Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
Revista Colombiana de Ciencias Pecuarias
Print version ISSN 0120-0690On-line version ISSN 2256-2958
Rev Colom Cienc Pecua vol.25 no.3 Medellín July/Sept. 2012
ARTÍCULOS ORIGINALES
Effects of group size on agonistic behaviors of commercially housed growing pigs¤
Efecto del tamaño del grupo sobre la conducta agonística de cerdos en crecimiento alojados en instalaciones comerciales
Efeitos do tamanho do grupo sobre a conduta agonística de suínos em crescimento alojados em instalações comerciais
Shin-Jae Rhim1*, PhD.
* Corresponding author: Shin-Jae Rhim, School of Bioresource and Bioscience, Chung-Ang University, Ansung 456-756, Korea, Tel: +82-31-670-4842, Fax: +82-31-676-4842, E-mail: sjrhim@cau.ac.kr.
1School of Bioresource and Bioscience, Chung-Ang University, Ansung 446-756, Korea.
(Recibido: 29 marzo, 2012; aceptado: 30 abril, 2012)
Summary
Objective: this study was conducted to understand the effects of group size on the agonistic behaviors of pigs reared at differing pen densities. Methods: thirty groups of pigs (a total of 350 individuals) housed at low, medium, and high group density (5, 10, or 20 individuals in 6.0 m x 6.0 m pens) were consecutively observed for 10 h on days 30, 90, and 180 with the aid of video technology. Results: the frequency of vocalizations was lower at low group density and higher at high group density on all investigated days. Pigs housed at high group density showed significantly more agonistic behaviors than those at low group density. Conclusions: the study reveals a higher level of aggression in older pigs and at high group density. It is concluded that group density is a major cause of the observed agonistic behaviors.
Key words: aggression, housing, pen density, swine, vocalization.
Resumen
Objetivo: este estudio busca comprender los efectos del tamaño del grupo sobre los comportamientos agonísticos de cerdos criados en corrales bajo diferentes densidades de animales por unidad de espacio. Métodos: treinta grupos de cerdos (un total de 350 animales) alojados en baja, media y alta densidad de animales (5, 10 ó 20 animales en corrales de 6.0 x 6.0 m) fueron observados durante 10 horas consecutivas en los días 30, 90, y 180 con la ayuda de tecnología de vídeo. Resultados: la frecuencia de vocalizaciones fue menor en el grupo de baja densidad de animales, y mayor en el grupo de alta densidad durante todos los días estudiados. Los cerdos alojados en grupos con alta densidad animal mostraron un comportamiento significativamente más agonístico que aquellos en baja densidad. Conclusiones: este estudio revela que existe un mayor nivel de agresión en los cerdos más viejos y en grupos con mayor densidad de animales. Se concluye que la densidad de alojamiento es una causa importante de la conducta agonística observada.
Palabras clave: agresividad, alojamiento, densidad en corral, porcino, vocalización.
Resumo
Objetivo: este estudo busca compreender os efeitos do tamanho do grupo acerca dos comportamentos agonísticos dos suínos criados em corrais sobre diferentes densidades de animais por unidade de espaço. Métodos: trinta grupos de suínos (um total de 350 animais) alojados em baixa, meia e alta densidade de animais (5, 10 ou 20 animais em corrais de 6.0 x 6.0 m) foram observados durante 10 horas consecutivas nos dias 30, 90 e 180 com a ajuda de tecnologia de vídeo. Resultados: a frequência de vocalizações foi menor no grupo de baixa densidade de animais, e maior no grupo de alta densidade durante todos os dias estudados. Os suínos alojados em grupos com alta densidade animal mostraram um comportamento significativamente mais agonístico que aqueles em baixa densidade. Conclusões: este estudo revela que existe um maior nível de agressão nos suínos mais velhos e em grupos com maior densidade de animais. Concluiu-se que a densidade de alojamento é uma causa importante da conduta agonística observada.
Palavras chave: agressividade, alojamento, comportamento agonístico, densidade, suíno.
