Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
Revista Colombiana de Ciencias Pecuarias
Print version ISSN 0120-0690On-line version ISSN 2256-2958
Rev Colom Cienc Pecua vol.24 no.4 Medellín Oct./Dec. 2011
Influenza virus A H5N1 and H1N1: features and zoonotic potential¤
Virus de Influenza A H5N1 y H1N1: características y potencial zoonótico
Os virus da Influenza A H5N1 e H1N1: características e potencial zoonótico
Diana M Echeverry1*, MV; Juan D Rodas2*, MV, PhD
1Grupo de investigación Biogénesis, Facultad de Ciencias Agrarias, Universidad de Antioquia, AA 1226, Medellín, Colombia.
2Grupo de investigación CENTAURO, Facultad de Ciencias Agrarias, Universidad de Antioquia, AA 1226, Medellín, Colombia.
(Recibido: 16 febrero, 2011; aceptado: 25 octubre, 2011)
Summary
Influenza A viruses which belong to the Orthomyxoviridae family, are enveloped, pleomorphic, and contain genomes of 8 single-stranded negative-sense segments of RNA. Influenza viruses have three key structural proteins: hemagglutinin (HA), neuraminidase (NA) and Matrix 2 (M2). Both HA and NA are surface glycoproteins diverse enough that their serological recognition gives rise to the traditional classification into different subtypes. At present, 16 subtypes of HA (H1-H16) and 9 subtypes of NA (N1-N9) have been identified. Among all the influenza A viruses with zoonotic capacity that have been described, subtypes H5N1 and H1N1, have shown to be the most pathogenic for humans. Direct transmission of influenza A viruses from birds to humans used to be considered a very unlikely event but its possibility to spread from human to human was considered even more exceptional. However, this paradigm changed in 1997 after the outbreaks of zoonotic influenza affecting people from Asia and Europe with strains previously seen only in birds. Considering the susceptibility of pigs to human and avian influenza viruses, and the virus ability to evolve and generate new subtypes, that could more easily spread from pigs to humans, the possibility of human epidemics is a constant menace. A recent example was the outbreak of H1N1 influenza in 2009 that crossed species and geographical borders affecting up to 20 to 40% of the people in some parts of the world. Vaccines and control measures are continuously being developed to address a threat that every year claims human and animal lives, and makes us fear for a new and more lethal strain of the virus.
Key words: H1N1, H5N1, influenza virus, pandemic, zoonotic.
Resumen
Los virus de influenza tipo A pertenecen a la familia Orthomyxoviridae, son envueltos, pleomórficos, y contienen 8 segmentos de ARN de cadena negativa. Dichos agentes poseen tres proteínas estructurales claves: Hemaglutinina (HA), Neuraminidasa (NA) y proteína de Matriz 2 (M2). La HA y la NA son glicoproteínas de superficie, que debido a su gran diversidad fueron usadas para crear la clasificación y nomenclatura de subtipos de virus de influenza, basada en la reactividad serológica contra ellas. De acuerdo con lo anterior, hasta la fecha se han identificado 16 subtipos de HA (H1-H16) y 9 subtipos de NA (N1-N9). De los virus tipo A, los denominados H5N1 y H1N1 han demostrado ser los más patógenos. El contagio directo de los virus de influenza A de las aves a los humanos se consideraba un evento raro y su dispersión se creía que era aún más limitada, percepción que ha cambiado desde 1997 y particularmente con los últimos brotes zoonóticos de influenza en Asia y Europa. Adicionalmente, si se considera que el porcino es una especie susceptible a virus de influenza provenientes de humanos y aves, y que estos agentes infecciosos tienen la capacidad de realizar un cambio antigénico al reasociarse generando así nuevos subtipos, la potencialidad de nuevas amenazas a la población humana es cada vez más tangible. Ejemplo de lo anterior es la reciente pandemia de influenza A H1N1 en el año 2009, que atravesó fronteras sin discriminación geográfica o de especie, afectando entre el 20 y el 40% de las personas en algunas partes del mundo. Vacunas y medidas de control se desarrollan en la actualidad para afrontar esta amenaza que cada año cobra vidas humanas y animales, y nos hace temer por una nueva y más mortífera epidemia en el futuro.
Palabras clave: H1N1, H5N1, pandemia, virus Influenza A, zoonosis.
Resumo
Os vírus da influenza A pertencem à família Orthomyxoviridae, são envolvidos, pleomórficas, e contem oito segmentos de RNA de cadeia negativa. Os vírus da influenza têm três principais proteínas estruturais: HA (hemaglutinina), NA (neuraminidase) e proteína de matriz 2M. A hemaglutinina e a neuraminidase são glicoproteínas de superfície, que dão nome a os tipos e subtipos dos vírus da Influenza até agora têm sido identificados 16 subtipos da HA (H1-H16) e nove subtipos da NA (N1-N9). Dos vírus tipo A, chamado H5N1 e H1N1 tem demostrado que são os mais patogênicos. A transmissão direta do vírus da influenza A das aves para os humanos foi considerado um evento raro e sua propagação parecia ser mais limitada, esta percepção mudou com os recentes surtos zoonóticos da influenza. Além disso, considerando que o suíno é uma espécie suscetível ao vírus da influenza de humanos e aves, e que esses agentes infecciosos são capazes de fazer uma alteração no antígeno ao associar novamente, gerando assim novos subtipos, o potencial para novas ameaças na população humana, esta tem-se tornando mais palpável. Um exemplo disso é a recente pandemia da influenza H1N1 no 2009, que atravessou as fronteiras sem discriminação geográfica ou de espécie afectando entre o 20 o 40% das pessoas em algumas partes do mundo. Vacinas e medidas de controle são atualmente desenvolvidas para tratar a pandemia que ainda hoje continua a afetando vidas humanas e animais, e o medo latente de uma nova e mais mortal ainda não chega.
Palavras chave: avicultura, pandemia, suinocultura, zoonose.
¤ To cite this article: Echeverry DM, Rodas JD. Influenza virus a H5N1 and H1N1: features and zoonotic potential. Rev Colomb Cienc Pecu 2011; 24:647-662
* Corresponding author: Juan David Rodas. Universidad de Antioquia, Ciudadela Robledo, Calle 75 # 65-87 Bloque 47-150. Medellín, Colombia. E-mail: juandavid.rodas@gmail.com.
Introduction
From the beginning of the XX century, influenza viruses became a human threat and drawback for economic development across the world; a zoonotic agent that through history has been one of the most frightening causes of pandemics, imposing high tolls on animal and human lives (Potter, 2001). Apparently, the first records of influenza infections in America occurred at the time when Columbus arrived to the American continent and, with him, the introduction of horses and pigs from Europe (Manrique et al., 2009).
Influenza viruses are classified into three antigenically distinct types: A, B and C, according to the differences in their nucleoproteins (NP) and matrix proteins (M1) (Cheung and Poon, 2007). Some of the most virulent type A influenza viruses (H1N1 and H5N1 among others) have been responsible for serious human pandemics and can be found in humans, pigs, birds, horses, water mammals, cats and dogs, raising the possibility of transmission from animals to humans and vice versa (Cheung and Poon, 2007; Iftimovici et al., 1979). Since the Spanish flu outbreak of 1918, which claimed millions of human lives, the influenza pandemics are appearing with intervals of 10 to 15 years, usually accompanied by variations in their pathogenicity (Hsieh et al., 2006). Different strains of type A influenza viruses are still circulating in animal and human populations, showing their ability to mutate, and generate new reassorted viruses, against which there is no immunological memory. Once mutated, they can spread quickly and easily from person to person, posing a worldwide threat to public health due to international traveling. Many efforts are currently underway in order to control and prevent the dissemination of these viruses. Common measures include the development of new vaccines, the promotion of hygiene practices, or even restrictions on international trips and bans on international commerce. Understandably, the unique features of influenza viruses are a constant challenge to the scientific community.
