INTRODUCTION
Preservatives are widely used and necessary in all products with risk of deterioration because by inhibiting the growth of bacteria and fungi they prevent microbiological contamination of the product, protecting the consumer from possible infection [1]. Although there are many molecules for this purpose, it has been reported that the majority generates allergic reactions and sensitization by contact, being necessary the search for stable antimicrobials, broad spectrum and pH range, non-toxic or irritating [2].
One of the preservatives commonly used in cosmetics, food and medicine is benzoic acid (figure 1). Despite being considered a harmless substance, its topical application can generate hives, erythema, and stinging, probably by induction in the skin of large amounts of prostaglandin D2 [3]. In this sense, possible strategies that reduce the allergic reactions normally presented by this preservative have been evaluated, and the formation of complexes with polyelectrolytes is an important alternative because they modify its physicochemical properties, functionality and could even improve its safety [4].
Because the skin represents a significant route for the non-invasive administration of compounds, the evaluation of the toxicity and activity of molecules such as preservatives must be assessed [5]. In vitro tests conducted in Franz cells are considered as one of the main tools to evaluate transepithelial permeation, which consist of two compartments, one donor and another recipient where it is sampled periodically to evaluate the amount of compound that has penetrated through tissue over time [6]. These compartments are separated by a synthetic or natural membrane [7, 8].
Consequently, permeation tests were performed with benzoic acid and with the complexes (B75C/25 and EuB 100 (figure 2) generated by the ionic interactions of the molecules involved, where Eu corresponds to the Eudragit E100 polyelectrolyte (figure 3), B to the benzoate ion and Cl to the chloride ion that act as counterions, the subscripts indicate the percentage at which each counterion is neutralizing the polymer; these complexes have shown in previous studies carried out within the research group, that their antimicrobial activity is maintained [4, 6]. This work is intended to assess whether there is a difference in the permeation behavior of complexes containing benzoic acid against the uncomplexed compound, from which it could be inferred if there is a difference in their biological safety.
MATERIALS AND METHODS
Materials
Benzoic acid (99%) Sigma Aldrich (U.S., lot: 09128 DCV); HPLC Tedia grade acetonitrile (U.S., lot: 905154); phosphoric acid (85%) Carlo Erba (E.U, lot: 8C109098E); sodium chloride J.T Baker (U.S., lot: t10c54); 2.39M hydrochloric acid; Eudragit® E100 (Germany, lot: B140101519); complexes E7uB75C/25 and EuB 100 HPLC grade water.
Biological material
For the development of the permeation tests, ear skin obtained from freshly slaughtered pigs weighing between 80-100 kg and with an average age of 4-6 months was used, these were removed and stored immediately in 1 M NaCl solution [9] under cooling conditions (4-8 °C). Once transported, it was verified that the skin did not present any type of lesion, spot, or tattoo making cuts of skin of 600 μm thickness to the samples with appropriate acceptance criteria. The integrity of the skin was determined through the measurement of transepithelial water loss (TEWL), using the Cutometer® dual MPA 580 with a Courage + Khazaka® model TM 300 tewameter, selecting pig skins with values below 35 g/h/m2 [10].
Operating conditions of the Franz cell system for transdermal permeation studies
Permeation assays were performed using Franz cells (PermeGear® brand) under two different conditions: infinite doses and finite doses. The first condition was used to establish the physicochemical parameters of permeation [11], which allowed to define the storage conditions of pig skin so that it would not be required to test immediately after the animal was slaughtered. Finite dose tests were performed simulating the conditions of use [11] in which benzoic acid would be used as a preservative, with the purpose of evaluating the safety profile of complexed benzoic acid (E7uB75Cl25, EuB 100 ), and free. The development of this work was endorsed by Comité de Etica de la Facultad de Ciencias de la Universidad Nacional de Colombia (Acta N° 07 de 2015). Below are the details of each methodology used.
Infinite doses
Through the infinite dose tests, an enough benzoic acid (>100 μg/cm2) was applied to reach the maximum flow per unit area [11], corresponding to 2.3 mg/mL solution's benzoic acid. Permeation tests were carried out in three Franz cells at 32 °C, 400 rpm stirring, 0.9% NaCl receptor medium adjusted to pH 7.4 [12], using pig skin cuts obtained from different pigs with 600 μm thickness (using the dermatome Aesculap-B.Braun model ACCULAN® 3Ti) [13] and diffusion area 1.75 ± 0.20 cm2. Before starting the test, the skin could equilibrate in the assembly for 15 minutes at 32 °C, with an environmental relative humidity of 60% and the TEWL was measured (to establish that the skin was suitable for study). The application of 500 μL of the benzoic acid solution (>100 μg/cm2) was carried out in the donor compartment, which remained occluded. The exposure time was 6 h, taking 500 μL aliquots of the receiving compartment at 0.5; 1; 1.5; 2; 3; 4 and 6 h, immediately replenishing with the receiving medium. Quantification of benzoic acid was performed by HPLC-DAD (Shimadzu® Prominence series) as soon as the aliquots were obtained in each sampling. The analytical methodology was previously validated using as phosphoric acid (1%)/acetonitrile (70:30) mobile phase, flow of 1 mL/min, isocratic temperature of 35 °C and injection volume of 10 μL. This procedure was followed with samples of the skin of ears of freshly slaughtered pigs and after stored for 72 h in 1 M NaCl at -2 °C.
