Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
Revista Colombiana de Entomología
Print version ISSN 0120-0488
Rev. Colomb. Entomol. vol.41 no.1 Bogotá Jan./June 2015
Growth and nutritional physiology of Spodoptera frugiperda (Lepidoptera: Noctuidae) fed on Meliaceae fractions
Crecimiento y fisiología nutricional de Spodoptera frugiperda (Lepidoptera: Noctuidae) alimentadas con fracciones de Meliaceae
ANGELINA MARIA MARCOMINI GIONGO1, JOSÉ DJAIR VENDRAMIM2, SÂMYA DANIELLE LIMA DE FREITAS3and MARIA FÁTIMA DAS GRAÇAS FERNANDES DA SILVA4
1 Ph. D. in Entomology. Escola Superior de Agricultura Luiz de Queiroz, Av. Pádua Dias, 11, 13418-090, Piracicaba, Brazil. ammarcomini@yahoo.com.br Corresponding author.
2Ph. D. in Entomology and Professor. Escola Superior de Agricultura Luiz de Queiroz, Av. Pádua Dias, 11, 13418-090, Piracicaba, Brazil.
3 M. Sc. in Chemistry. Universidade Federal de São Carlos, Rodovia Washington Luiz, km 235, São Carlos, Brazil.
4 Post Doctor in Chemistry and
Professor. Universidade Federal de São Carlos, Rodovia Washington Luiz, km 235, São Carlos, Brazil.
Abstract: The effect of hexane, dichloromethane, ethyl acetate and hydroalcoholic fractions obtained from ethanolic extract of Trichilia pallida, T. pallens and Toona ciliata on growth and nutritional physiology of Spodoptera frugiperda was evaluated. First instar larvae were fed on a diet treated with fractions for ten days. The most active fractions were applied on the diet and offered to larvae until the pupal stage to verify effects on growth and survival. To calcúlate the nutritional indices, fractions were incorporated into the diet offered to fourth instar larvae for four days. The dichloromethane fraction of T. pallens stems affected growth and survival of S. frugiperda larvae, and fractions of T. pallida leaves and stems, and of To. ciliata leaves and fruits affected larval growth. Nutritional analysis indicated that the fraction of To. ciliata leaves acts mainly as a phagodeterrent, while dichlorometane fractions of T. pallida stems and leaves, To. ciliata fruits and T. pallens stems cause toxic effects after ingestion, reducing the efficiency of food conversion and increasing the metabolic cost to the larvae.
Key words: Insect metabolism. Plant extracts. Sublethal effects.
Resumen: Se evaluó el efecto de fracciones de hexano, diclorometano, acetato de etilo e hidroalcohólica, obtenidas a partir del extracto de etanol de Trichilia pallida, Trichilia pallens y Toona ciliata sobre el crecimiento y fisiología nutricional de Spodoptera frugiperda. Larvas de primer estadio se alimentaron con dieta tratada con dichas fracciones durante diez días. Las fracciones más activas fueron aplicadas sobre la dieta y se ofrecieron a las larvas hasta pupa para verificar efectos sobre el crecimiento y sobrevivencia. Para calcular los índices nutricionales, la dieta con las fracciones fueron ofrecidas a las larvas de cuarto estadio durante cuatro días. La fracción de diclorometano de ramas de T. pallens afectó el crecimiento y la sobrevivencia de las larvas. Por otra parte, las fracciones de diclorometano de ramas y hojas de T. pallida y hojas y frutos de To. ciliata afectaron el crecimiento larval. El análisis nutricional indicó que la fracción de hojas de To. ciliata actúa principalmente como disuasor de alimentación mientras que las fracciones de diclorometano de ramas y hojas de T. pallens, frutas de To. ciliata y ramas de T. pallens causan efectos tóxicos después de la ingestión, reduciendo la eficiencia de conversión de alimento y aumentando el costo metabólico para las larvas.
Palabras clave: índices nutricionales. Fagodisuasión.
Introducción
One of the challenges for insect control is the development of more effective strategies than chemical insecticides, as there are insect populations resistant to most synthetics currently used, and also safer for man, the environment and natural enemies (Rattan 2010). Botanical insecticides are suggested as an alternative to synthetics (Akhtar et al. 2012), and the family Meliaceae is currently well investigated due to the occurrence of limonoid triterpenes, promising compounds that have biological activities on insects like insecticidal, phagodeterrent and growth regulating (Isman et al. 2002; Roy and Saraf 2006). Most work has been focused on the neem tree Azadirachta indica A. Juss. and the limonoid azadirachtin (Ntalli and Menkissoglu-Spiroudi 2011). Other bioactive compounds are available on genera Cedrela, Kha-ya, Trichilia, Chisocheton, Toona and Turaeu (Isman 1995; Abdelgaleil et al. 2001).
