Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
Revista de la Facultad de Medicina
Print version ISSN 0120-0011
rev.fac.med. vol.62 no.4 Bogotá Oct./Dec. 2014
https://doi.org/10.15446/revfacmed.v62n4.45593
DOI: http://dx.doi.org/10.15446/revfacmed.v62n4.45593
REVIEW ARTICLE
Dengue disease diagnosis: A puzzle to be solved
El diagnóstico en dengue: un rompecabezas a resolver
Jaime Eduardo Castellanos1,2, Carolina Coronel-Ruiz1
1 Grupo de Virología, Universidad El Bosque. Bogotá, Colombia.
2 Grupo de Patogénesis Infecciosa, Facultad de Odontología, Universidad Nacional de Colombia. Bogotá, Colombia.
Correspondence: Jaime E. Castellanos. Carrera 9 No. 131 A-02, Bloque D, Laboratorio 205. Grupo de Virología, Universidad El Bosque. Bogotá, Colombia. Telephone: (571) 6489066. E-mail: castellanosjaime@unbosque.edu.co.
Received: 22/07/2013 Accepted: 07/07/2014
Summary
Dengue is an infection caused by dengue virus and is the most important arthropod transmitted viral disease in the world, causing near 100 million cases and 50 000 fatalities each year. Health authorities believe that these numbers will grow in coming years. In Colombia, almost 600 municipalities are in regions with Aedes aegypti circulation, and the presence of four dengue serotypes has been demonstrated. Despite the increasing knowledge about disease pathogenesis and the dengue virus, some technical or scientific difficulties with diagnosing dengue remain, negatively affecting both public health surveillance and the appropriate attention to patients in health settings and hospitals. This paper reviews the principles and developments of the current diagnostic techniques for dengue, pointing out the difficulties with making accurate dengue diagnoses and case confirmations in public health and specialized laboratories. The principles and limitations of MAC-ELISA, IgG serology, viral NS1 detection and viral isolation by cell culture are presented. In addition, the review of immunochromatography techniques (rapid diagnostic tests) that have been put forward to help the point-of-care diagnosis is proposed. This paper is intended to bring forward some points of view about the issues related to dengue diagnosis and contribute to improve the discussion surrounding the strategies and techniques needed for reducing the impact of the disease and favoring its control.
Keywords: Dengue; Diagnosis; Flavivirus; Serology; Surveillance (MeSH).
Castellanos JE, Coronel-Ruiz C. Dengue disease diagnosis: A puzzle to be solved. Rev Fac Med. 2014;62(4):617-29. http://dx.doi.org/10.15446/revfacmed.v62n4.45593.
Resumen
La infección por el virus de dengue es la enfermedad viral transmitida por insectos más importante del mundo que genera grandes gastos a los sistemas de salud. En Colombia, en más de 600 municipios está presente el mosquito vector y se ha detectado la circulación de los cuatro serotipos del virus, haciendo previsible que la incidencia y prevalencia sigan aumentando. Aunque se han hecho grandes avances y desarrollos para entender la enfermedad, existen vacíos científicos y técnicos que dificultan su correcto diagnóstico y que debilitan tanto la vigilancia en salud pública como la correcta atención en las instituciones prestadoras de salud. En este documento se revisan los principios e interpretaciones de las herramientas de laboratorio disponibles para el diagnóstico de dengue, haciendo énfasis en las dificultades existentes para la correcta definición de los casos desde el laboratorio general y especializado. Se explican los fundamentos y limitaciones de las técnicas de ELISA de captura de anticuerpos IgM, serología para IgG, detección del antígeno viral NS1, aislamiento viral en cultivos celulares y las pruebas más recientes basadas en la inmunocromatografía –pruebas rápidas–. La revisión se propone, además de aportar a la discusión sobre las dificultades para el diagnóstico en dengue, ofrecer una idea actualizada sobre cómo abordar el diagnóstico tanto en los laboratorios de salud pública como en los laboratorios especializados y contribuir así a mejorar los estándares de atención de los pacientes, reducir el impacto de la enfermedad y favorecer su control.
Palabras clave: Dengue; Diagnóstico; Flavivirus; Serología; Vigilancia (DeCS).
Castellanos JE, Coronel-Ruiz C. El diagnóstico en dengue: un rompecabezas a resolver. Rev Fac Med. 2014;62(4):617-29. http://dx.doi.org/10.15446/revfacmed.v62n4.45593.
Introduction
The dengue virus, whose official acronym is DENV, is a small virus that contains a genome of positive-sense RNA. It is responsible for the most important infectious disease transmitted by mosquitoes in the world. This disease is widely distributed through more than 100 countries, mostly in tropical and sub-tropical zones (1). More than a million cases of dengue are reported around the world each year, and the number of deaths may be approximately 50 000 (2). Dengue infection has a wide spectrum of clinical manifestations that go from infections without symptoms to fatal cases. According to the new classification from the World Health Organization (WHO), in cases where symptoms appear, they can present as an undifferentiated febrile illness (dengue), or as a severe disease (severe dengue), characterized by plasmatic extravasations, hemorrhages, and, in some cases, compromising of organs like the brain, liver, or heart (3,4).
Patients with dengue consult health institutions motivated by symptoms of high fever, headache, retrobulbar pain, arthralgia, chills, and, in some cases, rash. These signs and symptoms define a probable clinical presentation of dengue. If, in addition, the patient presents some additional sign like abdominal pain, vomiting, edema, bleeding of the mucous membranes, hepatomegaly, or thrombocytopenia, the presentation is denominated “dengue with warning signs”. A clinical presentation of “severe dengue” is diagnosed if, in addition to the previous signs, there is evidence of plasma leakage (pleural effusion, ascites, shock), or severe hemorrhages, or severe damage in an organ (for example the liver, the nervous system, the heart, the kidneys). In all of these cases, the support of both general and specialized laboratories is required so that the event goes from “probable case” to “confirmed case” (5). Although, normally, severe disease in children is considered to have the worst prognosis, indicators of severity like vomit, abdominal pain, bleeding of the mucous membranes were significantly more frequent in adults than in children according to a recent study from a cohort in Vietnam (6)
National and international organisms consider disease due to dengue to be an event of interest for public health and notifying the appropriate authorities is obligatory (7). In Colombia, the Ministry of Health, through the National Health Institute (INS, Instituto Nacional de Salud), carries out surveillance following the guidelines of the PAHO/WHO with the goal of studying the areas in which cases occur and of proposing activities for controlling the disease when outbreaks or epidemics appear. The laboratories of clinics and hospitals fulfill an important role in this task, performing IgM antibody detection tests in blood serum samples of probable cases and the results are sent to the Regional Laboratories of Public Health, who confirm a percentage of the samples and consolidate the information that they should report to the national surveillance system. Some of these samples are sent to the Virology Laboratory of the INS for virological surveillance, viral isolation, or the detection of viral RNA through RT-PCR.
