Serviços Personalizados
Journal
Artigo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Acessos
Acessos
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares em
SciELO
Similares em
SciELO -
 Similares em Google
Similares em Google
Compartilhar
Revista Colombiana de Ciencias Químico - Farmacéuticas
versão impressa ISSN 0034-7418versão On-line ISSN 1909-6356
Rev. colomb. cienc. quim. farm. v.39 n.1 Bogotá jan./jun. 2010
Artículo de investigación científica
Apparent molar volumes of the anesthetic drugs procaine-HCl and lidocaine-HCl in water at temperatures from 278.15 to 313.15 K
Volúmenes molares aparentes de los anestésicos procaína-HCl y lidocaína-HCl en agua a temperaturas entre 278,15 y 313,15 K.
Daniel R. Delgado1, álvaro F. Jiménez Kairuz2, Rubén H. Manzo2, Edgar F. Vargas3, Fleming Martínez1*.
1 Grupo de Investigaciones Farmacéutico-Fisicoquímicas, Departamento de Farmacia, Facultad de Ciencias, Universidad Nacional de Colombia, A.A. 14490, Bogotá D. C., Colombia. * Corresponding Author: E-mail: fmartinezr@unal.edu.co
2 Departamento de Farmacia, Facultad de Ciencias Químicas, Universidad Nacional de Córdoba, Córdoba, Argentina
3 Laboratorio de Termodinámica de Soluciones, Departamento de Química, Facultad de Ciencias, Universidad de los Andes, Bogotá D. C., Colombia.
Recibido para evaluación: February 15, 2010 Aceptado para publicación: April 28, 2010
SUMMARY
Lidocaine-HCl and procaine-HCl are local anesthetic drugs widely used in minor chirurgic procedures, nevertheless, physicochemical information about their volumetric behavior, as well as for other aqueous properties, is not complete at present. In this context, in this article, densities of aqueous solutions of both drugs have been measured as a function of concentration (from 0.0500 to 0.5000) mol kg -1 at several temperatures, i.e. 278.15, 283.15, 288.15, 293.15, 298.15, 303.15, 308.15, and 313.15 K. The apparent molar volumes and partial molar volumes at infinite dilution for the electrolyte drugs were calculated, whereas, the partial molar volumes at infinite dilution and partial molar expansibilities for the molecular forms were also calculated. The dependence of these properties with temperature is shown. The results are interpreted in terms of interaction solute-solvent.
Keywords: Lidocaine-HCl, Procaine-HCl, Apparent molar volume, Molar expansibility, Solution thermodynamics.
RESUMEN
La lidocaína-HCl y procaína-HCl son anestésicos locales ampliamente usados en procedimientos quirúrgicos menores, sin embargo la información fisicoquímica acerca de su comportamiento volumétrico, así como de otras propiedades fisicoquímicas, aún es incompleta en la actualidad. Por esta razón, en este artículo se presentan los valores de densidad de algunas soluciones acuosas de estos dos fármacos en función de la concentración (desde 0,0500 hasta 0,5000) mol kg-1 a diferentes temperaturas (278,15, 283,15, 288,15, 293,15, 298,15, 303,15, 308,15 y 313,15 K). Así mismo se presentan los volúmenes molares aparentes y volúmenes molares parciales a dilución infinita de los fármacos como electrolitos, y de otro lado, los volúmenes molares parciales a dilución infinita de los fármacos moleculares y las expansibilidades molares, los cuales fueron calculados a partir de los valores de densidad y composición de las mezclas. Los resultados obtenidos se interpretan en términos de interacciones soluto-solvente.
Palabras clave: Lidocaína-HCl, Procaína-HCl, Volumen molar aparente, Expansibilidad molar, Termodinámica de soluciones.
INTRODUCTION
Local anesthetics drugs are amphiphilic compounds that have hydrophobic and hydrophilic domains that are separated by intermediate alkyl chains. The hydrophilic group corresponds to tertiary or secondary amine and the hydrophobic domain is an aromatic moiety. These compounds are classified in ester type and amide type, and it depends on the group that is bind to the aromatic residue. The nature of this bond determines several of their pharmacological properties (1).