There are reports of bioactivity of other Meliaceae likeTrichilia pallida Swartz, Trichilia pallens C. DC. and Toona ciliata M. Roemer extracts on some insect pests (Bezerra-Silva et al. 2010; Cunha et al. 2005; Pavarini et al. 2010), including Spodoptera frugiperda (J. E. Smith) (Lepidoptera: Noctuidae) (Rodríguez and Vendramim 1997; Roel et al. 2000; Bogorni and Vendramim 2003 2005). However there are no studies about fractionation of these extracts to evaluate the effect of the fractions on the fall armyworm. Considering the potential of these three Meliaceae for the control of insects, this study was carried to evaluate the effect of the fractions of ethanolic extracts on the growth, survival and nutritional physiology of S. frugiperda.
Materials and methodsThe chemical procedures were carried out at the Laboratório de Produtos Naturais of Universidade Federal de Sao Carlos (UFSCar), and biological tests with S. frugiperda at Laboratório de Plantas Inseticidas of Escola de Superior de Agricultura Luiz de Queiroz (ESALQ), in Sao Paulo, Brazil, from October 2010 to December 2012.
Insects rearing. S. frugiperda was maintained in a labora-tory under controlled conditions (25 ± 2 °C, RH = 60 ± 10% and 14-hour photoperiod). Larvae were fed on an artificial diet (Greene et al. 1976) and adults received honey solution 1%.
Plant material. Stems and leaves of T. pallida (voucher ESA 81288) were collected in October 2010, and stems, leaves and fruits of To. ciliata (voucher ESA 78420) in January 2011, both from the ESALQ campus. Stems and leaves of T. pallens (voucher ESA 81286) were collected in April 2011 from Lajeado das Orquídeas in Sapopema-PR. The material was dried in a drying oven with air circulation at 40 °C, ground in a Wiley mill and stored in glass vials.
Extracts and fractions. Ethanolic extracts were prepared using an Ika Ultra Turrax (T25) dispersing instrument. Five extractions of five minutes each at 8,000 rpm and room temperature were carried out, by adding 1 L of ethanol per 200 g of powdered plant material. The extracts were filtered through a filter paper and the solvent was eliminated under vacuum on a rotary evaporator (45 °C) until dry. Each re-sidue of ethanol extract (10 g) was dissolved in methanol (100 mL) and water (300 mL) and exhaustively extracted by consecutive liquid/liquid partition with solvents of increasing polarity (hexane, dichloromethane and ethyl acetate) using a separating funnel, which was shaken to mix the solvent with the extract and then allowed to stand for phase separation. The hexane, dichloromethane, ethyl acetate and last remaining hydroalcoholic fractions were evaporated under vacuum. At the end of the process, the following were obtained: hexane, dichloromethane, ethyl-acetate and hydroalcoholic fractions of T. pallida leaves, T. pallida stems, T. pallens leaves, T. pallens stems, To. ciliata leaves, To. ciliata stems and To. ciliata fruits, totaling 28 fractions.
Bioactivity assays. Fractions were evaluated in four bio-assays: 1) all fractions of T. pallida; 2) fractions of T. pallens stems; 3) fractions of T. pallens leaves; and 4) all fractions of To. ciliata. Hexane, dichloromethane and ethyl acetate fractions of three plants were redissolved in acetone and water (1:1) and hydroalcoholic fractions were redissolved in ethanol and water (1:1) at 1% (10,000 ppm) (weight by volume). The artificial diet was placed in multi-well plates (2 mL per well) and 30 gL of fractions at 1% were applied on the diet surface. After two hours for solvent evaporation, first instar larvae (24 h of age) were placed one per well. Three controls were used: water and acetone (1:1), water and ethanol (1:1), and only water to evaluate the effect of the solvents. Each treatment consisted of four repetitions of 24 larvae, except in the assay with fractions of T. pallens leaves, which consisted of five replicates of twelve larvae. Mortality was recorded daily, and after ten days the surviving larvae were weighed.