The role of health personnel in the management of the cases and breakouts is more and more important, especially in the detection of cases, their evaluation, and patient follow-up, as well as in the respective notification of the surveillance systems. Several studies have been performed in which the different signs, symptoms and data obtained in clinical laboratory tests are evaluated as elements that can be used for making earlier and acurate diagnoses (8-10). Nonetheless, it is clear that the clinical diagnosis of dengue is not specific and requires an additional effort to confirm probably cases in the laboratory. The problem of diagnosis becomes more complex, taking into account that many DENV infections are asymptomatic –although they may transmit the virus— and another large percentage of the infections present with a very light febrile presentation, such that often it does not require a medical consultation (11). In this document, we review the foundations of the laboratory tests that are recommended for confirming dengue cases, and we alert readers of the difficulties to dengue infection diagnosis confirmation, putting forward the need to reinforce the criteria of the diagnostic algorithm proposed. This can be useful both for the professionals that attend to the patients and for the national system of epidemiological surveillance.
Keys for laboratory diagnosis
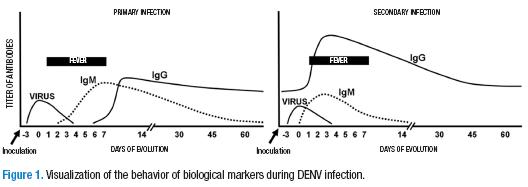
Behavior of the virus and immune response during infection
The mosquito vector — in Colombia, the species Aedes aegypti– upon feeding on the blood of an infected person, initiates the replication of the virus in its gastrointestinal system and salivary glands. When the mosquito bites a second person, it inoculates them with the virus that is present in its saliva and initiates the phase that is intrinsic of the replication of the virus in human beings. If the person comes in contact with the virus for the first time, it is called a primary infection. DENV is captured by Langerhans cells in the skin and is transported to the lymph nodes where it replicates and infects mainly the monocytes that, in turn, head toward the peripheral blood, producing more and more virus. This is called the viremic phase (12).
3 to 5 days after the mosquito bite, symptoms begin to appear. The day that fever starts is considered day 1 of the disease. The febrile phase may last from 3 to 7 days. The virus can be detected in the blood during viremia from before the temperature increase and in the first days of the fever (between days -3 and 3). During the viremic phase, DENV can be detected in 30% to 60% of attempts by amplifying the viral RNA in the blood serum (13, 17). However, the non-structural viral protein NS1 can be detected with an ELISA assay in approximately 60-80% of the tests even up to 3 or 4 days after the viremic phase since the viral protein is secreted into plasma during viremia and remains detectable even after the point in which the virus cannot be isolated (15,16).
In the acute period of the infection, the levels of IgM antibodies are undetectable in 50% of the patients. When the fever begins to abate, a period known as defervescence, in which the clinical presentation tends to solve itself, the viremia disappears, and the IgM titers increase in such a way that 93% of the patients at the sixth day are already positive and, after day 10, all patients have detectable levels of IgM antibodies (17). It is for this reason that the IgM antibody detection performed on two samples from the patient with an interval of 7 to 14 days (acute/convalescent) in which a four-fold increase in the titer can be seen in the second compared to the first is considered the definitive test for confirming a case (14).
It is important to highlight that the DENV-specific IgM antibody level remain high for 3 months. As such, their mere presence is not sufficient for confirming the diagnosis of dengue during a febrile presentation. The most frequent case of error is an infection by bacterias, parasites, or other viruses that causes a febrile presentation similar to that of dengue. Thus, if the patient had a dengue infection in the last 3 months, the IgM serology would be positive. However, it would be unrelated to the current presentation, constituting a classic false positive. Two to three weeks after the clinical presentation, an IgG-specific antibody response is generated, antibodies that can be detected by an ELISA test.
In reality, 4 different types of DENV exist (DENV-1 to DENV-4) that can infect a single patient at different times, since infection by one does not protect against infection by another serotype. In other words, a person in a hyperendemic area could suffer from up to 4 different dengue presentations over his lifetime. Worse still, it is know that having memory antibodies for one of the serotypes is a risk factor for developing severe dengue since, during an infection by a serotype that is different from that of the first infection, the virus-antibody complexes that are formed facilitate the infection of new mononuclear cells. The monocytes infected become the targets of the immune mechanisms and, upon being attacked, liberate chemical mediators that increase capillary permeability, activate the complement, secrete cytokines and chemokins, which together provoke physiopathological changes that are typical of severe dengue (18).
Later infection by a serotype different than the one that caused the primary infection is called secondary infection. It leads to a different immune response that affects the diagnostic strategy. The clinical presentation is similar to that of a primary infection, but the kinetics of the markers in the serum are different. The titer of the IgG antibodies rises rapidly, while the titer of the IgM antibodies increases, but not as much as in a primary infection (Figure 1). As occurs during a primary infection, in the acute phase of a secondary infection, the virus can be detected in the blood, either by amplification of the RNA or by the detection of the non-structural NS1 protein (19).
The viruses of the genus Flavivirus –including the Yellow Fever virus, the West Nile virus, the Japanese encephalitis virus, the Saint Louis encephalitis virus, etc.—, in their protein envelope, have similar sequences that can induce cross reactivity in the antibodies. This characteristic represents an additional problem for the serological studies of dengue. The serums that are positive for yellow fever or, even, the serums of individuals vaccinated with the live attenuated virus, generate an antibody response that is shared with dengue and that may turn out positive in tests in more than 40% of cases. This implies a further problem when evaluating serological tests for dengue (20).
Viremia can be detected, thanks to the viral antigen or the viral RNA, even before the appearance of fever and during the first days. The IgM antibodies in primary infections can remain at elevated levels for up to three months after the febrile presentation. In secondary infections, the IgG antibodies have greater affinity and are more easily detected than the IgM antibodies.
What are the diagnostic tests for dengue?
The laboratory diagnostic strategy for dengue infections is similar to the one used in other viral infections. In the case of dengue, it can be confirmed by the detection of the virus in the serum, either by detecting the soluble antigen (NS1), detecting viral RNA, or by isolating the virus in cell cultures (virological methods). A case can also be confirmed by evaluating the existence of a specific immunological response (serological methods), involving the apparition and detection of IgM antibodies in the acute phase, IgG antibodies in the convalescent phase, and neutralizing and/or inhibiting antibodies in hemagglutination (21).
