Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
Revista Colombiana de Entomología
Print version ISSN 0120-0488
Rev. Colomb. Entomol. vol.41 no.1 Bogotá Jan./June 2015
Compatibility of pesticides with the generalist predatory mite Amblyseius largoensis (Acari: Phytoseiidae)
Compatibilidad de plaguicidas con el ácaro depredador generalista Amblyseius largoensis (Acari: Phytoseiidae)
RAFAEL ROCHA DA SILVA1, ADENIR VIEIRA TEODORO2, MARIA DE JESUS DE SOUSA SILVA1, PAULO REBELLES REIS3 and SHÊNIA SANTOS SILVA4
1 M. Sc. Graduate Programme in Agroecology, Maranhão State University (UEMA), 65054-970, São Luís, MA, Brazil. rafael.rs@me.com, mjs.silva8@gmail.com.
2 Ph. D. Embrapa Coastal Tablelands, 3250 Beira-Mar, Jardins, 49025-040, Aracaju, SE, Brazil. adenir.teodoro@embrapa.br. Corresponding author.
3 Dr. EPAMIG/CTSM EcoCentro, 37200-000, Lavras, MG, Brazil. paulo.rebelles@epamig.ufla.br.
4 Dr. Visiting Researcher, Embrapa Coastal Tablelands, 3250
Beira-Mar, 49025-040, Aracaju, SE, Brazil. sheniass@yahoo.com.br.
Abstraet: The aim of this study was to evalúate the compatibility of synthetic and alternative pesticides to an important natural enemy associated with pest mites in citrus orchards, in Sao Luís Maranhao State, Brazil: the generalist predatory mite Amblyseius largoensis. The pesticides tested were abamectin, mineral oil, mancozeb, pyridaben, neem oil (Azadiractha indica) and crude cottonseed oil (Gossypiumspp.). Compatibility of pesticides to A. largoensiswas assessed as to adverse effects on mortality and fertility as well as to interference with growth rate. Overall, the results of adverse effects and growth rate were corroborative. Mancozeb, mineral oil and neem oil were classified as slightly harmful while the cotton oil and abamectin were classified as moderately harmful and pyridaben was considered harmful. The growth rate of A. largoensisfemales exposed to mancozeb, mineral oil, neem oil and cotton oil was positive indicating population growth. We conclude that neem oil, mineral oil and mancozeb are compatible with A. largoensisas they were less harmful to this predator.
Key words: Vegetal oils. Integrated pest management. Physiological selectivity. Toxicity.
Resumen: El objetivo de este estudio fue evaluar la compatibilidad de plaguicidas alternativos y sintéticos con el depredador generalista de ácaros Amblyseius largoensis, enemigo natural de plagas de ácaros en cultivos de cítricos en Sao Luís, Maranhao State, Brazil. Los plaguicidas evaluados fueron la abamectina, el aceite mineral, el mancozeb, el piridaben, y los aceites de neem (Azadiractha indica) y de algodón (Gossypiumspp.). La compatibilidad de los plaguicidas con A. largoensisse evaluó mediante la estimación de los efectos adversos en la mortalidad y fecundidad y también por la interferencia con la tasa de crecimiento. En general, los Resultados totales fueron complementarios a los efectos adversos de la tasa de crecimiento. El mancozeb, el aceite mineral y el aceite de neem se clasificaron como plaguicidas ligeramente dañinos, el aceite de algodón y la abamectina como moderadamente nocivos y, finalmente, el piridaben se consideró significativamente perjudicial. La tasa de crecimiento de las hembras de A. largoensisexpuestas a mancozeb, aceite mineral, aceite de neem y aceite de algodón fue positivo, lo que indica crecimiento de la población del depredador. Los Resultados en este estudio mostraron que el aceite de neem, el aceite mineral y el mancozeb fueron los plaguicidas más compatibles y que menos efectos dañinos provocaron en el depredador generalista de ácaros A. largoensis.
Palabras clave: Aceites vegetales. Manejo integrado de plagas. Selectividad fisiológica. Toxicidad.