Effect on larval growth and survival. In a second test, the most active fractions were selected to determine the effects on larvae growth and survival. Fractions dissolved at 1% were applied on the diet surface in glass tubes (8.5 x 2.5 cm) containing 3 mL of diet (40 gL/tube), and first instar larvae were individualized in tubes. Four replicates of five larvae each were performed. The parameters evaluated were mortality, duration of larval stage and pupal weight at 24 hours.
Effect on nutritional physiology. To understand the effects of the most active fractions on the nutritional physiology of S. frugiperda, nutritional indices of fourth instar larvae that ingested treated diet were calculated. Fractions were dissolved in acetone and incorporated into the artificial diet at a concentration of 1,000 ppm (1 mg/g diet). The artificial diet containing the fractions or acetone (control) was placed in glass tubes of known weight, which were weighed again to obtain the initial weight of diet. Fourth instar larvae were weighed and individualized in the tubes. Initial fresh and dry weights of an aliquot of the same instar larvae were determined. A diet containing only acetone was used as control. Twenty fourth instar larvae were used per treatment (independent replicates), and after four days of feeding, larvae and feces were separated from the remaining diet. Larvae were killed by freezing and all the material was placed in a drying oven at 50 °C for seven days, to obtain the dry weight, which was used for the determination of nutritional indices.
From these data the weights were obtained: food supplied, food remaining, food ingested (I), feces (F), larvae weight gain (B) and larvae mean weight (C). Then the following indices were calculated in the duration of feeding time (T): the relative consumption rate (RCR = I / (C x T)), the relative metabolic rate (RMR = M / (C x T)), relative growth rate (RGR = B / (C x T)), the efficiency of conversion of ingested food (ECI = (B / I) x 100) and digested food (ECD = (B / (I - F)) x 100), the approximate digestibility (AD = ((I - F) / I) x 100), the metabolic cost (MC = 100 - EcD), weight of the assimilated food (I - F) and weight of the metabolized food (M = (I - F) - B) (Scriber and Slansky Jr. 1981).
Data analysis. All bioassays were carried out in a completely randomized design. The nutritional indices were subjected to covariance analysis (Raubenheimer and Simpson 1992), in which the denominator was used as a covariate for each index. Means were compared by the Tukey-Kramer test (p < 0.05), with the statistical program SAS 9.2 (SAS Statistical Analysis System, 2009). For the other analyses, the statistical program R version 3.0.1 (R Development Core Team, 2013) was used. Data with binomial distribution (mortality) were analyzed by GLM (Generalized Linear Models) by cbind procedure using the chi-square test (p < 0.05) for comparison of means. The other data were subjected to Bartlett's test to check homogeneity of variances, and to the Shapiro-Wilk test to verify the normality of residuals. Then an analysis of variance was done (F test, p < 0.05), and means were compared by the Tukey test (p < 0.05), using the glm and glht procedures.
Results and discussionBioactivity assays. Among hexane, dichloromethane, ethyl acetate and hydroalcoholic fractions of T. pallida leaves and stems applied on the diet offered to the larvae of S. frugiperda (Table 1), the dichloromethane fraction of stems was the one that caused higher larval mortality than those recorded in the ethanol, acetone and water controls. For both stems and leaves in ethyl acetate, hexane and hydroalcoholic fractions, mortality did not differ from controls. There was no difference between the water control and the ethanol and acetone controls (solvents used to dissolve the fractions), indicating no effect of them on the larvae. Despite the low mortality levels, the weight of larvae that ingested the diet containing dichloromethane fractions of T. pallida leaves and stems was reduced after ten days, not reaching 10% of those recorded in the other treatments.
Similar results were observed with the fractions of T. pallens stems, where only the dichloromethane fraction caused larval mortality higher than in the three controls, and drastically affected larval growth, which reached a maximum of 1.5% of the weight recorded in all treatments including controls (Table 1). For ethyl acetate, hexane and hydroalcoholic fractions, larval mortality did not differ from the controls.
There was no difference between treatments with fractions of T. pallens leaves and controls in relation to larval mortality. For larval weight, the value obtained in the hexane fraction was lower than that obtained in control water, but did not differ from ethanol and acetone controls (Table 1). Fractions of To. ciliata stems, leaves and fruits did not reduce larval survival, but dichloromethane fractions of leaves and fruits affected the larvae development (Table 2).
