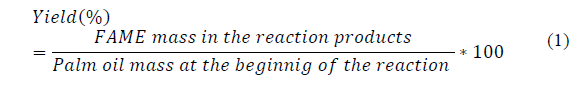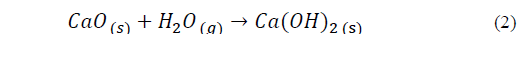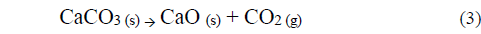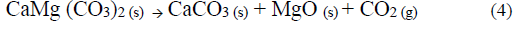1. Introduction
The global production of biodiesel has significantly increased over time due to the demand of modern society for clean energy sources with low environmental impact. In Colombia, between 2009 and 2014, the biodiesel production from palm oil increased from 169411 to 518092 tons, due to the growth of the production capacity of this industry [1]. According to Kiss, Dimian and Rothenberg [2], the main advantages of biodiesel against regular oil-based Diesel are: low toxicity, biodegradability, lubricity, and the absence of sulfides; moreover, it promotes rural economy and job generation.
The transesterification is the most effective chemical way for biodiesel production, since it allows the reduction of the molecular weight in approximately a third, compared to triglycerides, which improves the final product volatility [3]. This process can be performed using either homogeneous (same phase as the reactants) or heterogeneous (different phase from the reactants) catalysts, having the possibility to perform both types of catalysis in an acid or basic medium [4]. The alkaline homogeneous catalysis is common in the commercial biodiesel production processes, with yields higher than 97 % FAME (fatty acid methyl esters) in a short period of time (10 minutes to 2 hours), low temperatures (298 K - 343 K), using sodium or potassium hydroxide [5]. Heterogeneous catalysis conventionally uses alkaline earth metal oxides with yields higher than 80 % FAME at low temperature [5]. The main advantage of this catalysis compared with the homogeneous one is the simple catalyst recovering, which allows its reuse in posterior neutralization phases of the process [6]; furthermore, most of the times, it does not require neutralization phases, which avoids ample water contamination [7].
Multiple studies are focused on the research of alternative sources for biodiesel production, with lower costs and environmental impact. The Dolomite is considered as a promissory catalyst in the production of this biofuel because it is composed mainly by calcium and magnesium carbonates CaMg (CO3)2, being decomposed to calcium and magnesium oxides at high temperatures [8]. It is a common material in continental and marine rocks, being found along different territories. This characteristic makes dolomite an easy access, low cost, profuse material [9]. Authors such as Ilgen [10] have studied methanolysis with dolomite during a 3-hour reaction time obtaining 91.78 % FAME yield using canola oil (6:1 mole/mole, 3 % wt/wt a 333.15K). Wei et al., [11] obtained 75.3 % FAME yield in the olive oil transesterification during 3 hours with a 12:1 molar ratio using 6 % wt/wt of dolomite at 318.15 K. Ngamcharussrivichai, Nunthasanti, Tanachai, and Bunyakiat., [12], performed methanolysis at 333.15 K during 3 hours with palm kernel oil obtaining 98 % of the methyl ester content with 6 % wt/wt of dolomite and 30:1 molar ratio. Ngamcharussrivichai, Wiwatnimit, Wangnoi, [13], analyzed the palm kernel oil transesterification with methanol in presence of modified dolomite with Ca (NO3)2 achieving a 99.9 % FAME yield at 333.15 K with a 15:1 molar ratio, 10 % wt/wt catalyst concentration at 3 hours and Jaiyen et al., [14], incorporated SnO2 y ZnO on the dolomite having 98 % FAME yield. This shows that dolomite is a promising mineral in biodiesel production, having high FAME yields (>95 %) with high methanol-oil molar ratios (>15:1) in batch reactors.
In the current work, the transesterification of the RBD palm oil was studied in a heterogeneous system using dolomite as a catalyst, where the effect of the methanol-oil molar ratio and the amount of dolomite were examined at one-hour of reaction. To avoid the excess of methanol usage because of environmental and economic reasons the best process conditions which occur at the lowest molar ratio must be determined.
