Introduction
Brazilian production of peanut (Arachis hypogaea L.) during season 2013/2014 reached 289.4 million tons occupying 106.9 million hectares. São Paulo State is the major producer with 257.4 million tons (Conab 2014). Bahia, Sergipe, Ceará and Paraíba States are the main producers in the Northeast region, however, their production is not sufficient to supply the regional annual demand (50 million tons of pods), which represents the second consumer market in Brazil (Suassuna et al. 2006).
Diverse insects and mites attack peanut during growing. Their occurrence does not always represent a risk to peanut yield; it depends on the population level of these organisms and the damage caused by the pests (Almeida 2013). However, severe losses can occur if it overcome the tolerance capacity of the peanut crop and control measures are not taken correctly (Lourenção et al. 2007; Calore et al. 2012).
The main peanut pests are trips and the red necked peanut worm, but leafhoppers, caterpillars, mites, bugs, termites and scales may occur and cause serious yield losses (Almeida 2013).
Among scales, a cosmopolitan and polyphagous species, the mealybug Dysmicoccus brevipes Cockerell, 1893 (Hemiptera: Pseudococcidae), is worthy of attention. This mealybug lives in colonies and is found in protected places in the plant (Luz et al. 2005). Female are approximately three millimeters long, oval-shaped, pink colored, covered with white waxy and powdery secretion and surrounded by 17 pairs of waxy filaments, the four posteriors being stronger than the others (Santa-Cecília et al. 2007). Males are winged and have antennas with eight segments and a pair of caudal filaments similar to those seen in the females (Beardsley 1965). This insect is common in tropical regions and it is found in a wide variety of plants (Williams and Willink 1992; Huang and Ching-Yi 2014) in roots, leaves and fruits (Ben-Dov 1994; Santa-Cecília et al. 2007). The origin center of D. brevipes is uncertain, although it is believed to be Central or South America (Waterhouse 1998).
Despite the importance of D. brevipes as cosmopolitan pest, there is no information in peanut crop. Thus, this work aimed to assess the productivity losses caused by this mealybug in this crop.
Materials and methods
This work was carried out in the Laboratory of Entomology and in the Experimental Field of Embrapa Cotton, in Campina Grande, Paraíba State, Brazil, at 7º13’50”S 35º52’52”W. The soil where the crop was grown was analyzed by the Soil Laboratory of Embrapa Cotton and classified as eutrophic Regosol (Silva 2009), however, no need of fertilizers was found.
Dysmicoccus brevipes (Cockerell) was identified by Dr. Ernesto Prado (Visiting Researcher), based on adult female’s morphological characters. The identified specimens were stored in the vouchers collection at the Laboratory of Biological Control of Pests of the EPAMIG/URESM/EcoCentro for future and eventual comparisons.
Peanut cultivar BR1 seeds were sown in soil naturally infested by D. brevipes in a field area of 109.2 m2 (4.2 m x 26 m). The peanut plot consisted of rows spaced 0.7 m apart with 0.20 m between the plants in the row, keeping one plant per hole after emergence. Each experimental unit consisted of three rows of 2.5 m in length. All the agronomic practices for the conventional development of peanut crop were used.
We used completely randomized design because the physicochemical characteristics of the selected soil was enough homogeneous. Eighty randomly plants were harvested 90 days after planting. The percentage of roots and pods naturally infested by D. brevipes were evaluated assigning them a scale from one to five, which constituted the treatments. Weight and number of pods were also quantified to establish correlation with the number of mealybug per plant. The scale used was as follows: T1 = zero mealybug per plant; T2 = one to 20 mealybugs per plant; T3 = 21 to 40 mealybugs per plant; T4 = 41 to 80 mealybugs per plant and T5 = more than 80 mealybugs per plant. Five treatments and sixteen repetitions were used.
Productivity (kg.ha-1 pods) was determined through manual harvesting in a 2.0 m2 area for each experimental unit. Pods were weighted and the yield (kg.ha-1) was estimated.
The number and the weight of attacked peanut pods by adults and nymphs of the mealybug were subjected to analysis of variance and regression, using the Statistical Analysis System and Genetic (SAEG) of the Federal University of Viçosa (Ribeiro Jr. 2001).