Biology of the Influenza A Virus
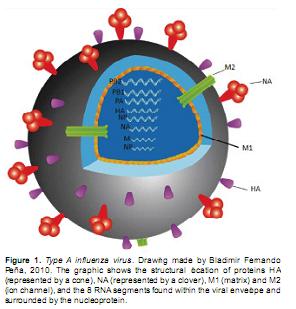
Influenza A is an enveloped pleomorphic virus that belongs to the Orthomyxoviridae family (Vincent et al., 2008; Cheung and Poon, 2007; Kalthoff et al., 2010). Flu viruses have their genome wrapped in helical capsids and adopt different shapes varying from small spherical to long filamentous forms. Its genome consists of 8 single stranded negative-sense segments RNA and the virus particle contains a lipid envelope which is acquired from the host cell membrane during the viral budding process (Cheung and Poon, 2007; Webster et al., 1992; Lamb and Krug, 2001). Three proteins: hemagglutinin (HA), neuraminidase (NA) and Matrix (M2) are included in its envelope, two of which, HA and NA, are surface glycoproteins.
Up to date, 16 subtypes of HA (H1- H16) and 9 subtypes of NA (N1-N9) have been identified. A typical structure of an influenza A virus with its outer envelope studded with glycoproteins HA and NA is shown in Figure 1 (Cheung and Poon, 2007). The first segment codes for one of the subunits of the viral polymerase PB2. This PB2 has an endonuclease activity and uses the host mRNA to generate cap primers. These primers are "stolen" from the cellular mRNA 5' end, and are used for the synthesis of viral mRNA through a mechanism known as "cap snatching" (Cheung and Poon, 2007).
The second segment codes for the polymerase PB1 subunit that is also part of the RNA polymerase. The third segment encodes the polymerase acidic protein (PA), which is the smallest subunit of the RNA polymerase complex. PA is essential for the transcription and viral replication, allowing nuclear accumulation of the PB1 subunit (Cheung and Poon, 2007). All the proteins that make part of the viral polymerase complex contain signals for nuclear localization that are required for their transportation to the nucleus. The fourth segment of the influenza virus genome encodes the HA molecule, this protein is responsible for the binding of viral particles to the host cell receptors that contain sialic acid (Lamb and Krug, 2001). The HA is synthesized as the HA0 polypeptide precursor, which undergoes posttranslational cleavage to generate subunits HA1 and HA2. HA cleavage is a requirement for the viral particle to be infectious and is considered to be the major determinant for virulence (Laver et al., 1984). The fifth segment encodes the nucleus protein (NP); which is an essential component for viral transcription and replication and also plays an important role for the nuclear transport of viral RNA (vRNA) (Kistner et al., 1989; Winter and Fields, 1981).
The sixth segment codes for NA, a surface glycoprotein with receptor-destroying activity by cleavage of the ligand alpha-quetosidic between a terminal sialic acid and an adjacent residue D-galactose or D-galactosamine residue (Matrosovich et al., 2004). Liu et al suggest that the NA molecule is not required for entry, replication and viral assembly; however, when its activity is inhibited, the progeny of viral particles remain stuck together or attached to the cell surface forming large aggregates (Matrosovich et al., 2004; Liu et al., 1995). Consequently, we can assume that NA plays an essential role in the release of the viral progeny from infected cells. The seventh segment encodes matrix proteins, M1 and M2. In the viral particle, the M1 protein forms a layer to separate the ribonucleoprotein (RNP) from the viral membrane and interacts with the viral RNA, vRNA, and the RNP protein components during the assembly and disassembly of the influenza A virus.
M1 it also plays a role on the binding to the cell membrane and the viral morphology. The M2 protein is an integral trans-membrane protein enclosing an ion channel essential for the pH regulation, the acidification inside the viral particle is necessary for the denudation step into the cell. M2 plays an essential role on viral replication and is a likely target for therapeutical intervention (Schnell and Chou, 2008). In addition, the endosomal acidification of the virus particle is required for its replication because it leads to the dissociation of virus ribonucleoproteins, vRNPs, from M1 proteins that enter to the nucleus. The ion channel activity of M2 is also important to maintain a high pH in the Golgi vesicles in order to stabilize the natural conformation of the new synthesized HA molecule during the intracellular transport for the viral assembly (Schnell and Chou, 2008; Pinto et al., 1992; Mould et al., 2003).
The eighth segment encodes two more proteins: NS1 and NS2. The NS1 is the only nonstructural protein of influenza virus (Bergmann et al., 2000; Qiu and Krug, 1994). Recently, it was discovered that NS1 from H5N1/97 gave the virus the ability to be less susceptible to the antiviral effects of the tumor necrosis factor and interferon (Qiu and Krug, 1994; Steidle et al., 2010). Some scientists claimed that NS1 may induce a cytokine imbalance and this probably contributes to the extreme pathogenicity of the avian influenza viruses in humans (Bergmann et al., 2000). Finally, the NS2 protein, considered the least abundant among the proteins synthesized by the influenzavirus, plays an important role in promoting the normal replication of the viral RNA, and performs and important function as a carrier protein for vRNA inside the nucleus and the processed form (viral mRNA) to the cytoplasm for later translation into proteins (nuclear traffic) (Qiu and Krug, 1994; Steidle et al., 2010).
RNA transcription and replication of the influenza virus type A
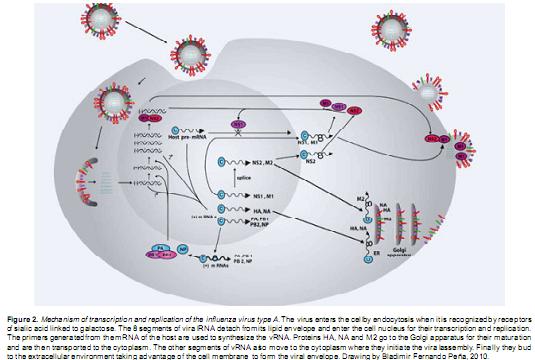
Right after the virus enters into a susceptible cell, viral disassembly takes place and cell nucleus becomes the target site for transcription and replication of the viral genome. The polymerase subunit PB2 binds to the 5' end of the host cell mRNA and the cleavage occurs to generate primers for the primary viral transcription (Fodor et al., 1995; Braam et al., 1983; Hoffmann et al., 2000). At this stage, the eight segments of mRNA are synthesized in equimolar quantities (Fodor et al., 1994; Hoffmann et al., 2000; Flick et al., 1996). The amount of viral proteins synthesized is dependent on the amount of corresponding mRNA in the infected cells. The secondary transcription can be divided into two phases, early and late phase. In the early phase the vRNA of NS1 and NP are preferentially synthesized, and as a result, NS1 and NP are the predominant viral proteins in infected cells at this point (Klumpp et al., 1997). During the late phase, vRNA are synthesized on equivalent amounts, as necessary for the genome of the viral progeny. Then, the NS1 protein is synthesized at a reduced level, meanwhile the mRNA of HA, NA and M1 are preferentially expressed.
In general, most of the mRNA completely processed and polyadenylated must be transported from the nucleus to the cytoplasm with the help of the NS2 protein for the protein synthesis. These membrane proteins (HA, NA and M2), pass also through the secretory pathway to the Golgi apparatus for a subsequent maturation: HA and NA proteins are post-translationally modified and transported to the cell surface for integration into the cell membrane (Matrosovich et al., 2004). The M1 protein forms a layer to separate the RNP of the viral membrane, and interacts with the vRNA and RNP components for the assembly and viral disassembly, and then binds to the cell membrane and becomes important in the acquisition of the virus morphology (Figure 2) (Hay et al., 1977; Hay et al., 1982; Shapiro et al., 1987).
Subsequently, once all the viral segments (genome) merge and the structural viral proteins inserted in the cellular membrane become part of the viral envelope, the virus assembly takes place to produce the new viral progeny.