Virological methods
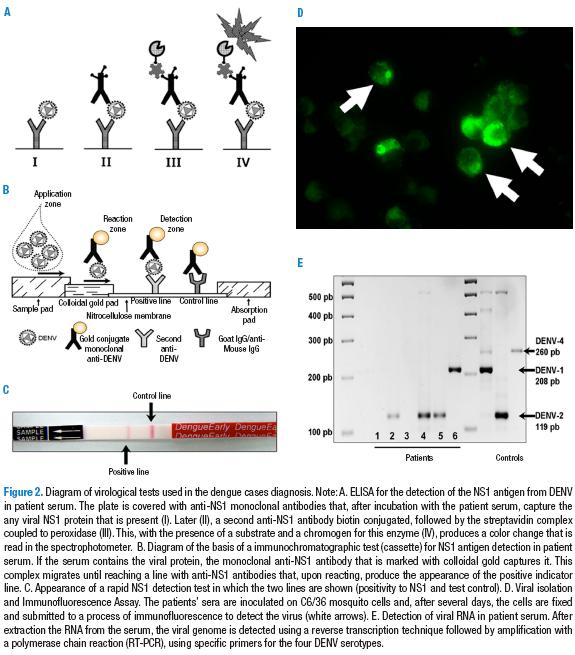
ELISA for NS1 detection
The nonstructural protein 1 (NS1) is a glycoprotein that is expressed in cells infected by DENV, either anchored to the membrane (mNS1) or freed into the extracellular medium in its soluble form (sNS1) (22). Based on this principle, an ELISA antigen-capture assay was implemented. This consists of an ELISA plate that has a monoclonal anti-NS1 antibody attached to it. The patient’s serum is placed on this plate, and, if it contains the NS1 protein, it will be recognized. After washing, it is incubated with an anti-NS1 polyclonal rabbit antibody followed by a secondary antibody coupled to the peroxidase enzyme that is responsible for generating color upon coming in contact with the substrate (hydrogen peroxide) (23). This ELISA antigen-capture assay can detect the viral antigen long before the IgM appears, and, if there is a positive result in a febrile individual, it can confirm the case of dengue.
Recently, in a previous study, the research team responsible for the present document demonstrated that 40% of serums coming from probably cases of dengue that were negative for IgM were positive for NS1. This confirms them as true positives, which makes us question the predictive value of IgM in a single serum sample (24). It is also probable that NS1 cannot be detected in samples taken from patients in days 5 to 7 after the beginning of the fever. As a result, in these cases, additional tests are required for confirmation. The detection of NS1 becomes complicated in secondary infections due to the fact that the antigen can form specific antibody complexes, which makes the capture assay difficult.
Rapid immunochromatographic NS1 test
The rapid test cassettes for the viral antigen are used more and more frequently. It allows health personnel to identify the presence of the NS1 viral protein in serum samples from febrile patients as part of the initial diagnosis in the point-of-care. The test is based on the same principle: if the viral antigen is present in the serum, it is recognized by a reactant of the system, an anti-NS1 antibody coupled with colloidal gold. This complex flows thanks to the buffer until finding and reacting with a second polyclonal anti-NS1 antibody that traps it on the test line, which can be visually perceived as a pink or purple line after a few minutes (Figure 2). The importance of this test is the possibility that it provides of detecting the presence of the virus in the serum (its soluble antigen) while the patient is febrile and at a point in which the increase in the titer of IgM antibodies is still undetectable. .
In a simultaneous evaluation of several NS1 detection systems in Colombian patients, it was found that the ELISA assay had a sensitivity of approximately 70%, while the rapid immunochromatography tests had a sensitivity of between 50% and 80%. In this evaluation SD Bioline Dengue Duo Test cassettes —which detects NS1, IgM and IgG— showed the best behavior with a sensitivity of 80,7% (25). It is important to emphasize that rapid tests are subject to the same interpretation as the rest of the serological tests, taking into account the day of the disease when the sample was taken from the patient, how it was handled, and complementary tests. The NS1 test may be negative if the sample is taken late during the febrile presentation. As such, the combination of various tests and formats is suggested for the confirmation of a case. In several reports it has been found that sensitivity is greater in primary infections than in secondary infections. This may be due to the presence of antibodies that form a complex with the NS1 protein, thereby avoiding its detection during secondary infections (26).
Detection of viral RNA en serum
For the molecular detection of DENV, a great variety of technical protocols have been designed based on blood, serum, plasma, or tissue samples with previous complementary DNA synthesis for a reverse transcription reaction (RT). The most frequently used protocol is that described by Lanciotti and cols (1992) and later modified by Chien et al. (2006). It consists of a nested RT-PCR, meaning that an amplification of a 511 pb fragment is performed using a pair of primers that recognize all of the DENV serotypes. Later, a re-amplification is done using this product and specific primers for each of the serotypes as a template. Each of the serotypes end in different sized amplicons (27,28). Due to its simplicity and ease, the RT-PCR for DENV has become the preferred method of many laboratories, since its sensitivity is much greater than that of viral isolation.
DENV diagnosis through molecular biology is more sensitive and specific. Although it is possible that false amplifications may occur (29), most specialized laboratories have the experience, the knowledge, and the equipment necessary for performing RT-PCR for DENV. Recently, real-time PCR techniques have been introduced that improve the sensitivity and specificity of molecular detection. They are also used for research into the pathogenesis of disease (30) and have been used to evaluate the viral burden of patients (31,32). The assay is based on the use of specific pairs of primers and a probe marked with fluorescent dye. The regions of viral cDNA, previously synthesized from RNA extracted from the sample, are amplified through a classic Taq polymerase reaction. The specific probes for each serotype are marked with a molecule that produces a different color. They hybridize with the cDNA region when they amplify. These probes are hydrolyzed causing a change in fluorescence that is detected by the equipment and allows the reporting of the presence of the virus in the respective sample (33). Although it is a method that is costly and requires a high degree of expertise, each day it is becomes more sensitive and rapid and less costly. As such, it is used more and more frequently.
Viral isolation and immunofluorescence
In this test, also known as viral culture, a dilution of patient serum taken during the acute phase is inoculated into a of mosquito cells culture. IF the sample is taken during viremia, the virus present in the serum or plasma infects the cells and is replicated (34). The test is also done with tissue samples in fatal cases (35). Due to the low levels of the virus in the blood, to isolate the virus, the sample must be taken early, between days 1 and 5 from the initiation of fever. Even in these cases, the success rate is 50% as a maximum, though it rarely is more than 20% to 30% (36). In general, it is a technique that requires specialized installations and personnel, that has high costs, and that provides results only 2 weeks after the initiation of the test. Thus, it is not very useful for the diagnosis and treatment of the patient. Although several types of primate cells are used for the isolation, (VERO, LLC-MK2), they are not the best for this process. In general, in dengue laboratories, patient serum is inoculated into C6/36 or AP-61 mosquito cells from the species Aedes albopictus and Aedes pseudoscutellaris respectively are used. After several days, cytopathic effects (as syncitya) are detected. An additional confirmation is always done using antibodies specific to each serotype (ATCC HB-46 to HB49) or PCR from the cell lysates (35).
