1. Introduction
Currently, there is a great interest on the lignocellulosic biofuels industry [1]. To achieve large-scale biotechnological production of ethanol at low costs that is environmentally safe, it is necessary to search abundant raw materials that do not compete against food security policies of populations and to not push the natural boundaries of primary and secondary forests. The raw materials should be easy to use, store, distribute, and transport and be attractive for use within the energy industry. Some lignocellulosic materials, such as sugarcane, corn, rice, beer, sweet sorghum, and wheat residue, fulfill these requirements [2]. Sugarcane is the most widely cultivated crop in subtropical countries, such as Colombia. Worldwide, the total area planted with sugarcane is 26 million hectares, producing 1.83 billion tons. The main product is sugar (92 %), followed by ethanol and animal feed [3].
Lignocellulosic residues contain 55(75 % dry matter basis (db) fermentable sugars. Besides, they are an emerging valuable resource for the generation of hemicellulose-based materials and products [4]. In biomass, the hemicellulose is joined to cellulose microfibrils through molecular interactions including hydrogen and van der Waals bonds. Also, hemicellulose has other functions, such as cell signaling and substance reserve. Hemicellulose molecules are branched polymers of low molecular weight with a degree of polymerization of 80-200. Hemicellulose isolation involves alkaline hydrolysis of the ester linkages to liberate them from the lignocellulose matrix [5]. Xylans and β-(1→3) and -(1→4) glucans are the principal components of hemicellulose in monocotyledonous plants like sugarcane. These components of hemicellulose comprise 15-20 % (db) of the cell wall [6]. A commercial process to hydrolyze these polysaccharides remains to be found. The likely cause is the strong ether and ester linkages that are between the lignin and the hemicellulose and the amount of hydrogen bonds within polysaccharides [7].
In Colombia, the highest percentage of agricultural residue (21.6 %) comes from sugarcane crops [8], where 54.9 % belongs to tops and leaves (around 9 million tons/year) with no specific use since it is burned or left in the field to decompose. Ethanol and different bioproducts can be obtained from this residue through the biorefinery concept. In order to use this residue in bioethanol manufacturing, before it is important to find an effective isolating method of the most abundant polysaccharides (cellulose and hemicellulose) that facilitates the enzymatic hydrolysis process to obtain fermentable sugars to biofuel because the direct hemicellulose combustion (heating value ≈13.6 MJ/kg) represents an inefficient use of the feedstock [9]. There are very few studies about hemicellulose characterization and extraction, specifically regarding top and leaf residue where molecular structures are not well known. However, there is some research that investigates hemicellulose characterization of sugarcane bagasse [4,10,11]. The goal of this study was to find an efficient method to extract hemicellulose selectively with the highest purity from residue of three sugarcane varieties (CC 8475, CC 8592, and V 7151), which are mainly harvested in the valley of the Cauca River. This research is important with respect to future investigations of ethanol production using this residue.
2. Materials and methods
2.1. Samples
Crop residue (tops and leaves) of the sugarcane varieties CC 8475, CC 8592 (CC: Cenicaña-Colombia) and V 7151 (V: Venezuela) was obtained from several dried agro-ecological zones (valley of Cauca River, Colombia). In this zone, the mean temperature is 23.6 °C and relative humidity is 73 % [8]. Residue was mechanically green-harvested and collected directly from each row (2 m distance between rows) from several commercial sugarcane fields (350 m × 100 m).
The residue of CC 8592 and V 7151 varieties was collected a few days after harvest, while the CC 8475 variety was collected after 30 days. All material of the same variety was first mechanically homogenized and then washed with distilled water (30 °C) to remove dirt and impurities. It was then dried to 45 °C to constant weight in an oven, milled, and sieved. In order to extract the hemicellulose, the residue was ground to a 0.8 mm size according to methods of Sluiter et al. [12].