Results and discussion
There were no infestations of other insects than mealybug or diseases on the peanut plants during the growing season.
Dysmicoccus brevipes specimens were observed infesting roots and pods in 84 % of the sampled peanut plants (Fig. 1), ranging from zero to 147.1 (Table 1). Peanut plants infested by D. brevipes had their growth paralyzed and presented yellowish, wilted and finally dried leaves (Figs. 1A, B). Green plants presented mealybug colonies with nymphs and adult females on roots and pods (Figs. 1C, D). On dried plants, mealybugs were rare, probably due to insect migration. This behavior was also observed in pineapple plants attacked by mealybugs (Lacerda et al. 2009).

Figure 1 Dysmicoccus brevipes (Hemiptera: Pseudococcidae) on peanuts. A. Yellowish leaves. B. Dried roots. C. Dried pods. D. Nymphs and adult females. Campina Grande, Paraíba, Brazil.
Table 1 Mean numbers of mealybugs, pods and weight of pods per plant, estimative of productivity of pods and losses of peanuts at five densities of Dymicoccus brevipes (Hemiptera: Pseudococcidae), at different ages. Campina Grande, Paraíba, Brazil.
| Treat. | Means ± Standard Error | Productivity | |||
|---|---|---|---|---|---|
| Mealybugs per roots and pods | Pods per plant | Pod Weight per plant (g) | Pods (Kg.ha -1 ) | Losses (%) | |
| T1 | 0.0 ± 0.0 | 32.1 ± 3.9 a* | 65.8 ± 10.0 a | 3,290.0 | - |
| T2 | 9.6 ± 1.6 | 28.6 ± 2.6 a | 51.4 ± 6.9 ab | 2,570.0 | 21.9 |
| T3 | 28.1 ± 1.7 | 31.9 ± 4.5 a | 49.5 ± 8.3 ab | 2,475.0 | 24.8 |
| T4 | 58.1 ± 3,3 | 31.3 ± 5.3 a | 43.3 ± 6.4 ab | 2,165.0 | 34.2 |
| T5 | 147.1 ± 11.1 | 21.1 ± 2.8 a | 33.9 ± 6.4 b | 1,695.0 | 48.5 |
| F | - | 1.82 | 3.05 | - | - |
| p-value | - | 0.13 | 0.02 | - | - |
| d.f.** | - | 4 | 4 | - | - |
Treatments (Treat.): T1= No infested (zero mealybug per plant); T2= from one to 20 mealybugs per plant; T3= from 21 to 40 mealybugs per planta; T4=from 41 to 80 mealybugs per plant and T5= more than 80 mealybugs per plant. *Means followed by the same lowercase letter in the column are not different by Tukey test. **Degree of freedom.
Pod mean number per peanut plant did not differ between treatments (Table 1). Seed number per pod and pod number per plant is a high heritability feature determined by fertilization (Smith 1956) and not affected by mealybugs at this stage of development.
The pod mean weight ranged from 32.1 g to 33.9 g (Table 1 and Fig. 2). The reduction of pod weight was directly related to the increasing number of mealybug (Table 1 and Fig. 2) and explained by the reduction of pod length and weight. Infested pineapple plants with D. brevipes had growth paralization, decrease of the leaves number, roots length and weigth of leaves and dry roots (Lim 1973; Colen et al. 2001). In vine, mealybugs cause yield losses mainly due to mealybugs feeding on the berries, together with sooty mold development on leaves and fruits, serving as subtract to saprophytic fungi development (González and Voloski 2004; Daane et al. 2008). Mealybug infestations also reduced the sugar content, development and colour of berries and plant vigour (Godfrey et al. 2002; Bertin 2010, 2013).