Transmission of Influenza Virus type A
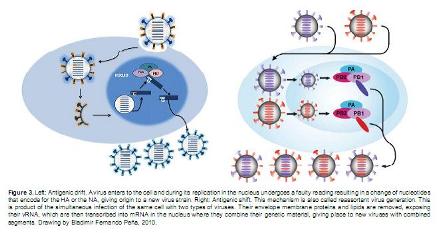
The direct transmission of the influenza A viruses from birds to humans used to be considered an extremely rare event. However, this perception began to change in 1997 when an H5N1 virus was transmitted directly from poultry to man, by direct contact with sick animals and/or their environment (Horimoto and Kawaoka, 2005). Even though during the last century there have been some reports of human infections with influenza virus subtype H1N1 and H3N2 coming from pigs, the risk of this kind of transmission has been considered very low (Vincent et al., 2008; Spronk, 2001). Nevertheless, these viruses pose a pandemic threat due to their extraordinary ability to evolve, producing new viral subtypes or viral strains for which human populations have no immunity (Horimoto and Kawaoka, 2005). The strategies that influenza virus displays to mutate and evolve come in two forms commonly known as antigenic "drift" and antigenic "shift". The first one (antigenic "drift"), consists of single point mutations of the genome which can alter HA or NA structure resulting in a different strain of the virus (Figure 3 left).
The second form (antigenic "shift"), consists of reassortment of segments which can affect the HA, NA, or other proteins (Figure 3 rigth). These changes usually confer the newly evolved virus a greater chance of aerosol transmission from person to person Direct contact with secretions and excretions from sick individuals has also been reported as a very easy way to get infected (Ungchusak et al., 2005; Horimoto and Kawaoka, 2005).
Even though, H5N1 virus transmission between humans has been considered a very limited event, only found between individuals in very close proximity, i.e. members of the same family or nurses and physicians taking care of ill people, it has been argued that this condition could change at any time due to the virus ability to generate antigenic changes and gain greater tropism for its entry and replication in other tissues, as it has been recently shown by the pandemic with the swine influenza A H1N1 virus (Van-Reeth, 2007; Newman et al., 2008).
Some years ago, this agent was also seen as a virus with limited transmission between humans, even though there were some reported cases of human disease and asymptomatic infections in the U.S. (Newman et al., 2008). Today, it has been recognized that certain factors, such as possession of pets and poor sanitation, may favor infection and dissemination of influenza virus on epidemic proportions. For example, some of the cities with the worst records in historic pandemics did not have good sanitary and hygienic conditions, let alone preparedness for emergencies and contingency plans. The spread of the influenza virus throughout the world is now facilitated by air plane traveling. The recirculation of air inside the cabin and small size of the virus to evade filters allows the virus to remain suspended in the air and a continuous exposure through the respiratory tract (Valderrama C, 2009).
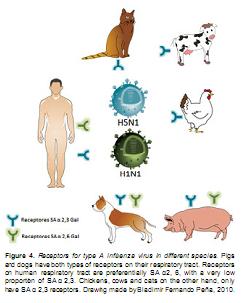
It has been described that when human influenza viruses enter the host, they are recognized by cellular receptors that contain sialic acid linked to galactose by the ligand 2,6 (SA α2, 6Gal) (Horimoto and Kawaoka, 2006). On the other hand, avian influenza viruses recognize mainly sialic acid cell receptors linked to galactose by the ligand 2.3 (SA 2, 3Gal) (Gambotto et al., 2008; Horimoto and Kawaoka, 2005; Horimoto and Kawaoka, 2006). It has also been shown that on the human tracheal/ bronchial epithelium the predominant receptors found on cells are type SA α2, 6 Gal and seemingly, this epithelium "in vitro" has a very low proportion of receptors type SA α2, 3 Gal (Shinya et al., 2006, Horimoto and Kawaoka, 2005; Horimoto and Kawaoka, 2006). Conversely, on birds, the cells of the respiratory and digestive tract contain mainly the receptor type SA α2, 3 Gal and it is thought that this fundamental difference may explain the preferential tropism of a viral subtype by one or the other species (see figure 4) (Gambotto et al., 2008; Horimoto and Kawaoka, 2005).
Pigs, on the other hand, have both types of receptors, SA α2,6 Gal and SA α2,3 Gal on their tracheal epithelial cells in almost equal proportion, which could explain why they are a " mixer vessel" susceptible to viruses from human and avian origin. An infection of pigs with viral subtypes from different sources could lead to the generation of reassortant agents, an after continuous passes and replications to the danger that easier transmission between humans has been acquired by the virus (Yu et al., 2009). It was recently found that dogs are also susceptible to infection by the H5N1 virus, replicating easily within the respiratory tract. The susceptibility of the canine species to this virus is given by the expression of receptors SA α2,3 and SA α 2,6 linked to glycans that are expressed mainly in the epithelial cells of the respiratory tract of dogs, similar to what happens in pigs (Chen et al., 2010; Giese et al., 2008; Maas et al., 2007). This finding alerts to the possibility that H5N1 virus could potentially be transmitted from canine to human.
Interestingly, during the outbreak of influenza A H5N1 in Asia in 2003, 4 felines in the zoo of Suphanburi (Thailand), became infected with this subtype and died after consuming chicken from a poultry farm of the same area. Receptors of sialic acid joined to galactose by the SA α 2,3 ligand which specifically binds to this subtype were found in the lower respiratory tract of the felines (Keawcharoen et al., 2004). Furthermore, since domestic cats have been experimentally infected with H5N1 virus developing a fatal disease, there is a chance that influenza in cats could be zoonotic and the virus was spread with their secretions (Thiry et al., 2007).
It has also been experimentally shown that influenza A virus can infect bovine species, and their secretions could be a source of transmission (Kalthoff et al., 2008). Likewise, some domestic rodents have been experimentally infected with the same virus (H5N1) with similar results, further adding to the list of potential sources of infection to humans (Thiry et al., 2007). In nature, many migratory birds are asymptomatic carriers of H5N1 virus and have traditionally been the primary reservoirs of avian influenza, increasing the risk of spreading the virus throughout the world and also transmitting the virus to susceptible domestic birds. The sport of duck/ goose hunting and then entering a poultry house has also been associated with outbreaks of influenza in turkeys and chickens (Van Reeth, 2007).
Epidemiology
Influenza type A viruses are the main, but not the only, etiologic agents of annual flu outbreaks and also the cause of pandemics that periodically affect humans and animals. Influenza type A is also considered a major biological threat due to its capacity for inter- species transmission (Franco-Giraldo and Álvarez- Dardet, 2009), and because zoonotic transmission of the most fatal H5N1 and H1N1 from animals to humans has increased during recent years (Perdue and Swayne, 2005; Franco-Giraldo and Álvarez- Dardet, 2009). It has been previously established that flu pandemics are likely to appear at intervals of 10 to 15 years with the emergence of new re-assorted viruses that spread rapidly within the world population (Kuri-Morales et al., 2006). In 1918, the pandemic known as the Spanish flu, caused by an H1N1 virus apparently of avian origin, produced the most deadly outbreak in human history, claiming the lives of 40 million victims in a single year (Potter, 2001). This was only comparable to the medieval pest (caused by Yersinia pestis), or the more recent AIDS pandemic (Acquired Immune Deficiency Syndrome caused by the Human Immuno-deficiency Virus, HIV) at the end of the XX century. The flu outbreak coincided with the appearance of the swine type H1N1influenza virus (SI) in the Midwest of United States (US), which was clinically recognizable but not isolated until 1930 (Vincent et al., 2008; Horimoto and Kawaoka, 2005; Zhou et al., 1999).
The Spanish flu was followed on the XX century by other less aggressive pandemics such as the 1957 Asian influenza, caused by an H2N2 type, with HA, NA and PB1derived from an avian virus and other gene segments derived from a previously described human influenza virus (Potter, 2001). About 10 years later (1968), a new pandemic initiated in Hong Kong by an H3N2 virus that possessed the HA and PB1 from avian origin in a background of genes from another human influenza virus (Horimoto and Kawaoka, 2005; Potter 2001). In this case the new HA from an avian precursor allowed the H3N2 to evade the human immune response (Horimoto and Kawaoka, 2006).