2.2. Selection of methods for hemicellulose extraction
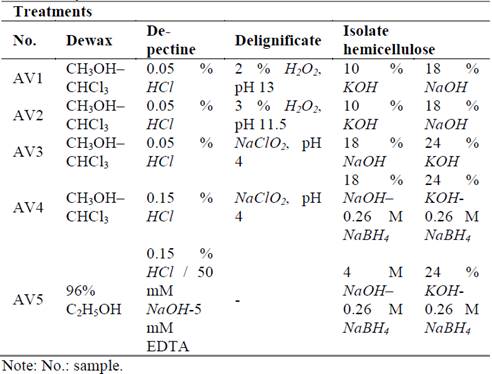
In this study, the hemicellulose was extracted and gravimetrically quantified from harvested sugarcane residue (CC 8592, CC 8475, and V 7151 varieties). Several sequential methods exist to obtain these materials in alkaline media (after removing soluble substances, pectin, and lignin) [13], but still it is not defined an efficient protocol for the isolation of pure hemicelluloses particularly from sugarcane residues; different yields and purities depend on each raw material [14]. For this reason, it was necessary to test different methodologies in relation to the most efficient methods found in the literature (Table 1) in order to select the best, starting with CC 8592 (the most cultivated in the Cauca River valley). Treatments were replicated twice.
Table 1 Different treatments used to extract hemicellulose from residue of the CC 8592 variety.
Source: The authors.
To extract mostly pectin, all samples were first dewaxed with methanol-chloroform or ethanol, and then treated with dilute acid. For lignin removal, alkaline peroxide was used based on reports of Sun et al. [10], who observed that this substance was a strong, effective agent for both hemicellulose delignification and solubilization of straws and grasses. For this reason, the assays were made at two pH levels (13 and 11.5). Also, it is important to ensure an adequate concentration of (HO.) and (O2-.) radicals, which are responsible for the delignification process [15], thus, two different concentrations (AV1 and AV2 treatments) were tested.
The oxidation with NaClO2 was considered (assays AV3 and AV5) because it acts specifically on the phenolic ring and produces changes in the phenolic nucleus of the lignin subunits and does not remove polysaccharides until at least 60 % delignification is achieved [16]. It also produces chlorine gas as an oxidizing agent, which is more stable than H2O2. A solution of NaBH4 was used in the AV5 assay, as it protects the hemicelluloses from depolymerization [17]. According to the research of Xiao et al. [14], there was no delignification in the AV4 assay when observing that only NaOH treatment was necessary for lignin and hemicellulose solubilization in alkaline media. After selecting the best hemicellulose extraction method with the CC 8592 variety, the process was replicated using the other varieties (CC 8475 and V 7151).
2.2.1. Extraction of liposolubles
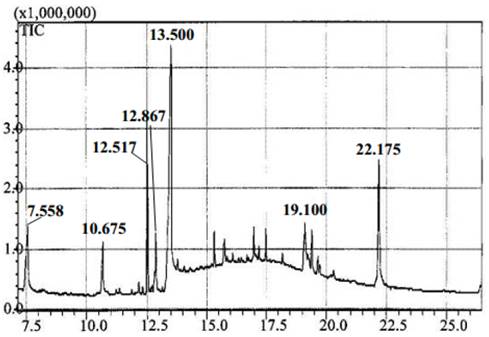
Two methods were used to clean or dewax materials. One employed chloroform-methanol (AV1, AV2, AV3, and AV4), and the other used 96 % ethanol (AV5). Pigments, waxes, and chlorophyll were analyzed by gas chromatography-mass spectrometry (GC-MS) in the supernatant that was extracted from each treatment. The GC-MS analysis was performed on a Shimadzu (Model QP 2010, Kyoto, Japan) system with a Restek XTI-5 capillary column (dimension: 30 m, ID: 0.25 mm, film thickness: 0.25 mm) programmed with a temperature at 240 °C with a 10 °C rise/min. The carrier gas was helium (99.999 %) at a constant flow of 1 mL/min. Electron impact mass spectra were taken at 70 eV with a scan speed of 0.5 scan/s from m/z 2(1024 amu. An ATAGO ABBE refractometer (Model NAR-3T, Washington, U.S.A.) was used to analyze °Brix in the supernatant obtained after dewaxing with 96 % ethanol.
In chloroform-methanol dewaxing, an amount of 10 g of sugarcane residue (CC 8592 variety) was mixed with 60 mL/g of chloroform-methanol (2:1 v/v) in a Pyrex Soxhlet extraction apparatus (Corning Incorporated,New York, U.S.A.) at 65 °C [16] to obtain total removal of cell wall liposoluble compounds (waxes, pigments, and chlorophyll). This process lasted 2(6 h until the solution became colorless. The insoluble residue was collected by filtration, washed with distilled water, and dried at 40 °C. The supernatant was obtained as mentioned previously in section 2.2.1 and analyzed by GC-MS and for °Brix.