2. Design of experiments and statistical analysis
In order to develop the present study, a complete random design with a 32 factorial structure was employed. The evaluated factors were: mass of dolomite calcined at 1073.15 K respect to the oil (levels: 4 %, 7 % and 10 % wt/wt) and the methanol-oil molar ratio (levels: 6:1, 9:1 y 12:1 mole/mole). The reaction performed with the natural dolomite (ND) at 10 % wt/wt in the different methanol oil molar ratios previously mentioned was defined as the control sample. Three repetitions per treatment were performed. The response variable was the amount of fatty acid methyl esters (FAME) estimated by gas chromatography, respect the amount of palm oil at the beginning of the reaction. A variance analysis (ANOVA) and the Tukey test of significance were performed. Furthermore, the postulation of normality for the data was verified. The analysis was carried out using the SAS V-9.3 software.
3. Materials and methods
3.1. Determination of physicochemical properties of palm oil
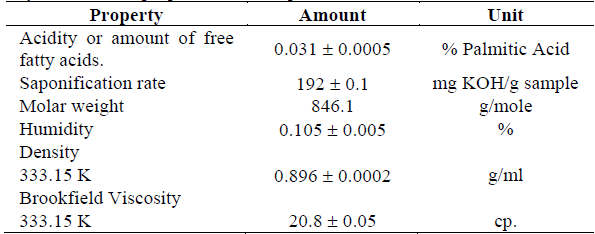
Palm oil was obtained from Intergrasas Ltda, located in Bogotá - Colombia. Physicochemical properties such as density, humidity, saponification index and acidity (amount of free fatty acids) were evaluated according to the standards NTC 336 [15], NTC 287 [16], NTC 335 [17] and NTC 218 [18], respectively. In addition, the viscosity was measured using a viscometer (Brookfield RVDV-II + P CP) at 333.15 K and 313.15 K.
3.2. Catalyst preparation
The dolomite manufactured by Inferhuila was acquired in an agricultural warehouse. Before being used as catalyst, the dolomite was ground on a cross beater mill Retsch - SK 100 and sieved using a mesh 325 ASTM (<45 μm), thus obtaining the Natural Dolomite (ND). The Calcined Dolomite (CD) was obtained calcinating the ND at 1073.15 K during two hours in a MM15 Terrigeno muffle furnace. The Stored Calcined Dolomite sample (CD-S) was prepared using the same procedures as the CD sample but it was stored for three weeks. To produce the Reactivated Dolomite sample (RD), the CD-S was calcined for two hours at 1073.15 K. Finally, the Washed Calcined Dolomite sample (WCD) was obtained by washing with methanol the CD used in a reaction cycle with a Soxhlet extractor, and then calcined at 1073.15 K for 2 hours. The WCD was used to evaluate the changes of the catalyst after a reaction cycle.
3.3. Catalyst characterization
The morphology and elemental composition of the catalyst, was evaluated using the scanning electron microscope (ESEM FEI-QUANTA 200) coupled to an EDX operated at 15 kV, obtaining images of the different catalysts surface (ND, CD and WCD) and a compositional analysis for WCD sample. The analyzes of surface area, pore diameter and pore volume were carried out using Quanta-Chrome NovaWin BET equipment. The FTIR tests for ND, CD, CD-S, RD and WCD samples were performed using Agilent CARY 630, operated in the range of 4000- 400 cm-1 (samples CD, RD and WCD were analyzed immediately after calcination at 1073.15 K).
The basicity of the catalyst was determined using the qualitative Hammett indicator test, measuring 0.001 g of the respective indicator and homogenizing it with 2 ml of toluene, 0.5 ml of methanol and 0.05 g of calcined dolomite (except for the indigo carmine which was prepared by adding 2 ml of methanol and 0.5 ml of toluene) [19] letting equilibrate 2h after which no further color changes were observed. The Hammett indicators used in this study were: bromothymol blue (H0= 7.2), phenolphthalein (H0= 9.8), indigo carmine (H0 = 12.2), 2.4-dinitroaniline (H0 = 15.0) and 4-chloro-2- nitroaniline (H0= 17.2).
