Introduction
Contamination of feed ingredients with Fusarium mycotoxins causes significant health problems in pigs and, consequently, economic losses for producers (Chaytor et al., 2011a). Fusarium mycotoxins such as deoxynivalenol (DON) are frequently found in various feedstuffs and can reduce feed intake and growth, and cause immune suppression (CAST, 2003; Avantaggiato et al., 2005; Jiang et al., 2010).
Although barley has lower energy content than corn, it is still a good grain source for swine diets because it contains greater concentrations of crude protein (CP), lysine, and available phosphorus compared with corn (NRC, 2012; Woyengo et al., 2014). However, a global survey indicated that the positive occurrence of DON in barley is 42% with average concentration being 1,677 mg/Kg, which is greater than the mycotoxin concentration guidance for swine diets, at 0.9 mg/Kg (CEC, 2006; Rodrigues and Naehrer, 2012).
The formation of DON in feedstuffs occurs at the field, prior to harvest. Under certain environmental conditions it is very difficult to completely avoid its development due to the impact of weather conditions. Thus, mycotoxin adsorbents such as yeast cell wall products (YCW) could be used to prevent the adverse effects of contaminated diets. However, their effects are still equivocal (Papaioannou et al., 2002; Weaver et al., 2013).
Therefore, the objective of the present study was to evaluate YCW as a mycotoxin adsorbent toprevent the adverse effects of Fusarium mycotoxin on growth performance, blood characteristic, and digestibility of dry matter (DM), organic matter (OM), CP, and energy for gilts fed diets containing barley naturally contaminated with Fusarium mycotoxins.
Materials and methods
All the experimental procedures used in the current study were approved by the Institutional Animal Care and Use Committee at Konkuk University.
Animals and experimental design
A total of 16 crossbred gilts averaging 61.7 Kg initial body weight (BW) (standard deviation = 5.6) were used in a 14-d study. The pigs were individually housed in metabolism crates (0.48 × 1.49 m2) in an environmentally controlled room and were grouped by BW into four blocks and randomly assigned to four treatments in a randomized complete block design.
Ingredients, diets, feeding, and sample collection
Corn-soybean meal (SBM)-based positive control (PC) and negative control (NC) diets contained 20% of control barley and barley naturally contaminated with Fusarium mycotoxins, respectively. Two additional diets were prepared by supplementing 0.2 or 0.4% YCW to the NC diet (NC1 or NC2) at the expense of corn starch (Table 1). The YCW product used in the present study was an esterified glucomannan polymer extracted from the cell wall of yeast. Average daily gain (ADG), average daily feed intake (ADFI), and gain:feed (G:F) were determined based on individual BW and feed consumption measured on d 0, 7, and 14. From d 10 to 14, fecal samples were collected quantitatively using the marker to marker method (Kong and Adeola, 2014).
On d 14, blood samples were collected via the jugular vein from all the pigs using a 21-gauge needle. A 10 mL evacuated tube spray coated with K2EDTA (BD Vacutainer® Plus Blood Collection Tubes, Becton, Dickinson and Company, Franklin Lakes, NJ, USA) was used for collecting blood samples. Blood samples for serum analysis were subsequently collected using a 5 mL evacuated tube coated with microscopic silica particles (VACUETTE® Z Serum Sep Clot Activator tube, Greiner Bio-one, Kremsmünster, Upper Austria, Austria). Collected blood samples were immediately delivered on ice to the laboratory and stored at 4 °C until centrifugation (3,500 rpm for 15 min at 4 °C). After centrifugation, a 2 mL aliquot was collected from each serum and plasma sample and stored at -20 °C for further analyses.
Table 1 Composition of experimental diets containing barley naturally contaminated with Fusarium mycotoxins (as-fed basis).
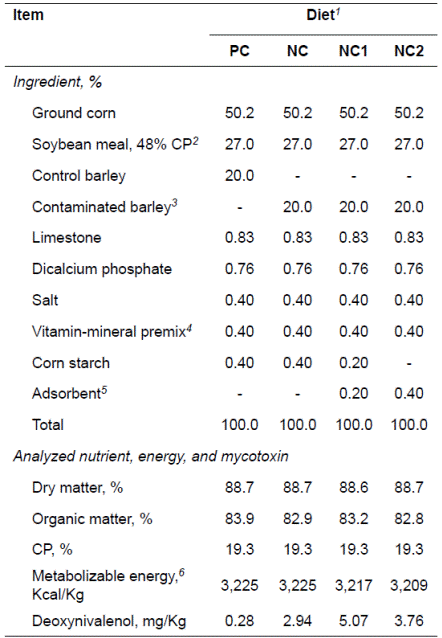
1. PC = corn-soybean meal based diet containing 20% of control barley; NC
= corn-soybean meal based diet containing 20% of contaminated barley;
NC1 = NC diet supplemented with 0.2% mycotoxin adsorbent; NC2 = NC
diet supplemented with 0.4% mycotoxin adsorbent.