Serological tests
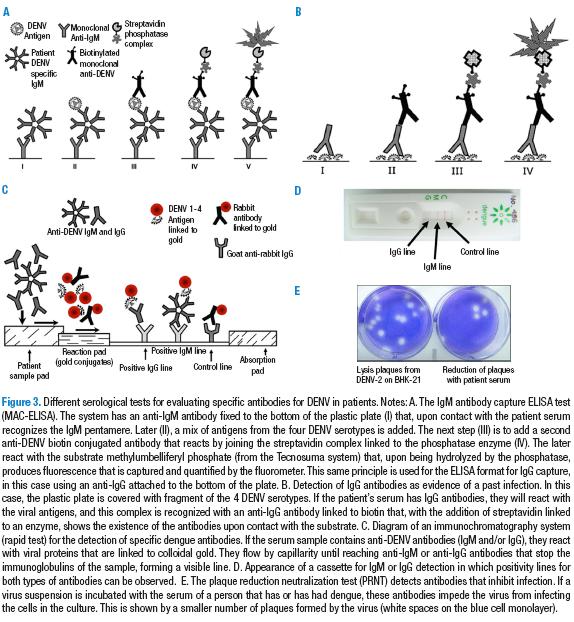
Serology for IgM
This is the primary and most frequently used methodology for diagnosis. This test (M antibody capture, MAC-ELISA) consists in an ELISA test to which an anti-IgM antibody is attached that will recognize immunoglobulin M when the problem serum is added. After incubation with a mix of antigens from the four DENV serotypes followed by the addition of a monoclonal anti-DENV antibody attached to an enzyme (phosphatase), it is incubated with the substrate that generates the signal (37) (Figure 3). Due to its relatively low cost, it is very useful both for surveillance and the study of epidemics. However, it does not work for identifying the DENV serotypes. If the evaluation is done in paired samples —obtained in the acute and convalescent phases— it can be an ideal diagnostic method. The sensitivity of the most frequently used tests is around 98% when previously confirmed dengue samples are used, and under these same conditions, the specificity is 90% on average (38). However, in field conditions, the probability that MAC-ELISA detects a case of dengue from a variety of febrile infections is approximately 60%, or less if the test is done in the first days of the fever (39). As noted above, we must insist on the difficulties of serological diagnosis due to the cross-reactivity that occurs in individuals with yellow fever or vaccinated against it (20).
Serology for IgG
For the detection of type-G immunoglobulins (pertaining to memory), two formats can be used. The first is the IgG indirect ELISA that uses a plate with DENV antigens attached onto which the problem serum is added followed by a monoclonal human anti-IgG linked to an enzyme. With the substrate, this generates a color change (increase in absorbancy). This system of indirect antibody detection is useful in studies of seroprevalence, no matter the time that has transcurred since the infection or the concentration of antibodies. That said, a different proposal is put forward in a second format: the IgG capture ELISA (40). In this case, a rabbit anti-human IgG antibody is adsorbed to the plate captures all serum IgG, and those that are specific to DENV recognize the mix of viral antigens that are added later. The next step consists of adding a monoclonal anti-DENV antibody linked to peroxidase that, upon contact with the substrate and the chromogen, causes a color change. This assay is based on the characteristics of secondary infections, where the response of IgM antibodies is slow, weak, and of a shorter duration than in primary infections; some times it is even undetectable. However, one or two days after the initiation of the symptoms, the IgGs rise rapidly to levels even higher than in the primary infection and remain at that point for more than a month. These antibodies have greater affinity, thus the cut-off point in the system is higher, allowing for a more specific diagnosis. A positive result confirms a current or recent presentation of secondary dengue. This improves the laboratory confirmation process.
Hemagglutination Inhibition Assay (HI)
The proteins from the surface of DENV have the capacity to agglutinate the erythrocytes of birds (of geese in particular) a reaction that can be observed with the naked eye.
The hemagglutination inhibition assay (HI) consists of mixing serial dilutions of patient serum with DENV antigens, and later putting the mix in contact with goose erythrocytes. If the patient has anti-DENV antibodies, they will attach to the viral proteins, impeding hemagglutination. The result is reported as the last dilution of serum that inhibits the HA. A titer of more than 1:2650 means that the concentration of anti-DENV antibodies is very high. This, in turn, means that the infection is in course or very recent. However, three months after an infection, the titer of hemagglutinating antibodies falls. Thus an HI assay will give results below 1:640 (41).Although this method has been used for a long time and has great sensitivity and specificity, it is time consuming and costly to perform. It requires a previous manipulation of the serums (to eliminate hemagglutinating agents), several dilutions of the serum must be made, and it requires the production of purified DENV antigens with different concentration and quality. For these reasons, this test is used with less and less frequency. As such, several laboratories have replaced the HI assay with the IgG capture ELISA in order to assess the increase in titers or to differentiate between primary and secondary dengue (42). That said, recently the monoclonal antibody based inhibition ELISA has been used. It is just as sensitive as the HI assay and has shown its usefulness in the laboratory (43).
Immunochromatographic tests (rapid tests cassettes for IgM and IgG antibodies)
The rapid tests for dengue —lateral-flowing immunochromatography—, has been developed with the goal of aiding in the diagnosis in sites where patients are attended. The principle is similar to that which was explained for the rapid NS1 test. In the IgM or IgG antibody detection cassettes, the system contains antigens of the 4 DENV serotypes linked to nanoparticles of colloidal gold and, as a control, rabbit IgG gold coupled.
The anti-DENV antibodies in the problem serum recognize the gold coupled DENV antigens and migrate along the nitrocellulose paper until it reaches a band to which anti-IgG or anti-IgM antibodies are attached. This band traps the respective antibodies that are flowing that, since they are attached to the viral antigen linked to the colloidal gold nanoparticles, can be seen with the naked eye as a pink or purple line (44). The control, a rabbit IgG linked to gold, continues to flow until finding its anti-antibody and stops to show the positive control line that indicates that the procedure was correct. In this way, a qualitative result can be found in less than 20 minutes regarding the presence of IgM or IgG in the patient’s serum. For example, in a study with 350 serums, the different rapid tests for IgM antibodies had a sensitivity of between 20% and 95%, with specificities of between 80% and 90%. A 40% of false positives presented in the serums of patients with malaria and cross-reactions with serums that were IgG positive for dengue (38).
Plaque reduction neutralization test
This assay is the most specific of the serological methods and has been used when high specificity detection of anti-dengue antibodies in a sample from the convalescent phase is required.
The PRNT is a functional assay theat measures the relative quantity of antibodies with the capacity of neutralizing the infection and reducing the number of infecting viral particles, thereby reducing the number of lytic plaques in a cell monolayer. With this method, it is possible to specifically quantify the titer of neutralizing antibodies for each of the 4 serotypes of DENV in the individual serum (45).
The test is based on incubating serial dilutions of the patient’s serum with a defined suspension of each of the DENV serotypes. In this way, the antibodies attach to the virus that later places itself on a cellular monolayer. If the virus is free (there are no antibodies), it infects the cells and produces plaques in the cell layer (holes) that can be easily counted. The number of plaques reduces as the concentration of the antibody increases (less dilution of the serum). The result is expressed as the dilution of serum that reduces 50% of the number of plaques. The great advantage of this method is that it can detect with which serotypes of the virus the individual has had contact, showing the specific “infection history”. It is the method used to quantify the protection in individuals immunized with vaccines that are being tried currently and, in addition, it is very helpful in seroepidemiology and in studies related to the immunopathogenesis of severe dengue (46). Despite its great robustness as a diagnostic method, it is a technique that is difficult to develop, having high costs and requiring great expertise and elements like viruses and cells that are all critical for obtaining correct and comparable results.