From these tests the diffusion parameters were obtained: permeation flow (equation 1) and permeability coefficient (equation 2), as well as the transepithelial water loss (TEWL) values of the two types of swine skin samples, establishing through the statistical analysis of the Kolmogorov Smirnov, Fisher and t Student tests (95% confidence level) if there was a significant difference between the diffusion and TEWL parameters of the pig samples at 0 and 72 h after slaughter.
Where, dQr is the amount of drug transported through the membrane inside the receptor compartment (μg), A is the active diffusion area (cm2) and dt is the exposure time (h).
Where, Jss is the flow calculated at steady state (mg/cm2/h) and Cd is the concentration of drug in the donor compartment (mg/cm3).
Finite doses
Solutions of 2.0 mg/mL benzoic acid (pH 5) were prepared in triplicate by adding the amount of equivalent complex to obtain the desired concentration. Permeation tests were performed in triplicate in Franz cells at 32 °C, 400 rpm agitation, 0.9% NaCl receptor medium adjusted to pH 7.4 [12], using pig skin cuts obtained from different pigs, 600 μm thick [13] and diffusion area 1.75 ± 0.20 cm2; the skin was used under the conditions previously mentioned (TEWL less than 35 g/h/m2). A volume of 17.6 μL of the solution (10 µg/cm2) was applied in the donor compartment without occlusion to simulate the conditions of use. The exposure time was 6 h, taking 500 µL aliquots of the receiving compartment at 0.5; 1; 1.5; 2; 3; 4 and 6 h, immediately replenishing with the receiving medium. The quantification of benzoic acid was performed by HPLC-DAD as soon as the aliquots were obtained in each sampling. The results were statistically analyzed, establishing whether there was a significant difference in the maximum permeated concentrations, using the Kolmogorov Smirnov, Fisher, and t Student tests assuming a 95% confidence level.
RESULTS
Infinite doses
The diffusion and TEWL parameters were established in pig samples from freshly slaughtered pigs and after stored for 72 h in 1 M NaCl at -2 °C. Regarding the maximum flow of benzoic acid in the steady state, values of125 μg/cm2/h and 106 μg/cm2/h were obtained in pig samples at 0 h and 72 h respectively. From the permeation flows obtained, the permeation coefficient of benzoic acid (Kp) was calculated, which was 0.0544 cm/h at 0 h and 0.0461 cm/h at 72 h. According to the Kolmogorov Smirnov and Fisher tests, it was determined that the data of the permeation flows and coefficients at 0 and 72 h follow a normal distribution, as well as homoscedasticity between the variances of the samples (table 1). Through the Student t test, it was obtained that the experimental statistic (t exp) is -2.01 for the flow and permeation coefficient, with a critical value of 2.78 (t tab) and probability of presenting a t experimental < t theoretical (P (T ≤ t) of 0.114 (>0.05), so there is no statistically significant difference in permeation parameters between samples of freshly slaughtered pig skins and those stored for 72 h in 1 M NaCl at -2 °C.
Table 1 Kolmogorov Smirnov, Fisher and t Student tests applied to the kinetic parameters (flow and permeation coefficient) of freshly slaughtered and stored swine skin for 72 h.
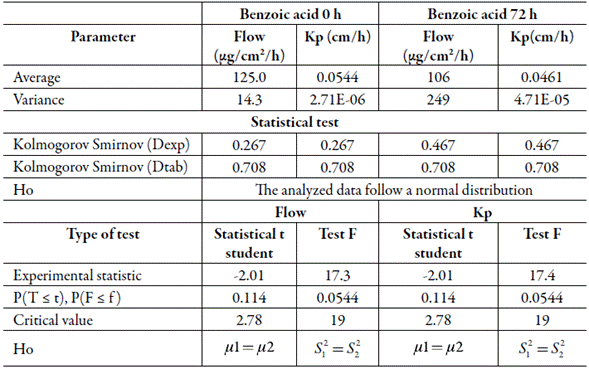
Simultaneously with the permeation test, the loss of transepithelial water (TEWL) was determined. For skins of freshly slaughtered pigs the loss of transepithelial water is 33.3 g/h/m2 and 31.7 g/h/m2 for pig samples stored for 72 h (table 2). However, after performing the statistical analysis, the Kolmogorov Smirnov, Fisher and t Student tests showed that there is no significant difference in the loss of transepithelial water, obtaining an experimental t of -0.315, a critical value of 2.78 and a probability of presenting an experimental t < theoretical t (P (T < t) of 0.768 (>0.05). In turn, the transepithelial water loss (TEWL) values in pig samples at 0 and 72 h, present a normal distribution and equality of variances (table 2)
Table 2 Kolmogorov Smirnov, Fisher, and t Student tests applied to the loss of transepithelial water (TEWL) of freshly slaughtered and stored swine skin for 72 h.