In ethanol dewaxing, sugarcane residue was mixed with 96 % ethanol (5 mL/g residue) at boiling temperature with constant agitation for different periods of time (5, 10, or 15 min). Each sample was then filtered and washed with 80 % ethanol until the solution became colorless [17]. The insoluble residue was dried at 40 °C. As previously explained, the supernatant was analyzed by GC-MS and for °Brix, but the ethanol was first evaporated on a rotary evaporator and the volume adjusted to 500 mL with distilled water.
2.2.2. Pectin extraction
After the liposolubles were extracted, two treatments were applied in order to dissolve the protein, soluble lignin, and pectin present in the dewaxed residue in acidic or alkaline media. In pectin extraction in acidic medium the soluble pectin was extracted in acidic media with a Soxhlet apparatus. Amounts of 0.05 % or 0.05 M hydrochloric acid (HCl) (250 mL/g of dewaxed sample) were used and recirculated with a peristaltic pump at room temperature for 2 h according to the modified protocol of Höije et al. [18].
In pectin extraction in alkaline media, an EDTA (Ethylenediaminetetraacetic acid) solution was used in diluted alkaline media to selectively extract the pectin according to the protocol of Flórez [17]. An amount (400 mL) of 50 mM NaOH with 5 mM EDTA was added to the dewaxed sample. The solution was then mixed with constant agitation for 1 h at 4 °C. The process was repeated by adding a new solution of 50 mM NaOH and 5 mM EDTA.
2.2.3. Delignification of sample
Dewaxed residue without liposolubles or pectins was delignified in order to ensure removal of most of the lignin. Two extraction methods were used for this purpose. The first used H2O2 in alkaline media, and the second used NaClO2 in acidic media.
In the first method, hydrogen peroxide was used as a mild agent to remove the lignin in the alkaline media. It is generally accepted that in this condition, it is an unstable molecule that produces hydroxyl and superoxide anion radicals. These radicals are thought to cause lignin structure oxidation, which leads to its dissolution. For this purpose, a modified method of Sun et al. [5] was used. The dried material was extracted with 3 % H2O2 to pH 11.5 or 2 % H2O2 to pH 13 (200 mL) in a Soxhlet apparatus, recirculating the extractant with a peristaltic pump. The pH was adjusted with 18 % NaOH. The treatment was made at 75 °C for 12 h to allow major disruption between lignin-hemicellulose bonds.
In the second method, a solution of NaClO2 at pH 4 (3.52 g/10 g dried material in 200 mL of solution) adjusted with glacial acetic acid was used at 75 °C for 2 h and constant agitation. The phases were separated by filtration and the solid dried to constant weight at 45 °C. The NaClO2 lignin was defined as the difference between the initial and final weights [18].
2.2.4. Alkaline extraction of hemicellulose
Solutions of NaOH and KOH were used for hemicellulose extraction [19]. The treatment in alkaline media causes cell wall swelling and hydrogen bond disruption between cellulose and hemicellulose. It also can disrupt ether bonds among lignin and hemicellulose, and to break ester linkages between hydroxycinnamic acid and hemicellulose [20]. The dried material was sequentially extracted with both alkaline substances, as shown in Table 1. The NaBH4 was added to AV4 and AV5 treatments with the goal of avoiding hemicellulose depolymerization in alkaline media by peeling reactions from the reducing end groups [21] and facilitating easy release from cell wall [17,18]. Therefore, 33 mL of 10 %- or 24 %-KOH solutions with or without 0.26 M NaBH4 were added to 1 g of dried material and mixed for 18 h. In addition, an alkaline solution of 4 M NaOH (50 mL/g dried material) with 0.26 M NaBH4 for 8 h, or 18 % NaOH (45 mL/g dried material) with or without 0.26 M NaBH4 for 18 h at room temperature was used [18]. Two different concentrations were tested because an increase in the concentration of alkalis can result in a major release of hemicellulose compactly embedded in the cell wall due to its propensity to reduce the molecular size of hemicellulosic polymers and disrupt the bonds between carbohydrates and lignin [22].