The next pandemic started in Russia on 1977 and was caused by a strain essentially identical deliberately reintroduced into the human population after being frozen for many years (Potter, 1998; Stieneke-Grob et al., 1992). More recently, in Hong Kong on 1997, a H5N1 virus of avian origin crossed for the very first time (as far as it is known) the "inter species barrier", infecting 18 people, and killing 6 of them. This unprecedented virus was transmitted directly from chickens to humans (Kalthoff et al., 2010). After that, on 1998 in the United States, triple reassortant viruses, i.e., viruses containing genes derived from human, swine and avian influenza, were isolated, but not until 2005 when a case of human infection with this virus was reported in the US (Newman et al., 2008).
Scientific reports show that between 1997 and 2001, the H5N1 virus has continued circulating in birds in the southwest of China, causing in 2003 two human deaths in Hong Kong and seriously affecting the Asian poultry industry (Horimoto and Kawaoka, 2005; Kalthoff et al., 2010; Sturm-Ramirez et al., 2004). In August 2006, the World Health Organization (WHO) reported 240 confirmed cases of H5N1 infection in humans around 10 countries with 141 of those dying, (59%) (Horimoto and Kawaoka, 2006). Some years earlier, in 2002, a new H9N2 subtype had been detected in swine of Hong Kong, and is now panzootic in poultry of Europe and Asia (Sturm-Ramirez et al., 2004), and caused, respiratory illness in at least one child of Hong Kong in 2003 (Horimoto and Kawaoka, 2005).
More recently, on April 11 of 2009 the first confirmed human case of influenza A H1N1 appeared in México, and spread to America, Western Europe, far East and Oceania, being diagnosed in more than 214 countries (WHO, 2009). On May 6th of that year, the first case of infection with the H1N1 virus was reported in Colombia and by the beginning of 2010 the number of cases had gone up to 152,560 of which 205 were fatal. By the middle of 2010 the WHO estimates that scaled up for the initially called swine H1N1 influenza virus scaled up to 18,366 deaths. At the end of that year was determined that the H1N1 strain was a triple re-associated genetic agent with segments of avian, swine and human strains, that mutated and jumped between species, i.e. from pigs to humans, and then became easily transmissible from human to human (WHO, 2009).
Regarding the human susceptibility toward these viruses, it is generally accepted that for common flu the most vulnerable human population to epidemic flu strains are the elderly (people over 60) and children. By contrast, pandemic influenza strains can affect any age group, and even harder the stronger individuals between ages of 20-40. In Colombia health authorities have estimated an approximate human lethality of 8% for influenza type A virus, related to seasonal rainy periods (Ramírez et a.l, 2009).
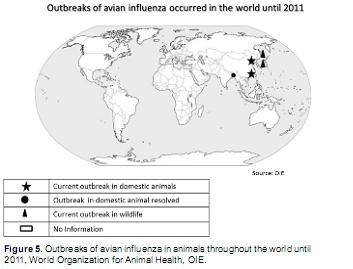
In some parts of the world, human outbreaks of influenza virus type A coincide with outbreaks in food animals. This has been a common scenario in Asia, where avian influenza subtypes remain circulating in populations of wild and domestic birds making it difficult to eradicate and control the human infection, and facilitating the persistence of the virus, which is considered to be endemic in several regions of that continent (see figure 5) (Rodríguez et al., 2006; Franco-Giraldo and Álvarez-Dardet, 2009; Gambotto et al., 2008, Horimoto et al., 2005, Hsieh Y et al., 2006). Concerning other domestic species, pig susceptibility to infection with avian and human influenza viruses is considered a serious public health risk, as it was shown on the recent pandemic of 2009 caused by the H1N1 virus, where swine where the initial link in the transmission chain. Accordingly, a study performed between 1958 and 2005 recorded 50 cases of human infection with influenza virus from pigs, and only in the U.S. during the year 2005 there were 11 cases of patients infected with a virus of swine origin (Alfonso, 2010). In 2009 viruses wholly or doubly re-associated strains of influenza virus were isolated from a swine farm in Canada (Pig- Human) (Vincent et a.l, 2009).
Despite of the serious human implications that swine influenza may have, the implementation of epidemiological surveillance for swine influenza has been inadequate, to the point that by the year 2010, this disease was not yet listed by the World Organization for Animal Health (OIE) as officially reportable disease. Likewise, the Colombian authorities represented by the Instituto Colombiano Agropecuario, ICA, have not yet implemented a program for the official control of this disease, starting with the free movement of pigs across the borders with neighboring countries (Hanssen, 1977). Nevertheless, it is worthwhile mentioning that, regarding the control of influenza virus in Colombia, the ICA has a surveillance program (only for Avian flu), and to date no cases of the highly pathogenic influenza A viruses (HVAF) have been reported.
Human clinical manifestations
Initial symptoms of infection by the influenza A H5N1 and H1N1 strains are the usual respiratory problems of any common cold, but soon progress to a more complicated form (Newman et al., 2008). In most cases of human H5N1 infection, persons show symptoms such as fever, cough and sneezing 2 to 4 days post-exposure, and then start exhibiting difficulty breathing. Evidence of pneumonia is also found in chest X-ray examination, and some also show extensive bilateral infiltration, lobular collapse and focal consolidation (Horimoto and Kawaoka, 2005; Ungchusak et al., 2005; Treanor et al., 1999).
Some of the patients infected with the H5N1 virus may develop gastrointestinal compromise, that may be accompanied by diarrhea, vomiting and abdominal pain. There have been reported cases of people who reach coma and others who develop encephalitis (Gamboto, 2008). Clinical complications of infection with influenza virus H5N1 may include also respiratory distress syndrome, renal dysfunction and/or multiple organ failure with or without REYE´s syndrome (Ungchusak et al., 2005). The pandemic strains of Influenza A H1N1 have shown very similar symptoms to the ones described for the H5N1 subtype, as it was described during the worst of these outbreaks on 1918 (Shenderovich et al., 1979). In Colombia during the recent outbreak of influenza A H1N1, the most common clinical signs were high fever, cough, tachypnea, hypoxia, pneumonia, headache, general malaise, myalgia and rhinorrhea and few cases reported gastrointestinal compromise (Ministerio de la Protección Social, 2009).
Clinical manifestations in animals
In chikens, the extremely variable virulence of infuenza type A viruses has given place to the classification of these agents in high and low pathogenic avian influenza viruses (HPAI and LPAI) (Iakhno et al., 1978). Some of the virulence factors of HPAI have been correlated with the presence of multiple basic amino acids close to the cleavage site of the HA, that allows it to be cleaved by a ubiquitous protease(s) found in tissues throughout the body (Iakhno et al., 1978). HPAI subtypes, H5 and H7, in birds produce swelling of microvascular endothelium, multifocal hemorrhage and thrombosis. Many birds may die rapidly without showing any symptoms. Others may develop symptoms such as depression, paralysis, tremors, abnormal movements and positions of the head. In some cases nasal and ocular liquid discharge and diarrhea before they dye can be seen before they die. This systemic infection leads to death within approximately 24 hours (Sturm-Ramirez et al., 2004). LPAI viruses rarely cause symptoms in birds and are found in many species; but depending on the strain may produce decreased egg production, and another may produce only excessive lacrimation or sinusitis. (Iftimovici et al., 1979).
Swine influenza virus has an incubation period from 1 to 3 days, after which pigs show acute febrile respiratory disease characterized by respiratory distress, cough, sneezing, nasal discharge, conjunctivitis and loss of appetite.
Animals tend to reduce their activity and they stay together. Fever is not always evident, but can also be very high reaching 40.5 and 42 ºC and when forced to move animals tend to develop severe respiratory distress (L'Vov et al., 2007; Spronk, 2001). Some animals may recover between 3 to 7 days postinfection unless the disease complicates with secondary infections. Febrile pregnant females will typically abort. In felines, symptoms are regularly associated with fever and include respiratory problems, neurological signs, anorexia and subsequent death. Finally, dogs show similar respiratory problems in some cases accompanied by conjunctivitis and diarrhea that could lead to death (Chen et al., 2010; Keawcharoen et al., 2004).
