-thaw of papaya samples are shown in Fig. 3. The DL increased significantly in all treatments (p<0.05) during the frozen storage time. Similar behavior was observed in papaya with osmotic pretreatment at 65°Brix [15], and in other fruits like apple and pear [8], melon [14] and strawberry [7]. It is possible that with increasing storage time in freezing a change arises in the size, number and shape of ice crystals (ice recrystallization), causing the output of the liquid phase or loss of the capacity for water retention of the fruit cell [26,27].
It is noted that the OTS treatments except OD45-30, had significantly (p<0.05) lower losses of DL as compared to the NTS. This behavior can be attributed to the lower amount of freezable water in fruit due to the application of OD, reducing the ice recrystallization process during frozen storage, and consequently presenting minor structural damages to the fruit cell matrix. A similar trend has been reported in Kiwi [12] and melon [14]. Otherwise, it can be said that since OTS shows a higher freezing rate, they make smaller crystal sizes within the cellular matrix, reducing structural damages and consequently less loss of liquid phase (drip loss) [9]. This result is an indicator of the cryoprotectant effect of osmotic treatment, previous to the papaya freezing process.
By comparing the drip loss between osmotic treatments, it is noted at lower moisture level, the samples presented lower losses of liquid phase (p<0.05), being the treatment DO5590 with the lowest moisture level (74.22 ± 0.43%, wet basis) which showed the lowest DL each time of frozen storage. This result corroborates the lowest freezable water, a decreasing ice recrystallization processing, and a decreasing structural s damage. Similar results were reported in papaya treated at 65°Brix [15] and Kiwi [23]. By comparing the osmotic solution concentration level, it is clear that for the same dehydration time, the dehydrated treatments at 55°Brix showed lower DL as compared to those of 45ºBrix, due to the lower moisture content reached in dehydrated samples.
4.4. Mechanical properties
A typical force-deformation curve (obtained from compression test) for treated and non-treated papaya samples are shown in Fig. 4. A typical fracture curve was not shown as described in Fig. 1. Curves were presented without showing a failure stress point (macroscopic break). This behavior means that the matrix of the papaya is flaccid, is not rigid as to present a breakpoint or macroscopic rupture [7], similar behavior has been observed in strawberries and kiwis [32].
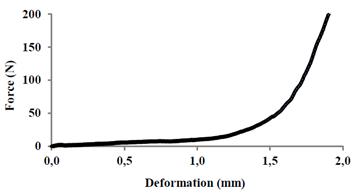
Source: The authors
Figure 4 Typical curve of stress and strain by uniaxial compression test in papaya.
For this reason, the mechanical properties of elastic modulus (EM) and stress of yield limit (σL), were analyzed, which are also suitable to characterize mechanical or viscoelastic food properties, which are associated with food hardness. The EM is associated with the feeling of hardness or resistance when a food is chewed [33]. The σL is associated to the linear elastic behavior change (solid) to nonlinear elastic or plastic (liquid), thus, a greater σL means a stronger or harder behavior [34].
4.5. Elastic modulus and stress of yield limit
Changes in elastic modulus of the different papaya treatments are shown in Fig. 5. It is noted that the EM decreases significantly (p<0.05) in all treatments during frozen storage, possibly due to structural damages to the matrix cell by the formation of ice crystals during storage [35]; however, when comparing the OTS and NTS, it is observed that the ones treated showed significantly (p <0.05) higher EM (harder), this behavior can be attributed to minor structural damages to contain less freezable water. Similar results have been investigated in mango [7] apple [8] and in tomato [36].
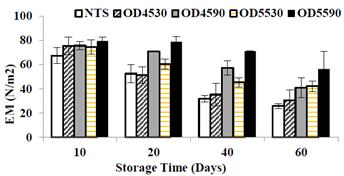
Source: The authors
Figure 5 Evolution of the elastic modulus in osmotically treated and untreated papaya samples during frozen storage.
When comparing the values of EM between osmotically treated samples, it is seen that the dehydrated samples performed the longest (90 min) for both treatments at 45 and at 55°Brix showed higher value of the elastic modulus. This behavior may be associated with lower moisture content reached. It is also noted that the samples treated at higher concentration (55°Brix) showed a higher elastic modulus than those of the less concentration (45°Brix) when compared to the same time of OD, also due to the lower moisture content and greater °Brix reached .
Therefore, the treatment with lower water level (DO55-90 with 74.22%±0.46) and higher content of soluble solids (22.5±0.43°Brix) was the one that presented the highest values of EM with 79.47±3.10,78.88±4.32, 70.97 0.22 and 56.40±14.43 43 N/m2 at 10, 20, 40 and 60 days respectively frozen storage. Similar behavior was observed at the osmo-freezing of papaya [37]. These results indicate that when papaya with less freezable water is frozen, the structural changes are affected to a lesser extent in the cell matrix, possibly because of presenting lower re-crystallization of ice [38]. According to [33], by reducing the moisture content of food in osmotic dehydration, the product reaches higher hardness in the osmo-freezing, due to the higher gain of solids and increased water loss. Fig. 6 shows the changes of the stress of yield limited in the different papaya treatments during frozen storage.
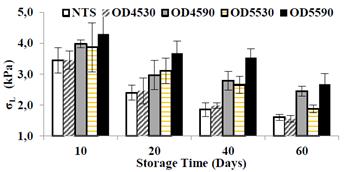
Source: The authors
Figure 6 Evolution of the stress of yield limit in osmotically treated and non-treated papaya samples during frozen storage.
Behavior of σL was similar to the modulus of elasticity above mentioned, it was also observed that σL decreased significantly (p <0.05) in all treatments during under freezing storage, which is an indicator of loss of hardness or softening of fruit by the effect of freezing-thawing process. This behavior is also due to cell damage caused by the ice recrystallization which weakening the mechanical strength of the fruit. However, the OTS showed significantly (p<0.05) higher values of σL as compared to NTS, this result indicates the positive effect of reducing the freezable water by the process of the OD to reach higher hardness or more solid fruit behavior. In fact, among samples osmotically treated it was also observed that treatment DO55-90 showed the highest value σL during storage, due to the less amount of freezable water.
5. Conclusions
The application of osmotic pretreatments followed by freezing process (osmo-freezing) had a cryoprotectant effect on papaya samples in terms of mechanical properties and drip loss. This provides more accurate and reliable estimates of papaya samples with lower content of freezable water and higher content of soluble solids have higher freezing rates, lower drip loss and higher hardness (firmness) than the non-treated samples during freezing storage, a possible explanation for this is due to less recrystallization ice. The freezing storage time influenced significantly (p<0.05) in the loss of papaya quality, are thought to contribute to ice recrystallization during storage, causing loss of cell contents. These results demonstrate that the osmo-freezing technique is effective in reducing quality loss in papaya samples during freezing storage.