2.3. Purification and precipitation of hemicellulose
The hemicellulose was first concentrated by vacuum evaporation and then precipitated with ethanol. Therefore, each alkaline extract that was obtained (described in section 2.2.4) was slowly acidified to 5.5(6.5 pH with 6 M HCl to stop any further hydrolysis [18]. Each extract was then concentrated by rotary evaporation at 45 °C to obtain a volume ranging from 100(300 mL. At this time, all hemicellulose parts were precipitated and filtrated. Pellets of hemicellulose were washed with 0.05 % HCl and dried to constant weight at 40 °C. Furthermore, the humidity was analyzed by the NREL/TP-510-42621.34 protocol [12]. The pellets of hemicellulose were macerated and stored in test tubes with a screw cap at 4 °C for further assays.
Supernatant solutions were further concentrated by rotary evaporation at 45 °C to obtain approximately 30 mL and were mixed with 3(4 volumes of 96 % ethanol to precipitate the remaining hemicellulose. The process was repeated to obtain colorless ethanol and avoid the presence of lignin in the precipitate. Subsequently, the precipitate was re-dissolved by heating in deionized water and dialyzed for three days (Spectra/Por 2 membrane, MWCO: 12-14,000, Spectrum Laboratories, Inc., Houston, U.S.A.) to remove dissolved salts [18]. The remaining solution was dried to a constant weight at 40 °C. Humidity was then analyzed via the NREL/TP-510-42621.34 protocol [12]. The hemicellulose pellets were macerated and stored in test tubes with a screw cap at 4 °C for further assays.
3. Results and discussion
3.1. Selection of hemicellulose extraction method
3.1.1. Extraction of liposolubles, pectin, and lignin in the CC 8592 variety
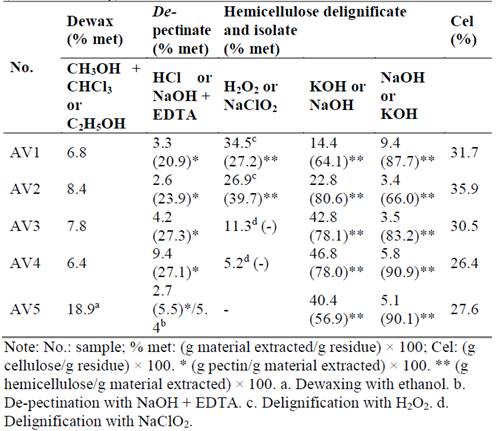
Hemicellulose extraction of CC 8592 sugarcane residue was made by five different treatments. The main objective was to solubilize liposolubles, pectin, and lignin compounds in order to find the treatment that selectively isolated the hemicellulose in alkaline media with the most purity and quantity with the introduction of a protector agent in two of the treatments, as shown in Table 2. Comparison between different treatments is necessary because the yield and composition of hemicellulose extracted depend on the separation method used due to the complex structure of different polysaccharides. Experimental error was <2 % in all assays.
Table 2 Percentages of dry material (w/w) and hemicellulose extracted per treatment (CC 8592 variety).
Source: The authors.
Based on GC-MS analysis (Fig. 1), which was done using the soluble fraction of dewaxed material, chloroform-methanol was found to be the most selective method to preferentially extract the waxes and pigments from sugarcane residues (waxes are normally deposited over the cell wall and are produced by plants to protect themselves from the effects of high and low temperatures, insect attack, and water loss [23]). It was found the light components of this fraction was mainly fatty acids including from the octanoic acid, the same as other autors [24], while treatment with ethanol dissolved liposolubles as well as sugars, since °Brix values increased from 0.7 in 5 min to 3.6 in 15 min. It is also probable that ethanol could have extracted other different compounds since it removed 18.9 % (dry matter) of the residue in 5 min (AV5, Table 2), whereas chloroform-methanol only removed 6.4(8.4 % of this material (AV1, AV2, AV3, and AV4).
In each extraction treatment, approximately 2.6(4.2 % of material (AV1, AV2, and AV3, Table 2) with 0.05 % HCl at room temperature was solubilized. According to FTIR analysis (data not shown), 20.9(27.3 % of this material corresponded to pectic substances. Also, during the acid-treatment process, the fiber was more susceptible to the posterior treatments by cellulose swelling [19] and/or some acid-labile linkages like arabinofuranosyl residue could be hydrolyzed [25]. These hemicellulosic polysaccharides are neutral side chains that attach to the rhamnose units in a galacturonan backbone (homogalacturonan or so-called smooth region) [26]. These extractions in acidic media depend on the HCl concentration.