Diagnosis
The diagnosis is mainly based on serological and molecular tests, applied to human and animal biological samples (Koopmans et al., 2004). Diagnosis in humans requires blood serum samples, throat or nasopharyngeal swabs and cerebrospinal fluid. However serological tests for the presence of antibodies against influenza virus are not always reliable due to the rapid appearance and evolution of symptoms, and the need to establish a rapid and effective treatment. In the case of the H5N1 virus, no specific antibodies can be detected until 14 days after the onset of symptoms, at which point the recommended treatment has no effect (Gambotto et al., 2008).
A method for a rapid detection of antigen is commonly used to diagnose human seasonal influenza, but this is not successful for the diagnosis of the H5N1 influenza virus because it has a low sensitivity and there is no test to distinguish between different subtypes of influenza (Uyeki et al., 2009; Weinberg and Walker, 2005). The hemagglutination inhibition test using equine red blood cells have shown high sensitivity for detecting subtype-specific antibodies to the HA antigen in humans (Crespo, 2000). A direct antigen search can be pursued through ELISA capture or less commonly through virus isolation (Crespo, 2000; Lange et al., 2009). More recently, the option of RT-PCR (PCR with Reverse Transcriptase) became available, and is nowadays considered one of the quickest and more specific diagnoses (Koopmans et al., 2004; Jiang et al., 2010; Gambotto et al., 2008). In birds, samples of blood, tracheal and cloacal swabs, feces and swabs of other organs are required (Swayne, 2008).
Treatment
Like most viral diseases, the course of the disease to influenza A virus depends on the host immune system since there are few options of specific drugs for treatment. Clinical symptoms appear at a time when most of the virus particles have replicated, and so, administration of drugs that block viral replication has limited effectiveness. However, some antiviral agents are useful as prophylactic agents and when administered within the first 24-48 hours of the onset of infection because they can reduce the intensity and duration of symptoms.
There are recommendations to treat the infection by the influenza A H1N1 virus with the blockers of M2 ion channel such as Amantadine and Rimantadine, and the neuraminidase inhibitors such as Oseltamivir and Zanamivir (Gambotto et al., 2008). Nevertheless, a single mutation in viruses from avian and human influenza conferred resistance to the Amantadine and Rimantadine, on some pandemics (Horimoto and Kawaoka, 2005). The neuraminidase inhibitors such as Oseltamivir, which interferes with the release of the virus from the infected cell, have proven to be effective against avian H5N1 virus in previous years, but its use is also limited when the treatment is not done within the first 24-48 hours postinfection because many patients will have developed irreversible lung damage (Hayden, 2006; Gambotto et al., 2008).
Survival rates increased when the treatment with Oseltamivir began during the first 5 days post-infection (Gambotto et al., 2008). Drug concentration is another important factor in controlling the disease, because the absorption of the drug decreases in patients with diarrhea (Gambotto et al., 2008). Unfortunately, influenza A viruses has acquired resistance to these antivirals as well (Gambotto et al., 2008). In the USA, i.e. resistance to Oseltamivir by almost all influenza A/H1 viruses has been reported between 2008 and 2009. Resistance was also observed in Asian patients treated with the same drug against the H5N1 virus. In these cases, a combination of neuraminidase inhibitor drugs and ion channel blockers would be advisable. In addition, since some viruses produce an acute inflammatory response, it has been proposed that corticosteroids treatment could lessen the damage caused by the aggressiveness of the immune response; however, no patient treated with this medicine has survived the infection (Gambotto et al., 2008).
Prevention and control
Antivirals and vaccines are the main strategy for the prevention and control of influenza A viruses. Besides their use in the treatment of infection, antivirals are also employed as prophylactics when outbreaks occur among unvaccinated individuals or even when the outbreak is due to an immunologic variant of the virus not covered by vaccines. Nevertheless, vaccines are the most effective way to control outbreaks of influenza (Gambotto et al., 2008). Currently, different approaches for developing vaccines are underway with the purpose of stimulating both, the cellular and humoral immune response (Gambotto et al., 2008). The original inactivated vaccines have been followed by the genetic engineered approaches with or without the use of vectors such as the baculovirus (Gambotto et al., 2008; Treanor et al., 1999).
Vaccines with inactivated virus are still preferred since they generate a robust antibody mediated immune response, however they also have a major disadvantage: for some viral subtypes (such as the HPAI H5N1), they are difficult to produce on a large scale with the traditional culture on embryonated eggs (Webby and Webster, 2003; Stephenson et al., 2005). As an alternative molecular approach, the influenza HA protein has also been produced in insect cells using an insect virus, the baculovirus as the recombinant vector. This vaccines either single or combined with adjuvant, generates an immune response mediated by antibodies, but so far has only proved to be effective in birds (Crawford et al., 1999; Treanor et al., 2006; Kang et al., 2003). On the other hand, viral particles used as vaccines have the advantage of generating a good local immune response at the mucosal level when administered by respiratory route. The immune response, also mediated by antibodies, can afford crossed immunity against different strains of avian influenza; however, production on large scale has been limited (Treanor et al., 2006; Kang et al., 2003). Among genetically engineered vaccines using other vectors, there are live attenuated vaccines based on adenovirus and paramyxovirus vectors (Van-Kampen et al., 2005).
Live attenuated virus vaccines generates good cellular and humoral immune responses both, systemically and at the mucosal level (immunoglobulin A), and are licensed for human use against influenza epidemics; however their administration in humans is effective only to the immune competent population (Kang et al., 2003). The adenoviral based vector vaccines have shown to induce not only humoral but also cellular immune response and generate a good protection in both animals and humans, but researchers still need to establish the possible side effects of repeated immunizations with this type of vaccines (Wesley et al., 2004).
Finally, vaccines have also been developed with reverse genetics methods, that is, by cloning of cDNA to each of the Influenza viral segments, using a system of plasmids (ranging from 4 to 12). These plasmids are then introduced in cell cultures in order to generate complete recombinant virus that can be used as vaccines (Drape et al., 2006; Neumann et al., 1999). There are some recombinant vaccines associated with lentogenic virus strains of New Castle that are, commercially available for immunization in poultry. These vaccines express the gene of the avian HA virus (from H5 or H7 strains), and they are also considered as an alternative for humans instead of the classical vaccines (Park et al., 2006; Bukreyev et al., 2005).
More recently, the development of transgenic birds resistant to the infection with influenza virus by the use of RNAi (RNA interference) has become an exciting possibility that was previously proven to be effective only against other viruses such as HIV, hepatitis B, poliovirus, among others (Chen et al., 2008). This technology consists on a double synthetic RNA of 21 nucleotides that would block transcription and replication of influenza virus. For this approach, there is already an extensive available library of viral RNA that has been screened using an ingenious Drosophila model, where RNAi that can be used to identify host genes which are indispensable for the replication of influenza virus in mammalian cells. These tools could become one of the most effective ways to control and treat this virus; however, the cost-benefit analysis may become an obstacle to its implementation (Hao et al., 2008; Hirsch, 2010). Vaccines used in the swine industry to generate immunity against influenza viruses are apparently very efficient, although their use early in life may be compromised by maternal antibodies (Vincent et al., 2008).
The new vaccine developed against the H1N1 influenza virus to control its spread was available on the market two months after the start of the 2009 pandemic and although the World Health Organization established its safety (showing only some mild side effects), its effectiveness was controversial (Seale, 2010). Among the side effects caused by the vaccine there were reports of headaches and muscle cramps. In a study conducted in Australia about the immunization against the new flu A H1N1, most people decline the vaccine for fear that it was unsafe and that the time to assess its effectiveness before being distributed was very short. Others concerns were that the vaccine could even produce the disease itself. Some people thought they had a sufficiently strong immune system capable to respond against the virus without any therapeutic intervention. Finally, some people who agreed to be immunized believed that the vaccine would protect them in any event from the seasonal influenza and that there was no real difference between the regular vaccines and this more specific one (Seale et al., 2010).