Bioactivity of these Meliaceae extracts against S. fru-giperda was already reported (Rodríguez and Vendramim 1997; Roel et al. 2000; Torrecillas and Vendramim 2001; Bogorni and Vendramim 2003, 2005). However, methods used to obtain the extracts and evaluate the bioactivity in these studies differ considerably from those used in this study, making it difficult to compare with the data obtained here. Some of these studies refer to aqueous or organic extracts obtained by different methods. the experimental techniques vary between tests with corn leaves treated daily with the extracts and the extract addition to the artificial diet. The concentrations used, the evaluation time and age of larvae are also important factors. Despite all these variables, the effects observed in larvae after ingestion of food treated with extracts of these plant species are similar: reduction in larval weight gain, prolonged larval stage, smaller pupae, and concentration-dependent mortality. The To. ciliata extracts, however, mainly caused sublethal effects, with low mortality even at higher concentrations, as reported by Rodriguez and Vendamim (1997) for S. frugiperda larvae. The most significant effect is the low larval weight gain. Some fractions caused large reductions in larval weight few days after feeding on treated diets. This may be a result of reduced consumption due to a phagodeterrent activity, or low nutritional adequacy of the food by the presence of xenobiotics. Another possibility is post-ingestion toxic effect, causing poor utilization of food by insects or inhibiting important vital processes, causing developmental delay. However, the mixture of compounds present in the fractions of the plant extracts makes it difficult to conduct a more detailed study of the mode of action.
The effect against insects of the genus Trichilia has been attributed to a group of tetranortriterpenoids that includes hirtin (Xie et al. 2004) and the trichilins (Champagne et al. 1989; Nakatani 2001). The limonoids from To. ciliata were related to the phytochemical resistance to the shoot borer Hypsipyla (Agostinho et al. 1994); and toonacilin, 6-acetoxitoonacilin (Kraus et al. 1978) and cedrelone (Arna-son et al. 1987; Koul and Isman 1992) have insecticidal and phagodeterrent properties.
The low weight gain observed in larvae after ingestion of dichlorometane fractions (except for T. pallens leaves) is an indication that this solvent is more efficient in the extraction of active compounds of these Meliaceae species. For example, Pérez-Flores et al. (2012) used dichloromethane to extract limonoids from leaves of some plants of this family. For Tuta absoluta (Meyrick, 1917) (Lepidoptera: Gelechiidae), the dichloromethane extract of T. pallida leaves was also more efficient than hexane and methanol extracts in hexane (Cunha et al. 2005). Dichloromethane is a solvent of medium polarity, extracting hydrophobic and amphipathic compounds, thus covering large amount of compounds (Ferracini et al. 2005). This feature is subject to the chemical composition of each plant species; that is, dichloromethane may not be a good solvent when apolar compounds are abundant. For Meliaceae species studied, however, this solvent was very efficient.
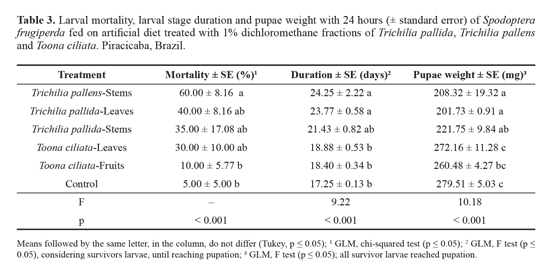
Effect on larval growth and survival. When larvae were exposed to the treatments during the entire larval stage, among fractions with higher bioactivity on S. frugiperda (dichloromethane fractions of T. pallens stems, T. pallida leaves and stems, and To. ciliata leaves and fruits), only the dichloromethane fraction of T. pallens stems caused larval mortality, which was different from that recorded in control. The fractions of T. pallida leaves and stems and To. ciliata leaves did not differ from the fraction of T. pallens stems, but also did not differ from the control. The fraction of T. pallens stems and the fraction of T. pallida leaves significantly affected the larval growth, causing a prolonged larval stage in approximately six to seven days compared to the control. In addition, pupal weight was lower than the control with these two treatments and with the fraction of T. pallida stems (Table 3). As previously verified, the fractions of To. ciliata leaves and fruits were those that least affected S. frugiperda survival and growth. A characteristic of many Meliaceae extracts is a slow and crescent effect over time, justifying the increased mortality observed in this assay compared to the previous one, which was only ten days long, mainly of T. pallens stems and of T. pallida leaves and stems.