As a result, when the concentration increased to 0.15 % HCl, the extraction of these compounds incremented more than two-fold (AV4, Table 2). On the other hand, as shown in Table 2, the material extraction using 0.15 % HCl at room temperature in treatment AV5 was minor perhaps because the ethanol in the prior treatment of dewaxing solubilized a part of the soluble compounds in this acid. Also, in this treatment, the chelating agent (EDTA) probably enhanced the solubilization of non-esterified pectins by complexing calcium and magnesium ions from their free carboxylic acid groups, while esterified pectic compounds and phenolic acids were hydrolyzed by diluted alkalis (50 mM NaOH) [27]. In AV5 the delignification was achieved in the posterior treatment of alkalis in conjunction with hemicellulose extraction.
It was observed that treatment AV1 extracted 28.3 % more biomass than the AV2 treatment (Table 2). This difference in quantity of biomass is probably related to the lignin and hemicellulose extracted. A smaller quantity of hemicellulose was solubilized in the treatment at pH 13 and 2 % H2O2 (27.2 %) compared to the treatment at pH 11.5 and 3 % H2O2 (39.7 %). Two factors could influence these results: the peroxide concentration and the pH. If there is a large concentration of this oxidant in the solution, the reaction of the decomposition is more complete, and more hydroperoxy anions (HOO-) are produced, which react with more peroxide to produce hydroxyl radicals (HO.) and superoxide anions (O2-.) [28]. The hydroperoxyl and hydroxyl radicals are thought to cause the oxidation of lignin structures, which leads to the introduction of hydrophilic (carboxyl) groups, cleavage of some inter-unit bonds, and eventually, lignin and hemicelluloses dissolution [10]. Therefore, 3 % H2O2 could promote major breakage between lignin-hemicellulose linkages, favoring a major solubilization of the hemicellulose at this stage (Table 2). On the other hand, the extraction with the alkaline solution at a pH higher than 13 (AV1 treatment) could produce major breaks between ester linkages of the p-hydroxycinnamic acids and ether linkages between lignin and hemicellulose, which could promote a higher dissolution of the lignin [20]. Also, the cellulose could be solubilized due to the higher concentration of the peroxide radicals produced [10]. It was observed that increasing the temperature from 25 to 75 °C increased the delignification rate, which may be due to an elevated active radical production, resulting in increased breakage between lignin-hemicellulose linkages [28]. The yield of lignin dissolution did not increase beyond 12 h of treatment.
Assays of delignification with chlorite (AV3 and AV4 treatments, Table 2) extracted 11.3 and 5.16 % of the biomass, respectively. If this biomass is added to the one extracted by HCl in different concentrations, there is no significant difference between the total biomass extracted in the different treatments (around 15 %). As shown in Table 2, the material solubilized by chlorite was less than that extracted with peroxide. However, it was observed that this material corresponded to almost all lignin, since hemicellulose was not found. The lignin solubilized by chlorite in the treatments was low, but favored in the subsequent hemicellulose extraction treatment with 18 % NaOH and 24 % KOH, besides the quality of easily-removed hemicellulose was not affected. The effectiveness of chlorite to delignify was corroborated by Gómez [29], who found that NaClO2 removed 90 % of lignin of the same CC 8582 variety of sugarcane residue, whereas 57 % was removed with the peroxide. Similar results were found by Kumar et al. [30] using poplar and switchgrass, who reported removal of more than 90 % lignin in both species. However, the authors noted that selectivity for lignin and carbohydrate extraction with NaClO2-acetic acid and peracetic acid was substrate- and pretreatment-dependent. The ability to remove lignin from this extractant may be because in NaClO2 solution, the free functional groups in lignin are progressively oxidized to carboxylic acid or CO2, and the ester and ether bonds between lignin and cellulose and other polysaccharides are cleaved [31].
For the above reasons and perhaps because it is the established and most popular laboratory method for lignin removal from biomass [32], the delignification utilizing acid-NaClO2 was selected to delignify the other varieties of sugarcane residue (CC 8475 and V 7151).