Despite the discussion about the pros and cons of vaccination, it is the ideal therapy to prevent the infection by the virus. Pending future discoveries and improvements of the available vaccines or the development of safer biological products that may provide cross protection against different influenza subtypes, the currently applied vaccines are typically directed to the most susceptible population including pregnant women in the second and third trimester of gestation, health personal at areas of risk, children and elderly, and adults with diseases such as diabetes mellitus, chronic lung disease, immunosuppressive diseases such as HIV, cancer or chronic renal failure (Seale et al., 2010).
Conclusions
In a world of rapid global mobilizations no other viral disease seems more pertinent to prevent and control than influenza A. The prospects of pandemics such as the Spanish flu at the animal, human and economic level are disproportionate when compared to any other disease and only close to the losses caused by natural disasters.
Beyond the scenes of desolation and destruction caused by weapons of mass destruction, nature has its own ways to control overpopulation, and in the middle of other threats such as climate change, poverty, energy crisis and loss of endangered species, zoonoses has claimed a great deal of attention as an increasing and important new actor to be considered.
Even though some of the previous thoughts reflect an obscure and worrisome outlook, there is still hope in science. Current technologies developed for the study of the pathogenesis of infection by the influenza virus, such as the production of infectious clones by recombinant DNA technology (genetic engineering), combination of PCR with restriction enzymes, plasmids and sequencing, represent key strategies for developing safer and more effective biological products (vaccines) for prevention of pandemics. Regardless of the plethora of new molecular methodologies, the impossibility of predicting the different viral subtypes that may cause outbreaks, demands alternative tools such as computational prediction and synthesis of new drugs or natural products that could block infection at any level (adhesion, penetration, transcription, replication or budding), or even technology based on RNA interference.
In the meantime, governments of all the nations must cooperate, be vigilant and create a global system of alert and communication, in order to respond quickly, steadily and cleverly to control de spread of air-borne diseases like influenza. Due to the epidemiological complexity of influenza virus, understanding its nature is one of the biggest challenges for basic and applied sciences of the XXI century, and a paradigm for the welfare and survival of future generations.
Acknowledgements
The authors would like to thank the grammatical and style corrections suggested by doctors David Villar and Jorge Agudelo and the financial support of the "Convocatoria de sostenibilidad 2009-2010, Universidad de Antioquia, which paid for the publication of this manuscript.
References
1.Alfonso P. Amenazas y oportunidades de reducción de riesgos por virus influenza en la interfase animal hombre. Rev Salud Anim 2010; 32:1-10. [ Links ]
2. Bergmann M, Garcia-Sastre A, Carnero E, Pehamberger H, Wolff K, Palese P and Muster T. Influenza virus NS1 protein counteracts PKR-mediated inhibition of replication. J Virol 2000; 74:6203-6206. [ Links ]
3. Braam J, Ulmanen I, Krug R. Molecular model of a eucaryotic transcription complex: functions and movements of influenza P proteins during capped RNA-primed transcription. Cell 1983; 34:611-618. [ Links ]
4. Bukreyev A, Huang Z, Yang L, Elankumaran S, St Claire M, Murphy B, Samal S, Collins P. Recombinant Newcastle disease virus expressing a foreign viral antigen is attenuated and highly immunogenic in primates. J Virol 2005; 79:13275. [ Links ]
5. Crawford J, Wilkinson B, Vosnesensky A, Smith G, Garcia M, Stone H, Perdue M. Baculovirus-derived hemagglutinin vaccines protect against lethal influenza infections by avian H5 and H7 subtypes. Vaccine 1999; 17:2265-2274. [ Links ]
6. Crespo M. El diagnóstico viral por el laboratorio. Revista Colombia Médica 2000; 31:135-150 [ Links ]
7. Chen J, Chen S, Stern P, Scott BB, Lois C. Genetic Strategy to Prevent Influenza Virus Infections in Animals. J Infect Dis 2008; 197:25-28. [ Links ]
8. Chen Y, Zhong G, Wang G, Deng G, Li Y, Shi J, Zhang Z, Guan Y, Jiang Y, Bu Z. Dogs are highly susceptible to H5N1 avian influenza virus. Virology 2010; 405:15-19. [ Links ]
9. Cheong H, Cheong C, Choi B. Secondary structure of the panhandle RNA of influenza virus A studied by NMR spectroscopy. Nucleic Acids Res 1996; 24:4197-4201. [ Links ]
10. Cheung T, Poon L. Biology of influenza A virus. Ann N Y Acad Sci 2007; 1102:1-25. [ Links ]
11. Drape R, Macklin M, Barr L, Jones S, Haynes J, Dean H. Epidermal DNA vaccine for influenza is immunogenic in humans. Vaccine 2006; 24:4475-4481. [ Links ]
12. Fodor E, Pritlove D, Brownlee G. The influenza virus panhandle is involved in the initiation of transcription. J virol 1994; 68:4092-4096. [ Links ]
13. Fodor E, Pritlove D, Brownlee G. Characterization of the RNA- fork model of virion RNA in the initiation of transcription in influenza A virus. J virol 1995; 69:4012-4018. [ Links ]
14. Franco-Giraldo A, Álvarez-Dardet C. Salud pública global: un desafío a los límites de la salud internacional a propósito de la epidemia de influenza humana A. Rev. Panam Salud Pública 2009; 25:540-547. [ Links ]
15. Gambotto A , Barratt-Boyes S, M de Jong, Neumann G, Kawaoka Y. Human infection with highly pathogenic H5N1 influenza virus. The Lancet 2008; 371:1464-1475. [ Links ]
16. Giese M, Harder T, Teifke J, Klopfleisch R, Breithaupt A, Mettenleiter T, Vahlenkamp T. Experimental infection and natural contact exposure of dogs with avian influenza virus (H5N1). Emerg infect Dis 2008; 14:308-310. [ Links ]
17. Hanssen H, Hincapié O, López J. Influenza en porcinos de Antioquia, Colombia y su repercusión en la salud humana. Estudio serológico. [Flu in swine from Antioquia, Colombia, and its repercussion in human health. Serological study]. Boletin Oficina Sanitaria Panamericana 1977; 35-43. [ Links ]
18. Hao L, Sakurai A, Watanabe T, Sorensen E, Nidom CA, Newton MA, Ahlquist P, Kawaoka Y. Drosophila RNAi screen identifies host genes important for influenza virus replication. Nature 2008; 454:890-893. [ Links ]
19. Hay A, Lomnicz i B, Bellamy A, Skehel J. Transcription of the influenza virus genome. Virology 1977; 83:337-355. [ Links ]
20. Hay A, Skehel J , McCauley J. Characterization of influenza virus RNA complete transcripts. Virology 1982; 116:517-522. [ Links ]
21. Hayden F. Antiv irals for influenza: historical perspectives, lessons learned. Antiviral Res 2006; 71:372-378. [ Links ]
22. Hirsch AJ. The use of RNAi-based screens to identify host proteins involved in viral replication. Future Microbiol 2010; 5:303-311. [ Links ]