Introduction
The use of pesticides that are compatible with natural enemies is pivotal to the success of biological control as selective pesticides have lower toxicity to beneficial organisms and they are therefore prioritized in Integrated Pest Management (IPM) programs (Varenhorst and O'Neal 2012; Abraham et al. 2013). Selectivity bioassays in which natural enemies are submitted to pesticides under controlled conditions help to classify such toxicants into innocuous and less harmful to harmful (Hassan et al. 1994; Reis et al. 2006). The use of selective pesticides is an important strategy of chemical control as such pesticides are less harmful to natural enemies (Reis et al. 1998; Abraham et al. 2013).
Predatory mites belonging to the family Phytoseiidae are major natural enemies of pest mites (Moraes 1992; McMurtry and Croft 1997; Sarmento et al. 2011; Hannef and Sadanandan 2013; Costa et al. 2014). The predatory mite Amblyseius largoensis (Muma, 1955) (Acari: Phytoseiidae) has been found inhabiting citrus orchards (Childers and Denmark 2011; Silva et al. 2013) associated with pests such as the citrus rust mite Phyllocoptruta oleivora (Ashmead, 1879) (Acari: Eriophyidae), the Texas citrus mite Eutetranychus banksi (McGregor, 1814) (Acari: Tetranychidae) and other tetranychid mites (Tanaka and Kashio 1977; Jamieson et al. 2005). This mite has a worldwide distribution and it is also found in South American countries including Brazil and Colombia (Moraes et al. 2004). Type III generalist phytoseiid mites like A. largoensis feed on pest insects and mites as well as on alternative food like pollen and nectar (McMurtry and Croft 1997; Carrillo et al. 2010) which allow them to survive in the field even in periods of low pest populations (Reis and Alves 1997; McMurtry and Croft 1997; Sarmento et al. 2011; Gerson and Weintraub 2012).
The control of pests is usually conducted with pesticides which may cause people poisoning and environmental contamination besides pest outbreaks and mortality of natural enemies (Gallo et al. 2002; Geiger et al. 2011; Abraham et al. 2013). Pesticides such as abamectin, mineral oil, mancozeb and pyridaben are registered to control citrus pests in Brazilian citrus orchards (Agrofit 2013). Alternative pesticides such as vegetal oils are usually less toxic to beneficial arthropods (Duso et al. 2008), however studies to assess their toxicity to natural enemies like the predatory mite A. largoensis are needed. Here, we aimed to evaluate the compatibility of synthetic and alternative pesticides to the generalist predatory mite A. largoensis.
Material and methodsThe predatory mite A. largoensis was collected from Oran-ge trees (Citrus sinensis L.) located in the city of Sao Luís (2°35'44.40"S 44°10'30.03"W), Maranhao state, Brazil. Mites were kept in plástic discs (7 cm diameter) floating on Petri dishes (10 cm diameter x 1.5 cm height) without lid filled with water under controlled conditions of tempe-rature (29 ± 5 °C) and natural relative humidity and photo-period. Mites were fed with castor bean pollen (Ricinus communis L.) and honey every other day (Reis and Alves 1997).
The pesticides abamectin (Abamectina DVA™ 18 EC, 18 g de a.i./L, dosage 0.3 ml/L), mineral oil (Iharol™ CE, 760 g de a.i./L, dosage 20 ml/L), mancozeb (Manzate™ WG, 750 g de a.i./kg, dosage 1.5 g/L) and pyridaben (SanmiteTM CE, 200 g de a.i./L, dosage 0.5 ml/L) were chosen because they are registered in Brazil to control the citrus rust mite P. oleivora (Agrofit 2013). The neem oil (Azadiractha indica A. Juss) (Sempre Verde Killer™, 3 ml a.i./L, dosage 15 ml/L) and the cotton crude oil (Gossypium spp.) (dosage 15 ml of oil + 10 ml of neutral detergent/L of water) (Ferreira and Michereff Filho 2002) were selected because they have been efficiently used to control pests of several crops. Pesticides were sprayed at their highest label dosage recommended to control the citrus rust mite while the neem oil was used in its maximum dosage to control pest mites. The cotton oil was used at the dosage recommended to control the coconut mite Aceria guerreronis Keifer 1965 (Acari: Eriophyidae) (Ferreira and Michereff Filho 2002).