The physicochemical characterization of local anesthetics in aqueous solutions has been object of study due to their widespread application in treatment of pain. It is widely accepted that the local anesthetics exert their pharmacological action by interacting with lipid molecules constituting the biological membranes of neurons. The mechanism of these interactions, however, is not clearly understood. The role of local anesthetics has been attributed to increase the surface pressure of the lipid layer that constitutes the nerve membrane, and therefore, closing the pores through the Na+, K+ or Ca2+ ions pass (1-8). It has been suggested that local anesthetics affect permeability by increasing the disorder degree of the membrane (9). Thus, the volumetric properties of anesthetics have an important role in the mechanism of anesthesia and the determination of volumetric properties of local anesthetics in aqueous solutions provides information needed to understand the mechanism of anesthesia.
On the other hand, it is well know that physicochemical characterization of drugs plays a crucial role in all the stages associated to design and development of pharmaceutical dosage forms, specially those intended to parenteral administration (10). Thus, in the literature, a few studies on the volumetric properties of local anesthetics have been reported (11-14).
In this context, as a contribution to generation and systematization of physicochemical information about drugs' aqueous behavior, the main goal of this study was to evaluate the effect of concentration and temperature on the apparent molar volume of lidocaine-HCl (Figure 1, LC-HCl) and procaine-HCl (Figure 1, PC-HCl) in water (15). Thus, the present investigation is a continuation of that reported previously (14). With that purpose, an interpretation in terms of solute-solvent interactions based on the corresponding volumetric behavior was developed.
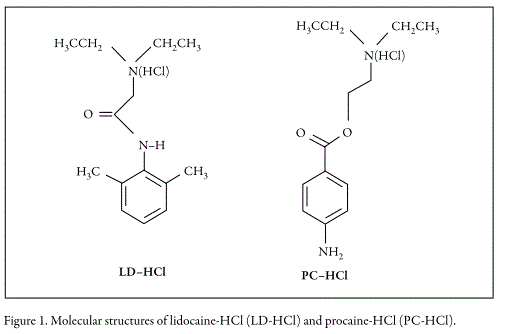
It is important to keep in mind that the study of molar volumes of drugs and other pharmaceutical compounds has been done to facilitate the design of pharmaceutical dosage forms as well as a useful physicochemical tool to propose possible mechanisms of the transfer processes of drugs across different biological membranes (16).
MATERIALS AND METHODS
Chemicals
In this investigation the following chemicals were used: lidocaine-HCl and procaine-HCl in USP quality (17) and distilled water (conductivity < 2 µS cm -1).
PPN-HCl aqueous solutions preparation
All LC-HCl and PD-HCl aqueous solutions were prepared in quantities of 30.00 g by mass using a Ohaus Pioneer TM PA214 analytical balance with sensitivity ±0.1 mg, in concentrations from 0.0500 mol kg-1 to 0.5000 mol kg-1, in order to study nine solutions. This procedure implied an uncertainty of ±2×10-5 in molality.
Density determination
This property was determined using a DMA 45 Anton Paar digital density meter connected to a Neslab RTE 10 Digital Plus (Thermo Electron Company) recirculating thermostatic water bath according to a procedure previously described (16). The equipment was calibrated according to Instruction Manual using air and water at the different temperatures studied (18). From the experimental density values and solution compositions all volumetric properties were calculated according to the equations presented earlier.
RESULTS AND DISCUSSION
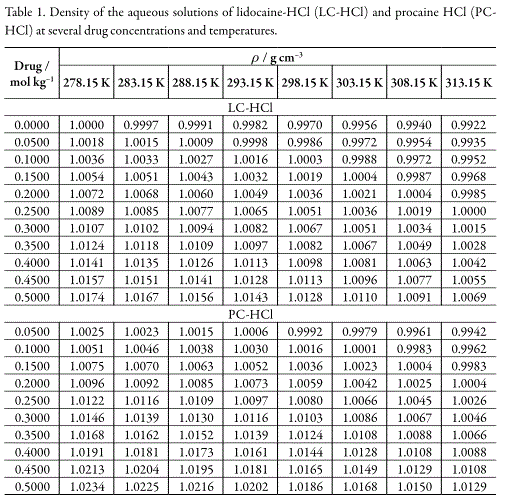
The experimental densities of both drugs solutions in water at (278.15 to 313.15) K are shown in Table 1. The apparent molar volumes (ΦV) were calculated by using equation 1.