23. Hoffmann E, Neu mann G, Hobom G, Webster R, Kawaoka Y. "Ambisense" Approach for the Generation of Influenza A Virus: vRNA and mRNA Synthesis from One Template. Virology 2000; 267:310-317. [ Links ]
24. Horimoto T, Kawaoka Y. Influenza: lessons from past pandemics, warnings from current incidents. Nat. Rev. Microbiol 2005; 3:591-600. [ Links ]
25. Horimoto T, Kawaoka Y. Strategies for developing vaccines against H5N1 influenza A viruses. Trends Mol Med 2006; 12:506-514. [ Links ]
26. Hsieh Y, Wu T, Liu D, Shao P, Chang L, Lu C, Lee C, Huang F, Huang L. Influenza pandemics: past, present and future. J Formos Med Assoc 2006; 105:1-6. [ Links ]
27. Iakhno MA, Isac henko VA, Molibog EV, Iamnikova SS, Vorkunova GK. Reversion in the natural variability of influenza A virus. Vopr Virusol 1978; 146-151. [ Links ]
28. Iakhno MA, Moli bog EV, Antonova IV, Scharfenhort H, Gericke E. Analysis of the influenza situation in the USSR and the GDR in nonepidemic (1978-1979) and epidemic (1979- 1980) seasons. Vopr Virusol 1985; 30:150-153. [ Links ]
29. Iamnikova SS, N imadava SP, Petrov NA, Vasilenko SK, Iakhno MA, Semenova NP, L'Vov DK. The characteristics of influenza A/H1N1 viruses related to A/PR/8/34 isolated in the Mongolian People's Republic. Vopr Virusol 1991; 36:188-1891. [ Links ]
30. Iftimovici R, Iacobescu V, Mihail A, Teodorescu D, Gianga I, Milici V, Tudor G, Moraru G, Bogos L, Gologan G, K. Z. Ghil, S. Vladescu. Serological investigations on the circulation of an influenza virus A(H1N1) strain in swine and horses. Virologie 1979; 30:19-23. [ Links ]
31. Iftimovici R, I acobescu v, Petrescu A, Mutiu A, Chelaru M. Isolation of influenza virus A/USSR 90/77 (H1N1) from wild birds. Virologie 1980; 31:243-247. [ Links ]
32. Jiang T, Kang X , Deng Y, Zhao H, Li X, Yu X, Yu M, Qin E, Zhu Q, Yang Q. Development of a real-time RT-PCR assay for a novel influenza A (H1N1) virus. J Virol Methods 2010; 163:470-473. [ Links ]
33. Kalthoff D, Hof fmann B, Harder T, Durban M, Beer M. Experimental Infection of Cattle with Highly Pathogenic Avian Influenza Virus (H5N1). Emerg Infect Dis 2008; 7:1132-1134. [ Links ]
34. Kalthoff D, Globig A, Beer M. (Highly pathogenic) avian influenza as a zoonotic agent. Vet Microbiol 2010; 140:237-245. [ Links ]
35. Kang S, Yao Q, Guo L, Compans R. Mucosal immunization with virus-like particles of simian immunodeficiency virus conjugated with cholera toxin subunit B. J Virol 2003; 77:9823-9828. [ Links ]
36. Keawcharoen J, Oraveerakul K, Kuiken T, Fouchier R, Amonsin A, Payungporn S, Noppornpanth S, Wattanodorn S, Theambooniers A, Tantilertcharoen R. Avian influenza H5N1 in tigers and leopards. Emerg Infect Dis 2004; 10:2189-2191. [ Links ]
37. Kistner O, Mull er K, Scholtissek C. Differential phosphorylation of the nucleoprotein of influenza A viruses. J Gen Virol 1989; 70: 2421-2426. [ Links ]
38. Klumpp K, Ruigr ok R, Baudin F. Roles of the influenza virus polymerase and nucleoprotein in forming a functional RNP structure. EMBO J 1997; 16:1248-1257. [ Links ]
39. Koopmans M, Wil brink B, Conyn M, Natrop G, Van der Nat H, Vennema H, Meijer A, Van Steenbergen J, Fouchier R, Osterhaus A. Transmission of H7N7 avian influenza A virus to human beings during a large outbreak in commercial poultry farms in the Netherlands. Lancet 2004; 363:587-593. [ Links ]
40. Kuri-Morales P, Galván F, Cravioto P, Zárraga L, Tapia-Conyer R. Mortalidad en México por influenza y neumonía (1990- 2005). Salud Pública Méx 2006; 48:379-386. [ Links ]
41. L'Vov DK, Shchelkanov M, Prilipov AG, Deriabin PG, Akanina DS, Suárez D. Molecular genetic characteristics of the strain A/ chicken/Moscow/2/2007 (H5N1) strain from a epizootic focus of highly pathogenic influenza A among agricultural birds in the neear-Moscow region. Vopr Virusol 2007; 52:40-47. [ Links ]
42. Lamb R, Krug R. Or thomyxoviridae: the viruses and their replication. Fields virol 2001; 1:1487-1531. [ Links ]
43. Lange E, Kalthoff D, Blohm U, Teifke J, Breithaupt A, Maresch C, Starick E, Fereidouni S, Hoffmann B, Mettenleiter T. Pathogenesis and transmission of the novel swine-origin influenza virus A/H1N1 after experimental infection of pigs. J Gen Virol 2009; 90:2119-2123. [ Links ]
44. Laver W, Colman P, We bster R, Hinshaw V, Air G. Influenza virus neuraminidase with hemagglutinin activity. Virology 1984; 137:314-323. [ Links ]
45. Liu C, Eichelberger M , Compans R, Air G. Influenza type A virus neuraminidase does not play a role in viral entry, replication, assembly, or budding. J Virol 1995; 69:1099-1106. [ Links ]
46. Maas R, Tacken M, Ruu ls L, Koch G, Van Rooij E, Stockhofe- Zurwieden N. Avian influenza (H5N1) susceptibility and receptors in dogs. Emerg Infect Dis 2007; 13:1219-1222. [ Links ]
47. Manrique F, Martínez A, Ospina J. La pandemia de gripe de 1918-1919 en Bogotá y Boyacá, 91 años después; La pandemia de gripe de 1918-1919. Infectio 2009; 13:182-191. [ Links ]
48. Matrosovich M, Matrosovich T, Gray T, Roberts N, Klenk H. Neuraminidase is important for the initiation of influenza virus infection in human airway epithelium. J Virol 2004; 78:12665- 12667. [ Links ]
49. Ministerio de la Protección Social. El ABC de la Influenza AH1N1, 2009. [Fecha de acceso: 25 de octubre de 2011]. URL: http://www.corpoica.org.co/sitioweb/intranet/Download/ Documentos/ABC%20A%20(H1N1).pdf [ Links ]
50. Mould J, Paterson R, Takeda M, Ohigashi Y, Venkataraman P, Lamb R, Pinto L. Influenza B virus BM2 protein has ion channel activity that conducts protons across membranes. Dev cell 2003; 5:175-184. [ Links ]
51. Neumann G, Watanabe T, Ito H, Watanabe S, Goto H, Gao P, Hughes M, Perez D, Donis R, Hoffmann E. Generation of influenza A viruses entirely from cloned cDNAs. Proc Natl Acad Sci U S A 1999; 96:9345-9351. [ Links ]
52. Newman A, Reisdorf E, Beinem ann J, Uyeki T, Balish A, Shu B, Lindstrom S, Achenbach J, Smith C, Davis J. Human case of swine influenza A (H1N1) triple reassortant virus infection, Wisconsin. Emerg Infect Dis 2008; 14:1470. [ Links ]
53. OMS. Alerta y respuesta ante Epidemias y Pandemias (EPR): 1 de mayo de 2009. Organización Muncial de la Salud 2009. [ Links ]
54. Park M, Steel J, García-Sastr e A, Swayne D, Palese P. Engineered viral vaccine constructs with dual specificity: avian influenza and Newcastle disease. PNAS2006; 103:8203-8208. http://www.pnas.org/content/103/21/8203.full.pdf+html. [ Links ]
55. Perdue M, Swayne D. Public health risk from avian influenza viruses. J Infor 2005; 49:317-327. [ Links ]
56. Pinto L, Holsinger L and Lamb R. Influenza virus M2 protein has ion channel activity. Cell 1992; 69:517-528. [ Links ]
57. Potter C. Chronicle of influen za pandemics. In textbook of influenza. Ed Nicholson 1998; 3-18. [ Links ]
58. Potter C. A history of influen za. J Appl microbiol 2001; 91:572- 579. [ Links ]