Pesticides were sprayed on plastic discs (5 cm diameter) through a Potter tower (Burkard, Rickmansworth, UK) at 5 psi/pol2 pressure with an 1.3 ml spray aliquot which resulted in an residue of 1.7 ± 0.25 mg/cm2 (Hassan et al. 1994). Control discs were sprayed only with distilled water. Sprayed discs dried up in open air for 1 hour before five adult females of A. largoensis in the beginning of their reproductive period (6 days old) and a male were transferred to them. Discs were placed to float on Petri dishes as described above. The experiment consisted of seven treatments (abamectin, mineral oil, mancozeb, pyridaben, neem oil, cotton oil and control) and six replicates in a randomized experimental design. Surviving predatory mites were daily fed with castor bean pollen and honey. The number of living mites and eggs laid were daily recorded over a period of 7 days.
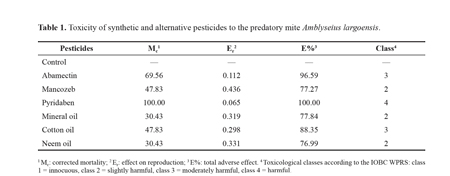
Our approach to determine pesticide compatibility with the predatory mite A. largoensis was based on the total adverse effect (E%) and the interference on growth rate. The total adverse effect (E%) was based on mortality and effect on reproduction using the equation: E% = 100% - (100% - Mc) x Er, where Mc = corrected mortality and Er = reproduction effect obtained from the quotient between the mean number of viable eggs in the treatment and the mean number of viable eggs in control. E% values were used to rank pesticides into classes from 1 to 4 according to the IOBC/WPRS (Hassan et al. 1994), i.e. class 1 = E < 30% (innocuous); class 2 = 30% < E < 79% (slightly harmful); class 3 = 80% < E < 99% (moderately harmful) e class 4 = E > 99% (harmful) (Reis et al. 1998).
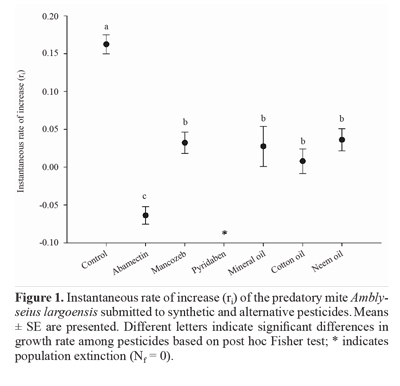
The instantaneous rate of increase (q), a proxy for growth rate, was used to assess the effect of pesticides on growth rate of the predatory mite A. largoensis using reproduction and mortality data according to the equation: ri = [ln (Nf/ N0)]/ At, where Nf is the final number of individuals, N0 is the initial number of individuals and At is the time elapsed between the onset and final of the experiment (7 days) (Stark et al. 1997). The ri is a snapshot of population growth in a given period and positive values mean population increase and negative values indicate population decline (Stark et al. 1997). The ri was calculated based on the number of eggs, immatures and adults daily added to the population over a period of 7 days. Anovas followed by post hoc Fisher tests were used to determine the influence of synthetic and alternative pesticides on the growth rate of and the number of descendants produced by A. largoensis using the software Statistica 10 (Statsoft Inc 1984-2011).
Results and discussionPyridaben (100%) and abamectin (69.56%) inflicted the highest mortality on females of the predatory mite A. largoensis while the mineral oil and the neem oil were less toxic (30.43%) (Table 1). Mancozeb, mineral oil and neem oil were considered slightly harmful (class 2), cotton oil and abamectin were ranked in class 3 as moderately harmful and pyridaben was classified as harmful (class 4) to A. largoensis. Although the mineral and the neem oil caused the lowest mortality on the predatory mite they were not considered innocuous because they negatively affected reproduction (Table 1).