Due to the difficulties associated with this technique, recently a method was developed that uses the Raji lymphoblast cell line that is infected with fluorescent viruses. In this way, the reduction in the number of cells infected when each viral serotype is treated with dilutions of patient serum can be seen through flow cytometry. This technique, though costly, allows for the assessment of antecedents of infection of a great number of samples (47).
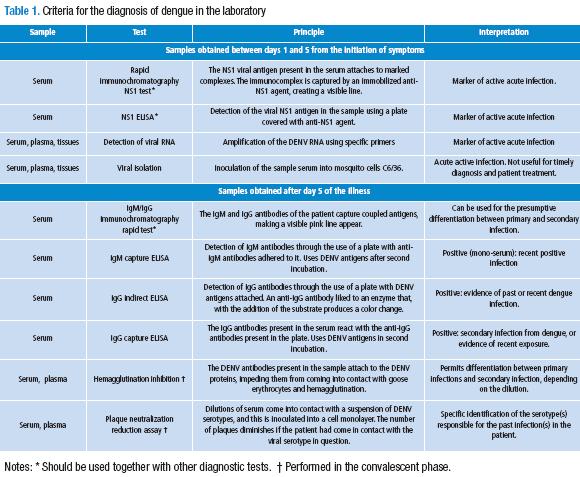
In Table 1, a summary of the different diagnostic tests and their meaning is shown.
The puzzle of dengue diagnosis
The early identification of dengue infection is a key element in providing timely care for its complications that usually occur around the 5th day of the disease (48). Therefore, delays in the diagnosis can affect the prognosis, since the inadequate management of these patients in early days (due to a poor diagnosis) could lead to late detection and serious or even fatal complications.
One of the main problems with the management of dengue in endemic zones is the difficulty associated with distinguishing it in a timely fashion from other infections with a similar febrile presentation, such as influenza, diarrheal diseases, measles, typhoid fever, malaria, yellow fever, and leptospirosis to name a few (49,50). Surveillance and diagnosis of dengue using only clinical criteria have a very low sensitivity and should be accompanied by a laboratory diagnostic process both of the health institution and of specialized laboratories (Colombian regional laboratories and the National Health Institute). In this review, each of the recommended tests for confirming a case of dengue in the laboratory have been described. As can be seen, some of these tests require trained personnel, specialized equipment, and special resources. The timely taking and appropriate handling of the clinical samples is also important, something that ensures higher sensitivity and specificity in the tests. As described earlier, the confirmation of the dengue cases presents many difficulties that should be strictly evaluated to ensure the attainment of a result (51).
These difficulties are related to the fact that there are 4 serotypes of DENV that can infect a patient at any point in time. It is well known that the antibodies of one serotype are only weakly effective against other serotypes. In fact, they function as opsonizing antibodies that increase the proportion of infected cells and as a result, the cytokine response. In the same way, diagnosis is made difficult by the cross-reaction that the anti-DENV antibodies have against other flaviviruses, such as the yellow fever virus and even the vaccine virus. The short viremia period also makes detection of the antigen or of viral RNA difficult in the acute phase. The most sensitive and specific test is assessment through the ELISA or the HI seroconversion or increase antibody titer tests, but it requires the analysis of two samples taken with an interval greater than 7 days (acute vs. convalescent). Lastly, the long-term persistence of memory antibodies may falsify the tests during acute infections.
Considering this panorama and the interest in confirming the cases in the laboratory, we have proposed various elements that may improve diagnoses. Operatively, a “probable case” is defined as an acute febrile episode with dengue symptoms (warning signs may appear) or severe cases with evidence of plasma leakage, severe hemorrhages, and/or damage in other organs (7). A “confirmed case” would be a probable case that has additional positive serological or virological evidence.
A positive result can be established if the viral protein NS1 is found (through the ELISA or immunochromatography test) or by amplifying the viral RNA through RT-PCR. This is possible in patients with less than 5 days of evolution, although the shorter the time since the initiation of the fever, the greater the probability of detection. The most specific test, the less sensitive, is the “viral isolation” test, however, it requires inoculating the patient’s serum into cell cultures and later confirming the diagnosis through fluorescence with monoclonal antibodies or by RT-PCR amplification. Due to the low sensitivity of these three techniques, a negative result does not rule out the dengue diagnosis. Therefore, whenever possible, it is recommended that at least two of the tests should be run simultaneously.
The gold standard test in the diagnosis is considered to the be the capture ELISA for the detection of IgM or IgG antibodies in two samples from the same patient taken with an interval of at least seven days. This test, however, may not be very useful for the patient. In this case, it can be shown that the serum from the acute phase is negative (or with a low absorbence signal) and that the sample from the convalescent phase shows the signal (seroconversion) or the signal increases significantly. In the great majority of the cases, only one sample of the patient is available (single serum) taken during the acute phase. If this single sample is positive for anti-dengue IgM or IgG capture, the case is considered “undetermined” unless a RT-PCR or NS1 detection test is done. Unfortunately, this is the most frequent state of definition of cases in Colombia. Frequently, in dengue endemic zones, there are patients that consult due to a febrile syndrome that is compatible with the symptoms of dengue and present a positive IgM serology. It is impossible with this single test to know if the febrile case is due to DENV, since the positive serology of a single serum could be due to a presentation of dengue that occurred in the last three months. This interpretation is valid both for the cases studied through IgM serology and for the rapid immunochromatographic test for IgM detection.
In cases of deceased patients who had a dengue compatible clinical presentation, the detection of the viral antigen by immunohistochemistry or the amplification of the viral RNA is recommended in samples of tissue fragments obtained from autopsies. Finally, the laboratory diagnostic must be complementary to a clinical evaluation that systematically investigates the disease through the signs and symptoms, as proposed by Díaz-Quijano et al. (2006). In their study, a score was assigned to each sign or symptom, noticeably improving the sensitivity and specificity of the process (42.4% and 96%, respectively) (8). In this way, a better clinical approach also favors laboratory confirmation, provides the benefit of immediate attention to the patient, and strengthens the institutional surveillance because can be avoided the variation dependent on the cases definition or the day when the samples were taken.