2. CP = crude protein.
3. Naturally contaminated with Fusarium mycotoxins containing 25.7 mg/Kg
deoxynivalenol.
4. Provided the following quantities per Kg of complete diet: vitamin A, 20,000
IU; vitamin D3, 3,200 IU; vitamin E, 40 IU; vitamin K, 4.0 mg; thiamin, 3.9 mg;
riboflavin, 8.0 mg; pyridoxine, 3.9 mg; vitamin B12, 0.05 mg; pantothenic acid,
30.0 mg; folic acid, 0.88 mg; niacin, 50 mg; biotin, 0.05 mg; Cu, 20 mg as copper
sulfate; Fe, 214 mg as iron sulfate; I, 4.0 mg as potassium iodate; Mn, 100 mg
as manganese sulfate; Se, 0.30 mg as sodium selenite; Zn, 251 mg as zinc
oxide; butylated hydroxytoluene, 40 mg.
5. Yeast cell wall product.
6. Calculated values based on NRC (2012).
Mycotoxins and chemical analyses
Enzyme-linked immunosorbent assay kits (AgraQuant®, Romer Labs Inc., Singapore, Republic of Singapore) were used to determine the concentration of DON in the ingredients and diets. Quantification ranges for analysis on DON were from 250 to 5,000 ng/mL, respectively.
All fecal samples and orts collected from individual pigs were dried in a forced-air oven at 55 °C to constant weight. Dry matter (Ahn et al., 2014) and nitrogen (method 984.13; AOAC, 2005) were determined in duplicate samples. Gross energy (GE) was determined using an isoperibol bomb calorimeter (Parr 1261 bomb calorimeter; Parr Instruments Co., Moline, IL, USA). The concentration of immunoglobulin (Ig) subsets (IgA, IgG, and IgM) in serum was determined using a Cobas Integra 800 analyzer (Roche, Mannheim, Germany). Blood chemistry measurements including albumin, globulin, total protein, alkaline phosphatase, alanine transaminase, aspartic acid transaminase, γ-glutamyl transferase, blood urea nitrogen (BUN), calcium, glucose, and phosphorus were determined on a Roche Cobas c702 automated blood chemistry analyzer (Roche, Mannheim, Germany).
Statistical analyses
Apparent total tract digestibility of DM, OM, CP, and GE was determined for each experimental diet (Kong and Adeola, 2014).
Experimental data were statistically analyzed using GLM procedures of SAS 9.2 (SAS Inst. Inc., Cary, NC, USA; 2008). Growth performance, nutrient digestibility, immunoglobulin subsets, and blood chemistry measurements were the response variables. Each animal was considered as an experimental unit. Orthogonal polynomial contrasts were used to test linear or quadratic effects of the mycotoxin adsorbent in the experimental diets, and a specific contrast was also used to compare PC and NC diets. The alpha level used for the determination of statistical differences was set at 0.05, and at 0.10 for tendencies.
Results
Mycotoxin analysis
Analyzed concentrations of DON in corn, SBM, control barley, or naturally contaminated barley were 0.6, 1.0, 0.3, or 25.7 mg/Kg, respectively. Analyzed concentrations of DON in PC, NC, NC1, or NC2 diet were 0.28, 2.94, 5.07, or 3.76 mg/Kg, respectively.
Growth performance
Pigs fed the PC diet had greater ADG and G:F (p<0.05) and tended to consume more feed (p = 0.061) compared with those fed the NC diet (Table 2) from d 0 to 7. The reduced growth performance was not recovered by the addition of the YCW except for final BW, which showed a tendency for a quadratic effect of YCW (p = 0.056). ADG, ADFI, and G:F were not affected by feeding the contaminated barley diet and there was no improvement in growth performance by adding the YCW to the NC diet from d 7 to 14. Pigs fed the NC diet had decreased ADG (p = 0.025) and tended to have decreased G:F (p = 0.096) compared to those fed the PC diet during the overall period.
Digestibility
Daily feed intake and nutrient digestibility were not different between PC and NC diets (Table 3). Fecal nutrient output tended to show quadratic responses (p<0.10) to the increasing addition of the YCW to the NC diet whereas nutrient intake and corresponding digestibility were not affected, except for CP intake, which showed a quadratic response (p = 0.041).