Sublethal effects were also observed, as delayed deve-lopment, characterized by a larval stage of longer duration, and pupal weight reduction. Bogorni and Vendramim (2005) observed longer duration of the larval stage of S. frugiperda after ingestion of corn leaves treated with aqueous extracts of T. pallida leaves and stems, and of aqueous extracts of T. pallens leaves. Torrecillas and Vendramim (2001) found the same effects with aqueous extracts of T. pallida stems. Such effects may be due to low food intake, caused by phagode-terrence or nutritional inadequacy of the food substrate (Rodríguez and Vendramim 1998), or by a deviation of part of the food ingested to detoxification metabolism (Tanzubil and McCaffery 1990). With low food intake and low biomass conversion, the insect takes longer to reach the critical weight for ecdysis, which implies an increase of the larval stage du-ration.
Effect on nutritional physiology. The analysis of covariance indicated the effect of treatments and covariates, but there was no interaction between the two factors, for any of the nutritional indices. Thus, the data were subjected to an analysis of variance.
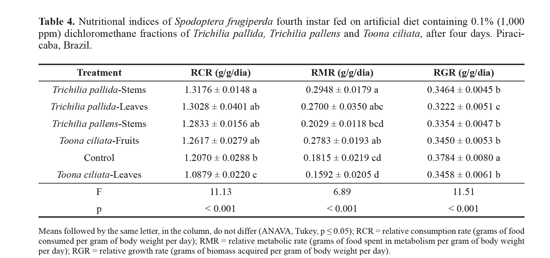
The relative consumption rate (RCR), which indicates the amount of food ingested by larva per gram of body weight per day, increased after ingestion of the fraction of T. pallida stems. The RCR was reduced in the larvae that ingested diets containing fraction of To. ciliata leaves, and the other fractions (T. pallida leaves, T. pallens stems and To. ciliata fruits) did not differ from the control (Table 4). The relative metabolic rate (RMR), which indicates the amount of food spent by the larva in metabolism per gram of body weight per day, increased after ingestion of a diet containing fractions of To. ciliata fruits and T. pallida stems. However, the relative growth rate (RGR), which indicates the gain of biomass by the insect in relation to body weight per day, was reduced after ingestion of all the fractions, and the greatest reduction was caused by the fraction of T. pallida leaves (Table 4). This may be due to a low food intake or to a toxic effect caused by the fractions, resulting in the use of food for purposes other than growth, such as detoxification enzymes synthesis.
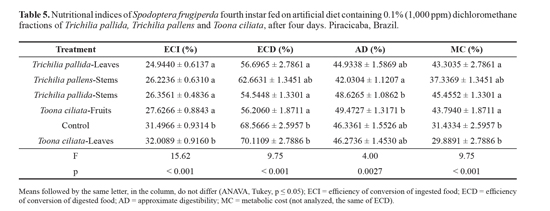
The efficiency of conversion of ingested food (ECI), which is the percentage of ingested food that is converted into biomass, was reduced in almost all treatments, except only the fraction of To. ciliata leaves, the only one that does not differ from the control (Table 5). The efficiency of conversion of digested food (ECD), which is the estimated percentage of assimilated food that was converted into biomass, decreased after ingestion of a diet containing fractions of To. ciliata fruits and T. pallida leaves and stems (Table 5). The low conversion of food into biomass justifies the delay in larval development observed with most of the fractions, and indicates that they cause toxic effects, except for the fraction of To. ciliata leaves, which caused no change in ECI and ECD. Fractions that caused reduction in ECD also caused increased metabolic cost (MC), i.e., the percentage of food metabolized into energy for the maintenance of life, increased. However, none of the fractions caused change in approximate digestibility (AD), which is the percentage of ingested food effectively assimilated by the insect (Table 5). This indicates that food was well assimilated by the larvae, but was mainly used for metabolism since the biomass conversion was low.