3.2. Hemicellulose isolation in the CC 8592 variety
Fig. 2 shows the total material extracted from dewaxed and de-pectinated sugarcane residue for both delignification and alkaline media extraction methods (18 % or 4 M NaOH, 10 % or 24 % KOH) (light gray bar). The total material varied from 50.9(58.3 % and referred to lignin and hemicellulose mixtures, which depended on the method used. Specifically, as shown in the same figure of the total material extracted by the AV1 treatment, only 47.3 % corresponded to the hemicellulose fraction, while with the AV4 treatment the fraction was 72.4 %. This difference shows that NaClO2 was more selective in extracting lignin than hemicellulose, as shown in Table 3. Hemicellulose was completely solubilized in the last treatments by NaOH and KOH (AV3, AV4, and AV5). In these cases, NaOH extracted more hemicellulose than KOH. On the contrary, in addition to lignin, peroxide extracted around 34 % of hemicellulose (AV1 and AV2, Table 3).
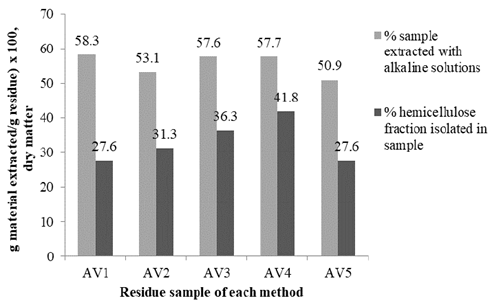
Source: The authors.
Figure 2 Total material and hemicellulose extracted through different treatments in the CC 8592 variety.
When comparing the extracting power of aqueous alkalis, NaOH is slightly more effective than KOH for the removal of hemicellulose, corresponding to the increasing alkali strength from KOH to NaOH [33]. In general, these hemicellulose molecules were not extracted with total purity. Lignin and/or cellulose were detected in many fractions. For this reason, depending on the extraction method used, the quantity of hemicellulose obtained varied from 27.6(41.5 %.
With respect to the AV4 treatment, it was observed that 41.8 % of hemicellulose was recovered (Fig. 2). It is possible that NaBH4 in this treatment acted as a protective agent in the alkaline media to minimize degradation of the reducing end groups in hemicellulose, avoiding its depolymerization [21], obtaining more intact structures, and resulting in greater precipitation. Fig. 3 shows that following treatment with NaClO2, the hemicellulose obtained by concentration in the rotary evaporator (samples A-2, A-3, and A-4) and ethanol (samples B-2, B-3, and B-4) remained whiter in color than those extracted by peroxide and without delignification. In these samples, the pigmentation is probably due to the presence of lignin associated with hemicellulose through ether linkages [34, 35].
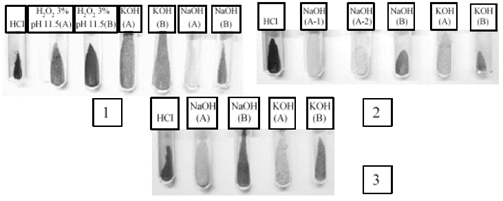
Source: The authors.
Figure 3 Hemicellulose from treatments of delignification with peroxide (1), chlorite (2), and NaOH in EDTA (3). The hemicelluloses in A were obtained by precipitation with a rotary evaporator concentration process. The hemicelluloses in B were obtained by precipitation with ethanol.
3.3. Fourier transform infrared spectra
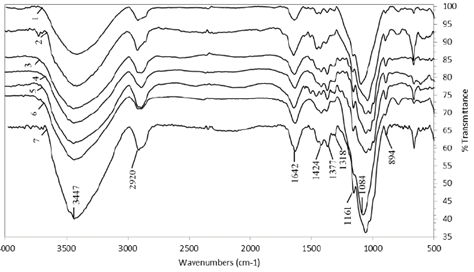
The FTIR spectra of the hemicellulose solubilized during different treatments with peroxide, chlorite, and NaOH-EDTA are shown in Figs. 4 and 5. In general, these spectra show the characteristics of lignin and hemicellulose bands for all the fractions isolated. As seen in Fig. 4, the prominent band around 3400 cm-1 represents the hydroxyl stretching vibrations (OH-1) of the hemicellulose and water involved in hydrogen bonding [10]. The C-H stretching vibrations of the methyl and methylene groups give a signal between 2919(2920 cm-1, while bending bands were found up to 1377(1378 cm-1[36]. This signal was more intense in the hemicellulose precipitated with ethanol. The bands at 1639(1642 cm-1 are indicative of the bending mode of the absorbed water, since holocellulose usually has a strong affinity for this compound. Also, in the solid state, these macromolecules may have disordered structures that can be easily hydrated [33].