where, M2 is the molar mass of solute, ρ0 and ρ are the densities of solvent and solution, respectively, and m is the drug concentration in the solution expressed as molality (13), (14).
Table 2 summarizes the results of the apparent molar volumes of LC-HCl and PCHCl, molal concentrations, and their uncertainties. Uncertainty values in apparent molar volumes were calculated according to the law of propagation of uncertainties (19), by means of equation 2, where the uncertainty in density measurements was ±0.0001gcm-3.
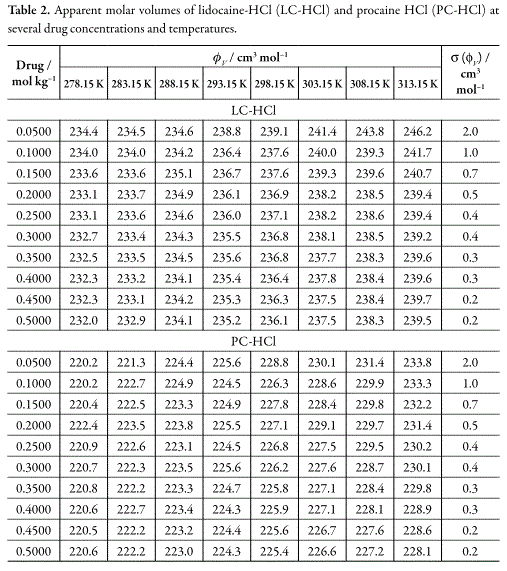

Table 2 shows that ΦV diminish for both drugs at all temperatures studied. This result is in opposite way to the PC-HCl behavior reported by Torres et al. (14) at 298.15 and 303.15 K. In this context, the ΦV dependence with respect to drugs molal concentration, at all temperatures studied, was fitted to equations of the following type (20):
where Φ0V= V0 Drug-HCl, is the apparent molar volume at infinite dilution (equal to the partial molar volume at infinite dilution) and Sv is the experimental parameter. Values of V0Drug-HCl and Sv were obtained by weighted least-squares method by using the ΦV numerical values together with their uncertainties. These values are shown in Table 3. The V0Drug-HCl values taken from the literature are also shown in this table
.
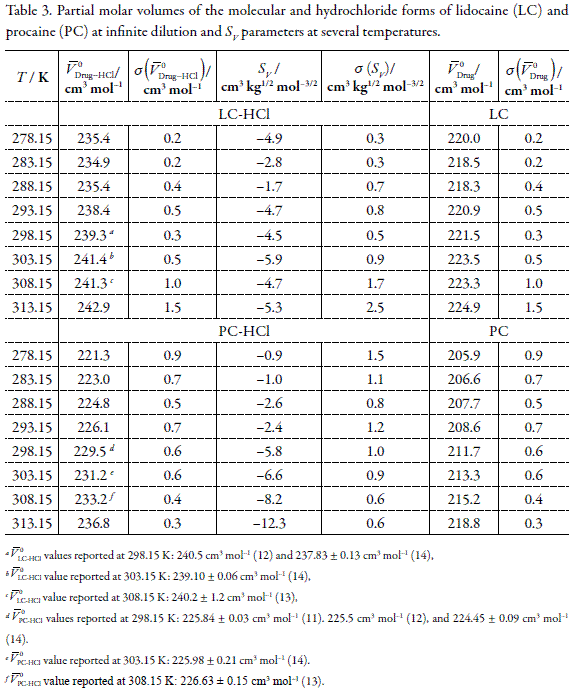
According to Table 3, a good agreement is found for V0LC-HCl with respect to the values reported in the literature at 298.15 (12, 14), 303.15 K (14), and 308.15 K (13), if and uncertainty near to 1.0 cm3 molâ1 is considered. Oppositely, for V0PC-HCl values great disagreement is found with respect to literature values finding differences of 5.0 cm3 mol-1. Nevertheless, no clear explanation is found for these discordances. Although it is necessary kept in mind that the concentration ranges studied in each case are so very different, which could lead to different kind of interactions.