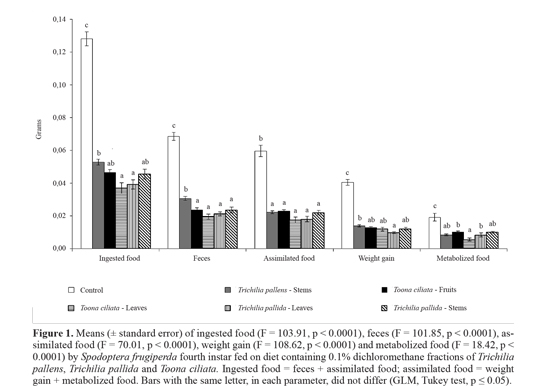
In all the fractions the amount of diet consumed decreased by more than half compared to the control (Fig. 1). As a result of food intake reduction, there was also reduced feces production and low weight gain, as well as a smaller amount of assimilated and metabolized food. Larvae that ingested a diet containing the dichloromethane fraction of To. ciliata leaves showed the least amount of food metabolized; that is, the highest amount of food was used for growth and not metabolism. Despite the reduction of total food consumption, the relative consumption to body weight (Table 6) was alte-red only with two fractions, increasing after ingestion of fraction of T. pallida stems, and decreasing with the fraction of To. ciliata leaves. In the first case, the increase indicates that there is not primary phagodeterrence (chemoreceptor-mediated), and the ECI and ECD reduction indicates that there is a post-ingestion toxic effect, which can be considered secondary phagodeterrence (reduced consumption caused by toxicity) (Mordue (Luntz) and Blackwell 1993). For the second fraction, phagodeterrence is responsible for reduction in relative consumption, since there was no change in the efficiency of food conversion, in metabolic cost or in relative metabolic rate, excluding the possibility of toxic effects, so the phagodeterrence is possibly primary. Therefore, for larvae fed on diets containing the fraction of To. ciliata leaves, despite lower food intake, ingested food was well used, but not enough for the growth to be equal to that of the control larvae. For the other fractions, the larvae continued feeding, probably as an attempt to compensate for the high metabolic cost.
Extracts of other Meliaceae also caused changes in the nutritional physiology of insects. When fourth instar Spo-doptera litura larvae were fed on a diet containing me-thanolic extract of branches of Trichilia americana (Sesse and Mocino) Pennington, the RGR, RCR, ECI and ECD all decrease. It is likely that this decrease in consumption rate is due to an phagodeterrent effect of the extract, and at least in part this contributes to the decrease in growth rate. However, a decrease in ECI and ECD indicates that T. americana extract also exhibits some post-ingestion toxicity (Wheeler and Isman 2001). Methanolic extracts of Melia azedarach L. leaves reduced RGR, RCR, ECD and ECI of Cnaphalocrocis medinalis Guenée (Lepidoptera: Pyralidae), while AD increased (Senthil-Nathan 2006). In these cases, it seems to be a mixture of phagodeterrence and toxic effects, due to simultaneous reduction in consumption and food conversion. In the present work, this was not observed. The only fraction that reduced RCR was To. ciliata leaves, but without reduction in ECI and ECD; thus the decrease in RGR was due to reduced consumption, not to a toxic effect. The other fractions caused toxic effects (reduced ECI and ECD), but caused no reduction in consumption, thus showing no phagodeterrence.
ConclusionsThe dichloromethane fraction of T. pallens stems affects growth and survival of S. frugiperda larvae; and fractions of T pallida leaves, of T. pallida stems, of To. ciliata leaves and of To. ciliata fruits affect growth. Bioactivity of the dichlo-romethane fractions of T. pallens stems, of T. pallida leaves, of T. pallida stems and of To. ciliata fruits is characterized by toxic effects after ingestion that can be referred to as secon-dary phagodeterrence, whereas fraction of To. ciliata leaves does not cause toxicity, but cause phagodeterrence, most likely primary.
AcknowledgementsThanks to the Instituto Nacional de Ciencia e Tecnologia -Controle Biorracional de Insetos Pragas (INCT-CBIP), to Coordenado de Aperfeigoamento de Pessoal de Nivel Superior (CAPES) and to the Fundado de Amparo á Pesquisa do Estado de Sao Paulo (FAPESP), for financial support.