Introduction
Mixing pigs is a common practice in modern swine husbandry. With the development of group housing systems for pigs, the benefits of group housing have been addressed in previous research (Weng et al., 2009; Baxter et al., 2010). However, the effects of group housing on behavior and welfare of pigs have not been well documented (Hillmann et al., 2003). The post-weaning regrouping of pigs is an especially highly stressful event and is one of the major welfare concerns of the pig industry (Arey, 1999). When mixing, nearly all unfamiliar individuals are involved in agonistic interactions (Frazer and Broom, 1997). The number of aggressive interactions following grouping correlates with the amount of lesions found on pigs (Barnett et al., 1992) and can provide a useful measure of welfare (Barnett et al., 1996). Aggression among pigs can be perpetuated and the welfare of subordinate group members may then suffer as a result of competition for food and space (Petheric and Blackshaw, 1987).
Anil et al. (2006) showed that the process of mixing unacquainted sows is associated with an increased stress level, which leads to a rise in salivary cortisol concentration. Lower ranking animals may be especially affected by impaired welfare due to social stress, which influences physiological and reproductive parameters (Hoy et al., 2009; Strawford, 2006). Under natural conditions unfamiliar pigs are hardly ever integrated into an established group (Gonyou, 2001).
Aggressive behavior can be observed in various behavioral contexts and occurs, for example, while competing for limited resources (Csermely and Wood-Gush, 1987) or in order to secure the social position of an individual animal and to establish a social structure within a group of individuals (Hagelso Giersing and Studnitz, 1996). Agonistic behaviors, such as opposing aggression and defensiveness, are part of the normal behavior patterns of pigs. As agonistic interactions inevitably occur when unfamiliar sows are grouped (Mendl and Deag, 1995; D'Eath and Turner, 2009), great challenges arise from keeping sows in groups (Gonyou, 2003). Severe manifestations of social stress can lead to delayed estrous, aborted fetuses, increased farrowing time, and failure to lactate (Varley and Stedman, 1993).
There is little evidence of an optimum group size at which fighting and aggression between pigs is minimized. Similarly, there is little evidence of an optimum space allowance to reduce fighting, although the provision of more space can decrease the levels of aggression over the long term. More work is needed to determine whether pigs adopt different agonistic strategies according to group size and composition. This study was conducted in order to assess the effects of group size (confined or loose housing) on agonistic behavior of pigs that are mixed during the growing period.
Methods
The experiment was conducted at a commercial swine farm located in Ansung (Gyeonggi Province, Korea) using a total of 350 pigs (Yorkshire x Landrace). Pigs were farrowed in 6.0 m x 6.0 m pens with solid concrete flooring and a heat lamp. Piglets were weaned at 20 (± 1.2) days of age and mixed at different densities: five (low density), ten (medium density), or twenty (high density) individuals in a 6.0 m x 6.0 m pen. The environmental control systems were the same among all housing facilities. The temperature in each room was controlled by ventilation fans and heaters and was maintained at approximately 20 ± 2 ºC. Pigs had ad libitum access to feed and water.
Ten replicates of each treatment were evaluated during 2006 and 2007. Two wide angle video cameras were installed at opposing corners at the ceiling of the stable so that the pen could be observed from two directions. The behavior of the pigs in the group was video-recorded continuously for 10 h per day during three non-consecutive days. Behaviors were analyzed from images digitally recorded from 08:00 to 18:00 h on days 30, 90, and 180 (day 0 = birth day). Video tapes were analyzed using a video recorder with jog-shuttle function. Instantaneous scan sampling was carried out at 2-min intervals. All video recordings were viewed by a trained observer who was blinded to the treatments to eliminate subjective bias and interindividual discrepancy (Li and Wang, 2011).
Vocalizations were registered by direct observation from the videos. Occurrences of the following behaviors were recorded: inactivity, chewing on other animals, locomotion, pen exploration, drinking, feeding, excretion, tail biting, belly nosing, agonistic behavior, fighting, and other social interactions and behaviors (Table 1). The frequency and duration (in seconds) of these behaviors was recorded from the videos on days 30, 90, and 180, together with which individual pigs were performing and receiving the behavior.