The sign of parameter Sv in equation 3 could be associated with the influence of the solute upon water. This structural influence can be described in terms of structure promotion or structure breaker effects of the solute on the surrounding water medium (21, 22). Negative values of Sv were found for some solutes such as tetraakylamonium salts, which are typical ionic surfactants, characterized by their water-structure promotion effect (22-25). In this type of solutes the hydrophobic effect become dominant compared with hydrophilic effect, therefore solvation around of the ionic moiety diminishes. According to Table 3 the value of Sv is negative for both drugs at all temperatures. These observations could be interpreted in terms of the structure promotion effect of both electrolyte drugs on the water-structure at temperatures. On the other hand, as it was already said, Torres et al. (14) reported positive Sv values for PC-HCl, which clearly is opposite to values showed in Table 3.

On the other hand, negative values of Sv were also found in aqueous solutions of propranolol and acebutolol (26), thioridazine hydrochloride (27), phenalalkylamines (28), n-alkylamine hydrobromides (29), and tetraalkylammonium bromides (20, 30).
On the other hand, by using the principle of volume additivity, the apparent molar volume at infinite dilution of Drug-HCl can be separated into individual ionic contributions according to the following expression (13, 31):
where V0Drug and V0HCl are the partial molar volume of free base and of HCl, respectively. Data of V0HCl were taken from the literature (32). The values of V0Drug obtained from equation 4 at each temperature are also shown in Table 3..

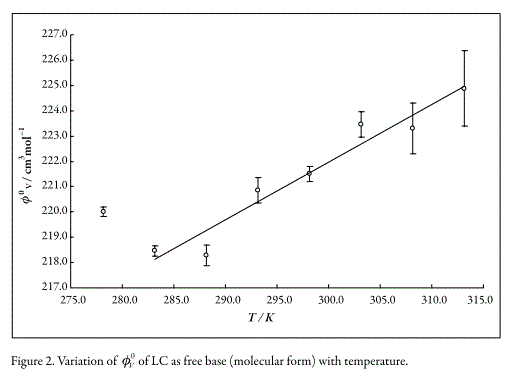
The variation of V0LC with temperature, without consider the value at 278.15 K (Fig. 2), was adjusted by method of weighted least squares to the following linear empiric equation (with r2 equal to 0.88),

where T is the temperature in Kelvin. Whereas, the variation of V0LC with temperature (Fig. 3) was adjusted by method of weighted least squares to the following parabolic empiric equation (with r2 equal to 0.98),
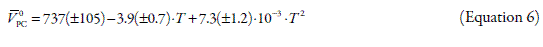
On the other hand, the partial molar expansibility at infinite dilution, E0Φ, can be calculated by differentiating equations 5 and 6 with respect to temperature, E0Φ= ∂V0Drug⁄∂T. The obtained E0Φ value for LC is 0.228 ± 0.019 cm3mol-1 K-1, while the respective values for PC are dependent on temperature, being as follows, 0.11 cm3mol-1 K-1 at 278.15 K, 0.26 cm3mol-1 K-1 at 288.15 K, 0.40 cm3 mol-1 K-1 at 298.15 K, and 0.55 cm3 mol-1 K-1 at 308.15 K.
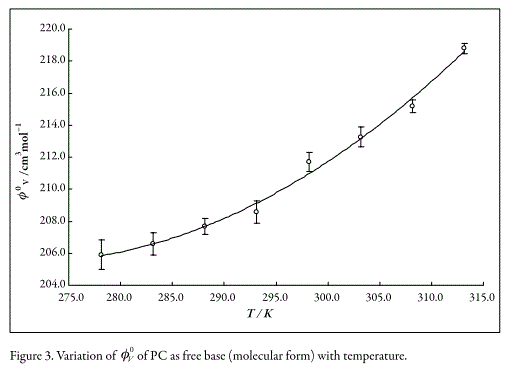
In Figures 2 and 3, it can be seen that tendency is such that V0Drug values for both drugs increases when the temperature increases. This result could be attributed to the breakage of solvent structure, which causes an increase in the structural molar volume (24). As was already said, E0Φ is positive which could be explained according to formation of "clathrate-like" structures as described by Wen and Saito (22). In this way, when the concentration of Drug-HCl increase the water cluster surrounding the ions tends to join with their neighbors and form flickering cages forcing the ions to get inside these cages. On heating these structures would breakdown, leading to the expansion of the whole system (25).
