Introducción
Adolescence is characterized not only by psychological maturity 1 but also by psychophysiological changes 2,3, which are important for optimal performance and social functioning. Furthermore, it is a determining period for the consolidation of self-control and regulation related with adequate social and personal welfare 4. However, disorders in optimal maturation, and inability of behavioral adaptation and plan elaboration could be associated to the outcome and manifestation of risk behaviors related to health 5-7.
Psychophysiological emotional response is not a basic process, but a complex function, which involves multiple cognitive and physiological processes for its elaboration, including the central and peripheral nervous systems 8,9. Historically, important findings can be identified in the psychophysiology of emotional response such as Walter Cannon, Klüver and Bucy, among other more recent studies, which are related to the amygdala as a center for emotions in the brain 10-14. Such response can be measured using different physiological sources 15,16 such as electromyography (EMG) 16, temperature, skin conductance (SC), heart rate (HR) 16-19, and electroencephalography 20.
Studies on psychophysiology suggest that emotional response can be measured through a wide induction domain. According to some authors, this response can be classified in four basic aspects: a) perception, including images, sounds, words, shock, smelling, among others; b) imagery, which could consist of emotional expression or text induced imagery; c) anticipation, consisting of punishment threat, cued reward, or gambling tasks, and d) actions, such as giving a speech, driving a car, parachuting, among others 21.
During adolescence, a cascade of endocrinal changes affect the function of the brain, and as a result, self-regulation is affected. Additionally, control 22 systems, such as the hypothalamic-pituitary-gonadal, which remained passive in previous years, become increasingly active with the beginning of the activation of reproductive functions during puberty 23. Furthermore, it has been proved that reactivity to cortisol also increases in adolescents with low-restriction in stressing situations 24, and that dysfunction in these systems could lead to psychiatric disorders 25, or to the risk behaviors in which adolescents frequently get involved such as drug use or abuse, accidents as result of impulsive behavior, or even death 1,26,27.
Previous studies in children with Attention Deficit Hyperactivity Disorder (ADHD), a disorder that includes impulsivity as one of the main symptoms 28, reported two autonomic abnormal reactions related to emotional induction or suppression 29. The first was related to respiratory arrhythmia, and the second to an altered response in cardiac pre-ejection, suggesting a parasympathetic inefficiency according to the authors. The objective of this work was to compare specific characteristics of psychophysiological emotional response among adolescents with risk behavior and a control group.
Methods
Participants
The subjects included in this study, RB and control group, were directly invited to participate. RB participants were adolescents who, according to their high school teachers and school psychologists, presented one or more of the behaviors of interest of this study (impulsivity, risk sexual behavior, alcohol, tobacco and drug use, and unhealthy physical inactivity). 44 adolescents were included in the study (n=44), with a mean age of 13.7 years (SD 1.36), 63.6% male, 36.4% female, all right handed, without history of psychological, psychiatric or neurological disorders, and 7.93±0.99 mean years of regular public middle school and high school education in rural areas of Baja California in México. The Montreal Cognitive Assessment was applied prior to each experiment to discard cognitive disabilities, and both parents and participants read and signed an informed consent.
Instruments and Materials
Risk behavior measures
All participants were subjected to a brief cognitive assessment, measured with the Montreal Cognitive Assessment (MoCA) to ensure the minimum executive performance needed for this study. The 60 items of the Risk Behavior Questionnaire (RB) created by the Centers for Disease and Prevention 30, translated into Spanish and adapted to the Mexican population, was used to identify the participants with higher RB. The dimensional groups were: a) impulsive behaviors that lead to accidents, b) alcohol and tobacco use; c) drugs use, d) sexual risk behavior that leads to undesired pregnancy or sexual transmission disease, and e) physical inactivity.
Psychophysiological emotional response measures
Time domain analysis of heart rate (HR) was used to calculate variance between R-R intervals in the electrocardiogram in bits per minute, skin conductance (SC) expressed in microsiemens, electromyiogram (EMG) expressed in mV, and temperature (Temp) in Fahrenheit degrees. Moreover, signals were also measured using a 12 channel Physiolab J&J Engineering with the Physiopilot USE-3 software.