3.4. Transesterification reaction
The transesterification reaction was carried out using a Batch reactor (AmAr A-2014) of stainless steel with mechanical agitation, PID controller for temperature, heated by resistances, cooled with water, and a maximum capacity of 1000 ml. Typically the CD sample was suspended in a required volume of methanol (99.95 % Panreac) defined in the experimental design and subsequently added to the reactor filled with 400 ml of palm oil. The same process was performed for the mixtures 4, 7 and 10 % wt/wt of CD in methanol and the control sample according the experimental design. Reaction conditions were: 333.15 K, 600 rpm and 1 hour [20]. The catalysts used in the transesterification reactions correspond to CD after calcination, due to the carbonation and loss of catalytic activity of the CD-S sample (Fig. 4). After the course of reaction, the catalyst was separated by centrifugation (centrifuge Hettich 1206-01) at 6000 rpm for 5 min and the reaction mixture (biodiesel, methanol and glycerin) was then loaded into a rotary evaporator (Tokyo Rikakikai 0SB-2100) at 313.15 K for 30 min at 33597.24 Pa, to recover the excess methanol. The biodiesel-glycerin mixture was kept in separatory funnels for a period of 12 hours until two phases appeared [19].
The content of methyl esters in the biodiesel was analyzed with a SHIMADZU G-C 2014 gas chromatograph equipped with a flame ionization detector (FID) and a column forte BP20 - WAX (60 m x 0.25 mm ID x 0.25 μm). The temperature of the column was first established at 403.15 K and increased to 453.15 K with a heating ramp of 15 K/min, (keeping this temperature for 0.5 min) and continued heating to 528.15 K with a heating ramp of 9 K/min. (keeping this temperature for 4 min) for a total time of 16.17 min. The samples to be analyzed were prepared by mixing: 500 μl of the phase rich in esters in hexane HPLC (10 mg / 10 ml), 200 μl of methyl heptadecanoate solution in hexane HPLC (10 mg / 10 ml) and 800 μl of hexane HPLC, to complete a volume of 1.5 ml. The amount of methyl esters produced (FAME yield % wt/wt) was calculated according to the equation reported by Suryaputra, Winata, Indraswati, and Ismadji [21] and Naureen, Tariq, Yusoff, Chowdhury and Ashraf [22] (Equation 1).
4. Results and discusion
4.1. Determination of physicochemical properties of palm oil
The properties of the RBD oil used as raw material in the transesterification are found in Table 1.
4.2. Catalysts size distribution
After treating ND thermally an incomplete calcination of the material was observed, because there is some gray color the sample (Fig. 1B) whereby, the size of the particle had to be reduced. The Dolomite was grinded using a cross beater mill and sieved with 130, 200, 230 y 325 mesh ASTM (106μm, 75μm, 63μm and 45μm respectively). Once this process was complete, it was found that particle sizes lower than 45 μm allow the complete calcination of the catalyst (Fig. 1C) which assures the presence of CaO and MgO in major amounts (active phases in the reaction of interest) Fig. 1C.
4.3. Catalyst characterization
4.3.1. X-ray dispersive spectroscopy
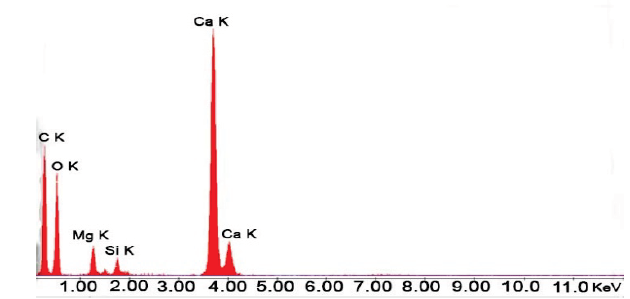
The X-ray Dispersive Spectroscopy resulted from the elemental chemical microanalysis performed over the WCD (Fig. 2) revealed the composition of the catalyst surface as: carbon, oxygen, magnesium, calcium and silicon, in proportional order Ca > C > O > Mg > Si, which coincides with the composition of the natural dolomite reported by Inferhuila [23] (55 % CaCO3, 39 % MgCO3 and 4 % other) defining the presence of silicon as impurity.