Literature citedABDELGALEIL, S. A.; OKAMURA, H.; IWAGAWA, T.; SATO, A.; MIYAHARA, I.; DOE, M.; NAKATANI, M. 2001. Kha-yanolides, rearranged phragmalin limonoide antifeedants from Khaya senegalensis. Tetrahedron 57: 119-126. [ Links ]
AGOSTINHO, S. M.; DAS, M. F.; DA SILVA, G. F.; FERNANDES, J. B.; VIEIRA, P. C.; PINHEIRO, A. L.; VILELA, E. F. 1994. Limonoids from Toona ciliata and speculations on their che-mosystematic and ecological significance. Biochemical Syste-matics and Ecology 22: 323-328. [ Links ]
AKHTAR, Y.; ISMAN, M. B.; NIEHAUS, L. A.; LEE, C.; LEE, H. 2012. Antifeedant and toxic effects of naturally occurring and synthetic quinones to the cabbage looper, Trichoplusia ni. Crop Protection 31: 8-14. [ Links ]
ARNASON, J. T.; PHILOGENE, B. J. R.; DONSKOV, N.; KUBO, I. 1987. Limonoids from the Meliaceae and Rutaceae reduce feeding, growth and development of Ostrinia nubilalis. Ento-mologia Experimentalis et Applicata 43: 221-226. [ Links ]
BEZERRA-SILVA, G. C. D.; VENDRAMIM, J. D.; SILVA, M. A.; DIAS, C. T. S. 2010. Efeito de extratos orgánicos de Meliaceae sobre Bemisia tabaci (Gennadius) biótipo B em tomateiro. Arquivos do Instituto Biológico 77 (3): 477-485. [ Links ]
BOGORNI, P. C.; VENDRAMIM, J. D. 2003. Bioatividade de extratos aquosos de Trichilia spp. sobre Spodoptera frugiperda (J. E. Smith) (Lepidoptera: Noctuidae) em milho. Neotropical Entomology 32 (4): 665-669. [ Links ]
BOGORNI, P. C.; VENDRAMIM, J. D. 2005. Sublethal effect of aqueous extracts of Trichilia spp. on Spodoptera frugiperda (J. E. Smith) (Lepidoptera: Noctuidae) development on maize. Neotropical Entomology 34 (2): 311-317. [ Links ]
CHAMPAGNE, D. E.; ISMAN, M. B.; TOWER, G. H. N. 1989. Insecticidal activity of phytochemicals and extracts of the Meliaceae. pp. 95-109. In: Arnason, J. T.; Philogene, B. J. R.; Morand, P. (Eds.). Insecticides of plant origin. American Chemical Society. Washington DC. USA. 224 p. [ Links ]
CUNHA, U.; VENDRAMIM, J. D.; ROCHA, W. C.; VIEIRA, P. C. 2005. Potencial de Trichilia pallida Swartz (Meliaceae) como fonte de substáncias com atividade inseticida sobre a traja-do-tomateiro, Tuta absoluta (Meyrick) (Lepidoptera: Gelechiidae). Neotropical Entomology 34 (4): 667-673. [ Links ]
FERRACINI, V. L.; QUEIROZ, S. C. N.; GOMES, M. A. F.; SANTOS, G. L. 2005. Método para a determinado de hexazinone e tebutiuron em água. Química Nova 28 (3): 380-382. [ Links ]
GREENE, G. L.; LEPPLA, N. C.; DICKERSON, W. A. 1976. Velvetbean caterpillar: a rearing procedure and artificial me-dium. Journal of Economic Entomology 69 (4): 487-488. [ Links ]
ISMAN, M. B.; ARNASON, J. T.; TOWERS, G. H. N. 2002. Che-mistry and biological activity of ingredients of other species of Meliaceae. pp. 827-833. In: Schmutterer, H. (Ed.). The neem tree Azadirachta indica A. Juss. and other meliaceous plants: sources of unique natural products for integrated pest manage-ment, medicine, industry and other purposes. Neem Foundation. Mumbai. India. 696 p. [ Links ]
ISMAN, M. B., 1995. Leads and prospects for the development of newbotanical insecticides. Reviews of Pesticide Toxicology 3: 1-20. [ Links ]
KOUL, O.; ISMAN, M. B. 1992. Toxicity of the limonoid allelo-chemical cedrelona to noctuid larvae. Entomologia Experimen-talis et Applicata 64: 281-287. [ Links ]
KRAUS, W.; GRIMMINGER, W.; SAWITSKI, G. 1978. Toona-cilin and 6-acetoxy toonacilin, two novel B-seco tetranortrit-erpenoids with antifeedant activity. Angewandte Chemie 17: 452-453. [ Links ]
MORDUE (LUNTZ), A. J.; BLACKWELL, A. 1993. Azadirachtin: an update. Journal of Insect Physiology 39 (11): 903-924. [ Links ]
NAKATANI, M. 2001. Limonoids from Meliaceae and their biolo-gical activities. pp. 527-554. In: Tringali, C. (Ed.). Bioactive Compounds from Natural Sources. Taylor & Francis. London. U. K. 693 p. [ Links ]
NTALLI, N. G.; MENKISSOGLU-SPIROUDI, U. 2011. Pesticides of botanical origin: a promising tool in plant protection. pp. 3-24. In: Stoytcheva, M. (Ed.). Pesticides - Formulations, Effects, Fate. InTech. Rijeka. Croatia. 808 p. [ Links ]
PAVARINI, R.; ANSANTE, T. F.; CACERES, D. P. 2010. Efeito do extrato aquoso de plantas sobre a broca da bananeira Cosmopo-lites sordidus Germar (Coleoptera: Curculionidae). Nucleus 7 (1): 203-208. [ Links ]
PÉREZ-FLORES, J.; EIGENBRODE, S. D.; HILJE-QUIROZ, L. 2012. Alkaloids, limonoids and phenols from Meliaceae species decrease survival and performance of Hypsipyla grandella lar-vae. American Journal of Plant Sciences 3: 988-994. [ Links ]
R DEVELOPMENT CORE TEAM. 2013. R: A language and envi-ronment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. Available in: http://www.R-pro-ject.org. [Review date: 12 March 2013] [ Links ].