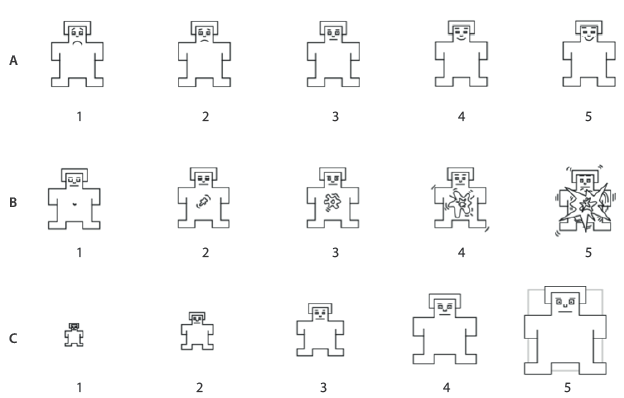
Emotional responses were produced by presenting emotional stimuli of the International Affective Picture System (IAPS) 31. Visual stimuli were divided in 18 pleasant, 18 neutral and 18 unpleasant stimuli in randomized order. Stimuli were presented in a 27-inch monitor (Figure 1). To measure the emotional valence assigned to each visual stimuli, the participants had to respond to the three scales from the Self-Assessment Manikin (SAM) in three different levels: pleasure, arousal, and dominance (Figure 2). The EEGxProc free software was used to present the visual stimuli.
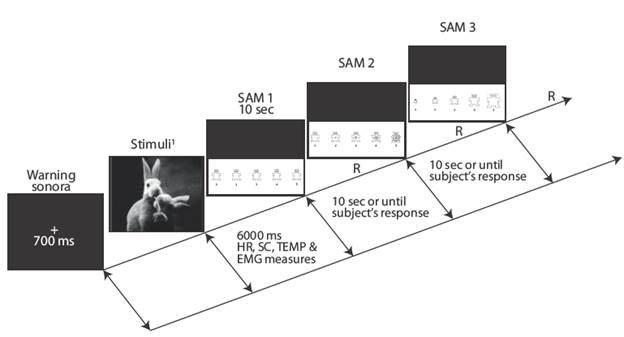
Figure 1 Stimuli sequential probe, psychophysiological measure, and cognitive SAM assignment. International Affective Picture System 31 stimuli numbers: pleasant, 32. Source: Own elaboration based on the data obtained in the study.
Source: Own elaboration based on the data obtained in the study.
Figure 2 Self-Assessment Manikin. a) pleasure, b) arousal, c) dominance.
Procedure
Each participant was assessed individually in a quiet room, with a controlled temperature of 75°F. An interview was first applied to identify the inclusion criteria previously described, followed by the application of the MoCA. After this, the participant was asked to answer each of the RB questionnaire items. Electrodes were placed on the participant while being awake and sitting in a comfortable as follows: three EKG electrodes (positive, negative, and ground) on the sternum and on the fourth and fifth intercostal space; two SC sensors located in the middle and index fingertips of the left hand; a temperature sensor attached to the internal surface of the left hand, and a positive and negative electrode located longitudinally over the abdominal surface for the EMG recording. Resistance lower than 5 kOhm was assured for all derivations.
Prior to the presentation of the emotional stimuli, a five-minute baseline was recorded while the participants were in a rest state with their eyes closed. They were instructed to remain quiet and not to move. After the baseline period, the subjects were instructed to look at the center cross of the monitor placed 50 cm in front of the subjects, and to keep sight of each stimuli for six seconds, during which psychophysiological emotional response was measured.
Immediately after, the participants were asked to answer in a numeric keyboard the three SAM emotional valence in a five option Likert scale 16,21,32. The experimental protocol and informed consent were properly presented to and approved by the Bioethical Committee from the Medicine and Psychology Faculty of the Autonomous University of Baja California, and followed the ethical procedures according to Helsinki Declaration of 2013 and the General Health Law for research in human participants of Mexico.
Data reduction and analysis
Psychophysiological emotional response was reduced by calculating the average of each autonomic signal (EMG, SC, HR, and temperature) during the six seconds of stimuli presentation, corresponding to each image group (pleasant, unpleasant, and neutral). Each SAM behavioral scale was estimated separately for each stimuli group. Statistical t-tests were applied to search for differences between independent samples. MANOVA tests were performed to determine differences between and within subjects.
Results
According to the RB questionnaire, the experimental group had 21 (47.7%) participants, who showed high risk behavior indicators in one or more of the five assessed dimensions (Table 1).
Table 1 Demographical characteristics of participants with RB (n=21) and without RB (n=23).