CONCLUSION
From all topics discussed previously it can be concluded that the volumetric behavior of LC-HCl and PC-HCl in aqueous media is dependent both on drug concentration and temperature. Based on the negative signs of the Sv term obtained it could be proposed that these drugs act as water-structure promoter due to hydrophobic effect around its non-polar moieties. Ultimately, it can be said that the data presented in this report expand the physicochemical information about electrolyte drugs in aqueous solutions.
ACKNOWLEDGEMENTS
We thank the DIB-DINAIN of the Universidad Nacional de Colombia (UNC) for financial support and the Department of Pharmacy of UNC for facilitating the equipment and laboratory required.
REFERENCES
1. S.P. Gupta, Quantitative Structure-Activity Relationship studies on local anesthetics, Chem. Rev., 91, 1109-1119 (1991). [ Links ]
2. W.A. Catterall, Common modes of drug action on Na+ channels: Local anesthetics, antiarrhythmics and anticonvulsants, Trends Pharmacol. Sci., 8, 57-65 (1987). [ Links ]
3. C. Frelin, P. Vigne, M. Lazdunski, Biochemical evidence for pharmacological similarities between alpha-adrenoreceptors and voltage-dependent Na+ and Ca++ channels, Biochem. Biophys. Res. Commun., 106, 967-973 (1982). [ Links ]
4. J.C. Matthews, A. Collins, Interactions of cocaine and cocaine congeners with sodium channels, Biochem. Pharmacol., 32, 455-460 (1983). [ Links ]
5. M.C. Fishman, I. Spector, Potassium current suppression by quinidine reveals additional calcium currents in neuroblastoma cells, Proc. Natl. Acad. Sci. USA, 78, 5245-5249 (1981). [ Links ]
6. G.T. Bolger, K.A. Marcus, J.W. Daly, P. Skolnick, Local anesthetics differentiate dihydropyridine calcium antagonist binding sites in rat brain and cardiac membranes, J. Pharmacol. Exp. Ther., 240, 922-930 (1987). [ Links ]
7. D.E. Coyle, N. Sperelakis, Bupivacaine and lidocaine blockade of calcium-mediated slow action potentials in Guinea pig ventricular muscle, J. Pharmacol. Exp. Ther., 242, 1001-1005 (1987). [ Links ]
8. A.M. Shanes, Electrochemical aspects of physiological and pharmacological action in excitable cells. I. The resting cell and its alteration by extrinsic factors, Pharmacol. Rev., 10, 59-164 (1958). [ Links ]
9. J.C. Metcalfe, A.S. Burgen, Relaxation of anesthetics in the presence of cytomembranes, Nature, 220, 587-588 (1968). [ Links ]
10. A. Martin, P. Bustamante, A.H.C. Chun, "Physical Chemical Principles in the Pharmaceutical Sciences", 4th edition, Lea & Febiger, Philadelphia, 1993. [ Links ]
11. M. Iqbal, R.E. Verall, Apparent molar volume and adiabatic compressibility studies of aqueous solutions of some drug compounds at 25°C, Can. J. Chem., 67, 727-735 (1989). [ Links ]
12. H. Matsuki, S. Hashimoto, S. Kaneshina, Surface adsorption and volume behavior of local anesthetics, Langmuir, 10, 1882-1887 (1994). [ Links ]
13. M. Iqbal, M.A. Jamal, M. Ahmed, B. Ahmed, Partial molar volumes of some drugs in water and ethanol at 35°C, Can. J. Chem., 72, 1076-1079 (1994). [ Links ]
14. D.R. Torres, L. Blanco, F. Martínez, E.F. Vargas, Apparent molal volumes of lidocaine-HCl and procaine-HCl in aqueous solution as a function of temperature, J. Chem. Eng. Data, 52, 1700-1703 (2007). [ Links ]