Serum immunoglobulin and blood characteristics
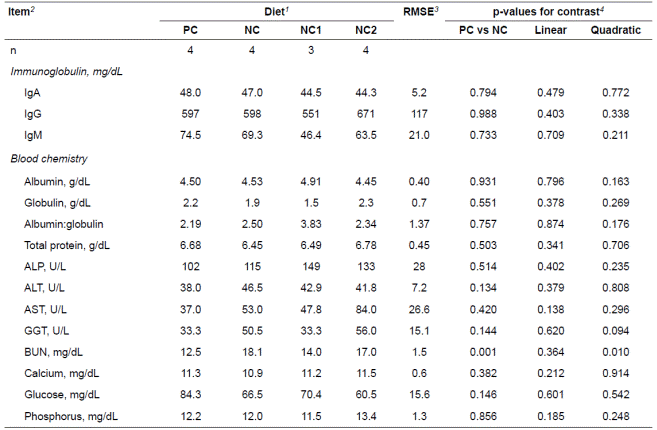
There was no effect of treatment on the concentrations of serum IgA, IgG, and IgM (Table 4). None of the blood chemistry measurements were altered by feeding the contaminated barley compared with the control barley, except BUN which was greater in the NC group (p = 0.001) compared with the PC group and showed quadratic responses to the increasing supplementation of YCW to the NC diets (p = 0.010).
Table 2 Growth performance of pigs fed experimental diets containing barley naturally contaminated with Fusarium mycotoxins.
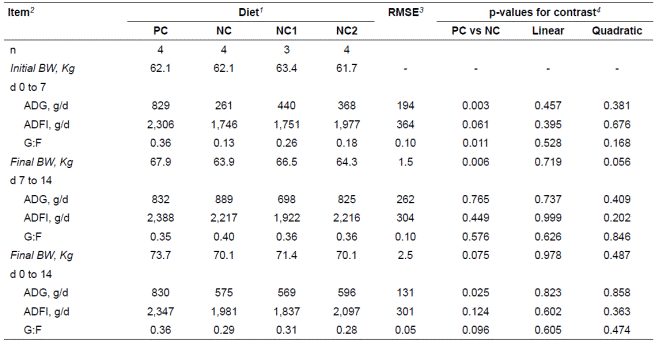
Table 3 Nutrients and energy digestibility of pigs fed experimental diets containing barley naturally contaminated with Fusarium mycotoxins.
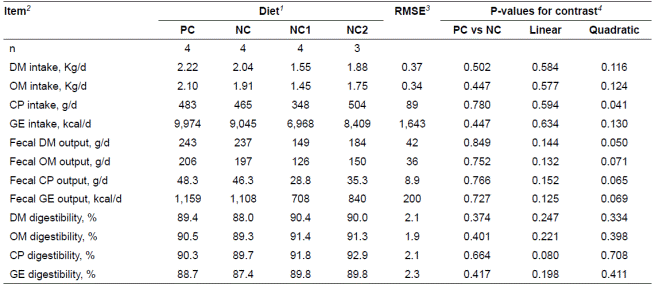
Table 4 Immunoglobulin and blood chemistry of pigs fed experimental diets containing barley naturally contaminated with Fusarium mycotoxins.
1. PC = corn-soybean meal based diet containing 20% of control barley; NC = corn-soybean meal based diet containing 20% of contaminated barley; NC1 =
NC diet supplemented with 0.2% mycotoxin adsorbent; NC2 = NC diet supplemented with 0.4% mycotoxin adsorbent.
2. ALP = alkaline phosphatase; ALT = alanine transaminase; AST = aspartic acid transaminase; GGT = γ-glutamyl transferase; BUN = blood urea nitrogen.
3. RMSE = root mean square error.
4. PC vs NC = contrast between the PC diet and the NC diet; Linear = linear effect of the graded concentration of the adsorbent; Quadratic = quadratic effect of the graded concentration of the adsorbent.
Discussion
Patience et al. (2014) reported analyzed DON concentrations of 0.25 mg/Kg in one diet and 4.6 mg/Kg in another diet when both diets were intended to include the same level of contaminated corn distillers dried grains with solubles. Distribution of Fusarium mycotoxin in ingredients is more likely to be uneven because Fusarium species may grow spottedly and produce mycotoxins under certain storage condition. This could have contributed to the uneven distribution of mycotoxins in the contaminated diets used in the present study.
Reduction of feed intake and consequent decrease of BW gain have been commonly observed when pigs are fed DON contaminated diets (Goyarts et al., 2005b; Chaytor et al., 2011b; Mok et al., 2013), and these were also observed in the present study. In addition, the ADFI of pigs fed the NC diet from d 0 to 7 was 24.3% less compared with those fed the PC diet, which was close to the predictive value (28.7%) of feed intake reduction calculated by the equation derived from the literature (Mok et al., 2013). However, growth depression was not observed from d 7 to 14, in agreement with Waché et al. (2009), who reported a transient negative effect of DON on growth performance of pigs fed diets with contaminated wheat.