All agonistic interactions were registered by recording the time of occurrence, the pigs involved, and the dominant and defeated animal resulting from the interaction. In this context, agonistic behavior was defined as follows: aggressive behavior between two pigs involving physical contact (biting, knocking, or lateral fighting with the opponents standing in antiparallel position, both performing bites or knocks) starting with the first physical contact and ending with submissive behavior (escape) shown by one of the opponents, or when both pigs moved away from each other (Borberg and Hoy, 2009; Krauss and Hoy, 2011).
In particular, fighting behavior within agonistic interactions was monitored. Fighting was considered to have begun when a pig bit another pig and the fight lasted for more than 1 s, and to have terminated when the pigs were separated for 5 s after a fight (Samarakone and Gonyou, 2009). The number of fights, duration of fights, and latency to the first fight was registered. Behavioral time values presented are the means and standard errors of the relative frequencies of each behavior, by calculating results obtained from each observation of each group.
All data were analyzed by using the Glimmix procedure of the SAS software (SAS Inst. Cary, NY) with the pen as the experimental unit. The residual data sets were tested for normal distribution using the Univariate Procedure of SAS. The data were not distributed normally and were transformed using the logarithm (X' = log 10(X + 0.5) + 0.5) to achieve normal distribution (Zar, 1999). The data were analyzed by ANOVA. Tukey's post hoc tests were used to determine pair-wise differences between treatments. P values are presented.
Results
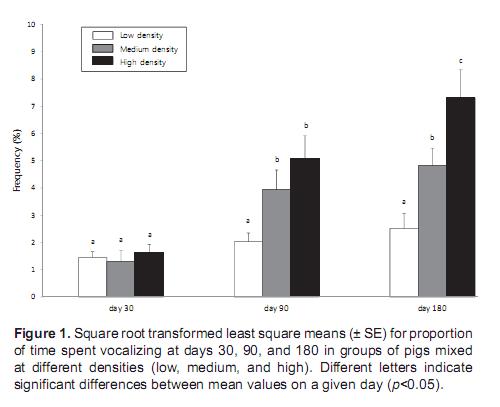
The frequency of vocalizations was significantly affected by density (ANOVA, F2,18 = 21.3, p<0.001) and time (F2,18 = 9.7, p<0.05). At medium and high density, the frequency of vocalizations was highest on day 180 and lowest on day 30. The frequency of vocalization on day 180 was higher at the high group density than at low and medium densities (Tukey's test, p<0.01) (Figure 1).
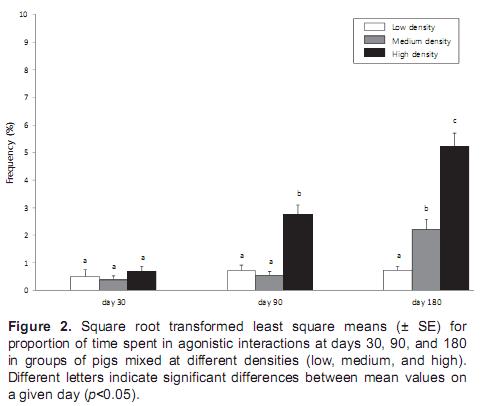
The agonistic interactions among pigs were also significantly affected by density (F2,18 = 6.4, p<0.05) and time (F2,18 = 17.5, p<0.01). On day 30, the time spent in agonistic interactions was not significantly different among the density groups (F2,18 = -2.5, p=0.21). However, on day 90 (F2,18 = 5.6, p<0.05) and day 180 (F2,18 = 19.2, p<0.01) agonistic interactions were significantly different among the density groups. Agonistic interaction was greater on day 180 than on days 30 and 90 (p<0.05) (Figure 2).