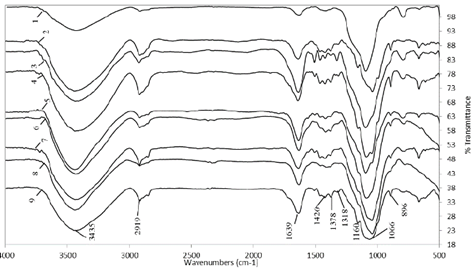
Source: The authors.
Figure 4 Hemicellulose extracted with NaOH. AV1, 2% NaOH, pH 13 (1); AV1, NaOH (2); AV2, 3% pH 11.5 (3); AV2, NaOH (4); AV3 (5); AV4 (6); AV5 (7); CC 8475 (8); and V 7151 (9).
A small band from 1720(1745 cm-1 in the region of carbonyl stretching is due to the acetyl, uronic, and ferulic ester groups of the polysaccharides. The absence of this signal in all sprectra in this region demonstrated that alkali treatment under the conditions used completely cleaved the ester bonds from the hemicellulose and did not significantly attack or oxidize the glycosidic linkages and hydroxyl groups among them. An intensive band near at 1420 cm-1 corresponds to the C=O stretch of carboxylic anions (salt) for uronic acids in hemicellulose [28]. The occurrence of a rather weak band range in all spectra from 1500(1523 cm-1 suggests the presence of only small amounts of bound lignin in these hemicellulosic fractions. The fractions precipitated with ethanol showed a less intense band in this region, which also occurred with the fraction that was not delignified (AV5 treatment). The hemicellulose that was delignified with chlorite and concentrated by rotary evaporation did not show this band range. This means that the white color they exhibited signaled the absence of lignin.
In the anomeric region (700-950 cm-1), a small sharp band in all spectra at 894(896 cm-1 was observed, due to the C-1 group frequency or ring frequency, which is characteristic of β-glycosidic linkages within sugar units of hemicellulose [28], specifically the 1→4 glycosidic bond between xylopyranose (Xylp) units of the main xylan chains [37]. On the other hand, a small peak at 792 cm-1 is indicative of α-anomers in side chains [10]. The broad band from 1000(1170 cm-1 was typical of arabinoxylans (AX) [36], since the presence of the arabinosyl side chains is shown by a high-intensity peak near 1030(1130 cm-1 in the spectrum of water-soluble hemicellulosic fractions and a relatively low intensity peak at 1161 cm-1 in the spectrum of holocellulose [33,37]. The AX has a linear backbone that is, in part, substituted by α-L-arabinofuranosa residues positioned either on O-3 or O-2 (mono-substitution) or on both O-2 and O-3 (di-substitution) of the Xylp monomer units; a characteristic of this compound is that the bulk of it can only be released by alkaline solutions [38].
3.4. Hemicellulose extracted from CC 8475 and V 7151 varieties
With respect to the previous results, the methods of using chloroform-methanol for dewaxing, HCl for de-pectination, and NaClO2 for delignification and NaOH and KOH were selected to isolate hemicellulose with NaBH4 as a protective agent in alkaline media. This procedure was replicated in CC 8475 and V 7151 varieties in order to extract hemicellulose, as shown in Table 4.
Table 4 Comparative results of hemicellulose extractions from CC 8475, V 7151, and CC 8592.
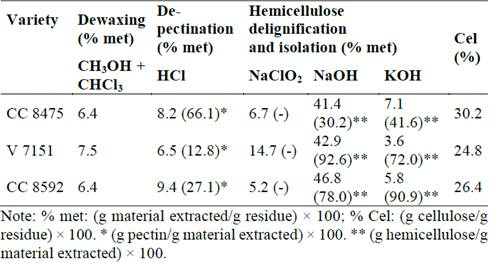
Source: The authors.