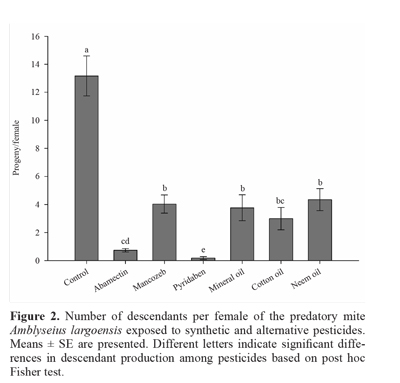
The growth rates of A. largoensis exposed to mancozeb, cotton oil, mineral oil and neem oil were significantly re-duced in comparison with control (F5,20 = 19.24; P < 0.05), yet positive r values were obtained indicating population growth (Fig. 1). Mites exposed to abamectin had negative ri values suggesting population decline, while pyridaben elimi-nated the population of the predatory mite after 7 days (Nf = 0) (Fig. 1). The number of descendants per female of A. largoensis was affected by the pesticides (F6,35 = 28.20; P = 0.00) (Fig. 2). Abamectin and pyridaben, which were the most toxic pesticides to the predatory mite, also caused the highest reduction in descendants. Mancozeb, cotton oil, mineral oil, and neem oil also reduced the number of descendants (Fig. 2).
Our combined approach of total adverse effect and interference on growth rate allowed us to assess the compatibility of synthetic and alternative pesticides to the predatory mite A. largoensis. In general, total adverse effects matched growth rate results. The oils of neem and mineral as well mancozeb were considered slightly harmful (class 2) to the predatory mite A. largoensis. In fact, growth rates of A. largoensis exposed to these pesticides were positive indicating population increase. Alternative pesticides like the neem oil have additional advantages such as low toxicity to mammals, fast degradation in the environment and relative selectivity to natural enemies (Erler et al. 2010; Nicoletti et al. 2012). Neem pesticides are efficient against pest mites (Venzon et al. 2008) besides being selective to the predatory mites Neoseiulus californicus (McGregor, 1954) and Phytoseiulus macropilis (Banks, 1904) (Acari: Phytoseiidae) (Bernardi et al. 2013). The mineral oil has been used to control insect pests (Najar-Rodriguez et al. 2008; Chueca et al. 2009), and no resistance has been identified (Najar-Rodriguez et al. 2008). Contrasting to our results, Reis et al. (1998) ranked mineral oil as harmful (class 4) to another natural enemy found in citrus orchards, the predatory mite Iphiseiodes zuluagai Denmark and Muma, 1972 (Acari: Phytoseiidae). The fungicide and acaricide mancozeb exerts it activity through contact and by inhibiting the enzyme acetylcholinesterase in the nervous system (Pang et al. 2009). Mancozeb inflicted mortality and reduced oviposition to A. largoensis colonies in comparison with control. Adults of the predatory mite Typhlodromus pyri Scheuten, 1857 (Acari: Phytoseiidae) treated with mancozeb had low mortality and oviposition reduction (Gadino et al. 2011) while this pesticide caused total mortality on I. zuluagai populations (Reis et at. 1998).
The cotton oil and abamectin were classified as mo-derately harmful (class 3) and negatively affected the growth rate of A. largoensis. Mites exposed to abamectin had negative ri values suggesting population decline towards extinction. Together with pyridaben, abamectin drastically reduced the fecundity of the predator as shown for Er values (Table 1) and the number of descendants (Fig. 2). The cotton oil is an alternative pesticide which has been used against pests of several crops such as the coconut mite A. guerreronis (Ferreira and Michereff Filho 2002), and more studies on this oil are needed to assess its toxicity to pests and natural enemies alike. The abamectin is a broad-spectrum acaricide that have been used against pest mites in citrus orchards (Agrofit 2013). Similarly to pyridaben, abamectin causes mortality and reduces ovipositon of A. largoensis. The res-ponse to a given pesticide is species-specific. For example, abamectin is considered moderately harmful (class 3) to the predatory mite Euseius alatus De Leon, 1966, slightly harmful (class 2) to Euseius citrifolius Denmark & Muma, 1970 and innocuous (class 1) to Amblyseius herbicolus (Chant, 1959) and I. zuluagai (Acari: Phytoseiidae) (Reis et al. 2006). Pyridaben was classified as harmful (class 4) and caused the extinction of the population of A. largoensis (Nf = 0). Pyridaben is a contact acaricide that inhibits complex I of the mitochondrial respiratory pathway with a fast initial action and has been considered a non selective pesticide model for its high mortality and sharp fecundity reduction to predatory mites (Reis et al. 2006; Meyer et al. 2009; Park et al. 2011).