Several mycotoxin adsorbing agents have been used for preventing the adverse effects of mycotoxins on pigs (Chaytor et al. 2011a). These products include YCW, natural clays, and cellulose products, and were evaluated through in vitro and in vivo studies (Weaver et al., 2013; Kong et al., 2014; Patience et al., 2014). Among adsorbents, YCW products showed a particular affinity for Fusarium mycotoxins and are hypothesized to prevent mycotoxin absorption in the gastrointestinal tract (Huwig et al., 2001; Kong et al., 2014). Several studies have shown the mycotoxin prevention effects of YCW in pigs, broilers, layers, and horses (Raymond et al., 2003; Swamy et al., 2003; Chowdhury and Smith, 2004). However, the reduced growth of pigs fed diets containing barley contaminated with Fusarium mycotoxins in the present study was not recovered by adding YCW to the contaminated diets. This is in agreement with previous pig studies in which YCW products were not able to recover the adverse effects of DON contamination on growth (Swamy et al., 2002; Mahan, 2010; Patience et al., 2014).
The percentage of in vitro adsorption of aflatoxin by the YCW was 92.7%, whereas that of DON was only 22.9% (Kong et al., 2014), indicating that adsorption efficacy of YCW is dependent on the type of mycotoxins (Patience et al., 2014). This difference was also pronounced in previous in vivo studies (Avantaggiato et al., 2005; Weaver et al., 2013). Grenier and Applegate (2013) demonstrated that 90% of DON absorption takes place in the proximal small intestine and thus YCW does not have enough time to adsorb DON. This may be attributed to the inability of YCW to recover the reduced growth performance resulting from feeding contaminated diets.
Excretion of cell wall degrading enzymes by Fusarium culmorum (Kang and Buchenauer, 2000) as well as increased activity of protease, amylase, and non-starch polysaccharide-degrading enzymes in wheat inoculated with F. culmorum (Matthäus et al., 2004) were observed. In addition, fungus protein may improve protein quality of the contaminated ingredient (Hamilton et al., 1988). Several studies have reported a DON-related improvement in nutrient digestibility (Goyarts and Dänicke, 2005a). In the present study, however, feeding pigs diets with barley naturally contaminated with Fusarium mycotoxins did not have a significant effect on nutrient digestibility, in agreement with Dänicke et al. (2004) who reported no effect of feeding DON-contaminated diets to pigs on nutrient digestibility. The reason for this discrepancy in results is still unclear. There was a quadratic effect of YCW on CP intake and quadratic trends for nutrients output. Because DON concentration varied among contaminated diets, it was difficult to interpret the effect of YCW on nutrient digestibility. Analyzed DON concentration in NC1 and NC2 diets was 5.07 and 3.76 mg/Kg, respectively, whereas it was 2.94 mg/Kg in the NC diet. Thus, it may be speculated that relatively high DON concentrations in diets with YCW might have confounded the effect of YCW on nutrient intake and output in the present study. A similar effect was observed in a previous study in which relatively high concentration of DON (6 mg/Kg) in the glucomannan polymer supplemented diets compared to that in contaminated diet alone (4.6 mg/Kg) confounded the effect of the glucomannan polymer (Swamy et al., 2002).
Inconsistent results in serum immunoglobulin subsets were observed when pigs were fed diets contaminated with Fusarium mycotoxins. Several studies have showed the up-regulation of serum immunoglobulin subsets in pigs fed diets containing Fusarium mycotoxins (Swamy et al., 2002; Goyarts et al., 2005b) whereas other studies showed no alteration of immunoglobulin subsets (Swamy et al., 2003; Chaytor et al., 2011b). In the present study, immunoglobulin subsets were not altered by feeding pigs with contaminated diets. Differences in experimental procedures, source and level of mycotoxins, as well as duration of toxin exposure might have caused the observed variation among studies (Döll and Dänicke, 2011). Blood urea nitrogen, indicative of renal health, in pigs fed the NC diet was higher than that of pigs fed the PC diet, and the pigs quadratically responded to increasing YCW levels from 0 to 0.4%. However, the values determined from all the treatments were within the reference range of BUN in pigs (8.2 to 25 mg/dL; Latimer et al., 2003). Other blood serum measurements did not change by feeding the NC diet compared with the PC diet, which is in agreement with the study conducted by Cote et al. (1985) who observed no alterations in hematology and serum chemistry in starter pigs fed diets containing DON (from 0.7 to 5.8 mg/Kg).
In conclusion, feeding diets containing barley naturally contaminated with Fusarium mycotoxins resulted in growth depression of growing gilts, whereas nutrient digestibility, immunoglobulin subsets, and blood chemistry were not affected. Furthermore, YCW supplemented to contaminated diets failed to ameliorate the negative effects of dietary Fusarium mycotoxins on growth performance of pigs.