It is expected that the expression of recombinant viral proteins could improve the specificity of the new systems by avoiding the epitopes shared by flaviviruses. Due to the great variability of the sensitivity and specificity values that are reported in the publications, a call has been made to the researches to evaluate the existing diagnostic systems in multi-center field tests. In view of the fact that serious cases are on the rise, we also suggest that the circulating serotypes be identified along with primary cases versus secondary cases, since this will positively affect epidemiological surveillance. Unfortunately, in order to evaluate the current diagnostic systems, reactants are needed (viruses, antigens), as well as standardized positive and negative serums, that can be used for the standardization of the country’s diagnostic laboratories. This, without counting the difficulties associated with the more complex tests, like molecular detection and typification through RT-PCR or viral isolation in cell cultures, tests which should be standardized in more laboratories in order to widen the coverage of specific diagnoses.
The research groups and the companies that produce diagnostic kits are moving toward the development of more sensitive and specific systems. Nevertheless, it is also important that they have a lower cost, allowing health institutions in endemic zones to have access to the products, and to be able to further the tests and support the diagnosis of the cases. The new systems have the challenge of aiming toward earlier diagnosis of the individuals and eliminating interference from the response of other flaviviruses. New systems must be able to make a serological diagnosis of the infecting serotype and allow for genomic studies of the viruses or even the assessment of the viral burden. It also is proposed that new systems should be able to work with different kinds of samples (plasma, saliva, urine, etc.)
In addition, tests for the assessment of biomarkers are in development. These tests could be associated with a better prognosis and aftercare. Interested laboratories should start to share information, reactants, antibodies, positive and negative control serums, viral isolates, and cell cultures in order to standardize robust and appropriate processes for the existing diagnostic kits, and for those of the future (52).
Conclusions
Despite the advances in most recent years in immunological, virological, molecular, and cellular techniques, objective difficulties still exist with regard to the confirmation of dengue cases. Operative problems range from, for example, the appropriate time to take a sample, to its handling and transport to the laboratory, to the tests to be performed depending on the laboratory’s level of expertise and the time required for the results to be ready. Furthermore, the difficulty of a highly specific clinical diagnosis, and the little experience of first and second level laboratories with the topic, should be dealt with so as to create new spaces where the task of integral diagnosis can be performed.
The IgM capture ELISA should become a routine technique in all hospitals and clinics, taking advantage of the fact that it is included among the benefits of the health insurance system. The use of rapid tests in care centers (since they have not shown greater sensitivity than the ELISA test) should be accompanied by correct training of the personnel for the identification of the signs and symptoms of a mild clinical presentation and warning signs. With increased incidence, an effort should be made to collect and process samples with serological and virological techniques in specialized laboratories in order to verify the real magnitude of the disease in the country and contribute to its control. As can be seen here, this is an effort that includes all actors of the general health system, and tasks should be set out that lead us to better dengue diagnoses.
Conflict of interests
None declared by the authors.
Financing
Study carried out as part of the program “Multidisciplinary Research Network for the Preventiona and Control of Vector-Borne Diseases”, financed by Colciencias (Contract 360-2011) and the Universidad El Bosque.
Acknowledgments
To Dr. Myriam Velandia Romero and Dr. Sigrid Camacho Ortega of the Virology Group at the Universidad El Bosque for reviewing the manuscript.
References
1. Velandia ML, Castellanos JE. Virus del dengue: estructura y ciclo viral Dengue virus : structure and viral cycle. Infectio [Internet]. 2011;15:33-43 [cited 2013 Sept 14]. Available from: http://goo.gl/CQ6vre. [ Links ]
2. Guzman MG, Halstead SB, Artsob H, Buchy P, Farrar J, Gubler DJ, Hunsperger E, et al. Dengue: a continuing global threat. Nat Microbiol Rev [Internet]. 2010;8:S7-16 [cited 2013 Sept 14]. Available from: http://goo.gl/9w6Bfc. [ Links ]
3. Organización Mundial de la Salud. Dengue. Guias para el diagnóstico, tratamiento, prevención y control [Internet]. 2009 [cited 2014 Aug 12]. Available from: http://goo.gl/R45ATw. [ Links ]
4. Barniol J, Gaczkowski R, Barbato EV, da Cunha R V, Salgado D, Martínez E, et al. Usefulness and applicability of the revised dengue case classification by disease: multi-centre study in 18 countries. BMC Infect Dis [Internet]. 2011;11:1-12 [cited 2013 Sept 14]. Available from: http://goo.gl/cDElGs. [ Links ]
5. Horstick O, Jaenisch T, Martinez E, Kroeger A, See LL, Farrar J, et al. Comparing the Usefulness of the 1997 and 2009 WHO Dengue Case Classification: A systematic literature review. Am J Trop Med Hyg [Internet]. 2014; 91 [cited 2014 Oct 6]. Available from: http://goo.gl/nBKJjt. [ Links ]
6. Dinh-The T, Le-Thi-Thu T, Nguyen-Minh D, Tran-Van N, Tran-Tinh H, Nguyen-Van-Vihn C, et al. Clinical features of dengue in a large Vietnamese cohort: intrinsically lower platelet counts and greater risk for bleeding in adults than children. PLoS Negl Trop Dis [Internet]. 2012;6:e1679 [cited 2013 Sept 17]. Available from: http://goo.gl/VAV3LP. [ Links ]
7. Ministerio de la Protección Social, Instituto Nacional de Salud, Organización Panamericana de la Salud. Guía de atención clínica integral del paciente con dengue [Internet]. Bogotá: INS y Ministerio de la Protección Social; 2010 [cited 2014 March 23]. Available from: http://goo.gl/8c0DMM. [ Links ]