59. Qiu Y, Krug R. The influenz a virus NS1 protein is a poly (A)-binding protein that inhibits nuclear export of mRNAs containing poly (A). J Virol 1994; 68:2425. [ Links ]
60. Ramírez A, Mendoza A, Montoya J, Cótes K, López J, Herrera D, Rey G, De la Hoz F. Mortalidad asociada con las temporadas de mayor circulación de los virus de la influenza en Bogotá, Colombia, 1997-2005. Rev Panam Salud Pública 2009; 26:435- 436. [ Links ]
61. Rodríguez A, Izquierdo M, Moros M, H eras C. Medidas de vigilancia y contención de la influenza aviar en aves. Implicaciones para la salud pública Surveillance and Control Measures of avian influenza in birds. Implications for Public Health. Rev Esp Salud Publica 2006, 80:621-630. [ Links ]
62. Schnell J, Chou J. Structure and me chanism of the M2 proton channel of influenza A virus. Nature 2008; 451:591-595. [ Links ]
63. Seale H, Heywood A, McLaws M, Ward K , Lowbridge C, Van D, MacIntyre C. Why do I need it? I am not at risk! Public perceptions towards the pandemic(H1N1) 2009 vaccine. BMC Infect Dis 2010; 10:91-98. [ Links ]
64. Shapiro G, Gurney T, Krug R. Influen za virus gene expression: control mechanisms at early and late times of infection and nuclear-cytoplasmic transport of virus-specific RNAs. J Virol 1987; 61:764. [ Links ]
65. Shenderovich S, Molibog EV, Iakhno MA, Zakstel'skaia L, Zhdanov VM. Antigenic and biological characteristics of the A (H1N1) strains that caused the 1977-1978 epidemic. Vopr Virusol 1979; 480-486. [ Links ]
66. Shinya K, Ebina M, Yamada S, Ono M, Kas ai N, Kawaoka Y. Avian flu: influenza virus receptors in the human airway. Nature 2006; 440:435-436. [ Links ]
67. Spronk G. Swine influenza virus. Adv Po rk Prod 2001; 12:51- 54. [ Links ]
68. Steidle S, Martinez-Sobrido L, Mordste in M, Lienenklaus S, Garcia-Sastre A, Staheli P, Kochs G. Glycine 184 in the non- structural protein NS1 determines virulence of influenza A virus strain http://jvi.asm.org/content/early/2010/10/06/JVI.00701-10.full.pdf+html [ Links ]
69. Stephenson I, Bugarini R, Nicholson K, Podda A, Wood J, Zambon M, Katz J. Cross-reactivity to highly pathogenic avian influenza H5N1 viruses after vaccination with nonadjuvanted and MF59-adjuvanted influenza A/Duck/Singapore/97 (H5N3) vaccine: a potential priming strategy. Vaccine 2005; 191:1210- 1215. [ Links ]
70. Stieneke-Gröber A, Vey M, Angliker H, Shaw E, Thomas G, Roberts C, Klenk H, Garten W. Influenza virus hemagglutinin with multibasic cleavage site is activated by furin, a subtilisin- like endoprotease. EMBO J 1992; 11: 2407-2412. [ Links ]
71. Sturm-Ramírez K, Ellis T, Bousfield B, Bissett L, Dyrting K, Rehg J, Poon L, Guan Y, Peiris M, Webster R. Reemerging H5N1 influenza viruses in Hong Kong in 2002 are highly pathogenic to ducks. J Virol 2004; 78: 4892-4901. [ Links ]
72. Swayne D. Avian influenza. Isolation and Identification of Avian Path ogens 2008; 128-134. [ Links ]
73. Thiry E, Zicola A, Addie D, Egberink H, Hartmann K, Lutz H, Poule t H, Horzinek M. Highly pathogenic avian influenza H5N1 virus in cats and other carnivores. Vet Microbiol 2007; 122:25-31. [ Links ]
74. Treanor J, Kotloff K, Betts R, Belshe R, Newman F, Iacuzio D, Wittes J, Bryant M. Evaluation of trivalent, live, cold- adapted (CAIV-T) and inactivated (TIV) influenza vaccines in prevention of virus infection and illness following challenge of adults with wild-type influenza A (H1N1), A (H3N2), and B viruses. Vaccine 1999; 18:899-906. [ Links ]
75. Treanor J, Schiff G, Couch R, Cate T, Brady R, Hay C, Wolff M, She D, Cox M. Dose-related safety and immunogenicity of a trivalent baculovirus-expressed influenza-virus hemagglutinin vaccine in elderly adults. The J Infec Dis 2006; 93:1223-1228. [ Links ]
76. Ungchusak K, Auewarakul P, Dowell S, Kitphati R, Auwanit W, Puthava thana P, Uiprasertkul M, Boonnak K, Pittayawonganon C and Cox N. Probable person-to-person transmission of avian influenza A (H5N1). New Engl J Med 2005, 352:1427-1434. [ Links ]
77. Uyeki T, Prasad R, Vukotich C, Stebbins S, Rinaldo C, Ferng Y, Mors e S, Larson E, Aiello A, Davis B. Low sensitivity of rapid diagnostic test for influenza. Clin Infect Dis 2009; 48:89-92. [ Links ]
78. Valderrama C. Transmisión de enfermedades en los vuelos comerciales: del mito a la realidad. Infectio 2009; 13:203-216. [ Links ]
79. Van-Kampen K, Shi Z, Gao P, ZhanG J, Foster K, Chen D, Marks D, Elm ets C, Tang D. Safety and immunogenicity of adenovirus-vectored nasal and epicutaneous influenza vaccines in humans. Vaccine 2005; 23:1029-1036. [ Links ]
80. Van-Reeth K. Avian and swine influenza viruses: our current understand ing of the zoonotic risk. Vet Res 2007; 38:243-260. [ Links ]
81. Vincent A, Ma W, Lager K, Janke B, Richt J. Swine Influenza Viruse s: A North American Perspective. Adv Virus Res 2008; 72, 127-154. [ Links ]
82. Vincent A, Ma W, Lager KM, Gramer MR, Richt JA, Janke BH. Characterization of a newly emerged genetic cluster of H1N1 and H1N2 swine influenza virus in the United States. Virus Genes 2009; 39:176-185. [ Links ]
83. Wang C, Lamb R and Pinto L. Direct measurement of the influenza A viru s M2 protein ion channel activity in mammalian cells. Virology 1994; 205:133-140. [ Links ]
84. Webby R, Webster R. ¿Are we ready for pandemic influenza? Science 2003; 302:1519-1524. [ Links ]
85. Webster R, Bean W, Gorman O, Chambers T, Kawaoka Y. Evolution and e cology of influenza A viruses. Microbiol Mol Biol Rev 1992; 56:152-159. [ Links ]
86. Weinberg A, Walker M. Evaluation of three immunoassay kits for rap id detection of influenza virus A and B. Clin Vaccine Immunol 2005; 12:367-370. [ Links ]
87. Wesley R, Tang M, Lager K. Protection of weaned pigs by vaccination with human adenovirus 5 recombinant viruses expressing the hemagglutinin and the nucleoprotein of H3N2 swine influenza virus. Vaccine 2004; 22:3427-3434. [ Links ]
88. WHO. Casos confirmados del nuevo virus influenza A H1N1 a nivel mundia l al 11 de julio de 2010. 2010. World Organization for Animal Health - OIE. Outbreaks of avian influenza in anmals throughout the world until 2011. [Fecha de acceso: Abril 22 de 2011]. URL: http://www.oie.int/wahis/public.php?page=disease_outbreak_map&disease_ type=Terrestrial&disease_id=15&empty=999999&newlang=3 [ Links ]
89. Winter G, Fields S. The structure of the gene encoding the nucleop rotein of human influenza virus A/PR/8/34. Virology 1981; 114:423-428. [ Links ]
90. Wright P, Webster R. Orthomyxoviruses. Fields virol 2001; 1:1533-1579. [ Links ]
91. Yu H, Zhou Y, Li G, Zhang G, Liu H, Yan L, Liao M and Tong G. Further evidence for infection of pigs with human-like H1N1 influenza viruses in China. Virus res 2009; 140:85-90. [ Links ]
92. Zhou N, Senne D, Landgraf J, Swenson S, Erickson G, Rossow K, Liu L, Yoon K, Krauss S, Webster R. Genetic reassortment of avian, swine, and human influenza A viruses in American pigs. J Virol 1999; 73:8851-8856. PR8 without affecting the host interferon response. J Virol 2010. [ Links ]