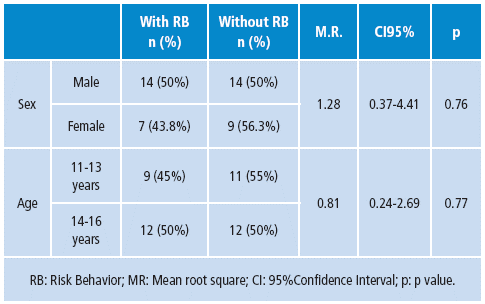
Source: Own elaboration based on the data obtained in the study.
The results were compared for each autonomic signal, and each stimuli type was compared between the groups. Regarding pleasant stimuli, RB participants showed increased abdominal EMG compared with the control group. Similar results were observed when analysing EKG; the control group showed a significant decrease of bits per minute (BPM) when compared with RB adolescents. SC showed significant increase of microsiemmens (μS) in the RB group compared with controls. There were no significant differences observed for temperature parameters between groups.
The mean comparison of EKG showed significantly lower values in both groups (mean=84.27, t=60.77, p<0.05) for unpleasant stimuli compared to neutral (media=87.03, t=70.64, p<0.05) and pleasant (mean=88.09, t=68.87, p<0.05) stimuli (Table 2).
Table 2 Psychophysiological comparison between subjects with RB (n=21) and without RB (n=23).
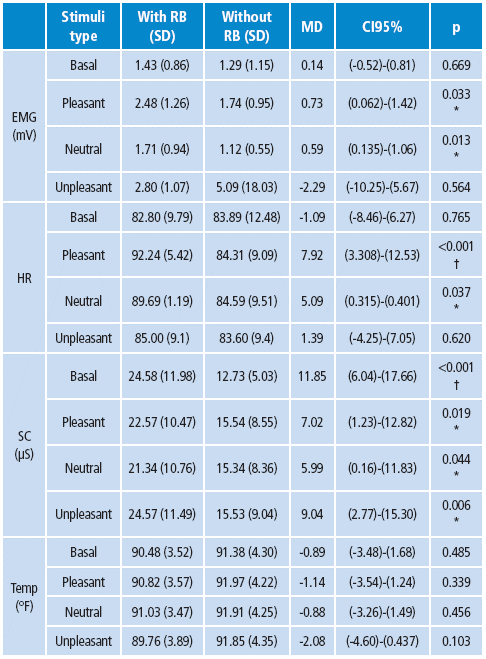
Source: Own elaboration based on the data obtained in the study.
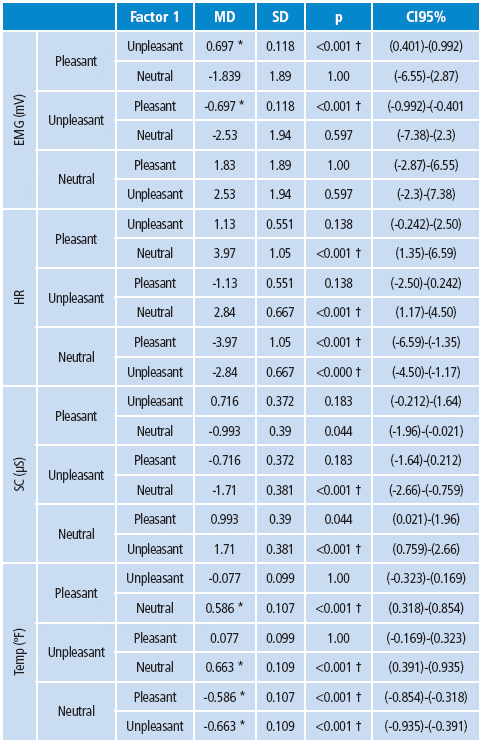
Both groups showed a close emotional valence assessment in relation to the observed stimuli, as well as in the pleasure and domain scales for pleasant stimuli. However, the arousal scale presented significant differences; the control group reported higher levels of arousal compared to the RB group. The control group showed lower pleasure and dominance assessment to unpleasant stimuli compared to RB, while no differences were found in the arousal scale (Table 3).
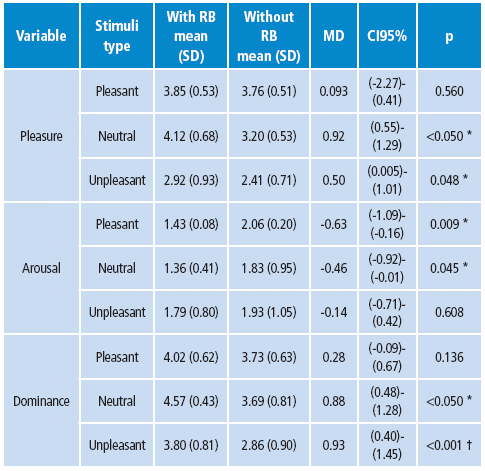
RB: Risk behavior, SD: standard deviation, MD: difference between the means, CI: 95% confidence interval, p: p value.