On day 30, fight latency (F2,18 = -2.6, p=0.12), total duration of fighting (F2,18 = -4.3, p=0.34), and fight frequency (F2,18 = -9.4, p=0.31) were not significantly different among the density groups. At day 90, fight latency (F2,18 = 6.9, p<0.05) and total duration of fighting (F2,18 = 3.8, p<0.05) were significantly higher in the high-density group compared with groups in the low and medium densities (p<0.05). Fight frequency was not significantly different among the density groups on day 90 (F2,18 = -3.7, p=0.37). All variables (fight latency, F2,18 = 6.5, p<0.05; total duration of fighting, F2,18 = 18.2, p<0.01; fight frequency, F2,18 = 12.7, p<0.01) were significantly different among the density groups on day 180 with higher values in the high-density group (p<0.05) (Table 2).
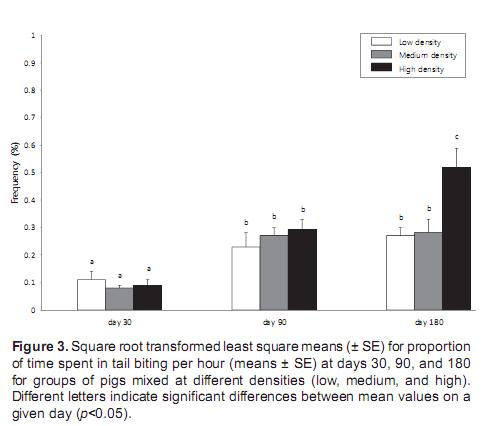
Frequency of tail biting did not differ among the density groups on day 30 (F2,18 = 0.2, p=0.26) and day 90 (F2,18 = 1.3, p=0.14), but was significantly higher on day 90 compared with day 30 (p<0.05). On day 180, tail biting frequency was significantly different among the density groups (F2,18 = 7.2, p<0.05), with a significantly higher frequency of tail biting among pigs in the high-density group than in those at low and medium densities (p<0.05). There was no difference in tail biting frequency between the low and medium densities (Figure 3). Both density (ANOVA, F2,18 = 5.3, p<0.05) and day (F2,18 = 4.7, p<0.05) had a significant effect on tail biting, but there was no interaction between these factors (F2,18 = 0.5, p=0.08).
Discussion
The main objective of this study was to conduct a detailed investigation of the effect of group size on the agonistic behavior of pigs at different ages. Of particular interest was the impact that high density had on vocalization, agonistic interactions, fighting, and tail biting. The results of this study indicate that agonistic behavior increases as pigs grow. In particular, the older pigs (day 180) in the highdensity group were more aggressive than those at low density.
Generally, fighting occurs between pigs that are unfamiliar with each other (Arey and Edwards, 1998). Fighting establishes relative social ranking (Meese and Ewbank, 1973) thereby reducing the need for outright aggression to settle future disputes between animals; however, there is a higher level of agonistic behavior among pigs in the groups of medium and high densities. Spacing between individual animals in a test pen with the high density group was previously shown to be associated with agonistic behavior (Rushen, 1988; Moorse et al., 1994; Jensen et al., 1996). Thus, aggressive interactions may continue between familiar animals, but they do so at much lower levels unless resources such as food (Baxter, 1989) or space (Arey et al., 1992) become limited.
Fighting for resources among pigs usually lasts longer than fighting for dominance hierarchy (Langbein and Puppe, 2004). In the present study, fighting among pigs in the high-density group was Fighting for resources among pigs usually lasts longer than fighting for dominance hierarchy (Langbein and Puppe, 2004). In the present study, fighting among pigs in the high-density group wasmore vigorous on day 180, with longer duration and higher fight frequency. On day 30, few fights among pigs were observed for all three densities because pen size was large enough for the piglets. However, by day 180 the older pigs were suffering from space stress and the number and frequency of agonistic interactions were higher at high group density compared with low group density.
Vocalization may be an indicator of status for pigs (Rhim et al., 2008). A large number of calls occurred in the groups that were housed at high density. Moreover, vocalization increased with the age of the pigs. The frequency of vocalization was the highest in the high-density group on day 180.
In conventional swine production, pigs are usually mixed at weaning and during the early growing stage to efficiently utilize housing facilities. Mixing of unacquainted pigs induces aggression and, consequently, can result in injuries (O'Connell et al., 2005). Mixing can also cause a setback in growth rate (Li and Johnston, 2009). Since fights among older pigs are more intensive and cause more injuries than fights among younger pigs (Weary et al., 2002), animals in commercial growing systems may modify their behavioral response to mixing during later stages of production.