ConclusionsThe oils of neem and mineral as well as mancozeb are compatible with the predatory mite A. largoensis as they are less harmful to this predator. The cotton oil had an intermediate selectivity while pyridaben and abamectin were more harmful to A. largoensis and therefore should be avoided in IPM programs.
AcknowledgmentsWe thank Dr. Ester Azevedo da Silva for mite identification and the National Council for Scientific and Technological Development (CNPq) for funding.
Literature citedABRAHAM, C. M.; BRAMAN, S. K.; OETTING, R. D.; HINKLE, N. C. 2013. Compatibility of pesticides with natural enemies for pest management in greenhouse gerbera daisies. Journal of Economic Entomology 106 (3): 1590-1601. [ Links ]
AGROFIT. 2013. Sistemas de Agrotóxicos Fitossanitários. Ministério da Agricultura, Pecuária e Abastecimento - Coordenajao-Geral de Agrotóxicos e Afins/DFIA/SDA. Available: http://extranet.agricultura.gov.br/agrofit_cons/principal_agrofit_cons. Accessed: 12 March 2013. [ Links ]
BERNARDI, D.; BOTTON, M.; CUNHA, U. S.; BERNARDI, O.; MALAUSA, T.; GARCIA, M. S.; NAVA, D. E. 2013. Effects of azadirachtin on Tetranychus urticae (Acari: Tetranychidae) and its compatibility with predatory mites (Acari: Phytoseiidae) on strawberry. Pest Managment Science 69 (1): 75-80. [ Links ]
CARRILLO, D.; PENA, J. E.; HOY, M. A.; FRANK, J. H. 2010. Development and reproduction of Amblyseius largoensis (Acari: Phytoseiidae) feeding on pollen, Raoiella indica (Acari: Te-nuipalpidae), and other microarthropods inhabiting coconuts in Florida, USA. Experimental and Applied Acarology 52: 119-129. [ Links ]
CHILDERS, C. C.; DENMARK, H. A. 2011. Phytoseiidae (Acari: Mesostigmata) within citrus orchards in Florida: species distribution, relative and seasonal abundance within trees, associated vines and ground cover plants. Experimental and Applied Acarology 54 (4): 331-371. [ Links ]
CHUECA, P.; GRAFTON-CARDWELL, E. E.; MOLTO, E. 2009. Influence of spray equipment and water volume on coverage of citrus and control of citricola scale, Coccus pseudomagnoliarum (Hemiptera: Coccidae) with mineral oil. Journal of Economic Entomology 102 (1): 296-303. [ Links ]
COSTA, E. C.; TEODORO, A. V; RÉGO, A. S.; PEDRO-NETO, M.; SARMENTO, R. A. 2014. Functional response of Euseius concordis to densities of different developmental stages of the cassava green mite. Experimental and Applied Acarology 64 (3): 277-286. [ Links ]
DUSO, C.; MALAGNINI, V; POZZEBON, A.; CASTAGNOLI, M.; LIGUORI, M.; SIMONI, S. 2008. Comparative toxicity of botanical and reduced-risk inseticides to mediterranean popula-tions of Tetranychus urticae and Phytoseiulus persimilis (Acari Tetranychidae, Phytoseiidae). BioControl 47 (1): 16-21. [ Links ]
ERLER, F.; CETIN, H.; SARIBASAK, H.; SERTTAS, A. 2010. Laboratory and field evaluations of some botanical pesticides against the cedar leaf moth, Acleris undulana Journal of Pest Science 83 (3): 265-272. [ Links ]
FERREIRA, J. S.; MICHEREFF FILHO, M. 2002. Produjao integrada de coco: práticas fitossanitárias. Aracaju: Embrapa Tabuleiros Costeiros, 107 p. [ Links ]