8. Martínez-Vega RA, Díaz-Quijano FA, Villar-Centeno LA. Dificultad para el diagnóstico clínico temprano del dengue en un área endémica y su impacto sobre el manejo médico inicial. Rev Med Chil [Internet]. 2006;134:53-60 [cited 2013 Sept 17]. Available from: http://goo.gl/3Vaylp. [ Links ]
9. Díaz A, Kourí G, Guzmán M, Lobaina L, Bravo J, Ruiz A, et al. Description of the clinical picture of dengue hemorrhagic fever/dengue shock syndrome (DHF/DSS) in adults. Bull Pan Am Health Organ. 1988;22:133-44. [ Links ]
10. Kaushik A, Pineda C, Kest H. Diagnosis and management of dengue fever in children. Pediatr Rev [Internet]. 2010;31:E28-35 [cited 2013 Dec 9]. Available from: http://goo.gl/pjkfBC. [ Links ]
11. Endy TP, Chunsuttiwat S, Nisalak A, Libraty DH, Green S, Rothman AL, et al. Epidemiology of inapparent and symptomatic acute dengue virus infection: a prospective study of primary school children in Kamphaeng Phet, Thailand. Am J Epidemiol [Internet]. 2002;156:40-51 [cited 2013 Sept 17]. Available from: http://goo.gl/vEQBLj. [ Links ]
12. Rothman AL, Medin CL, Friberg H, Currier JR. Immunopathogenesis versus protection in dengue virus infections. Curr Trop Med Rep [Internet]. 2014;1:13-20 [cited 2014 septiembre 18]. Available from: http://goo.gl/ITqxMt. [ Links ]
13. Yamada KI, Takasaki T, Nawa N, Kurane I. Virus isolation as one of the diagnostic methods for dengue virus infection. J Clin Virol [Internet]. 2002;24:203-9 [cited 2013 Sept 17]. Available from: http://goo.gl/gzMnW2. [ Links ]
14. Weg CAM Van De, Gorp ECM Van, Supriatna M, Soemantri A, Osterhaus ADME, Martina BEE. Evaluation of the 2009 WHO Dengue case classification in an Indonesian pediatric cohort. Am J Trop Med Hyg [Internet]. 2012;86:166-70 [cited 2013 Sept 19]. Available from: http://goo.gl/eI2U21. [ Links ]
15. Hang VT, Nguyet NM, Trung DT, Tricou V, Yoksan S, Minh N, et al. Diagnostic accuracy of NS1 ELISA and lateral flow rapid tests for dengue sensitivity, specificity and relationship to viraemia and antibody responses. PLoS Negl Trop Dis [Internet]. 2009;3:1-7 [cited 2013 Sept 13]. Available from: http://goo.gl/dFXoca. [ Links ]
16. Huhtamo E, Hasu E, Uzcátegui NY, Erra E, Nikkari S, Kantele A, et al. Early diagnosis of dengue in travelers: comparison of a novel real-time RT-PCR, NS1 antigen detection and serology. J Clin Virol [Internet]. 2010;47:49-53 [cited 2013 Sept 13]. Available from: http://goo.gl/9pLGed. [ Links ]
17. Kuno G, Gómez I, Gubler DJ. An ELISA procedure for the diagnosis of dengue infections. J Virol Methods [Internet]. 1991;33:101-13 [cited 2013 Aug 28]. Available from: http://goo.gl/DJdmP4. [ Links ]
18. Houghton-Triviño N, Salgado DM, Rodríguez JA, Bosch I, Castellanos JE. Levels of soluble ST2 in serum associated with severity of dengue due to tumour necrosis factor alpha stimulation. J Gen Virol [Internet]. 2010;91:697-706 [cited 2013 Aug 28]. Available from: http://goo.gl/BiLbgY. [ Links ]
19. Shu P, Huang J. Current advances in dengue diagnosis. Clin Diagn Lab Immunol [Internet]. 2004;11:642-50 [cited 2013 Aug 28]. Available from: http://goo.gl/0gB0Fa. [ Links ]
20. Houghton-Triviño N, Montaña D, Castellanos JE. Dengue-yellow fever sera cross-reactivity; challenges for diagnosis. Rev Salud Publica (Bogotá) [Internet]. 2008;10:299-307 [cited 2013 Aug 28]. Available from: http://goo.gl/gP1U8U. [ Links ]
21. Oliveira de Paula S, Lopes da Fonseca BA. Dengue: A review of the laboratory test a clinician must know to achieve a correct diagnosis. Braz J Infect Dis [Internet]. 2004;8:390-8 [cited 2013 Aug 28]. Available from: http://goo.gl/xEnMLV. [ Links ]
22. Young PR, Hilditch PA, Bletchly C, Halloran W. An antigen capture enzyme-linked immunosorbent assay reveals high levels of the dengue virus protein NS1 in the sera of infected patients. J Clin Microbiol [Internet]. 2000;38:1053-7 [cited 2013 Aug 30]. Available from: http://goo.gl/fBkML1. [ Links ]
23. Alcon S, Talarmin A, Debruyne M, Falconar A, Deubel V, Flamand M, et al. Enzyme-Linked Immunosorbent Assay specific to dengue virus type 1 nonstructural protein ns1 reveals circulation of the antigen in the blood during the acute phase of disease in patients experiencing primary or secondary infections. J Clin Microbiol [Internet]. 2002;401-7 [cited 2013 Aug 28]. Available from: http://goo.gl/zk173d. [ Links ]
24. Prada Arismendy J, Buitrago JA, Beltrán J, Chavarro OL, Castellanos JE. Evaluación del valor diagnóstico de la detección de NS1 en pacientes con dengue agudo. Revista Salud Bosque [Internet]. 2012;2:7-16 [cited 2013 Aug 28]. Available from: http://goo.gl/LDNxaA. [ Links ]
25. Osorio L, Ramirez M, Bonelo A, Villar LA, Parra B, Dengue P, et al. Comparison of the diagnostic accuracy of commercial NS1-based diagnostic tests for early dengue infection. Virol J [Internet]. 2010;7:361 [cited 2013 Dec 9]. Available from: http://goo.gl/4ZlSQc. [ Links ]
26. Blacksell SD. Commercial dengue rapid diagnostic tests for point-of-care application: recent evaluations and future needs? J Biomed Biotechnol [Internet]. 2012;2012:1-12 [cited 2013 Sept 17]. Available from: http://goo.gl/1A7Zu6. [ Links ]
27. Lanciotti RS, Calisher CH, Gubler DJ, Chang G-J, Vorndamt VA. Rapid detection and typing of dengue viruses from clinical samples by using reverse transcriptase-polymerase chain reaction. J Clin Microbiol [Internet]. 1992;30:545-51 [cited 2013 Sept 17]. Available from: http://goo.gl/0cnyjn. [ Links ]
28. Chien L, Liao T, Shu P, Gubler DJ, Chang GJ, Chien L, et al. Development of real-time reverse transcriptase PCR assays to detect and serotype dengue viruses. J Clin Microbiol [Internet]. 2006;44:1295-304 [cited 2013 Sept 17]. Available from: http://goo.gl/zN22mw. [ Links ]
29. Wiwanitkit V. Dengue fever: diagnosis and treatment. Expert Rev Anti Infect Ther [Internet]. 2010;8:841-5 [cited 2013 Sept 17]. Available from: http://goo.gl/EGhWsI. [ Links ]
30. Prada Arismendy J, Castellanos JE. Real time PCR. Application in dengue studies. Colombia Médica [Internet]. 2011;42:243-58 [cited 2013 Sept 17]. Available from: http://goo.gl/adl59k. [ Links ]