It was observed in the present study that limiting space tends to increase aggression among growing pigs. In addition, aggression levels are greater at higher stocking rates. The aggression that occurs during mixing results in stress responses, and such responses could have detrimental effects on productive parameters.
References
1. Anil L, Anil SS, Baidoo SK, Waler RD. Effect of group size and structure on the welfare and performance of pregnant sows in pens with electronic sow feeders. Can J Vet Res 2006; 70:128-136. [ Links ]
2. Arey DS. Time course for the formation and disruption of social organization in group-housed sows. Appl Anim Behav Sci 1999; 62:199-207. [ Links ]
3. Arey DS, Edwards SA. Factors influencing aggression between sows after mixing and the consequences for welfare and production. Livest Prod Sci 1998; 56:61-70. [ Links ]
4. Arey DS, Petchey AM, Fowler VR. The peri-parturient behaviour of sows housed in pairs. Appl Anim Behav Sci 1992; 34:49-59. [ Links ]
5. Barnett JL, Cronin GM, McCalum TH, Newman EA, Hennessy DP. Effects of grouping unfamiliar adult pigs after dark, after treatment with amperozide and by using pens with stalls, on aggression, skin lesions and plasma cortisol concentration. Appl Anim Behav Sci 1996; 50:121-133. [ Links ]
6. Barnett JL, Hemsworth PH, Cronin GM, Newman EA, McCallum TH, Chilton D. Effects of pen size, partial stalls and method of feeding on welfare-related behavioural and physiological responses of group-housed pigs. Appl Anim Behav Sci 1992; 34:207-220. [ Links ]
7. Baxter EM, Lawrence AB, Edwards SA. Alternative farrowing systems: design criteria for farrowing systems based on the biological needs of sows and piglets. Animal 2010; 5:580-600. [ Links ]
8. Baxter MR. Design of a new feeder for pigs. Farm Build Progr 1989; 96:19-22. [ Links ]
9. Borberg C, Hoy S. Mixing of sows with or without the presence of a boar. Livest Sci 2009; 125:314-317. [ Links ]
10. Csermely D, Wood-Gush DGM. Aggressive behavior of grouped sows in different contexts. Appl Anim Behav Sci 1987; 17:368-369. [ Links ]
11. D'Eath RB, Turner SP. The natural behavior of the pig. In: Marchant-Forde JN (Ed). The welfare of pigs. New York: Springer; 2009. p 13-45. [ Links ]
12. Frazer AF, Broom DM. Farm animal behaviour and welfare. Oxon: CABI Publishing; 1997. [ Links ]
13. Gonyou HW. The social behavior of pigs. In: Keeling LJ, Gonyou HW (Ed). Social behavior in farm animals. Oxon: CABI Publishing; 2001. p 147-176. [ Links ]
14. Gonyou HW. Group housing: alternative systems, alternative management. Adv Pork Prod 2003; 14:10-107. [ Links ]
15. Hagelso Giersing M, Studnitz M. Characterization and investigation of aggressive behaviour in the pig. Acta Agric Scand A: Anim Sci Suppl 1996; 27:56-60. [ Links ]
16. Hillmann E, von Hollen F, Bunger B, Todt D, Schrader L. Farrowing conditions affect the reactions of piglets towards novel environment and social confrontation at weaning. Appl Anim Behav Sci 2003; 81:99-109. [ Links ]
17. Hoy S, Bauer J, Borberg C, Chonsch L, Weirich C. Impact of rank position on fertility of sows. Livest Sci 2009; 126:69-72. [ Links ]
18. Hötzel MJ, Machado Filho LCP, Wolf FM, Costa OAD. 2004. Behaviour of sows and piglets reared in intensive outdoor or indoor systems. Appl Anim Behav Sci 2004; 86:27-39. [ Links ]
19. Jensen P, Forkman B, Yngvesson J, Furuhaug IL. Assessment in pig conflicts. In: Duncan IJH, Widowski TM, Waley DB (Ed). The 30th International Congress of the International Society for Applied Ethology. Guelph: The Colonel K.L. Campbell Centre for Study of Animal Welfare; 1996. [ Links ]