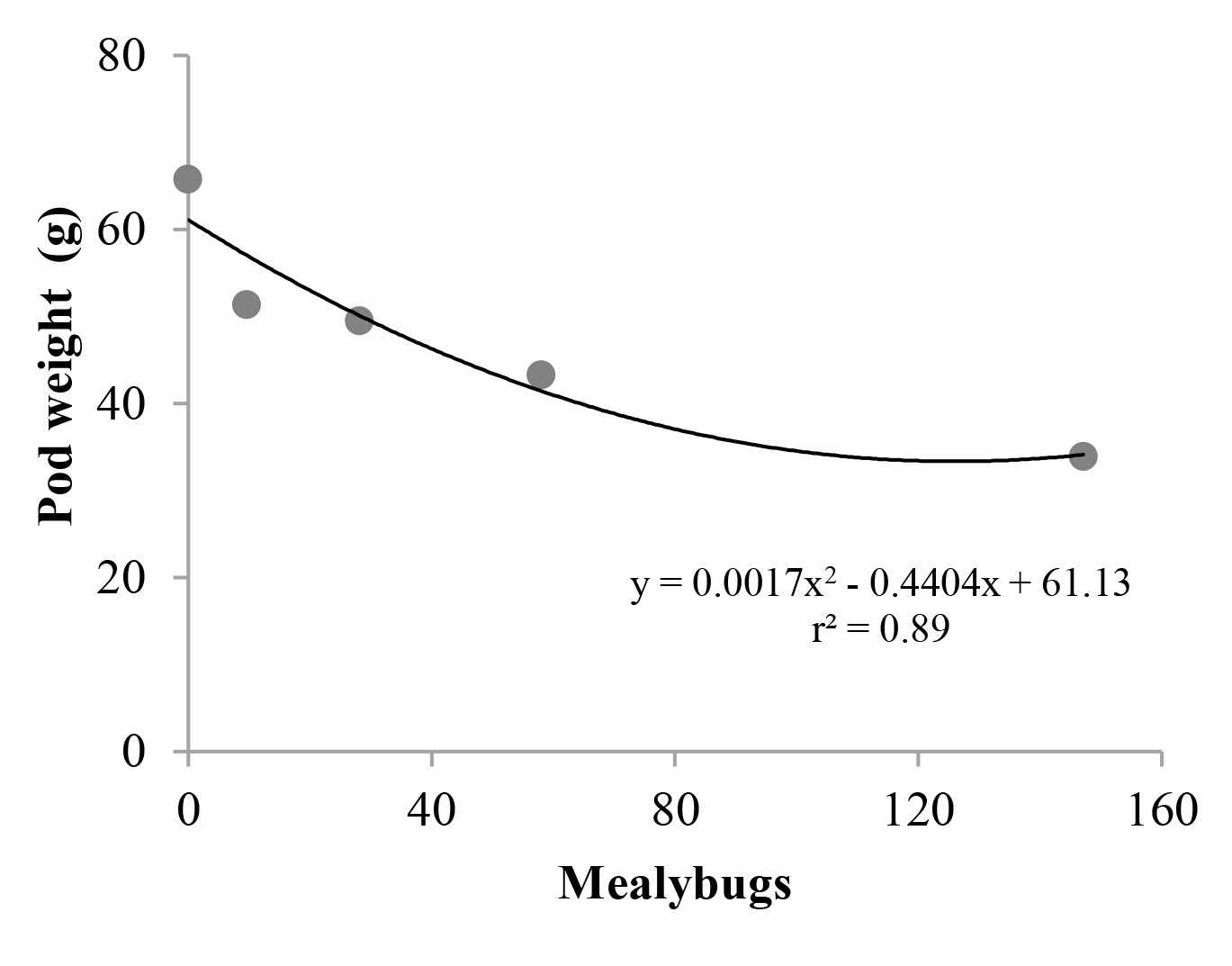
Figure 2 Dysmicoccus brevipes (Hemiptera: Pseudococcidae) infestation effect on peanut pod weight. Campina Grande, Paraíba, Brazil.
A loss of 48.5 % of productivity was observed between the highest infestation level (T5) and un-infested treatment.
The yield reduction showed here justifies control measures of this mealybug at high population level. However, it is still unknown the exact economic injury level and economic threshold of this pest on peanut. Our data indicated that high pineapple mealybug infestations are able to reduce yield as high as 47 %.