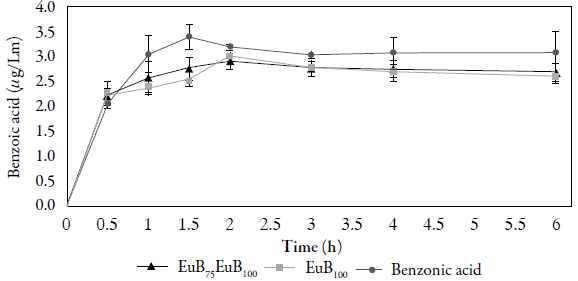
Finite dose
The maximum concentrations of benzoic acid in the E7uB75Cl25,E7uB100 and free were determined. For the complexes E7uB75Cl25 and E7uB100 the maximum concentrations of benzoic acid were 2.88 μg/mL and 2.99 μg/mL respectively, with a maximum value of 3.43 μg/mL in uncomplexed benzoic acid (table 3). The results obtained show normality in the distribution of the samples of the three compounds, an experimental value (D exp) of 0.267 and a critical value (D tab) of 0.708 for the averages of the maximum concentrations of benzoic acid in the complexes EEuB 75 Cl 25 , EauB 100 and free benzoic acid.
Table 3 Kolmogorov Smirnov test applied to the maximum concentrations ofbenzoic acid, EuB 75 Cl 2 EuB 100 complexes.
In relation to the Fisher test (table 4), it is observed that for the EuB 75 Cl 25 ,EuB100 complex the experimental F is 3.07, and 2(5.9 in the EuB100, with a critical value of 19 for both compounds, presenting homecedasticity only for the complex EuB75Cl25.
Table 4 Fisher test applied to the maximum concentrations of the complexes EuB 75 Cl 25 , EuB1 comparing equality of variances with benzoic acid.
The Student t test of equal variances was applied for EuB75Cl25, and of unequal variances for the EuB100 complex. Table 5 shows the data calculated in the Student t test; for the EuB75Cl25 comp0exthere is a statistically significant difference between the maximum concentration of the benzoic acid of the EuB 75Cl25 œmplexand that of the uncomplexed benzoic acid, the experimental t is 2.91, with a critical value of 2.78 and the probability of presenting an experimental t < theoretical t (P (T ≤ t) is 0.0435 (< 0.05).
Table 5 Student t test applied to the maximum concentrations of the complexes EuB 75Cl25 and EuB 100 comparing equality of means with benzoic acid.
In the complex ,EuB 100, the t statistic is 2.65, the critical value is 2.92 and the probability of presenting an experimental t < theoretical t (P (T ≤ t) is 0.0888 (>0.05). Therefore, there is no significant difference between the averages of concentrations of free and complexed benzoiu acid in EuB 100.
DISCUSSION
Infinite doses
In the tests carried out at infinite doses, an amount of benzoic acid was applied with which the maximum flow per unit area was reached and maintained. Under these conditions, it was possible to determine the diffusion parameters such as the permeability coefficient (Kp) and the maximum flow in the steady state [11]. In figure 4, the cumulative amount of benzoic acid (permeated amount) is observed as a function of time, whose slope represents the maximum permeation flow, from which the permeation coefficient (Kp) [14, 15] is calculated.
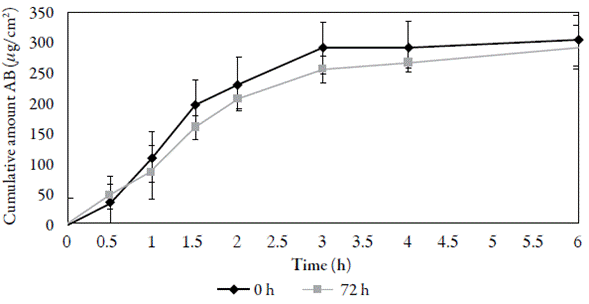
Figure 4 Permeation profile of benzoic acid (AB) using pig skin at 0 h and 72 h after the animal is slaughtered.