4.3.2. Scanning Electron Microscopy (SEM)
SEM images of the dolomite reveal an increase in the roughness of the dolomite surface after calcination due to the decomposition of the carbonate groups present in the ND [14]. The ND sample (Fig. 3 A ) liberates CO2 continuously, which causes a morphological variation observed in CD and WCD samples (Figs. 3B y 3C respectively). Additionally, the individual surface of the particles goes flatter after the thermal treatment [11]. According to Marques, Sousa, Loureiro, Rodriguez and Silveria, [24], a heterogeneous distribution can be observed after the calcination (irregular dimensions) as well as a less compact distribution in the calcined sample; nonetheless, it was more evident in the WCD sample.
4.3.3. Texture analysis (BET)
Table 2 shows the values of surface area, pore diameter and pore volume obtained when analyzing the different dolomite samples. According to Yoosuk, Udomsap, and Puttasawat [25], the increase in surface area and pore volume is attributed to the formation of holes during thermal decomposition, in addition, Jaiyen et al. [14] indicates that the increase in the parameters evaluated is directly influenced by the calcination temperature, reaching its maximum value at 1073.15 K.
4.3.4. Fourier-transform infrared spectroscopy (FTIR)
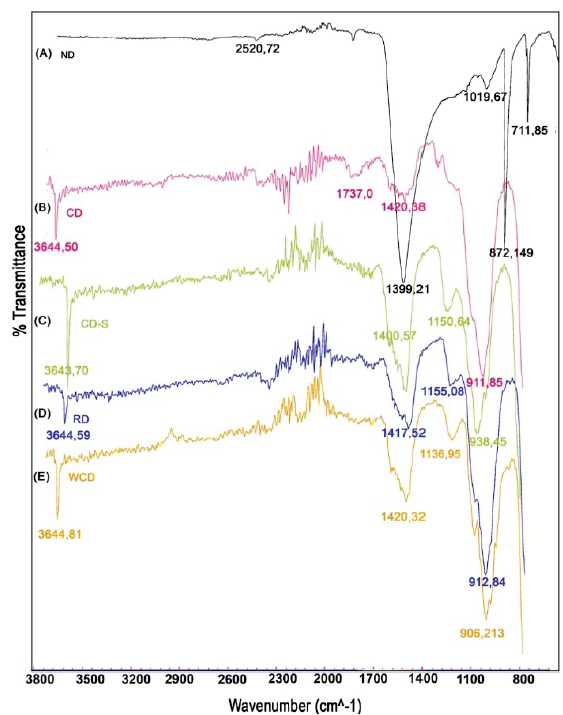
The characteristic FTIR spectrum of the ND is observed in Fig. 4A, which shows the asymmetric stretching vibrations of the carbonate groups at 1399.21 cm-1, out of the plane and in plane bending modes at 711.85 and 872.14 cm-1, respectively. Besides the internal modes, the combination of the previous bending modes was evidenced at 2520.72 cm-1, along with a peak corresponding to silicates (1019.67 cm-1) which is understood as impurity of the sample [26]. Authors such as Lavat and Graselli [27], report the presence of bands at 3400.16 and 1575 cm-1 attributed to the vibrations of the O-H bond of water, which are absent in the ND sample.
The samples CD, CD-S, RD and WCD (Figs. 4B, 4C, 4D and 4E respectively) reveal the presence of the hydroxyl functional group in the bands between 3643 to 3645 cm-1, belonging to the vibrations of the -OH group of Calcium Hydroxide and Magnesium Hydroxide, as consequence of the interaction between the oxides and the environmental humidity (Eq.2) [28]. The bands present between 1400 and 1421 cm-1 represent the stretching and vibrations of the carbonate complex bonds on the surface of the mixed oxide [29].
When comparing the spectra of Figs. 4-D and 4-B, it can be inferred that the second calcination (RD) does not generate a variation of the functional groups, since the same bands of the carbonate and hydroxyl groups are present. The storage of dolomite (Fig. 4-C ) considerably increases the vibrations of carbonates present in the sample, which decreases its catalytic activity as the proportion of calcium and magnesium oxides available for the reaction decreases (band of 1400.57 cm-1). Finally, the spectrum obtained from the analysis on the WCD sample (Fig.4-E ), indicates that the transesterification process followed by solvent washing and calcination does not modify the functional groups of the dolomite, since it presents a similar spectrum to the CD and RD samples.