15. S. Budavari, M.J. O'Neil, A. Smith, P.E. Heckelman, J.R. Obenchain Jr., J.A.R. Gallipeau, M.A. D'Arecea, "The Merck Index, An Encyclopedia of Chemicals, Drugs, and Biologicals", 13th ed., Merck & Co., Inc., Whitehouse Station, NJ, 2001. [ Links ]
16. F. Martínez, A. Gómez, C.M. ávila, Volúmenes molales parciales de transferenciade algunas sulfonamidas desde el agua hasta la mezcla agua-etanol (X = 0.5), Acta Farm. Bonaerense, 21, 107-118 (2002). [ Links ]
17. US Pharmacopeia, 23rd edition, United States Pharmacopeial Convention, Rockville, MD, 1994. [ Links ]
18. O. Kratky, H. Leopold, H. Stabinger, "DMA45 Calculating Digital Density Meter, Instruction Manual", Anton Paar, KG, Graz, Austria, 1980. pp. 1-12. [ Links ]
19. B.N. Taylor, C.E. Kuyatt, "Guidelines for evaluating and expressing the uncertainty of NIST measurement results", Technical Note 1297, United States Department of Commerce, National Institute of Standards and Technology, Gaithersburg, MD, 1994. [ Links ]
20. F.J. Millero, The molal volumes of electrolytes, Chem. Rev., 71, 147-176 (1971). [ Links ]
21. F. Franks, H.T. Smith, Apparent molal volumes and expansibilities of electrolytes in dilute aqueous solution, Trans. Faraday Soc., 63, 2586-2598 (1967). [ Links ]
22. W.Y. Wen, S. Saito, Apparent and partial molal [ Links ]
23. R. De Lisi, S. Milioto, R.E. Verrall, Partial molar volumes and compressibilities of alkylmethylammonium bromides, J. Solution Chem., 19, 665-692 (1990). [ Links ]
24. L.H. Blanco, Y.P. Salamanca, E.F. Vargas, Apparent molal volumes and expansibilities of tetraalkylammonium bromides in dilute aqueous solutions, J. Chem. Eng. Data, 53, 2770-2776 (2008). [ Links ]
25. R. Gopal, M.A. Siddiqi, The variation of partial molar volume of some tetraalkylammonium iodides with temperature in aqueous solutions, J. Phys. Chem., 70, 1814-1817 (1968). [ Links ]
26. J.M. Ruso, A. Gonzalez-Perez, G. Prieto, F.A Sarmiento, Volumetric study of two related amphiphilic beta-blockers as a function of temperature and electrolyte concentration, Coll. Surf. B: Biointerfaces, 11, 165-175 (2004). [ Links ]
27. M.A. Cheema, S. Barbosa, P. Taboada, E. Castro, M. Siddiq, V. Mosquera, A thermodynamic study of the amphiphilic phenothiazine drug thioridazine hydrochloride in water/ethanol solvent, Chem. Phys., 328, 243-250 (2006). [ Links ]
28. F. Shahidi, Partial molar volumes of phenalkylamines and their physiologically active derivatives in water, Can. J. Chem., 65, 1924-1926 (1987). [ Links ]
29. P.A. Leduc, J.L. Fortier, J.E. Desnoyers, Apparent molal volumes, heat capacities, and excess enthalpies of n-alkylamine hydrobromides in water as a function of temperature, J. Phys. Chem., 78, 1217-1225 (1974). [ Links ]
30. L.H. Blanco, E.F. Vargas, Apparent molar volumes of symmetric and asymmetric tetraalkylammonium salts in dilute aqueous solutions, J. Solution Chem., 35, 21-28 (2006). [ Links ]
31. Y. Marcus, Ionic volumes in solution, Biophys. Chem., 124, 200-207 (2006). [ Links ]
32. R. Pogue, G. Atkinson, Apparent molal volumes and heat capacities of aqueous HCl and HClO4 at 15-55°C, J. Chem. Eng. Data, 33, 495-499 (1988). [ Links ]