20. Krauss V, Hoy S. Dry sows in dynamic groups: an investigation of social behavior when introducing new sows. Appl Anim Behav Sci 2011; 130:20-27. [ Links ]
21. Langbein J, Puppe B. Analysing dominance relationships by sociometric methods – a plea for a more standardized and precise approach in farm animals. Appl Anim Behav Sci 2004; 87:293-315. [ Links ]
22. Li Y, Wang L. Effects of previous housing system on agonistic behaviors of growing pigs at mixing. Appl Anim Behav Sci 2011; 132:20-26. [ Links ]
23. Li YZ, Johnston LJ. Behavior and performance of pigs previously housed in large groups. J Anim Sci 2009; 87:1472-1478. [ Links ]
24. Meese GB, Ewbank R. The establishment and nature of the dominance hierarchy in the domesticated pig. Anim Behav 1973; 21:326-334. [ Links ]
25. Mendl M, Deag JM. How useful are the concepts of alternative strategy and coping strategy in applied studies of social behavior? Appl Anim Behav Sci 1995; 44:1107-1121. [ Links ]
26. Moore AS, Gonyou HW, Stookey JM, McLaren DG. Effect of group composition and pen size on behaviour, productivity and immune response of growing pigs. Appl Anim Behav Sci 1994; 40:13-30. [ Links ]
27. O'Connell NE, Beattie VE, Watt D. Influence of regrouping strategy on performance, behaviour and carcass parameters in pigs. Livest Prod Sci 2005; 97:107-115. [ Links ]
28. Petheric JC, Blackshaw JK. A review of the factors influencing the aggressive and agonistic behaviour of the domestic pig. Aust J Exp Agric 1987; 27:605-611. [ Links ]
29. Rhim SJ, Kim MJ, Lee JY, Kim NR, Kang JH. Characteristics of estrus-related vocalizations of sows after artificial insemination. J Anim Sci & Technol 2008; 50:401-406. [ Links ]
30. Rushen J. Assessment of fighting ability or simple habituation: what cause young pigs (Sus scrofa) to stop fighting. Aggress Behav 1988; 14:155-167. [ Links ]
31. Samarakone TS, Gonyou HY. Domestic pigs alter their social strategy in response to social group size. Appl Anim Behav Sci 2009; 121:8-15. [ Links ]
32. Statham P, Green L, Bichard M, Mendl M. A longitudinal study of the effects of providing straw at different stages of life on tail-biting and other behaviour in commercially housed pigs. Appl Anim Behav Sci 2011; 134:100-108. [ Links ]
33. Strawford ML. Social factors that affect the behavior and productivity of gestating sows in an electronic sow feeding system. Thesis of MSc, University of Saskatchewan, Saskatoon, 2006. [ Links ]
34. Varley M, Stedman R. Stress and reproduction. In: Cole DJA, Wiseman J, Varley MA (Ed). Principles of pig science. Nottingham: Nottingham University Press; 1993. p 277-296. [ Links ]
35. Weary DM, Pajor EA, Bonenfant M, Fraser D, Kramer DL. Alternative housing for sows and litters. Part 4: effects of sowcontrolled housing combined with a communal piglet area on pre- and post- weaning behaviour and performance. Appl Anim Behav Sci 2002; 76:279-290. [ Links ]
36. Weng RC, Edwards SA, Hsia LC. Effect of individual, group or ESF housing in pregnancy and individual or group housing in lactation on sow behavior. Asian-Aust J Anim Sci 2009; 22:1574-1580. [ Links ]
37. Zar JH. Biostatistical analysis, 4th ed. Upper Saddler River: Prentice-Hall Inc.; 1999. [ Links ]
Notas
¤ To cite this article: Shin-Jae R. Effects of group size on agonistic behaviors of commercially housed growing pigs. Rev Colomb Cienc Pecu 2012; 25:353-359.