4.3.5. Hammett indicators
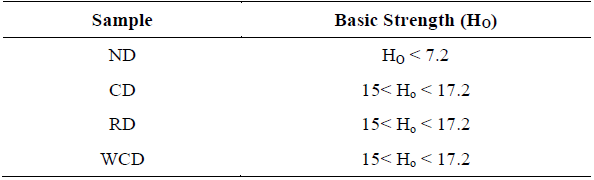
The basic strength of the different samples was analyzed using the qualitative Hammett indicator technique. According to Table 3 the ND has a low basic strength, since the catalyst in its natural state is composed mostly by alkaline earth metal carbonates, which have the following order of basicity: oxide> hydroxide> carbonate [25] because of this the ND sample CaMg (CO3)2 does not have catalytic activity in the transesterification of palm oil with methanol [24]. On the other hand, all the other samples to which a thermal treatment was carried out at 1073.15 K are in a basicity range of 15 <Ho <17.2, since the dolomite at that temperature is oxidized to a phase of mixed oxides (CaO + MgO) according to equations 3 and 4 [30]. The samples WCD and RD shows that washing the dolomite with methanol or submitting it to a second calcination process does not alter the basic strength of its surface. This is important because the reaction activity depends on the number of basic sites present in the catalysts as well as on the basic strength [12].
These results are comparable to those reported by Ilgen [10] (15 <Ho <18.4), Tamaddon, Tayefi, Hosseini and Zare [31] (15 <Ho <18.4), and are superior to the data of Yoosuk [25] ( 9.8 <Ho <12.2), Wang, Hu and Han [32] (8.2 <Ho <10.1), Wei et al. (2014) (11 <Ho <15) and Li, Li, Tong, Wang and Chen [33] (9.8 <Ho <15) who used similar conditions to those used in this study.
4.4. Transesterification reaction
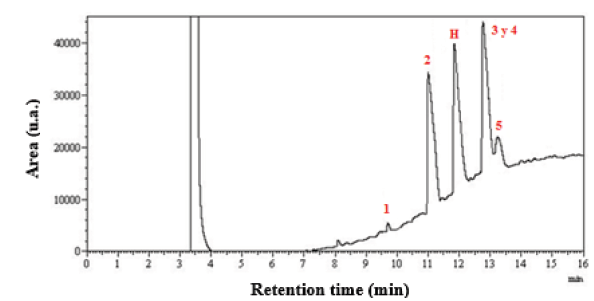
The quantification of methyl esters of fatty acids was developed by adding the contributions of 5 methyl esters identified: N°1 methyl myristate (C14: 0), N°2 methyl palmitate (C16: 0), N°3 and N°4 methyl stearate (C18: 0) + methyl oleate (C18: 1) and N°5 methyl linoleate (C18: 2), at retention times in the chromatographic column of 9.7, 11.0, 12.8 and 13.2 min respectively (Fig. 5). All samples of biodiesel were injected in triplicate into the chromatograph and the coefficient of variation of the different fatty acids areas was calculated.
Methyl heptadecanoate (C17: 0) was used to evaluate homogeneity in the detection of the esters under study by the chromatograph (H). A coefficient of variation of less than 10% in this fatty acid represented an adequate treatment of the samples by the equipment, since this fatty acid is not found in palm oil.
Table 4 Influence of the amount of catalyst on FAME yield. Reaction conditions: methanol/oil molar ratio 9:1 mole/mole, temperature, 333.15 K, reaction time 1 h and 600 rpm.

Source: The Authors.
Table 5 Effect of the methanol/oil molar ratio on the FAME yield. Reaction conditions: catalyst amount, 4 % (wt/wt), temperature 333.15 K reaction time 1 h and 600 rpm.

Source: The Authors.