GADINO, A. N.; WALTON, V. M.; DREVES, A. J. 2011. Impact of vineyard pesticides on a beneficial arthropod, Typhlodromus pyri (Acari: Phytoseiidae), in laboratory bioassays. Journal of Economic Entomology 104 (3): 970-977. [ Links ]
GALLO, D.; NAKANO, O.; SILVEIRA NETO, S.; CARVALHO, R. P. L.; BATISTA, G. C.; BERTI FILHO, E.; PARRA, J. R. P.; ZUCCHI, R. A.; ALVES, S. B.; VENDRAMIN, J. D.; MARCHINI, L. C.; LOPES, J. R. S.; OMOTO, C. 2002. Entomologia Agrícola, Fealq. 920 p. [ Links ]
GEIGER, F.; BENGTSSON, J.; BERENDSE, F.; WEISSER, W. W.; EMMERSON, M.; MORALES, M. B.; CERYNGIER, P; LIIRA, J.; TSCHARNTKE, T.; WINQVIST, C.; EGGERS, S.; BOMMARCO, R.; PAERT, T.; BRETAGNOLLE, V; PLAN-TEGENEST, M.; CLEMENT, L. W.; DENNIS, C.; PALMER, C.; OÑATE, J. J.; GUERRERO, I.; HAWRO, V; AAVIK, T.; THIES, C.; FLOHRE, A.; HAENKE, S.; FISCHER, C.; GOE-DHART, P. W.; INCHAUSTI, P. W. 2011. Persistent negative effects of pesticides on biodiversity and biological control potential on European farmland. Basic and Applied Ecology 11 (2): 97-105. [ Links ]
GERSON, U.; WEINTRAUB, P. G. 2012. Mites (Acari) as a factor in greenhouse management. Annual Review of Entomology 57: 229-247. [ Links ]
HANEEF, S.; SADANANDAN, M. A. 2013. Survey of predatory mites (Acari: Phytoseiidae) associated with economically important plants of north Kerala. Biological Forum - An International Journal 5 (2): 119-122. [ Links ]
HASSAN, S. A.; BIGLER, F.; BOGENSCHÜTZ, H.; BOLLER, E.; BRUN, J.; CALIS, J. N. M.; COREMANS-PELSENEER, J.; DUSO, C.; GROVE, A.; HEIMBCH, U.; HELYER, N.; HOK-KANEN, H.; LEWIS, G. B.; MANSOUR, F.; MORETH, L.; POLGAR, L.; SAMSOE-PETERSEN, L.; SAUPHANOR, B.; STÁUBLI, A.; STERK, G.; VAINIO, A.; VEIRE, M. V; VIGGIANI, G.; VOGT, H. 1994. Results of the sixth joint pesticide testing programme of the IOBC/WPRS. Entomophaga 39 (1): 107-119. [ Links ]
JAMIESON, L. E.; CHARLES, J. G.; STEVENS, P. S.; MCKEN-NA, C. E.; BAWDEN, R. 2005. Natural enemies of citrus red mite (Panonychus citri) in citrus orchards. New Zealand Plant Protection 58: 299-305. [ Links ]
MCMURTRY, J. A.; CROFT, B. A. 1997. Life-styles of phytoseiid mites and their roles in biological control. Annual Review of Entomology 42: 291-321. [ Links ]
MEYER, G. A.; KOVALESKI, A.; VALDEBENITO-SANHUEZA, R. M. 2009. Seletividade de agrotóxicos usados na cultura da macieira a Neoseiulus californicus (McGregor) (Acari: Phyto-seiidae). Revista Brasileira de Fruticultura 31 (2): 381-387. [ Links ]
MORAES, G. J. 1992. Perspectivas para o uso de predadores no controle de ácaros fitófagos no Brasil. Pesquisa Agropecuária Brasileira 27 (1): 263-270. [ Links ]
MORAES, G. J.; McMURTRY, J. A.; DENMARK, H. A.; CAMPOS, C. B. 2004. A revised catalog of the mite family Phyto-seiidae. Zootaxa 434: 1-494. [ Links ]
NAJAR-RODRÍGUEZ, A. J.; LAVIDIS, N. A.; MENSAH, R. K.; CHOY, P. T.; WALTER, G. H. 2008. The toxicological effects of petroleum spray oils on insects: Evidence for an alternative mode of action and possible new control options. Food and Chemical Toxicology 46 (9): 3003-3014. [ Links ]