31. Houghton-Triviño N, Martín K, Giaya K, Rodríguez JA, Bosch I, Castellanos JE. Comparación de los perfiles de transcripción de pacientes con fiebre de dengue y fiebre hemorrágica por dengue que muestra diferencias en la respuesta inmunitaria y claves en la inmunopatogénesis. Biomédica [Internet]. 2010;30:587-97 [cited 2014 Sept 17]. Available from: http://goo.gl/f4tmJ4. [ Links ]
32. Waggoner JJ, Abeynayake J, Sahoo MK, Gresh L, Tellez Y, Gonzalez K, et al. Single-reaction, multiplex, real-time RT-PCR for the detection, quantitation, and serotyping of dengue viruses. PLoS Negl Trop Dis [Internet]. 2013;7:1-9 [cited 2014 Aug 23]. Available from: http://goo.gl/Jfbmju. [ Links ]
33. Santiago GA, Vergne E, Quiles Y, Cosme J, Vazquez J, Medina JF, et al. Analytical and clinical performance of the CDC real time RT-PCR assay for detection and typing of dengue virus. PLoS Negl Trop Dis [Internet]. 2013;7:e2311 [cited 2014 Sept 13]. Available from: http://goo.gl/OouxcE. [ Links ]
34. Kao CL, King CC, Chao DY, Wu HL, Chang GJ. Laboratory diagnosis of dengue virus infection: current and future perspectives in clinical diagnosis and public health. J Microbiol Immunol Infect [Internet]. 2005;38:5-16 [cited 2014 Sept 18]. Available from: http://goo.gl/iDnyoj. [ Links ]
35. Medina F, Medina JF, Colon C, Vergne E, Santiago GA, Muñoz-Jordán JL. Dengue Virus: isolation, propagation, quantification and storage. Curr Protoc Microbiol. 2012;15D.2.1-15D.2.24. [ Links ]
36. Gubler DJ, Suharyono W, Tan R, Abidin M, Sie A. Viraemia in patients with naturally acquired dengue infection. Bull World Health Organ [Internet]. 1981;59:623-30 [cited 2013 Aug 28]. Available from: http://goo.gl/rZHCVw. [ Links ]
37. Nawa M, Takasaki T, Yamada K. Development of dengue IgM-capture enzyme-linked immunosorbent assay with higher sensitivity using monoclonal detection antibody. J Virol Methods [Internet]. 2001;92:65-70 [cited 2013 agosto 23]. Available from: http://goo.gl/UyhSsZ. [ Links ]
38. Hunsperger EA, Yoksan S, Buchy P, Nguyen VC, Sekaran SD, Enria D, et al. Evaluation of commercially available anti-dengue virus immunoglobulin M tests. Emerg Infect Dis [Internet]. 2009;15:436-40 [cited 2013 Sept 17]. Available from: http://goo.gl/4vzGXK. [ Links ]
39. De Oliveira-Poersch C, Pavoni DP, Queiroz MH, de Borba L, Goldenberg S, dos Santos CN, et al. Dengue virus infections: comparison of methods for diagnosing the acute disease. J Clin Virol [Internet]. 2005;32:272-7 [cited 2013 Sept 19]. Available from: http://goo.gl/DWhB7c. [ Links ]
40. Cuzzubbo AJ, Vaughn DW, Nisalak A, Solomon T, Kalayanarooj S, Aaskov J et al. Comparison of PanBio Dengue Duo IgM and IgG capture ELISA and Venture Technologies Dengue IgM and IgG Dot Blot. J Clin Virol [Internet]. 2000;16:135-44 [cited 2013 Sept 19]. Available from: http://goo.gl/3zAyJD. [ Links ]
41. Gubler DJ. Serologic diagnosis of dengue/dengue haemorrhagic fever. Dengue Bulletin [Internet]. 1996;20:20-3 [cited 2013 Aug 23]. Available from: http://goo.gl/tz8lyK. [ Links ]
42. Chungue E, March G, Plichart R, Boutin JP, Roux J. Comparison of immunoglobulin G enzyme-linked immunosorbent assay (IgG-ELISA) and haemagglutination inhibition (HI) test for the detection of dengue antibodies. Prevalence IgG-ELISA antibodies in Tahiti. Trans R Soc Trop Med Hyg [Internet]. 1989;83:708-11 [cited 2013 Sept 17]. Available from: http://goo.gl/AS1djg. [ Links ]
43. Vázquez S, Bravo JR, Pérez AB, Guzmán MG. ELISA de inhibición. Su utilidad para clasificar un caso de dengue. Rev Cubana Med Trop [Internet]. 1997;49:108-12 [cited 2013 Aug 23]. Available from: http://goo.gl/Dgnri4. [ Links ]
44. Cuzzubbo AJ, Endy TP, Nisalak A, Kalayanarooj S, Vaughn DW, Ogata SA, et al. Use of recombinant envelope proteins for serological diagnosis of dengue virus infection in an immunochromatographic assay. Clin Diagn Lab Immunol [Internet]. 2001;8:1150-5 [cited 2013 Sept 17]. Available from: http://goo.gl/Kbr3FF. [ Links ]
45. Thomas SJ, Nisalak A, Anderson KB, Libraty DH, Kalayanarooj S, Vaughn DW, et al. Dengue plaque reduction neutralization test (PRNT) in primary and secondary dengue virus infections: How alterations in assay conditions impact performance. Am J Trop Med Hyg [Internet]. 2009;81:825-33 [cited 2014 Oct 18]. Available from: http://goo.gl/3cohWy. [ Links ]
46. Roehrig JT, Hombach J, Barrett ADT. Guidelines for plaque-reduction neutralization testing of human antibodies to dengue viruses. Viral Immunol [Internet]. 2008;21:123-32 [cited 2013 Sept 23]. Available from: http://goo.gl/eI4jFx. [ Links ]
47. Mattia K, Puffer BA, Williams KL, González R, Murray M, Sluzas E, et al. Dengue reporter virus particles for measuring neutralizing antibodies against each of the four dengue serotypes. PLoS One [Internet]. 2011;6:e27252 [cited 2014 Oct 21]. Available from: http://goo.gl/KuSsBA. [ Links ]
48. Martínez-Vega RA, Díaz-Quijano FA, Coronel-Ruiz C, Yebrail S, Villar-Centeno LA. Evaluación de la utilidad de la prueba rápida de casete por inmunocromatografía para el diagnóstico de dengue en una región endémica colombiana. Biomédica [Internet]. 2009;29:616-24 [cited 2013 Aug 12]. Available from: http://goo.gl/uaW7gM. [ Links ]
49. Díaz-Quijano FA, Martínez-Vega RA, Villar-Centeno LA. Criterios clínicos para diagnosticar el dengue en los primeros días de enfermedad. Biomédica [Internet]. 2006;26:22-30 [cited 2013 Jul 16]. Available from: http://goo.gl/nyW7CT. [ Links ]
50. Gubler DJ. Dengue, urbanization and globalization: the unholy Trinity of the 21th century. Trop Med Health [Internet]. 2011;39:1-9 [cited 2013 Sept 17]. Available from: http://goo.gl/sXVWY7. [ Links ]
51. Shu P, Chen L, Chang S, Su C, Chien L, Chin C, et al. Dengue virus serotyping based on envelope and membrane and nonstructural protein NS1 serotype-specific capture immunoglobulin M enzyme-linked immunosorbent assays. J Clin Microbiol [Internet]. 2004;42:2489-94 [cited 2013 Sept 17]. Available from: http://goo.gl/zxZXRW. [ Links ]
52. Peeling RW, Artsob H, Pelegrino JL, Buchy P, Cardosa MJ, Devi S, et al. Evaluation of diagnostic tests: dengue. Nat Rev Microbiol [Internet]. 2010;8:S30-7 [cited 2013 Sept 17]. Available from: http://goo.gl/A3GcSW. [ Links ]