* 95% probability.
† 98% probability.
Source: Own elaboration based on the data obtained in the study.
The Repeated Measures Generalized Linear Model was used to find differences between stimuli type and group. Three levels for each type of stimuli were used as factor one among the subjects, and two levels per group variable were used between subject factors. Bonferroni correction was applied to confidence intervals when comparing the main effects. All physiological variables showed significant differences depending on the stimuli to which they were subjected.
According to the group factor, significant differences were found between the studied groups for each kind of stimuli in HR, SC and temperature. EMG did not show differences between groups per stimuli (Table 4).
Table 4 Differences in psychophysiological measures between groups based on the type of stimuli.
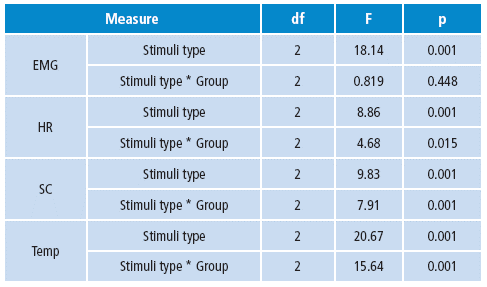
df: degrees of freedom, F: F value, p: p value.
Source: Own elaboration based on the data obtained in the study.
Within subjects, tests showed significant differences of the psychophysiological emotional reaction depending on the observed stimuli (Table 5).
Discussion
Emotional psychophysiological response has been described previously by other authors in relation to behavioral disorders such as Attention Deficit Hyperactive Disorder (ADHD) 29. Musser reported ADHD as a syndrome that involves altered emotional regulation associated with parasympathetic mechanisms. This study did not report any differences between ADHD and control groups, which coincides with the findings in RB adolescents.
Considering the hypothesis on the highly emotional arousing created by stimuli that illustrate threat, violence, death or eroticism 16, it was expected that SC responses would present higher parameters, and a more pronounced ECG deceleration compared to neutral stimuli. Similar results were found in this study, which were also more prominent in the risk behavior group.
In other studies 17,21, this psychophysiological reaction has been interpreted as the activation of a defensive survival behavior that increases in the presence of stimuli considered as an attack or threat. This suggest that, compared to the control group, adolescents with risk behavior could experience higher psychophysiological signs of threat. In behavioral terms, when perceiving threat stimuli, both groups report higher arousal for unpleasant stimuli, which coincides with Bradley 21.
Furthermore, it was found that the risk behavior group report less threats caused by unpleasant arousing stimuli compared to control group; nevertheless, in behavioral measures, the risk behavior group show psychophysiological signs of SC corresponding to higher arousal.
The ECG response to unpleasant threat stimuli was characterized by an initial significant deceleration when compared to the other stimuli groups, and did not show any differences between groups, which is consistent with the reports of other authors proposing that the most common response to a threatening stimuli in ECG is initial bradycardia as a preparation for defense, accompanied by late cardiac acceleration (after six seconds) 16,17,21,32.
Other studies have also reported that adolescents with anxiety symptoms show higher ECG responses compared to their pairs without these symptoms 33. For future studies, these findings lead to propose, as demonstrated in previous studies 18, that ECG could be a useful source for training programs on emotional response to reduce physiological emotional instability.
Conclusions
Risk behavior in the sample of adolescents studied from a psychophysiological emotional response approach can provide relevant information about sympathetic stability related to the experience on emotional information. The risk behavior group presented signs of major psychophysiological reaction to emotionally loaded stimuli in comparison with control group. The control group was characterized by initial bradycardia, which was more evident than in the risk behavior group. However both groups showed pronounced ECG deceleration to unpleasant stimuli, with no differences between them. Additionally, the electromyogram showed high levels of response to pleasant stimuli in the risk behavior group. The same group was characterized by higher levels of skin conductance and to the three types of stimuli as well.
Psychophysiological emotional reaction might be recognized as one of reliable variable to use as a diagnosis measure regarding the presence of risk behavior in adolescents. However, more research is needed to support such an idea, and prognosis based on cohort studies as well. This may demonstrate the continuity or evolution of the presence of psychophysiological parameters found in this study.