NICOLETTI, M.; MARIANI, S.; MACCIONI, O.; COCCIOLET-TI, T.; MURUGAN, K. 2012. Neem cake: chemical composition and larvicidal activity on Asian tiger mosquito. Parasitology Research 111 (1): 205-213. [ Links ]
PANG, Y. P; SINGH, S. K; GAO, Y.; LASSITER, T. L.; MISHRA, R. K.; ZHU, K. Y; BRIMIJOIN, S. 2009. Selective and irreversible inhibitors of aphid acetylcholinesterases: steps toward humansafe insecticides. Plos One 4 (2): 1-13. [ Links ]
PARK, J. J.; KIM, M.; LEE, J. H.; SHIN, K. I.; LEE, S. E.; KIM, J. G.; CHOO, K. 2011. Sublethal effects of fenpyroximate and pyridaben on two predatory mite species, Neoseiulus womersleyi and Phytoseiulus persimilis (Acari, Phytoseiidae). Experimental and Applied Acarology 54 (3): 243-259. [ Links ]
REIS P. R.; ALVES, E. B. 1997. Biologia de Euseius alatus De-leon (Acari: Phytoseiidae). Anais da Sociedade Entomológica Brasileira 26 (2): 359-363. [ Links ]
REIS, P. R.; CHIAVEGATO, L. G.; MORAES, G. J.; ALVES, E. B.; SOUSA, E. O. 1998. Seletividade de agroquímicos ao ácaro predador Iphiseoides zuluagai Denmark & Muma (Acari: Phytoseiidae). Anais da Sociedade Entomológica Brasileira 27 (2): 265-273. [ Links ]
REIS, P. R.; FRANCO, R. A.; PEDRO NETO, M.; TEODORO, A. V. 2006. Selectivity of agrochemicals on predatory mites (Phytoseiidae) found on coffee plants. Coffee Science 1 (1): 64-70. [ Links ]
SARMENTO, R. A.; RODRIGUES, D. M.; FARAJI, F.; ERASMO, E. A. L.; LEMOS, F.; TEODORO, A. V.; KIKUCHI, W. T.; SANTOS, G. R.; PALLINI, A. 2011. Suitability of the predatory mites Iphiseiodes zuluagai and Euseius concordis in controlling Polyphagotarsonemus latus and Tetranychus bastosi on Jatropha curcas plants in Brazil. Experimental and Applied Acarology 53 (3): 203-214. [ Links ]
SILVA, R. R.; SILVA, M. J. S.; SILVA, E. A.; GALVAO, A. S.; REIS, P. R. 2013. Acarofauna em pomar cítrico com enfase em Phytoseiidae. Magistra 25 (3/4): 197-203. [ Links ]
STARK, J. D.; TANIGOSHI, L.; BOUNFOUR, M.; ANTONELLI, A. 1997. Reproductive potential: its influence on the sus-ceptibility of a species to pesticides. Ecotoxicology and En-vironmental Safety 37 (3): 273-279. [ Links ]
STATSOFT Inc (1984-2011) Statistica for Windows (Software-system for data-analyses) Version 10.0. Tulsa. [ Links ]
TANAKA, M.; KASHIO, T. 1977. Biological studies on Amblyseius largoensis Muma (Acarina: Phytoseiidae) as a predator of the citrus red mite, Panonychus citri (McGregor) (Acarina: Tetranychidae). Bulletin of the Fruit Tree Research Station 1: 49-67. [ Links ]
VARENHORST, A. J.; ONEAL, M. E. 2012. The response of natural enemies to selective insecticides applied to soybean. Environmental Entomology 41 (6): 1565-1574. [ Links ]
VENZON, M.; ROSADO, M. C.; MOLINA-RUGAMA, A. J.; DUARTE, V. S.; DIAS, R.; PALLINI, A. 2008. Acaricidal efficacy of neem against Polyphagotarsonemus latus (Banks) (Acari: Tarsonemidae). Crop Protection 27 (3/5): 869-872. [ Links ]
Received: 3-Dec-2014
Accepted: 1-May-2015
DA SILVA, R. R.; TEODORO, A. V; DE SOUSA SILVA, M. D. J.; REIS, P. R.; SILVA, S. S. 2015. Compatibility of pesticides with the generalist predatory mite Amblyseius largoensis(Acari: Phytoseiidae). Revista Colombiana de Entomología 41 (1): 7680. Enero-Junio 2015. ISSN 0120-0488.