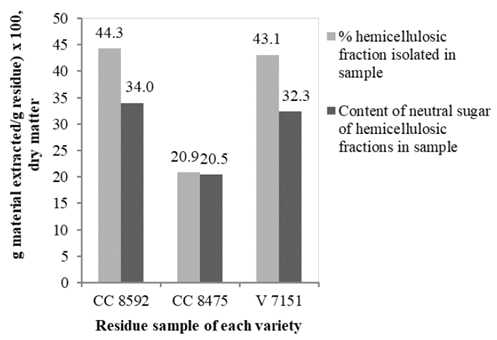
The percentage of material extracted by different solvents is similar except for lignin that was solubilized with NaClO2 in the V 7151 variety, which was, on average, 145 % superior to the other two varieties. In the same residue, Gómez [29] did not find significant differences in the composition among the three varieties, whose residue was harvested at the same time with little time in the field. More representative change in the de-pectination process was observed in the CC 8475 variety due to all material being extracted in acidic media; 66.1 % corresponded to pectic substances. In the other two varieties, this material only represented 12.8 or 27.1 % of the total. It was found the NaClO2 did not extract hemicellulose in CC 8592. The total material extracted was similar in the three varieties, but the critical aspect in all the processes was the quantity of hemicellulose extracted in alkaline media with NaOH and KOH. The hemicellulose obtained was about half that of the other two varieties (Fig. 6).
More significant change occurred in the fraction precipitated with ethanol, which was only 4 % compared to 28 % isolated in the Venezuelan variety and CC 8592. In relation to the hemicellulose precipitated by the rotary evaporation process, the difference among the three varieties was only 3 %. The treatment with NaClO2 in CC 8475 likely could not delignify as effectively as the other two varieties mainly due to the strong interactions between hemicelluloses and lignins often hinders hemicellulose extraction [13], which were more recalcitrant due to their longer time in the field (> 30 days). It is well known abiotic and biotic stresses increase lignin content of grasses, [39] and lignification continues postharvest in plants [40]. Also, it is likely that ethanol was added for this reason, where a significant part of hemicellulose combined with large amount of lignin could not precipitate and remained in the solution without recovery. The FTIR analysis of the hemicellulose of CC 8475 and V 7151 showed similar spectra to CC 8592 (numbers 8 and 9, Fig. 4, and 6 and 7, Fig. 5) and, therefore, are the same type or structure. In this hemicellulose, no lignin was detected from 1507(1523 cm-1.
Source: The authors.
Figure 5 Hemicellulose extracted with KOH. AV1 (1), AV2 (2), AV3 (3), AV4 (4), AV5 (5), CC 8475 (6), and V 7151 (7).
Source: The authors.
Figure 6 Yield and content of neutral sugar of hemicelluloses extracted in three varieties of sugarcane residue.
The amount of hemicellulose in the majority of plants ranges from 15(35 % (dry matter) [35], in sugarcane bagasse ranges between 25-28 % [41] and the CC8475, CC8592 y V7151 varieties of sugarcane residues showed hemicellulose concentrations between 20-34 %. In this research, it is possible to find in many isolated hemicelluloses other polluting components, such as residual lignin, cellulose, and pectins (frequently linked to arabinans and galactans on their side chains [34]. In addition, some water can remain, which can be embedded among the branches of the hemicelluloses, making it difficult to release.
4. Conclusion
The dewaxing method with methanol-chloroform, de-pectination with 0.15% HCl, delignification with NaClO2, and extraction of hemicellulose with 18% NaOH and 24% KOH in 0.26 M NaBH4 resulted in the greatest yield of hemicellulose extraction among the five different methods tested in this study using the CC 8592 variety. This procedure was replicated using CC 8475 and V 5171 varieties. It was found that, on average, 41% of hemicellulose extraction was possible gravimetrically with exception of CC 8475. However, the extracted hemicellulose was not totally pure. Furthermore, arabinoxylans, small portions of lignin, cellulose, and pectin were extracted, as evidenced by FTIR analysis. Residue of the CC 8475 variety was collected 30 days after harvest, and, because of this, it was presumed that the lignin-hemicellulose complex was more recalcitrant and was therefore not recovered after precipitation with ethanol. It is believed that leaves and tops that remain in the field exposed to environmental factors for several weeks undergo cellular structure and composition alterations. Therefore, it would be interesting to perform studies to observe these variations on residue remaining in the field over time. It is also important to adjust hemicellulose extraction methods to use alkaline media to separate hemicellulose with greater purity.