RATTAN, R. S. 2010. Mechanism of action of insecticidal secon-dary metabolites of plant origin. Crop Protection 29 (9): 913920. [ Links ]
RAUBENHEIMER, D.; SIMPSON, S. J. 1992. Analysis of cova-riance: an alternative to nutritional indices. Entomologia Ex-perimentalis et Applicata 62: 221 -231. [ Links ]
RODRIGUEZ, C. H.; VENDRAMIM, J. D. 1997. Avaliação da bioatividade de extratos aquosos de Meliaceae sobre Spodoptera
frugiperda (J. E. Smith). Revista de Agricultura 72 (3): 305-318. [ Links ]
RODRIGUEZ, C. H.; VENDRAMIM, J. D. 1998. Uso de índices nutricionales para medir el efecto insectistático de extractos de meliáceas sobre Spodoptera frugiperda. Manejo Integrado de Plagas 48: 11-18. [ Links ]
ROEL, A. R.; VENDRAMIM, J. D.; FRIGHETTO, R. T. S.; FRIGHETTO, N. 2000. Efeito do extrato acetato de etila de Trichilia pallida Swartz (Meliaceae) no desenvolvimento e sobrevivencia da lagarta-do-cartucho. Bragantia 59 (1): 53-58. [ Links ]
ROY, A.; SARAF, S. 2006. Limonoids: overview of significant bio-active triterpenes distributed in plants kingdom. Biological and Pharmaceutical Bulletin 29 (2): 191-201. [ Links ]
SAS STATISTICAL ANALYSIS SYSTEM. 2009. SAS User's Guide: Statistics. SAS Institute, Cary, NC, USA. [ Links ]
SCRIBER, J. M.; SLANSKY JR., F. 1981. The nutritional ecology of immature insects. Annual Review of Entomology 26: 183211. [ Links ]
SENTHIL-NATHAN, S. 2006. Effects of Melia azedarach on nu-tritional physiology and enzyme activities of the rice leaffolderCnaphalocrocis medinalis (Guenée) (Lepidoptera: Pyralidae). Pesticide Biochemistry and Physiology 84: 98-108. [ Links ]
TANZUBIL, P. B.; MCCAFFERY, A. R. 1990. Effects of azadi-rachtin and aqueous neem seed extracts on survival, growth and development of the African armyworm, Spodoptera exemp-ta. Crop Protection 9: 383-386. [ Links ]
TORRECILLAS, S. M.; VENDRAMIM, J. D. 2001. Extrato aquoso de ramos de Trichilia pallida e o desenvolvimento de Spodoptera frugiperda em genótipos de milho. Scientia Agricola 58 (1): 27-31. [ Links ]
WHEELER, D. A.; ISMAN, M. B. 2001. Antifeedant and toxic activity of Trichilia americana extract against the larvae of Spodoptera litura. Entomologia Experimentalis et Applicata 98: 9-16. [ Links ]
XIE, Y S.; ISMAN, M. B.; GUNNING, P.; MACKINNON, S.; AR-NASON, J. T.; TAYLOR, D. R.; SANCHEZ, P.; HASBUNT, C.; TOWERS, G. H. N. 1994. Biological activity of extracts of Trichilia species and the limonoide hirtin against Lepidoptera larvae. Biochemical Systematics and Ecology 22: 129-136. [ Links ]
Received: 28-Sep-2014
Accepted: 10-Dec-2014
Suggested citation:
MARCOMINI GIONGO, A. M.; VENDRAMIM, J. D.; FREITAS, S. D. L.; SILVA, M. F. G. F. 2015. Growth and nutritional phys-iology of Spodoptera frugiperda (Lepidoptera: Noctuidae) fed on Meliaceae fractions. Revista Colombiana de Entomología 41 (1): 33-40. Enero-Junio 2015. ISSN 0120-0488.