Simultaneously, the measurement of transepithelial water loss (TEWL) was performed, this evaluates the integrity of the skin assuming an increase in water loss when the skin is affected [10], therefore, it is necessary to guarantee optimal skin conditions in the performance of the tests avoiding excessive prediction in the permeation of the compounds. According to the values reported by the equipment manufacturers, the TEWL for healthy human skin is 10 to 25 g/h/m2; however, in the literature, values of 30.21 ± 1.27 g/h/m2 are reported for pig skin [10], data close to the TEWL of the samples used. Even so, there is a discrepancy in the values reported in other groups (2-10 g/h/m2) which may be a consequence of differences in equipment calibration methods, values reported by manufacturers and conditions of use, for these reasons, the standardization of TEWL measurement methods is required to obtain reproducible data that can be compared.
As mentioned previously, diffusion parameters and transepithelial water loss (TEWL) were used as comparison criteria to assess whether there was a significant difference in their values after performing permeation tests with ear samples obtained from freshly slaughtered pigs and with samples stored for 72 h in 1M NaCl at -2 °C. With this in mind, the statistical analysis carried with the Student t test required that the values of the samples have a normal or Gaussian distribution (Kolmogorov Smirnov test), as well as a prior determination of the homoscedasticity between the groups to be compared (Fisher test) to establish the type of test to be applied (Student t test for equal or unequal variances) [16, 17]. After performing the statistical analysis, it was evidenced that there is no significant difference in the diffusion parameters in the TEWL measurements between the pig samples of freshly slaughtered pigs and samples stored for 72 h in 1 M NaCl at -2 °C. Therefore, storage ensures proper maintenance of biological samples for permeation studies.
The storage method used is the cooling and use of a saline medium to preserve the samples. Skin cooling is a simple, cost-effective and easily available method for short-term storage as is the interest of the study, reduces the metabolic rate of cells and nutritional demands [18], inhibits bacterial proliferation and together with a medium such as NaCl allows the viability of the samples for a certain time. The use of an ion exchange preservation medium (1 M NaCl solution, -2 °C for a maximum of three days) was carried out based on previous studies of the research group, where it was shown that it has several advantages over other means of preservation (MEM, Williams, RPMI and PBS buffer), since in addition to maintaining the skin with an adequate level of integrity, it facilitates the separation of the epidermis, it has a simpler composition than other media whose components could interfere with the analytical determination [9] and in turn is cost effective.
Consequently, the tests can be carried out without the need for pigs to be slaughtered immediately, which facilitates the operational conditions for carrying out the studies and, in turn, through proper storage the structural and functional integrity of the skin is guaranteed to obtain reliable results [18].
Finite doses
Because the concentrations of benzoic acid as a preservative are used from 0.1 to 0.2% in topical formulations [19], 2 mg/mL benzoic acid solutions were used to simulate the conditions of use. In figure 5, the maximum concentrations reached by benzoic acid (free and EuB 75 Cl 25 and EuB100 complexes) are observed in permeation tests carried out over a period of 6 h.
According to the statistical analysis performed, it was obtained that the maximum permeate concentration of benzoic acid in the EuB75Cl25 complex is significantly different from that of free benzoic acid. This allows us to conclude that the EuB75Cl25 complex has a greater safety index since the permeate concentration of benzoic acid from the complex is lower than that of free benzoic acid (figure 5). For the EuB 100 complex the concentration is not significantly different from that of the uncomplexed benzoic acid, despite not presenting a statistically significant difference, it is observed in figure 5, that its safety profile could be better than that of benzoic acid without complexing, since the maximum concentration permeated in the EuB 100 is lower, being necessary to carry out additional tests that allow to conclude respect to the permeation of the same.
This difference in permeation behavior of free benzoic acid with respect to complexing can be explained by the strong electrostatic interaction between the positive charges of Eudragit® E100 and the negative charges of benzoic acid that are present in the complexes, as a consequence of the acid base reaction that is given between them for their training. This interaction hinders the diffusive process of benzoic acid, which as part of the complex is included in a macrostructure with high molecular weight, which does not easily pass through the skin, but only to the extent that benzoic acid is released from the complex in its free form, which generates a decrease in the permeation rate, compared to uncomplexed benzoic acid [20].
CONCLUSIONS
Benzoic acid is partially absorbed dermally (between 22 and 89%) and at low doses irritation symptoms may appear [3], in this sense the use of complexes with polyelectrolytes and benzoic acid could be an alternative to the current preservative (without complexing), since it would improve its safety profile by decreasing the percentage that this compound can permeate the skin, without losing its antimicrobial activity.