Table 4 shows the effect of the amount of catalyst on FAME yield. In this case there were not, significant differences between the evaluated levels (4 %, 7 % and 10 % wt/wt), which indicates that the yield is not influenced by the amount of CD added to the reaction. This result is comparable with the information reported by Ilgen [10], who studied calcined dolomite at 1123.15 K in the transesterification of canola oil with methanol, where an increase in the FAME yield from 77.16 % to 91.60 % among 0.5 % and 1.5 % (wt/wt) of catalyst was found, nonetheless, increasing this amount to 6 % (wt/wt) did not produce a significant increase in the yield. Ngamcharussrivichai et al. [12], evaluated the transesterification of palm kernel oil with calcined dolomite at 1073.15 K and observed that catalyst amounts greater than 6 % wt/wt did not represent significant increases in the production of methyl esters (98 % FAME, 3 hours of reaction). The control sample made with ND did not catalyze the transesterification of palm oil with methanol thus, they were not considered for the analyzes performed.
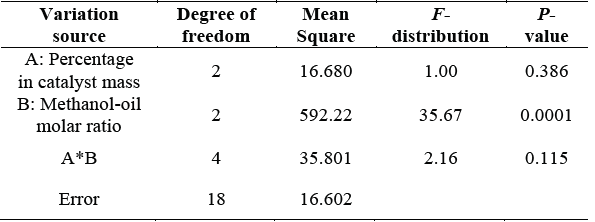
Regarding the influence of the amount of methanol on the FAME production (Table 5), there were not found significant differences between the levels 9: 1 and 12: 1 (mole / mole), however, these were significantly higher than the level 6: 1 (mole / mole) with a 95 % of confidence (Table 6 and 7). These results agree with Marques et al. [24], research who studied the transesterification with methanol (4h) of canola oil (6: 1 mole / mole, 1 % wt/wt) and sunflower oil (9: 1 mole / mole, 2 % wt/wt) achieving 98.11 % and 96.52 % FAME yield respectively. Ilgen [10], reports yield equal to 91.78 % FAME with 3-hour reaction in the transesterification of canola oil with methanol (6: 1 mole / mole, 3 % wt/wt).
Comparing these results with the investigations of Ngamcharussrivichai et al. [13], 99 % FAME at 3 h of reaction (15: 1 mole/mole, 10 % wt/wt) and Ngamcharussrivichai., et al, [12], 98 % FAME at 3 hours of reaction (30: 1 mole/mole, 6 % wt/wt), it can be observed that it is feasible to achieve high yields in biodiesel production with low amounts of methanol, which represents environmental and economic benefits.
Additional factors such as oil acidity and moisture affect the transesterification reaction. When the oil humidity is low and the value of free fatty acids calculated is less than 0.5 % (Table 1), the saponification is lower and the biodiesel production increases [34,35].
In this study, the highest FAME yield was 82.62 % at 1-hour, molar ratio methanol oil equal to 9:1 (mole/mole) and 4% wt/wt of CD. The response variable (FAME yield) fulfills the normality assumption defined by the Shapiro-Wilk test where the P-value of the tests performed is higher than 0.05 (0.61005 for the mass percentage of catalyst and 0.211845 for the methanol-oil molar ratio) with 95 % confidence. Finally, although the performance obtained is lower than those mentioned above, the reaction time in the present study was lower than those reported by other authors.
5. Conclusions
The calcination process at 1073.15 K for 2 hours is possible when the particle sizes in the dolomite are <45μm, which allows the formation of calcium and magnesium oxides, the improvement of the base strength and its superficial area increase.
The amount of catalyst added to the reaction did not show significant differences under the conditions evaluated. However, for the methanol-oil molar ratio the level 6:1 (mole / mole) is significantly lower than levels 9:1 and 12:1 (mole /mole).
For the transesterification, the dolomite must be used immediately after the calcination process since prolonged storage times favor the carbonation of the material. In addition, recalcining the sample does not affect the functional groups on its surface.
Dolomite is a potential catalyst for the production of biodiesel since it is an economical and easily processable compound with low toxicity and good FAME yields at moderate methanol-oil ratios.
In the present study, we obtained a yield of 82.67% FAME at 1 hour of reaction, using 4% wt/wt of calcined dolomite and a 9:1 (mole /mole). methanol-oil molar ratio. This is a promissory result for the transesterification with methanol of Colombian RBD palm oil.